4-Nitrobenzamide: A Comprehensive Perspective
Historical Development
Chemists working in the late nineteenth and early twentieth centuries often pursued new aromatic amide compounds, motivated by curiosity and the hunger for new dyes and pharmaceuticals. 4-Nitrobenzamide, with its distinct nitro group attached to the para position of the benzene ring, emerged as a logical extension of early work on aniline dyes and related nitroaromatic chemicals. Researchers found that attaching nitro groups could tune the biological and physical properties of benzamides. Over decades, refinements in nitration and amidation methods moved the synthesis away from cumbersome batch reactions to more precise, scalable chemical processes suitable for industry. As patents documented new pathways, the entry of 4-nitrobenzamide into specialty chemical markets could not be overlooked.
Product Overview
4-Nitrobenzamide finds use primarily as an intermediate in chemical synthesis, thanks to both the electron-withdrawing nitro group and the reactivity of its amide function. The yellow crystalline powder often lands in research labs and manufacturing setups pursuing specialty dyes, pharmaceutical precursors, and materials science experiments. The distinct odor, reminiscent of other nitroaromatic compounds, marks its presence in the workplace. Unlike its simple parent benzamide, the nitro substituent dramatically shifts its applications by altering both reactivity and toxicity profiles. Secondary uses show up in agricultural chemistry and, to a lesser degree, in pigment technology where robust light-fastness is crucial.
Physical & Chemical Properties
Solid at room temperature, 4-nitrobenzamide displays a melting point near 210–213°C and carries a yellow hue, with moderate solubility in organic solvents such as ethanol, acetone, and dimethylformamide. In water, its solubility drops drastically, aligning with many other nitroaromatic compounds. Chemical stability comes from the resonance stabilization of the aromatic ring alongside a strong amide bond, but the nitro group remains an active center for reduction and nucleophilic attack. A molecular formula of C7H6N2O3 and a molar mass near 166.14 g/mol allow straightforward calculation in lab settings. Its logP value suggests limited bioaccumulation, but the presence of the nitro group requires care because this moiety often introduces higher toxicity compared to unsubstituted benzamides.
Technical Specifications & Labeling
Professional suppliers catalog 4-nitrobenzamide using CAS Number 619-01-2 and a unique EINECS identifier. Labels clearly indicate it as a hazardous substance due to the nitro group, accompanied by hazard pictograms for irritant and potential environmental risks. Storage recommendations point toward cool, dry places, away from oxidizers and strong reducers. Specifications often demand a purity upwards of 98 percent, with moisture content and melamine impurities tightly controlled. Analytical reports usually include HPLC, NMR, and IR scan results to confirm identity and quality, which reassures researchers trying to avoid unwanted side products in syntheses.
Preparation Method
The preparation of 4-nitrobenzamide follows reliable chemistry: start with nitration of benzamide or, more commonly for scale, use 4-nitrobenzoic acid as the starting material. Benzamide nitration poses challenges with regioselectivity and over-nitration, so converting 4-nitrobenzoic acid to its acid chloride, followed by reaction with aqueous or alcoholic ammonia, produces the amide cleanly. Careful control of temperature and stoichiometry during conversion helps minimize decomposition and formation of byproducts. Sometimes, small-scale preparations draw on direct aminolysis of esters of 4-nitrobenzoic acid, which avoids the harsher conditions of acid chloride chemistry while sacrificing some yield. The final product often undergoes recrystallization from ethanol to purify the bright yellow crystals before storage and shipping.
Chemical Reactions & Modifications
In synthetic work, 4-nitrobenzamide's most prominent reactivity centers on the nitro group. Reduction steps, employing agents such as iron filings and hydrochloric acid or catalytic hydrogenation, transform the nitro functionality to an amine for further derivatization. Adding substituents to the aromatic core, such as halogenation or sulfonation, faces limitations due to potential interference from both the nitro and amide groups. Amide hydrolysis, promoted under acidic or basic conditions, liberates the parent 4-nitrobenzoic acid. Multi-step syntheses in medicinal chemistry often turn the nitro function into an entry point for heterocycle formation or cross-coupling reactions, assisted by contemporary palladium-catalyzed methods. Chemists working on conjugation to peptides or polymers look to the amide bond for strategic chemical handles, exploiting the balance of stability and reactivity provided by the molecule’s structure.
