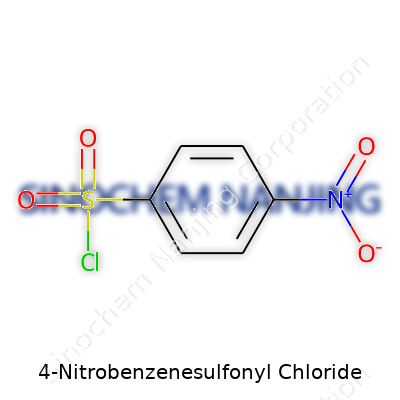
4-Nitrobenzenesulfonyl Chloride: An In-Depth Commentary
Historical Development
Chemists didn’t stumble on 4-nitrobenzenesulfonyl chloride by accident. Early work with aromatic chemistry in the late 19th century unlocked quite a few sulfonation routes. Researchers drew upon evolving nitration and sulfonylation methods to introduce both the nitro and sulfonyl chloride functionalities onto the benzene ring. Over the next decades, it became a predictable tool for protecting amines in peptide work, thanks in part to pharmaceutical research that demanded more stability and selectivity in syntheses. My early years in the lab were peppered with stories from mentors about benzenesulfonyl chloride derivatives, always coupled to tales of glassware fogged with fumes and protocols scrawled on yellowing pages. That sense of chemical tradition still hovers over bottles of this compound on stockroom shelves.
Product Overview
Known commonly as 4-nitrobenzenesulfonyl chloride or p-nitrobenzenesulfonyl chloride, the compound usually presents as a faint yellow crystalline powder. The label “p-Nosyl chloride” turns up often in peptide chemistry circles, shorthand that speaks volumes in research discussions. With a molecular formula of C6H4NO5SCl and a molar mass around 237.63 g/mol, it stands out as a reliable reagent for a narrow but critically important set of applications. Hardly an item for the general public, this is a specialty reagent engineers and scientists purchase in 25-gram bottles, watching expiration dates and humidity warnings closely. I once saw an entire batch ruined after someone failed to seal a bottle properly; clumping and hydrolysis don’t forgive mistakes in a tightly run synthesis campaign.
Physical & Chemical Properties
In the bottle, 4-nitrobenzenesulfonyl chloride’s yellow color hints at its nitro group, and its sharp, acrid odor leaves a quick impression. Melting point hovers around 142-145°C, but beware the fumes that waft off near that temperature—serves as a real reminder of the need for proper fume hoods. Solubility favors polar aprotic solvents like dichloromethane or acetone, though exposure to water leads to rapid hydrolysis, yielding the sulfonic acid. Its density rolls in at 1.65 g/cm³. High reactivity with nucleophiles, especially amines and alcohols, sets it apart from milder sulfonyl chlorides. In practice, the fast, exothermic release of HCl during reaction can catch new hands off-guard. Even small spills leave stains, an echo of its vibrant chemistry.
Technical Specifications & Labeling
Suppliers commit to purity, generally quoting figures of 98-99% as a minimum. Packaging must guard against moisture as even ambient humidity can trigger partial decomposition. Labels clearly display the hazardous nature of the reagent: corrosive to tissue and a respiratory irritant, with warnings outlined in several languages. Accurate batch numbers, shelf life, recommended storage conditions (preferably under argon or nitrogen, temperature below 30°C), and emergency procedures come standard. In my own practice, a secondary desiccator for the opened bottle has saved not just the material, but also the quality of sensitive downstream reactions.
Preparation Method
Most industrial processes build 4-nitrobenzenesulfonyl chloride from p-nitrochlorobenzene through sulfonation using chlorosulfonic acid. The resultant sulfonic acid couples directly with thionyl chloride or phosphorus pentachloride to yield the sulfonyl chloride derivative. Scale-up brings its own headaches: heat management, corrosion of equipment, and strict venting controls. From a bench-scale perspective, precise temperature control and steady stirring matter just as much as having anhydrous conditions; water’s presence ruins yields. My own missteps in the early days involved slow addition rates and underestimating exothermic spikes, which taught me to respect both the process and the substance.
