4-Nitrobenzenearsonic Acid: Development, Uses, Safety, and the Road Ahead
Historical Development
Interest in organoarsenic compounds stretches back over a century. The search for effective compounds against infectious diseases gave rise to several derivatives, and 4-Nitrobenzenearsonic acid stood out during the rise of synthetic chemistry in the early 20th century. Chemists explored arsonic acids for their medicinal and agricultural promise, and over time, regulatory demands shaped how these substances came to the table. Reports from the 1940s and 1950s, found in agricultural bulletins and pharmaceutical research, hint at the initial applications and growing public health concerns. Experience shows that historical evaluations helped push for more controls and precise formulation standards, steering the use of arsonic acids into clearer boundaries.
Product Overview
Known for its distinct yellow crystalline form, 4-Nitrobenzenearsonic acid serves as a building block in both research and industry. Its structure supports a variety of downstream chemical processes, which has drawn attention from synthesis chemists as well as formulation specialists in the animal health sector. Commercial samples often appear as pure-grade crystalline solids, handled using strict internal protocols due to toxicity. For every batch entering a lab, each gram brings with it a legacy of industrial application and regulatory scrutiny.
Physical & Chemical Properties
4-Nitrobenzenearsonic acid displays a melting point around 245–250°C, signaling thermal stability within moderate ranges. The compound’s molecular formula, C6H6AsNO5, yields a molecular weight just above 260 g/mol. Slightly soluble in water, its nitro and arsonic acid groups offer reactivity with bases and reducing agents. Appearance sticks out – fine, yellow, odorless crystals, which tend to clump if stored under humid conditions. From experience in handling hazardous fine chemicals, storage inside well-sealed vials, away from light and moisture, avoids degradation and keeps reactivity predictable.
Technical Specifications & Labeling
Producers supply 4-Nitrobenzenearsonic acid with clear documentation. Typical specifications report content above 98% purity, checked by HPLC or titration. Impurities like aniline or related nitroaromatics, if found, stay below 0.5%. Every container carries clear hazard warnings: skull-and-crossbones pictograms, UN number, and handling guidelines, as required by GHS and international transport codes. Labeling describes storage recommendations, including protection from light, humidity, and temperature extremes. Chemical identity, batch number, manufacturing date, and safety phrases appear boldly – without exceptions – as required by national and international regulations.
Preparation Method
Lab synthesis usually takes the route of diazotization followed by arsonation and nitration on a benzene core. Starting with aniline, chemists convert the amino group through diazotization, then couple with sodium arsenite. Final nitration with concentrated nitric acid introduces the nitro group at the para position, controlled by temperature and stoichiometry. In industrial settings, reactors under negative pressure and automated reagent addition allow for precise control of the process. Years of running bench-scale trials reveal the need for close monitoring: an uncontrolled exotherm can trigger dangerous byproducts or violent reactions. Effluent handling protocols avoid arsenic contamination of wastewater, sticking to well-tested procedures.
Chemical Reactions & Modifications
The structure of 4-Nitrobenzenearsonic acid offers sites for further functionalization. The nitro group undergoes reduction to form aminobenzenearsonic acids, which paves the way for pharmaceuticals with antibiotic potential. The arsonic acid group itself supports metal complex formation and can swap hydrogen atoms for alkyl groups. During derivatization, choosing the right catalyst and reagent concentrations changes yields significantly. Reductive conditions turn the characteristic yellow powder into deeper colors, depending on substituents. From direct experience, avoiding metal contamination during these reactions remains crucial, given the tendency of arsonic acids to complex with trace metals.
Synonyms & Product Names
Chemists sometimes find themselves dealing with a long list of names for the same compound. For 4-Nitrobenzenearsonic acid, synonyms such as p-Nitrobenzenearsonic acid, p-Nitrophenyl arsonic acid, and 4-Nitrophenylarsonic acid show up in literature, catalogs, and shipping documents. The compound appears in older regulatory texts under trade names tailored for livestock additives, and in research monographs under systematic nomenclature. Knowledge of these names stops mix-ups in ordering and boosts clarity during regulatory submissions or literature searches.