Synonyms & Product Names
Beyond “4-nitrobenzamide,” this molecule pops up as p-nitrobenzamide in older chemical literature, as well as 4-nitrobenzenecarboxamide and sometimes 1-amino-4-nitrobenzeneamide. Commercial sources may abbreviate it to PNBA, while international trade often leans on its CAS number (619-01-2) for universal identification. In pharmaceutical research, alternate designations may reflect its use as a building block or test compound under various project codes. Reference chemicals in regulatory filings should always check against the most up-to-date synonym lists to avoid cross-contamination in safety documentation.
Safety & Operational Standards
Handling 4-nitrobenzamide means paying close attention to standard safety procedures for nitroaromatic compounds. Direct contact with skin or inhalation of dust can provoke irritation and, over prolonged periods, cause health issues that resemble those from better-known nitrobenzenes. Facilities dedicated to its use maintain robust local exhaust ventilation, personal protective equipment (often including gloves, safety goggles, and dust masks), and authorized chemical waste disposal. In Europe and North America, workplace exposure guidelines keep airborne concentrations low through regular monitoring and rigorous training. Emergency procedures for accidental spills prioritize limiting environmental release and minimizing personal exposure by using absorbents and sealed waste containers. Even with material safety data sheets highlighting fire and reactivity risks, actual incidents remain scarce due to mature storage and handling protocols. Disposal usually routes through incineration at specialized facilities due to the molecule’s resistance to biodegradation in regular waste streams.
Application Area
Laboratories focused on new materials, agricultural chemicals, and small-molecule pharmaceuticals regularly pull 4-nitrobenzamide off the shelf as a versatile intermediate. Medicinal chemists value the combination of inertness in most reactions and the ease with which the nitro group can be swapped for other functions as ideas mature. In pigment and dye work, the molecule's structure contributes stability to formulations targeting applications exposed to intense light or oxidative conditions. Synthetic chemistry courses in higher education may feature it in multi-step synthesis exercises, teaching students the subtleties of functional group transformations. Agrochemical developers lean on its intermediate properties for small-scale production of specialized herbicidal agents that need further chemical tailoring before use in the field.
Research & Development
Advanced research projects employ 4-nitrobenzamide far beyond simple synthetic routes. Academic groups probing amide isomerization analyze how the nitro group tweaks bond rotation and hydrogen bonding patterns using NMR and crystallographic data. In computational chemistry circles, the molecule serves as a model to calibrate quantum mechanical predictions for electron distribution. Drug discovery projects, particularly in oncology, assess the biological activity of derivatives formed by further modification of either the nitro or amide function. Some polymer research projects incorporate this compound’s rigid aromatic core for structure–property investigations, coupling it to flexible chains for performance testing. Funding for applied research often revolves around finding new conversions of the core molecule that can drive down cost or open up access to molecules that otherwise require more expensive or less environmentally friendly starting materials.
Toxicity Research
Scientists never skip toxicity screening for new or specialty chemicals, nitroaromatic compounds included. Studies in both cell lines and animal models repeatedly flag 4-nitrobenzamide as more hazardous than its benzamide precursor, mostly due to the nitro group’s known links to hemotoxicity and potential mutagenic properties. Short-term exposures at high concentrations produce respiratory and mucosal irritation. Chronic effects, though less defined, have led regulatory agencies to flag compounds with similar structures for careful handling and restricted environmental release. Environmental studies warn about resistance to standard water treatment protocols, so research continues on advanced oxidative methods designed to break down aromatic nitro groups before discharge into municipal systems. Despite these warnings, strict operational standards keep any large-scale risk minimal, provided facilities do not cut corners on basic chemical hygiene.