Chemical Reactions & Modifications
The nitro group’s electron-withdrawing power makes the sulfonyl chloride even more reactive toward nucleophiles. In peptide chemistry, it serves as a reliable platform for introducing nosyl protecting groups onto amines, since it forms sulfonamide bonds with relative ease. That same nitro group on the para position tunes the electron density, making selective displacement reactions possible in multi-step synthetic chains. Beyond amine protection, chemists rely on 4-nitrobenzenesulfonyl chloride to activate alcohols for further conversion or to create complex sulfonate esters. The carbon skeleton stands up well to rigorous manipulations, so researchers blend it into both classic mechanisms and modern, catalytically driven transformations. Yield improvements over less reactive benzenesulfonyl chlorides have changed the design of synthetic strategies, particularly in medicinal chemistry.
Synonyms & Product Names
Call it 4-nitrobenzenesulfonyl chloride, p-nitrobenzenesulfonyl chloride, or p-Nosyl chloride and you hit the same target. Some catalogs abbreviate as NOS-Cl, and older literature may stick with terms like para-nitrobenzenesulfonyl chloride. Differences in naming reflect the global nature of reagent supply; chemists from China to Germany all recognize the structure, even if the pronunciation falters. A surprising array of trade names, usually centered on the “nosyl chloride” stem, appear across suppliers, forcing buyers to cross-check synonyms before ordering replacements. This confusion can clog communication until everyone agrees to stick with structural formulas in the final manuscript.
Safety & Operational Standards
Experience teaches respect for safety data sheets and actual accident reports. Skin burns, severe eye irritation, and potential for lasting respiratory harm set a serious tone. In routine use, full PPE—nitrile gloves, goggles, lab coat, and a well-maintained fume hood—becomes non-negotiable. Emergency protocols require calcium gluconate at hand for accidental skin contact and eye rinse stations ready for splashes. Training sessions in my lab always stress rapid cleanup, secondary containment trays, and pre-planned disposal routes for unreacted material and contaminated glassware. Even with precautions, users can’t afford to treat it casually; one slip with a spatula or a cracked bottle recasts a day’s work into a safety drill.
Application Area
4-Nitrobenzenesulfonyl chloride finds its main foothold in research chemistry, particularly synthetic organic and medicinal chemistry. Laboratories turn to it for installing protecting groups on amines and activating alcohols or phenols. Pharmaceutical development teams watch how this reagent unlocks selective transformations that boost the efficiency of multi-step syntheses. Outside peptide construction, it enters specialty polymer projects and environmental analysis, both as a derivatization agent and as a building block for more complex structures. Its heightened reactivity wins it a slot in new protocols for constructing heterocycles, a backbone of many drug discovery ventures. Not a compound for casual labs, its reach shows up wherever speed, selectivity, and robust downstream reactivity matter more than cost or ease of handling.
Research & Development
Ongoing research probes deeper into the selectivity, efficiency, and environmental impact of 4-nitrobenzenesulfonyl chloride’s chemical behavior. Universities and startups look at tweaking the basic structure to lower toxicity or enhance solubility in greener solvents. In peptide chemistry, efforts to minimize deprotection byproducts and tune reactivity carve out new chapters in protocol design. Automation and in situ monitoring streamline both safety and yield, encouraging new entrants to the field. Publishing trends show a steady rise in application notes featuring this reagent, usually focusing on higher purity, lower cost syntheses, or smarter, more sustainable waste management. My contacts in startups often share stories of adopting tailored versions for highly specialized targets, drawing in expertise from both academia and process engineering.