Safety & Operational Standards
Contact with 4-Nitrobenzenearsonic acid requires strict control measures. Direct skin contact often leads to irritation or worse; inhalation presents risks of systemic arsenic poisoning. Labs assign fume hoods, gloves, and full-length lab coats as basic requirements. Spillage triggers immediate containment, using inert absorbents, followed by disposal under hazardous waste guidelines. Occupational exposure limits stem from OSHA and NIOSH data, but real safety comes from daily discipline in following SOPs. Fire characteristics differ from common flammables – the compound itself resists ignition, yet decomposes at high heat to toxic gases. Regular audits and continued staff training stand out in keeping accident rates low. Having watched regulations evolve, adopting international standards like ISO 45001 lifts both lab safety and workplace morale.
Application Area
4-Nitrobenzenearsonic acid found a place as a feed additive in the livestock industry, enhancing weight gain in poultry and swine in decades past. Some drug formulations capitalized on its unique chemical structure to tackle protozoal infections. More recently, the shift in public opinion and regulatory policy has forced a retreat from agricultural applications, citing concerns about arsenic residues in food products. Nonetheless, the compound persists in research – as a reference standard, as a chemical intermediate in the synthesis of pharmaceuticals, and in analytical method development. Scientists across academia still use it when probing arsonic acid chemistry, though today’s backdrop leans heavily toward evaluating risks as much as benefits.
Research & Development
The history of 4-Nitrobenzenearsonic acid tracks closely with changing expectations from both the marketplace and public health. Research often targets safer modifications, routes for detoxification, and greener synthesis. University groups explore arsonic acids for their coordination chemistry, chasing new catalysts or bioactive molecules. For researchers I’ve met, every discovery ties back to safe handling – innovation means nothing without rigorous evaluations of both efficacy and toxicity. On the practical front, developing sensitive detection methods to track parts-per-billion trace levels in food and water supports regulatory shifts and environmental assessments.
Toxicity Research
Arsenic compounds speak a hard language; their risks show up in every standard toxicology manual. Acute exposure causes symptoms resembling heavy metal poisoning. Chronic exposure, either through the workplace or contaminated environments, links directly to organ damage and cancer. Animal studies document the accumulation of arsenic in organ tissues following exposures, and human epidemiology supports efforts to reduce all non-critical uses. Regulatory reviews since the late twentieth century spotlighted the need for robust alternatives in animal husbandry, based largely on these toxicity data. Disposal concerns dominate technical discussions – accidental releases spark lasting environmental worries, as arsenic leaches into soil and groundwater. Having spoken with workers in remediation projects, arsenic extraction and soil washing create burdens that take years (and millions of dollars) to address if not managed from the start.
Future Prospects
The story of 4-Nitrobenzenearsonic acid seems to underscore a larger transition in both science and stewardship. Shifts toward greener chemistry create hurdles and set a higher bar for both development and application. The search for less hazardous analogues continues, with research groups focusing on modifying the arsonic acid core or seeking non-arsenic alternatives altogether. Many technical groups now set their sights on remediation methods that can pull arsenic efficiently from industrial effluents. Every year, international guidelines call for more accountability, and the lessons from 4-Nitrobenzenearsonic acid play a role in shaping future priorities, from synthesis through end-of-life disposal. The compound serves as a touchstone in the dialogue between chemical possibility and public responsibility, showing that progress means more than just new molecules — it means safer lives and cleaner environments.
What is 4-Nitrobenzenearsonic Acid used for?
A Closer Look at 4-Nitrobenzenearsonic Acid
4-Nitrobenzenearsonic acid doesn't usually show up in everyday conversation, even among folks who work with agricultural chemicals. Yet for years, its presence on commercial feed labels has mattered a lot more than people realize. This compound, originally developed as a feed additive for livestock, shows how chemistry and farming often get tangled up in ways that affect both food safety and the environment.
From Lab to Feed: Why Use It?
Farms started using 4-nitrobenzenearsonic acid to promote growth in poultry and swine. The science behind it isn’t just about getting animals to bulk up faster; the additive also helps prevent certain diseases in crowded or unsanitary living conditions. Back in the 20th century, many producers relied on arsenic-based additives like this one to guard flocks against infections that easily spread. The European Food Safety Authority and the U.S. Food and Drug Administration reviewed these practices time and again. At one point, millions of tons of feed contained such compounds globally.