Future Prospects
4-Nitrobenzamide may never grab headlines as a blockbuster drug or consumer product, but its role in supporting technological progress looks set to expand. As green chemistry campaigns gain ground, researchers experiment with milder, less polluting synthesis paths for its preparation, minimizing waste and hazardous byproducts. Pharmaceutical and agricultural innovators still see room for derivatives built from its backbone, especially where selective reactivity and stability under field conditions remain valuable. The wave of automation and miniaturization in chemical research will likely expand the number of laboratories using small batches for high-throughput screening platforms. Some academic chemists champion its use as a standard molecule for benchmarking new analytical techniques, arguing that a widely studied, structurally simple yet reactive compound brings value beyond its immediate commercial impacts. Regulations continue to push for tighter safety margins and more detailed long-term toxicological tests, driving investments in better detection methods and remediation technologies. For a specialty chemical etched into over a century of laboratory manuals, 4-nitrobenzamide’s relevance endures in the hands of today’s careful chemists and tomorrow’s innovators.
What is 4-Nitrobenzamide used for?
What Is 4-Nitrobenzamide?
4-Nitrobenzamide sounds like something you only run across in a textbook, but it has roots in everyday science that quietly shape the world. In its purest form, it looks like a yellowish powder. Its appearance may not impress, but those humble grains help craft many products we use without even realizing it.
The Powerhouse Behind Organic Synthesis
Lab benches across the world see jars of 4-Nitrobenzamide because researchers rely on it as a building block. The compound starts with a benzene ring, one of the basic structures in chemistry, but attaches a nitro group and an amide. That small change makes a big difference. Researchers have found ways to tweak molecules like 4-Nitrobenzamide to create everything from dyes to pharmaceuticals.
I remember my own days as a chemist in a medicinal chemistry lab. Our projects often relied on compounds just like this. Some folks think of the benchwork as just tinkering, but the stakes get real quickly. You want a drug that targets a cancer cell and leaves healthy cells alone? The scaffolding often comes from these fundamental chemicals. For 4-Nitrobenzamide, that means using its nitro and amide groups to build molecules that later become active ingredients in medicines or agricultural products.
Why It Matters for New Medicines
Scientists appreciate 4-Nitrobenzamide because it's reliable and accessible. It's often one of the first options picked for designing "intermediates"—these are the skeletons that chemists use to stitch together more complex compounds. For instance, drug discovery projects use it for constructing molecules that interact with key proteins in the body. Cancer research sometimes depends on related structures, making every step—from raw material to finished tablet—matter for patient health.
Looking back at industry reports, the rise of custom chemical synthesis matches up with discoveries made using fundamental chemicals. The Chemical Abstracts Service lists thousands of journal articles that mention 4-Nitrobenzamide, often in the context of finding new routes to create target drugs. When researchers at pharmaceutical companies need to scale up a candidate compound for a clinical trial, they return to chemicals like 4-Nitrobenzamide and ask: Can we make it better, faster, or safer?
Role in Dyes and Pigments
Not every project ends up as a medicine. Dyes, pigments, and photographic chemicals count among the surprising products derived from 4-Nitrobenzamide. That connection matters for more than just color. For example, the textile industry faces tight environmental rules. The process for creating dye intermediates like 4-Nitrobenzamide often determines how much waste gets left behind and whether workers stay safe around reactive chemicals. Safer syntheses not only protect people but also help companies meet environmental standards.
Environmental and Safety Concerns
Using 4-Nitrobenzamide isn't risk-free. It carries its own hazard, as do many chemicals in synthesis labs. Handling it without proper protection may bring health concerns. Some researchers have moved toward green chemistry: using less harmful solvents, recycling materials, and limiting exposure. The European Chemicals Agency and other regulatory bodies monitor how these compounds are manufactured and handled because safety oversight saves lives and keeps pollution in check.