Toxicity Research
Researchers do not take the toxicology of 4-nitrobenzenesulfonyl chloride lightly. Acute effects, especially from inadvertent skin contact or inhalation, include burns and respiratory discomfort. Animal studies have flagged potential for longer-term health impacts, especially regarding pulmonary exposure to vapors. Regulatory bodies in Europe, the US, and East Asia track both occupational exposures and downstream environmental fate. Waste disposal draws scrutiny, thanks to the reactivity and persistence of sulfonated byproducts in the water stream. I’ve watched labs invest thousands in containment, neutralization, and analytical tracking, driven less by regulation than by the real-world risk to staff and students. Shared experience says: never trust risk tables alone—eyewitness accounts of the fallout from unplanned exposure shape best practices more than any written guideline.
Future Prospects
Interest surges in greener, safer alternatives for standard reagents in organic synthesis, and 4-nitrobenzenesulfonyl chloride isn’t immune to this trend. Bio-based solvents, milder activation protocols, and digital monitoring technology stir up new developments. Researchers already tinker with structural analogs to lower hazard potential and reduce persistent organic pollutants in waste. Expect regulatory pressure to push the market toward lower-impact formulations or process tweaks that minimize handling risk. Education in safety, technology upgrades, and cross-disciplinary work all shape where this compound goes next. Growth in pharmaceuticals and organic electronics keeps demand steady, but the edge belongs to those who can advance its use without compounding legacy environmental or health risks. Collaborative efforts among academic, industrial, and regulatory sectors look set to drive slow but meaningful change.
What is 4-Nitrobenzenesulfonyl Chloride used for?
A Closer Look at the Role of 4-Nitrobenzenesulfonyl Chloride
Plenty of chemicals quietly move the wheels of innovation, and 4-Nitrobenzenesulfonyl chloride ranks among those behind-the-scenes helpers. Its yellowish appearance seems nondescript, but it fits right into complex chemical reactions in laboratories and plants. Chemists see it as “nosyl chloride” for short, and the compound delivers a punch when tackling tough protection problems during synthesis. In my own experience in the lab, it often showed up during steps that required us to carefully shield parts of molecules, so unwanted reactions didn’t ruin months of work.
The world of pharmaceutical research might look crowded with unfamiliar names and technical jargon, but small chemicals such as nosyl chloride enable big leaps forward. It serves primarily as a sulfonylating agent, meaning it adds a sulfonyl group to a molecule—a key step to alter a material’s physical and chemical behavior. If you’ve ever worked through routes to create antibiotics or antiviral drugs, you know careful control over such steps determines whether a project survives the hurdles or grinds to a halt. I’ve seen colleagues lean on nosyl chloride to protect amines, especially during multi-step syntheses. That protection makes it possible to introduce specific atoms in exactly the right spots, a must for drugs that interact with cells in very targeted ways.
Why Precision Matters in Synthesis
One claim is that over 90% of new medicine projects rely on these chemical “protectors” to keep delicate reactions on track. Without reliable reagents like 4-nitrobenzenesulfonyl chloride, chemists would hit dead ends far more often, resulting in lost money and time. Proven facts support this: famous drugs like ampicillin and certain anti-cancer agents trace their lineage back through syntheses that call for amine protection and deprotection steps. Multiple peer-reviewed studies point to how nitrobenzenesulfonyl derivatives open up clean routes to important building blocks for peptides and nucleoside analogs. That’s not just a matter for research labs—a safer, more precise drug synthesis pathway translates to safer medicine on pharmacy shelves. Mistakes in these steps may result in harmful byproducts or drugs that don’t work as intended.
Risks, Safety, and Solutions Ahead
No chemical arrives without baggage. With 4-nitrobenzenesulfonyl chloride, health and safety concerns center around toxicity and its tendency to irritate skin, eyes, or lungs if mishandled. I learned the hard way in my first month in a lab to double up on gloves and make sure the fume hood fan stayed working. Responsible use depends on education, proper equipment, and updated workplace guidelines. Occupational data from regulatory bodies, such as OSHA and the European Chemicals Agency, show that clear instructions, training, and personal protection can prevent almost all accidents linked to nosyl chloride exposure.