While the early results made sense to industry—healthy, heavier chickens and pigs made for more food on tables—growing concerns followed. Reports revealed that some residues stuck around in animal tissue, and those didn’t just vanish when people cooked meat. Evidence also showed these chemicals could leach from animal waste into soil and groundwater. Over time, researchers established clear links between some arsenic-based compounds and health risks, including the development of cancer in both animals and humans.
Regulation, Reputation, and the Push for Alternatives
No one likes the idea of unnecessary chemicals lingering in the food supply or seeping into rivers. Once those risks made headlines, the public and regulators started pushing back. Groups like the World Health Organization published studies stressing that chronic exposure—even at low levels—can carry real dangers. Governments around the world started revising rules on feed additives, with many banning or tightly restricting 4-nitrobenzenearsonic acid and similar substances.
The phase-out has forced feed manufacturers to rethink routines. To this day, some countries permit limited use, but stories from farmers and veterinarians keep showing up about switching practices, re-examining sanitation, and investing in vaccines and better nutrition to keep livestock healthy without arsenic. That process is messy and expensive, with smaller outfits especially feeling the pinch.
Finding Safer Solutions
Looking back, using chemicals like 4-nitrobenzenearsonic acid highlights a classic tradeoff. Farmers needed to protect animals and run sustainable businesses, but society had to weigh those benefits against the health of the environment and people eating the meat.
The only way forward comes from attention to facts and willingness to adapt. A better understanding of pathogen control, dietary supplements, and animal management can help fill the gap. On my own family’s small farm, we ditched chemical additives a decade ago, betting instead on vaccination, cleaner pens, and regular vet check-ins. It took time and cost more, but the results made the headaches worth it—the animals stayed healthy, and customers felt more confident.
Researchers worldwide are still looking for new answers. Natural extracts, probiotics, and genetic selection offer some promise, but every farm has to deal with its own realities. To make real progress, governments, scientists, and farmers need to keep talking and share results—both successes and screwups. With more transparency and support, everyone stands a better chance at moving away from compounds like 4-nitrobenzenearsonic acid for good.
What is the chemical structure and formula of 4-Nitrobenzenearsonic Acid?
The Molecular Makeup
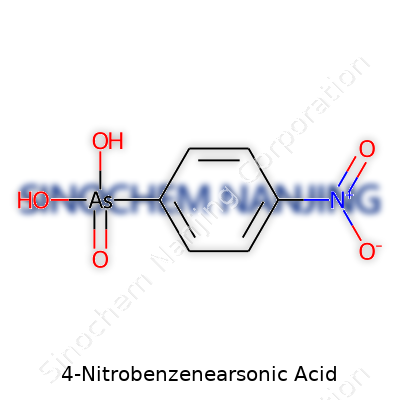
Science class might feel distant now, but the interest in how these molecules form the backbone of so much modern life sticks with me. 4-Nitrobenzenearsonic acid packs quite a punch for such a small molecule. Its formula reads as C6H6AsNO5. Looking at its structure, you find a benzene ring—a flat, hexagonal shape made from six carbon atoms and decorated with alternating double and single bonds. Most chemists spot the benzene ring right away; it’s the foundation of multiple industrial chemicals. In 4-nitrobenzenearsonic acid, this ring carries a nitro group (-NO2) at one position and an arsonic acid group (-AsO(OH)2) directly opposite it.
In my own college lab days, drawing out molecules helped concepts stick. Imagine orienting yourself around that benzene ring. Place the nitro group on the fourth carbon. Sling the arsonic acid on the first carbon. The molecule almost feels purposeful, like each part has its own job to do—one end for reactivity, the other for binding or carrying atoms with a heavy punch. Scientists call this “para” substitution, just meaning the substituents sit across from each other on the ring.
Why Chemists and Food Producers Watch This Compound
You often find this molecule in the agricultural sector. That caught my eye, given its history in the world of feed additives. Some poultry and swine producers once used related arsonic acids as feed supplements, claiming improved growth or disease control. As a science writer and curious onlooker, trends like these make me pause and dig in further.