What Lies Ahead
Innovation keeps growing. Organic chemists have started developing new reactions that use less energy, waste, and hazardous material. 4-Nitrobenzamide still serves as an important building block, but as industry moves toward cleaner technologies, it takes on new meaning. My experience in the lab showed me that progress often comes down to how we handle the details—the little things every researcher does in pursuit of something bigger.
What are the safety precautions when handling 4-Nitrobenzamide?
Why Working Safely with 4-Nitrobenzamide Matters
Anyone who’s spent time in a lab knows chemicals demand respect. 4-Nitrobenzamide may sound like something found only in dusty chemistry texts, but it turns up in plenty of research labs. It’s a solid, pale yellow powder that gets used in organic synthesis and as an intermediate for dyes or pharmaceuticals. Even though it isn’t one of the most notorious hazards, ignoring safety practices around it makes no sense. One splash, one sniff, or a gram spilled on bare skin can send a promising experiment sideways—or worse.
Personal Protective Equipment Isn’t Optional
In my early days as a lab tech, a mentor once handed me a pair of chemical splash goggles and said, “If you can’t see, you can’t do science.” Gloves, goggles, and a lab coat form the frontline against chemicals like 4-nitrobenzamide. This compound can irritate the skin and eyes, and inhaling its dust leads to breathing problems. Choose nitrile gloves, fit the goggles snugly, and keep those sleeves buttoned down. Even the best PPE won’t make up for careless handling, but skipping it shows disrespect for yourself and your colleagues.
Smart Engineering Controls
Working with powders, especially organics, gets messy fast. A chemical fume hood beats any other work surface for transferring, weighing, or cleaning up. Once, while weighing out what looked like a harmless yellow solid, I caught a whiff and lost my voice for half an hour. Turns out, the dust had drifted out of the beaker and straight up my nose. Arranging your workspace helps: small neat piles, dampened filters, and properly sealed containers keep the chaos contained. Always use a vacuum system with a HEPA filter if you need to clean up a spill—never a broom or compressed air.
Know the Hazards—Don’t Guess
Material Safety Data Sheets (MSDS) never made exciting reading, but they tell you exactly where trouble starts. With 4-nitrobenzamide, toxic effects come not just from direct exposure, but slowly, through repeated contact or poor storage. It’s classified as harmful if swallowed or inhaled, and animal studies highlighted its toxic properties. Never treat a solid like it’s safe just because it doesn’t splash.
Safe Disposal and Spills
Accidents won’t give you time for a rehearsal. Walk through the process ahead of time. For 4-nitrobenzamide, gather up contaminated materials with damp absorbents and place them in labeled, closed containers for hazardous waste pickup. Never flush it down the drain or toss it in the regular garbage. Storing chemicals in sealed, labeled bottles cuts down on confusion—nothing ruins a day faster than opening a jar of unknown powder.
Building Safe Habits for the Long Haul
Safety in the lab doesn’t depend on a checklist; it grows out of daily habits. Ask questions, double-check bottles, talk things over with colleagues. Teach new lab members to respect every new reagent, even if it seems plain. The chemical won’t care about your deadlines or research goals, but it will react if handled wrong. Small steps—washing hands, sealing bottles, reporting near-misses—add up. The science works better for it, and everyone goes home healthy.
What is the chemical structure and formula of 4-Nitrobenzamide?
Breaking Down the Chemical Identity
Digging into chemical compounds often gets technical fast, but every small detail in a molecule shapes how it behaves and interacts with the world. In the case of 4-Nitrobenzamide, the structure and formula guide everything from laboratory research to potential use in medicine or industry.
4-Nitrobenzamide belongs to the family of aromatic amides. The formula for this compound is C7H6N2O3. Looking at it, each part offers a clue about the molecule. The base, benzamide, starts with a benzene ring—six carbons linked in a circle, with hydrogen topping off each point. Swapping out one hydrogen for a CONH2 group brings you benzamide. Now comes the “4-nitro” twist: a nitro group (NO2) attaches itself to the fourth carbon of that ring, directly across from the amide. This makes the full name make sense—4-Nitrobenzamide.