Disposal and environmental impacts deserve mention, too. Like many sulfonyl chlorides, improper disposal can harm waterways and wildlife. The answer comes from better protocols in hazardous waste handling and investment in greener alternatives. Modern research explores ways to develop less hazardous sulfonylating agents, though right now, nosyl chloride’s reliability still keeps it in many toolkits.
The Real-World Impact
After talking with colleagues in both academia and drug manufacturing, I’ve come to see 4-nitrobenzenesulfonyl chloride as an enabler with sharp edges. Its ability to protect amines, create sensitive intermediates, and support complex syntheses gives immense value. Safe and informed handling, along with fresh research into sustainable chemistry, will keep this old workhorse relevant without adding unnecessary risk to people or the planet.
What are the storage conditions for 4-Nitrobenzenesulfonyl Chloride?
Recognizing Hazards Beyond the Label
Anyone who has worked in a lab knows that 4-nitrobenzenesulfonyl chloride demands a little more care than most shelf chemicals. It reacts quickly with water and gives off hydrogen chloride gas, which can corrode metal and sting your lungs faster than you expect. If you let humidity get to it, or cap the bottle improperly, you’ll notice clumping or a sharp smell before you see warning signs in your experimental results. One day, after rushing to weigh out a sample, I skipped the gloves. The irritation started within minutes. It’s sticky and unforgiving, so storage stops being a box-ticking exercise and becomes a real personal priority.
Temperature and Humidity Make a Big Difference
This compound doesn’t like heat or moisture. Most suppliers recommend storing it below 25°C but not freezing. In day-to-day lab practice, that means finding a shelf away from sun-exposed windows and heating vents. Labs in tropical areas often have to run air conditioners just to protect their more sensitive reagents. Even storing it on a top shelf can raise the temperature by a few degrees and shorten the chemical’s life. If you suspect high humidity in the room, it’s not excessive to double-bag the container or throw in silica gel packs to keep moisture at bay. Just last year, our building’s HVAC system broke down, and within a week the crystal structure of our open chemicals changed dramatically, making them unreliable for analysis.
Container Materials Matter
I learned early that not all containers are created equal. Glass makes the best choice as it doesn’t interact with the chlorinated sulfonyl group. I’ve seen plastic degrade subtly over time, especially lids, leading to tiny leaks. Always check for discoloration or warped caps. Repackaging the powder into sealable amber glass bottles can prevent UV light from accelerating decomposition. Never reuse old containers without cleaning them thoroughly first, or cross-contamination could undercut your experiment and health in unseen ways.
Labeling and Secondary Containment
The best label is one that can’t fade or rub off. Write hazards clearly in permanent ink. We once had a spill because a coworker misread a faded label and opened the jar outside the fume hood. Secondary containment, like a plastic bin with a snap lid, protects against spills and keeps everything organized. If you keep water-reactive compounds together—and separate from your acids and bases—you’ll avoid a lot of cross-reaction trouble. One quick tip: a splash of fertilizer or soap nearby can give away storage of reactive materials, so keep it simple and isolated.
Handling and Disposal Reflect Storage Practice
Gloves and goggles are non-negotiable. Try to handle it in a fume hood every time, as your clothes alone won’t stand a chance against fumes in case of an accident. If the bottle looks crusty, check the expiration before using it; degraded material should go into hazardous waste, not the regular trash. The U.S. Occupational Safety and Health Administration (OSHA) sets clear guidelines for handling and disposal—following these keeps your space legal and safe, and keeps clean-up costs low for everyone.
Invest in Regular Checks
Just setting storage conditions once won’t cut it. Regularly scheduled safety audits, monthly inspections, and quick training refreshers for new staff can catch problems early. Keep an inventory, and rotate your stock. These small steps, rooted in simple habits, make a world of difference for long-term lab safety and chemical stability.