There’s a lot at stake: Human health stands at risk if breakdown products show up in meat, water, or soil after widespread use. Research shifted quickly in the last decade. Back in the 2010s, the U.S. Food and Drug Administration started phasing out arsenic-based additives. Safety data piled up, raising eyebrows. Chronic arsenic exposure isn’t something you gamble with, given its link to cancer and organ damage.
The Bigger Environmental Picture
It’s easy to dismiss the impact of a single molecule, but, in practice, these atoms move through soil and water with lasting effects. Even tiny remnants influence regions downstream from farms and plants. From personal experience hiking and paddling around agricultural areas, you see how runoff and leaching really spread these molecules far and wide. One small drop in a feed trough eventually becomes part of a river or groundwater supply.
Ongoing monitoring remains essential. Universities and public health agencies keep a close eye on arsonic acid residues and breakdown products in food and drinking water. Many labs run regular mass spectrometry to spot these toxins at the part-per-billion level. These checks help guard public health and prevent problems up the food chain.
Building Safer Alternatives
As a writer who cares about the ripple effect of chemistry, seeing researchers look for better solutions inspires some hope. Teams now design new feed additives and crop protection agents with less risk and more scrutiny around breakdown paths. Instead of repeating old mistakes, modern chemists work alongside biologists and environmental scientists. Success requires pairing technical know-how with practical farm input and daily realities from the field.
For anyone seeking safer food and water, it’s clear that looking at molecular details has real-world payoff. Molecular formulas, group placements, and reaction paths still matter far beyond the test tube. Genuine progress means connecting molecular insight with healthy farms, cleaner groundwater, and safer plates at dinner.
Is 4-Nitrobenzenearsonic Acid hazardous or toxic?
Looking at the Dangers Up Close
4-Nitrobenzenearsonic acid, once used widely in animal feed additives, doesn’t get much attention outside scientific circles. People tend to focus on newer chemicals, but this compound sticks around in discussions about safety, health, and the environment. Chemicals related to arsenic rarely come with a friendly reputation, and this one follows that rule.
The Human Health Angle
Exposure brings worries for anyone working with it. Skin contact leads to irritation. Breathing dust means possible nose and throat problems. Arsenic-based compounds link to even more serious outcomes. Some animal studies point to organ damage from repeated doses, affecting kidneys and liver. Long-term effects remain a concern, especially considering the compound’s core—a blend of nitro and arsonic groups—means double the trouble for toxicity. The arsonic part is especially shady. In my own lab days, safety conversations about anything with arsenic always included gloves, special waste containers, even specific storage areas. There’s a reason: Chronic contact, even tiny amounts, adds up over time. The nitro group gives extra fuel to the risk—compounds with this group sometimes link to cell damage or disruption.
What Happens to the Environment
Everything dumped down the drain ends up somewhere. Chemicals like 4-nitrobenzenearsonic acid don’t just vanish. Studies measuring soil and water runoff from farms using additives with this ingredient show buildup. Certain plants and fish absorb this stuff and don’t let go. Wildlife can end up with more arsenic in their bodies than nature intended. It’s all part of the food chain now—another reason to give this chemical careful thought. Nobody wants a heavy metal legacy in their backyard.
Real-World Examples and Facts
Research from the early 2000s documented increased arsenic concentrations in soils and waterways near farms using these additives for chickens and pigs. Even the FDA recommended limits, pushing the industry toward safer alternatives. Many countries banned or restricted the use, not just out of precaution but as a direct response to measured soil and water contamination. Back in school, projects comparing different farm runoffs always ranked samples from treated areas highest in arsenic. These weren’t distant numbers—soil samples came straight from nearby agricultural towns.
Safer Paths Forward
Switching to feed additives without arsenic stopped the buildup problem. Farmers, including some in my own extended family, saw small bumps in cost but much less red tape. Regular soil tests now catch any contamination before it spreads. Research outfits keep tracking how old deposits move through the environment. Simple steps—solid waste handling, safe storage, personal protection—reduce health risks for those still handling older stocks.
A bigger solution grows from awareness and routine control. Regulation makes the difference. Countries keeping tighter controls on arsenic contamination see less risk, easier clean-up, and healthier people and land. Kids in school don’t worry about playing near fields. Local water sources run safer. It’s not just government that matters. Communities pitch in by pushing for better monitoring, education, and alternatives. In the end, what we know shapes how safely we live.