The Shape and Influence of Structure
Each piece contributes to the nature of this compound. The nitro group stands out as a strong electron-withdrawing force. It tugs on the electrons swirling around the benzene ring, changing the chemistry game. This changes reactivity, shifts how the molecule interacts with other players, and tweaks solubility. The amide group means it can form hydrogen bonds with ease, which opens doors in both chemistry labs and biological systems.
Some might brush off small molecular details, but a simple change in where a nitro group lands on the ring will shift properties, from melting point to reactivity. That’s why chemists pore over every atom’s position while designing experiments. I’ve seen firsthand how a single tweak in structure leads to new routes in synthesis, along with unexpected results. Safety in the lab depends on knowing this structure too. Nitro compounds need special attention so experiments run smoothly and safely.
Why This Compound Matters
4-Nitrobenzamide doesn’t just look good in a textbook. It steps into roles as a building block in drug research, intermediate for dyes, or as a starting point for more complex chemicals. Nitro groups in general have a history in developing pharmaceuticals—for example, some antibiotic families lean on such arrangements for their action. In the research space, 4-Nitrobenzamide has helped scientists study aromatic substitution, helping shape the foundation of organic chemistry.
Accuracy about this compound’s structure and formula makes all the difference. Getting the numbers wrong spells trouble later, whether setting up a reaction, checking safety data, or searching out toxicity risk. At the university level, students learning to draw and interpret this molecule build habits that prevent laboratory mistakes years later. Hands-on practice—pencil to paper, ball-and-stick kits—cements these relationships. Misplaced groups have led to real-world accidents, underscoring the need for care and knowledgeable supervision.
Pushing Chemistry Forward with Reliable Knowledge
Training younger chemists calls for attention to these molecular details. Resources need clear diagrams and consistent naming practices. Digital tools make it easier to double-check formulas and visualize new compounds. Sharing reliable structural drawings and tested approaches helps everyone work smarter and stay safer.
There’s always room to get better at teaching molecular structure. Open communication between labs and universities, plus updated, accessible safety protocols, has driven down preventable mishaps. By keeping the basics clear—from C7H6N2O3 to the specific nitro-and-amide layout—we support better science and a safer lab culture.
How should 4-Nitrobenzamide be stored?
Understanding 4-Nitrobenzamide’s Characteristics
4-Nitrobenzamide shows up in many labs as an intermediate in chemical synthesis and research projects. The compound appears as a yellow crystalline powder, and its handling comes with a responsibility that can’t be brushed aside. While it doesn’t burst into flames at the slightest provocation, it poses real health risks if mishandled, including respiratory and skin irritation. Anyone who’s spent time working near dry chemicals knows how easy it is to ignore the details until something goes wrong, typically when shortcuts sneak into daily routines.
What Proper Storage Looks Like
Storing chemicals is less about fancy equipment and more about staying alert and consistent. I’ve worked with teams who prided themselves on minimal incidents, and their labs always followed the basics: keep it dry, keep it cool, and keep it away from incompatible substances. The same applies to 4-Nitrobenzamide. A tight-sealing, clearly labeled glass or high-density polyethylene container keeps moisture out and confusion down to zero.
Direct sunlight and temperature swings chip away at chemical purity. The best spot for this compound often turns out to be a designated chemical cabinet set apart from any acid, base, or reducing agents—never just the closest shelf. A flame-resistant storage cabinet, kept away from the main workspace, adds an extra layer of protection when handling larger quantities.
Labeling Matters—For Safety, Not Just Compliance
Mistaken identity with powders looks just as dangerous as mishandling outright. Over the years, I’ve watched accidents unfold not from product failure but from labels fading or falling off. The name, the date received, and pertinent hazard symbols make a difference. That small investment in attention helps others—students, technicians, or an emergency responder—understand at a glance exactly what sits in each bottle.