What is the chemical formula and molecular weight of 4-Nitrobenzenesulfonyl Chloride?
Understanding 4-Nitrobenzenesulfonyl Chloride
Anyone who’s spent time in a chemistry lab or dived into how specialty chemicals shape modern manufacturing runs into names like 4-Nitrobenzenesulfonyl Chloride. Its formula, C6H4NO5SCl, gives a pretty clear picture: this compound brings together a benzene ring, a nitro group, a sulfonyl chloride, and keeps the structure manageable. The molecular weight checks in at 237.63 g/mol. For researchers, students, and folks in the pharmaceutical or materials sectors, these aren’t just numbers on a paper. Knowing a formula and weight often means the difference between success and disaster at the bench or in a production line.
Real Impacts of Small Details
Back in my days preparing organic intermediates, precision ran the show. Getting the composition wrong sets off a domino effect—yields drop, side reactions spike, or environmental impact rises without warning. 4-Nitrobenzenesulfonyl Chloride serves as a protecting group and activating agent in synthetic chemistry, especially for crafting complex molecules. Its clean molecular formula is not just a chemistry trivia fact; it tells pros exactly how to calculate stoichiometry, predict reactivity, and most importantly, how to make their processes safer. Never underestimate the importance of that molecular weight either. A half-gram error in weighing, multiplied by thousands in a plant, leads to mountains of wasted material and, if the compound is hazardous, bigger headaches with disposal and compliance.
Sourcing, Storage, and Safety
In one plant I worked at, sourcing 4-Nitrobenzenesulfonyl Chloride wasn’t hard, but handling it safely always drew extra attention. Dry conditions matter since sulfonyl chlorides react with water, kicking off corrosive fumes and making a mess of your yield. Labs and warehouses must run a tight ship—good ventilation, personal protective equipment, and proper labeling all make a difference. The nature of the compound, with its nitro and sulfonyl chloride functionalities, shows up in every safety data sheet. Both chemists and warehouse teams have to understand not just what it is, but the why behind the careful protocols.
Bigger Picture: Regulation and the Environment
Too many times, chemical handling skips a meaningful look at environmental impact. 4-Nitrobenzenesulfonyl Chloride isn’t something you chuck down the drain. Its degradation products include nitrophenol derivatives, which bring their own hazards. Down-to-earth stewardship involves minimizing spills, collecting and neutralizing waste, and making sure downstream processes fully consume or treat what’s left. Regulations push for this kind of care but culture reinforces it—building teams that know their chemicals by memory, not just by the label.
Addressing Challenges Head-On
Solutions depend on details. Correct storage containers, up-to-date inventory controls, and well-trained staff help reduce incidents and waste. It’s easy for companies to cut corners—modern regulation keeps most folks honest, but it still comes down to what teams do every day. Simple tactics like smaller bottles for portioning, regular safety checks, and electronic record-keeping sharpen awareness and keep everyone honest. Cutting corners on a small-molecule chemical like this one rarely ends well.
What safety precautions should be taken when handling 4-Nitrobenzenesulfonyl Chloride?
Understanding the Risks
Handling 4-Nitrobenzenesulfonyl Chloride feels like walking through a kitchen with a pan of simmering hot oil—one false move, and you could get burned. The sharp, irritating odor makes it hard to ignore, and its tendency to react with water or moist air turns a careless moment into a serious problem. I’ve worked with chemicals that seem tame on paper but bite back hard in reality, and this one earns respect for good reason. It causes nasty skin burns, hurts the eyes, and, if you inhale the dust, can turn a day at work into a hospital visit.
Protecting Your Body and Lungs
Gloves do more than keep your hands clean. Nitrile or neoprene stand up against 4-Nitrobenzenesulfonyl Chloride. I remember a colleague who tried latex once with a similar chemical and learned the lesson with a blistered hand. Double up if you’re handling large amounts or spills look possible. Long sleeves, closed shoes, and a sturdy lab coat let you focus on your work instead of worrying about splashes.