How should 4-Nitrobenzenearsonic Acid be stored and handled?
Respecting a Hazardous Chemical
Something that stands out right away about 4-nitrobenzenearsonic acid is the risk it brings along. Any compound with arsenic commands extra focus. Many people look at a name like this and move on, but workers in research labs, agriculture, or specialty manufacturing—these folks are dealing with the reality, not just a label.
Label the Danger: Keep Communication Clear
Mislabeling or poor documentation turns a risky chemical into a trap, especially when a lab gets busy or staff turns over. Labels should show hazard warnings, date of receipt, and key safety notes—not just the name. I’ve seen researchers breeze through bottles after a long shift, so clear signage, even a bold red warning, actually saves mistakes.
Keep It Dry and Sealed
Moisture and air make some chemicals break down or react. 4-nitrobenzenearsonic acid falls in this group. Glass containers with airtight screw caps make the best choice. Avoid metal lids—acids corrode, and then you have a bigger mess. Tight seals keep vapors inside, too, which helps protect everyone’s lungs.
Control the Climate—Stay Cool and Away from Light
Heat and sunlight can cause problems with chemical stability. Storing this compound below 25°C, away from direct light, helps prevent breakdown. Temperature swings cause condensation, ruined substances, and potential leaks. I keep my personal approach simple: store it at eye level in a dedicated cabinet, well labeled, with no source of ignition nearby. Never stack incompatible chemicals. Careless mixing leads to dangerous surprises.
Personal Protective Gear: No Compromise
Handling arsenic-based chemicals requires strict protocols. Lab coat, splash goggles, and proper gloves become non-negotiable. One slip can expose skin or eyes, so I always double-check gear before work begins. Clothes with sleeves, close-toed shoes—no shortcuts. Safety showers and eyewash stations belong within reach. Quick rinses can prevent lasting injuries if spills or splashes happen.
Ventilation and Containment Matter
Work done with powdered or volatile compounds means fine particles or vapors may escape. Fume hoods keep air clean and help workers avoid breathing anything harmful. Always open vessels within this controlled space. Good ventilation is not just a comfort; it’s health protection, especially for long shifts in the same workspace.
Safe Disposal: Ending Well
You can’t treat arsenic waste like household trash. Specialized containers for acids and heavy metals, kept separate from other waste, prevent accidental exposure or contamination. I’ve seen labs send samples to licensed hazardous waste handlers, which feels like a hassle but prevents far bigger, longer-term headaches.
Culture of Respect
Storing and handling 4-nitrobenzenearsonic acid is not just about compliance. It’s about looking out for others—your team, the environment, and whoever comes next. Training saves lives. Treating every step as crucial—labeling, containment, protections, and disposal—proves that safety isn’t just paperwork. It’s a choice everyone makes together, every day the bottle is in the building.
Where can I purchase 4-Nitrobenzenearsonic Acid and is it regulated?
What is 4-Nitrobenzenearsonic Acid?
4-Nitrobenzenearsonic acid stands out in chemistry circles and lab supply catalogs because of its unique properties. For folks working in specialized organic synthesis or toxicology research, it comes across the bench every so often. The compound owes its importance to a structure that features both a nitro group and an arsonic acid group attached to a benzene ring—an unusual combination that isn’t easy to come by outside of controlled supply networks.
Buying 4-Nitrobenzenearsonic Acid: Who Qualifies and Where to Look?
Most chemical suppliers do not keep this compound in general inventory, and for good reason. No local hardware store or standard online retailer like Amazon or eBay carries it for public sale. To find 4-Nitrobenzenearsonic acid, researchers and authorized labs often need to go through specialty chemical suppliers such as Sigma-Aldrich, Alfa Aesar, TCI, or ChemSpider’s listings. These companies cater to verified institutional accounts—think universities, government research labs, or registered corporate users with applicable credentials.
From my own years working in academic research, I saw firsthand how tight regulations work. Placing an order usually means navigating through supplier vetting, providing institutional documentation, and sometimes signing declarations about intended use. Private individuals rarely succeed in buying any arsenic-derived compound without a paper trail. This scrutiny stems from safety and misuse concerns, which suppliers take seriously since their reputations and legal standing are at stake.