Why Environmental Controls Can’t Be Skipped
Humidity invites clumping and slows down the work, but exposure to water can sometimes trigger irritation issues, depending on the chemical. A room with reliable ventilation and climate control protects not just the chemicals but the people spending long hours handling them. Air filters and exhaust systems pull away harmful dust and vapors before they spread.
In reviewing accidents at different research facilities, I’ve seen how poor ventilation or poorly set up storage spaces magnified the consequences of small spills. It is worth investing in a good airflow system, especially in an active organic chemistry lab.
Disposal and Emergency Readiness
At my first job, nobody actually looked at the disposal protocol until a serious spill happened. That delay created a mess costly in both time and money. Each container with leftover 4-Nitrobenzamide should go into a clearly marked waste station specifically built for hazardous chemicals. Access to absorbent materials, eyewash stations, and detailed emergency instructions needs to stay within easy reach.
Training new staff and regular drills help everyone stay primed for a surprise. Documentation of where to store and what to do in case of contact turns a prepared team into the front line of prevention.
Building a Culture of Awareness
It’s tempting to believe that routine equals safety, but the real results come from repeating simple, proven steps every day. Safe storage of 4-Nitrobenzamide forms just one piece of the lab safety puzzle. Clear protocols, reliable equipment, and ongoing training shape a workplace where people trust the system and each other. Safety isn’t just a checklist—it’s a reputation built on action.
Is 4-Nitrobenzamide hazardous or toxic?
Why People Care About 4-Nitrobenzamide
Chemicals pop up everywhere—in labs, factories, classrooms, and sometimes where we least expect them. 4-Nitrobenzamide isn’t a household name, yet it finds a spot in research and the specialty chemical trade. Anytime I hear someone ask about safety or toxicity, it’s easy to guess there’s uncertainty. Nobody likes grabbing a bottle without knowing what it could do to skin, lungs, or the planet.
The Real Hazards In Plain Terms
Here’s what you get with 4-Nitrobenzamide: a bright yellow powder, little aroma to notice, but it packs more risk than it shows. Scientific data reveals that nitro compounds—especially if the nitro group sits directly on a benzene ring—can pose health hazards. The main issues stem from skin and eye irritation. Handle 4-Nitrobenzamide without gloves, a splash could mean a rash.
Breathe in the dust, and it can irritate your respiratory tract. Swallow it, and nobody recommends that route. Some closely related chemicals go deeper, showing links to organ damage with prolonged exposure. Toxicity figures from standardized animal studies usually fall in the “moderately harmful” zone, yet these numbers don’t mean harmless. Chronic exposure—spills at work, airborne particles over months—build risk, not just a one-time headache.
Ecological Impact—Not Just a Lab Problem
I’ve seen cleaning practices in old university labs that would make any environmentalist sweat. Many nitroaromatics resist breaking down, meaning they stick around if released into rivers or soil. Some chemicals transform as they degrade, turning into something even nastier. Specific data for 4-Nitrobenzamide points to low-to-moderate toxicity to aquatic life, but that figure isn’t an excuse for careless dumping. Water treatment facilities can't work miracles against persistent substances. Once it gets into the ground or air, it can stay for a long time, showing up far from the original source. “Dilution is the solution to pollution” sounds clever until the same chemical comes back through the water supply years later.
Working Smart With Risk
You can’t talk safety without mentioning accident prevention. Any time I’ve worked with new chemicals, I’ve learned to dig for the Safety Data Sheet. 4-Nitrobenzamide often calls for gloves and goggles, lab coats and fume hoods. Splash-proof barriers turn a risky process into a routine one. Keeping an eyewash station within reach says more about a company’s safety culture than a poster on the wall ever could.