Goggles or a full-face shield mean no quick dash in and out with half-zipped protection. Face the stuff as if your eyesight depends on it, because it does. If you feel tempted to skip this for even a moment, ask anyone who needed an eye wash after a surprise splash—those minutes last forever. A snug-fitting mask or certified respirator blocks the fine dust; 4-Nitrobenzenesulfonyl Chloride doesn’t care about carelessness, so neither should you.
Keeping a Clean Bench
Every morning, before opening the containers, make sure the fume hood’s fan pulls with steady force. There’s no shortcut. Always keep the bottles inside the hood or a well-ventilated setup. Avoid open beakers on your workspace; lids off only for the necessary moment, then seal it tight. Paper towels, not sponges, work best if a spill happens; static and liquid don’t mix well around these compounds.
I learned early to label every bottle and flask—no handwriting games, no abbreviations. Rescue stations, eyewash, emergency showers—a quick look before starting your experiment can buy precious seconds in a crisis. Place everything you need within arm’s reach before you measure, pour, or stir.
Storage Habits that Matter
Keep 4-Nitrobenzenesulfonyl Chloride stored in a cool, dry cupboard, far away from acids, bases, and anything flammable. Humidity sneaks in through loose lids, turning a solid chemical into a wet, dangerous mess. Tighten those caps. Check containers regularly for leaks, cracks, or that faint hint of yellow on the rim—change them out fast if you spot trouble.
Dealing with Waste
Never pour leftovers or cleaning rinses down the drain. Solvents and residues need their own labeled waste bin, sealed and separate. Regular pickup schedules make sure nobody is tempted to ‘deal with it later.’
Training Makes the Difference
Experience builds confidence, but only proper training keeps everyone safe. Whenever new folks join the group, go over the protocol together. Don’t just hand them the safety sheet—walk through the steps, explain why shortcuts cause pain or paperwork, and insist on a safety drill at least once each semester.
Safety with chemicals isn’t just a checklist. It comes from habits, attention, and a stubborn refusal to take risks with your health.
How should 4-Nitrobenzenesulfonyl Chloride be disposed of?
Why This Chemical Matters
4-Nitrobenzenesulfonyl chloride pops up in research labs, pharmaceutical development, and chemical manufacturing. People who use it know it can be a useful tool, reacting efficiently for certain transformations. What many overlook is what the leftovers mean for people who never saw the first experiment. I remember the first time handling it — the yellow solid, the faint acrid smell — and the stern warning from my supervisor: “Respect the byproducts as much as you respect the reactant.” This stuck with me.
Health Risks and Environmental Impact
Breathing in its dust or touching the powder can cause health issues — irritation in eyes, throat, and skin. It isn’t just about direct contact, either. Mixed into water streams, leftover 4-nitrobenzenesulfonyl chloride doesn’t break down in a helpful way. The sulfonyl chloride group reacts with moisture, producing hydrochloric acid and generating heat — a recipe for burns, fumes, and secondary hazards. Nobody wants hydrochloric acid showing up uninvited in a river or a pipe system.
Improper disposal also creates another risk: persistent nitro-containing residues. These can disrupt bacteria and aquatic life, and some breakdown fragments linger for years. Stories circulate about minor spills causing headaches, skin rashes, or lingering lab odors, and these experiences aren’t urban legends. Workers deal with the fallout, from annoying to dangerous.
Proper Disposal: Responsibility in Practice
Labs with good safety culture treat this chemical as hazardous waste, not as general garbage. Here’s what practice looks like:
- Never pour it down the drain or mix it with regular trash.
- Keep it in sealed containers, clearly labeled, away from heat and moisture. I’ve seen leaky jars turn into mini-nightmares, so tight seals are non-negotiable.