Regulation: Public Health and Safety at the Forefront
4-Nitrobenzenearsonic acid isn’t just another chemical in a glass bottle—it’s an organoarsenic compound, and that classification means heavy regulation. In the United States, the Environmental Protection Agency (EPA) and the Drug Enforcement Administration (DEA) pay close attention to substances containing arsenic, especially ones that could enter environmental runoff or get misused. Outside the US, places like the European Union and Australia enforce their own restrictions and require buyers to have permits or specific professional credentials.
The 1970s and 1980s brought tighter control of arsenic agents in animal feed and water supplies. Stories of environmental contamination and public health scares around arsenicals led governments to clamp down. That past experience shapes current rules. Today’s chemical supply world puts human health first—purchasing or importing an arsonic acid compound often requires permits, record-keeping, and ongoing monitoring.
Challenges and Solutions in Sourcing for Legitimate Research
Researchers running into hurdles with acquisition sometimes feel frustrated. Even the right credentials do not guarantee smooth approval. More cooperation between suppliers, regulatory bodies, and institutional purchasing offices can help legit users gain access without needless delays. Digital databases that link regulatory clearance with purchasing systems can clear up paperwork bottlenecks and help track supply chain security all the way to end use.
Another practical solution includes establishing centralized institutional procurement. Researchers pool needs and resources, maintain secure storage, and handle waste properly. This not only meets compliance but also reassures the public that high-risk chemicals never leave responsible hands. As regulators continue to update guidelines, involving scientists in the process can create smarter, science-based policies. That’s the best way to ensure continued progress in chemical research while keeping communities and the environment safe.
| Names | |
| Preferred IUPAC name | 4-nitrophenylarsonic acid |
| Other names |
PASA Arsanilic acid p-Nitrophenylarsonic acid 4-Nitrophenylarsonic acid |
| Pronunciation | /ˈfɔːr ˌnaɪtroʊ ˌbɛnziːn ɑːrˈsɒnɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | 98-88-4 |
| Beilstein Reference | 1209696 |
| ChEBI | CHEBI:34680 |
| ChEMBL | CHEMBL20458 |
| ChemSpider | 21106275 |
| DrugBank | DB14055 |
| ECHA InfoCard | 100.006.148 |
| EC Number | 207-622-9 |
| Gmelin Reference | 8188 |
| KEGG | C13957 |
| MeSH | D009601 |
| PubChem CID | 6923 |
| RTECS number | CF9625000 |
| UNII | EH7C4I94WY |
| UN number | 1571 |
| Properties | |
| Chemical formula | C6H6AsNO5 |
| Molar mass | 265.08 g/mol |
| Appearance | White to light yellow crystalline powder |
| Odor | Odorless |
| Density | 1.74 g/cm3 |
| Solubility in water | slightly soluble |
| log P | 1.1 |
| Vapor pressure | 1.83 x 10^-7 mmHg (at 25 °C) |
| Acidity (pKa) | 1.92 |
| Basicity (pKb) | pKb ≈ 8.1 |
| Magnetic susceptibility (χ) | -69.5×10⁻⁶ cm³/mol |
| Dipole moment | 3.91 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 222.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -191.1 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1271.6 kJ mol⁻¹ |
| Pharmacology | |
| ATC code | P53AG10 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes serious eye irritation. Causes skin irritation. May cause damage to organs through prolonged or repeated exposure. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS07,GHS09 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H332, H372 |
| Precautionary statements | Precautionary statements: P261, P264, P270, P271, P272, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P312, P321, P330, P363, P405, P501 |
| NFPA 704 (fire diamond) | 3-1-2-OX |
| Flash point | 275°C |
| Explosive limits | Lower: 0.2% ; Upper: 2.0% |
| Lethal dose or concentration | LD50 oral (rat) 1,800 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 > 4640 mg/kg |
| NIOSH | TTJ19790RD |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.5 mg/m3 |
| IDLH (Immediate danger) | IDLH: 25 mg/m³ |
| Related compounds | |
| Related compounds |
Arsanilic acid 4-Aminobenzenearsonic acid p-Nitrophenylarsonic acid Roxarsone Nitrobenzene |