Safe storage habits matter, too. Keep it cool, dry, and airtight. A little moisture could cause unexpected reactions, especially with other lab ware nearby. Clean up spills right away, using separate tools just for these kinds of jobs. Don’t let powder sit on the floor—every footstep spreads it further.
Responsibility Beyond the Workplace
Many people assume chemicals in industry are safely locked away. History disagrees. Accidents, illegal dumping, and plain old neglect have left lasting damage. Laws in the US and Europe push for better control of hazardous chemicals, but personal and professional vigilance does more than legislation. I’ve learned to ask questions, check bottles twice, and keep waste in designated containers. It’s not just protecting myself—it’s about looking out for co-workers, neighbors, and the place we call home. Knowledge, not fear, drives safer handling and better choices with every dangerous compound.
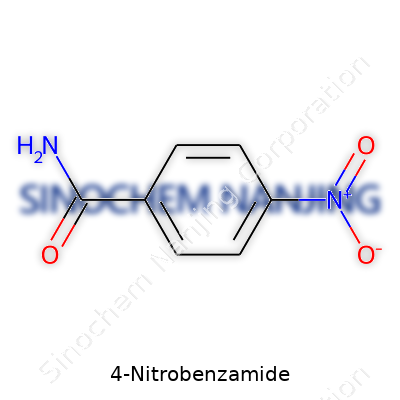
| Names | |
| Preferred IUPAC name | 4-nitrobenzamide |
| Other names |
4-Carbamoylnitrobenzene P-Nitrobenzamide Benzamide, 4-nitro- NSC 85290 4-nitro-benzamide |
| Pronunciation | /ˈfɔːr ˌnaɪ.trəʊ.bɛnˈzæm.aɪd/ |
| Identifiers | |
| CAS Number | 619-48-1 |
| 3D model (JSmol) | 4-Nitrobenzamide JSmol 3D model string: ``` NC(=O)c1ccc(cc1)[N+](=O)[O-] ``` |
| Beilstein Reference | 87253 |
| ChEBI | CHEBI:76011 |
| ChEMBL | CHEMBL3241509 |
| ChemSpider | 12394 |
| DrugBank | DB07703 |
| ECHA InfoCard | 100.031.403 |
| EC Number | 211-234-5 |
| Gmelin Reference | 85909 |
| KEGG | C14320 |
| MeSH | D009822 |
| PubChem CID | 7587 |
| RTECS number | KO5250000 |
| UNII | Z7F18Y61UC |
| UN number | UN1660 |
| Properties | |
| Chemical formula | C7H6N2O3 |
| Molar mass | 152.13 g/mol |
| Appearance | Light yellow to yellow crystalline powder |
| Odor | Odorless |
| Density | 1.41 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 1.13 |
| Vapor pressure | 5.19E-6 mmHg at 25°C |
| Acidity (pKa) | 11.9 |
| Basicity (pKb) | 14.06 |
| Magnetic susceptibility (χ) | -68.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.653 |
| Viscosity | 1.34 mPa·s (at 20 °C) |
| Dipole moment | 3.90 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 247.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -34.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2042.2 kJ/mol |
| Hazards | |
| Main hazards | Toxic if swallowed, causes skin and eye irritation |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS05,GHS07, GHS09 |
| Signal word | Warning |
| Hazard statements | H315, H319, H335 |
| Precautionary statements | Precautionary statements: P261, P264, P270, P271, P301+P312, P304+P340, P312, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-1-ox |
| Flash point | Flash point: 230 °C |
| Autoignition temperature | Autoignition temperature: 410°C |
| Lethal dose or concentration | LD50 oral rat 640 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 > 2000 mg/kg |
| NIOSH | RN/619-17-0 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 4-Nitrobenzamide: Not established |
| REL (Recommended) | 125 mg/L |
| Related compounds | |
| Related compounds |
Benzamide 4-Nitrobenzoic acid 4-Aminobenzamide 4-Nitroaniline 4-Nitrobenzyl alcohol 4-Nitrobenzoyl chloride |