- Consult the Material Safety Data Sheet (MSDS) and local hazardous waste guidelines. In some places, regulations demand specialized collection contractors come in and remove it — for good reason.
Disposal often means sending waste to facilities equipped for chemical destruction — commonly incinerators designed for toxic substances. Combustion at high temperature breaks down sulfonyl chlorides safely. Still, this doesn’t happen in a backyard or a university dumpster. Only certified hazardous waste handlers should take on this step. I've watched professionals kit up, collect waste drums, and cart them to facilities that carefully track each kilogram.
Building Better Habits
One thing I learned early on: minimize leftover chemicals from the start. Plan syntheses to avoid excess. Always neutralize small spills right away with sodium bicarbonate, then collect the solid waste separately. It takes a few extra minutes after an experiment, but it dodges headaches for janitorial staff, maintenance workers, and city employees.
Community Responsibility and Transparency
The question of proper disposal isn’t just for experts. Everybody, from the bench chemist to the environmental health officer, plays a part. Publicly available records, posted signs in labs, and regular staff training all help prevent chemical mishaps. When in doubt, it’s safer to ask a professional than to guess or hope for the best.
Throwing a cautionary eye at leftover 4-nitrobenzenesulfonyl chloride isn’t just bureaucracy. It’s about respecting the community, water supply, and the people down the hall who just want to get home safe. If managed responsibly, this chemical stays powerful without tipping into trouble.
| Names | |
| Preferred IUPAC name | 4-nitrobenzenesulfonyl chloride |
| Other names |
4-Nitrobenzenesulfonyl chloride p-Nitrobenzenesulfonyl chloride 4-Nitrobenzenesulphonyl chloride p-Nitrobenzenesulphonyl chloride 4-Nosyl chloride p-Nosyl chloride |
| Pronunciation | /ˈfɔːr ˌnaɪ.troʊ ˌbɛn.ziːnˈsʌl.fə.nɪl ˈklɔːr.aɪd/ |
| Identifiers | |
| CAS Number | 98-63-5 |
| Beilstein Reference | 1209284 |
| ChEBI | CHEBI:51838 |
| ChEMBL | CHEMBL15444 |
| ChemSpider | 8826 |
| DrugBank | DB14037 |
| ECHA InfoCard | 100.013.567 |
| EC Number | 220-899-4 |
| Gmelin Reference | 96962 |
| KEGG | C14333 |
| MeSH | D009725 |
| PubChem CID | 7477 |
| RTECS number | DB6475000 |
| UNII | W779N8AJZ6 |
| UN number | UN2811 |
| Properties | |
| Chemical formula | C6H4ClNO4S |
| Molar mass | 235.63 g/mol |
| Appearance | White to pale yellow crystalline powder |
| Odor | Odorless |
| Density | 1.53 g/cm³ |
| Solubility in water | Insoluble |
| log P | 0.5 |
| Vapor pressure | 0.00017 mmHg (25 °C) |
| Acidity (pKa) | -6.5 |
| Basicity (pKb) | 12.02 |
| Magnetic susceptibility (χ) | -62.0 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.620 |
| Viscosity | Viscous liquid |
| Dipole moment | 3.25 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 336.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –164.4 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -942 kJ·mol⁻¹ |
| Hazards | |
| Main hazards | Harmful if swallowed, causes severe skin burns and eye damage, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS05, GHS07, GHS08 |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H315, H318, H317, H334, H335, H410 |
| Precautionary statements | Precautionary statements: P261, P264, P271, P272, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362+P364, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-1-OX |
| Flash point | 91 °C |
| Lethal dose or concentration | LD50 oral rat 640 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 669 mg/kg |
| NIOSH | GE8575000 |
| REL (Recommended) | 5 mg/m³ |
| Related compounds | |
| Related compounds |
Benzenesulfonyl chloride p-Toluenesulfonyl chloride 4-Nitrobenzenesulfonic acid 4-Nitroaniline Sulfanilamide |

