4-Nitroanisole: Substance Profile, Handling, and Practical Impact
Historical Development
4-Nitroanisole holds a peculiar spot in the context of early synthetic chemistry. Chemists started paying real attention to nitroanisoles in the late 1800s. Around the turn of the 20th century, the increasing demand for dyes and intermediates drove up the need for specialty nitro compounds. Early synthesis sometimes followed rough methods using direct nitration, but over time, researchers grew savvy about the role of catalytic conditions and selectivity. Those who handled the compound in early decades often found its persistent smell and strong yellow tint tough to ignore. Over the years, as analytical techniques improved, understanding of its roles in making dyes, pharmaceuticals, and other organic compounds shaped how the industry approached production and safety guidelines.
Product Overview
4-Nitroanisole, recognizable by its sunlight-yellow crystals, emerges most often in labs working on specialty organic intermediates. Its full chemical name, 1-methoxy-4-nitrobenzene, makes it clear: you get a nitro group sitting para to a methoxy group on a benzene ring. This small change to the molecular arrangement impacts both chemical reactivity and physical handling, giving 4-nitroanisole its particular value in target synthesis. Unlike general-use solvents or additives, this compound sits among niche intermediates, tuned for use in dye and pesticide synthesis, research projects, and custom molecule development. People working with the substance pay close attention to batch purity since contaminants throw off results in sensitive reactions.
Physical & Chemical Properties
Workers easily spot 4-nitroanisole by its sharp, crystalline form and distinct pale-yellow color. With a melting point commonly around 56 to 57 degrees Celsius, it doesn’t take much to turn it into a free-flowing liquid. Its molecular formula, C7H7NO3, pulls a molar mass of about 153.14 g/mol. It smells faintly sweet and can evoke a hay-like tone in concentrated form. Solubility marks an important line: it dissolves in ethanol, ether, and benzene, but sits stubbornly in water. The presence of a nitro group and a methoxy group makes the molecule both electron-rich and electron-deficient, which lets you push its reactivity in more than one direction. It resists oxidation in mild conditions, yet under basic or reducing setups, it jumps into new transformations.
Technical Specifications & Labeling
Bottles show clear hazard labeling because skin or eye contact isn’t trivial. You’ll usually see purity at 98% or higher, checked by both TLC and HPLC against an established reference standard. Dense labeling includes the CAS number (100-17-4), proper UN numbers for transport, and hazard pictograms that meet GHS standards. Suppliers who care about traceability list lot numbers, batch dates, and warnings about proper storage—cool, dry, and out of direct sunlight. Some research-grade packaging tucks a small desiccant pack in with each shipment, since clumped crystals mean exposure.
Preparation Method
Preparation of 4-nitroanisole most often starts from p-nitrophenol or anisole. Common practice today relies on the nitration of anisole using a mixture of concentrated nitric and sulfuric acids under careful temperature control. This ensures the nitro group lands at the para position, avoiding unnecessary byproducts. The reaction gets monitored for temperature swings since overheating triggers more substitution than intended, making purification harder. Post-nitration, chemists typically use cold water or alcohols to help the product crystallize out. After thorough washing, drying under vacuum eliminates residual acids and solvents. The final product usually passes through a round of recrystallization to sharpen up purity.
Chemical Reactions & Modifications
4-Nitroanisole serves as more than just a static intermediate. In the lab, reduction of the nitro group gives p-anisidine, which sees heavy use in manufacture of azo dyes and some pharmaceuticals. On the other hand, demethylation swaps the methoxy group for a hydroxy, shifting it to nitrophenol territory. Skilled chemists also tap into the nitroanisole’s versatility for coupling reactions and nucleophilic substitution, tailoring the molecule to suit pesticides and complex organic materials. Its performance in electrophilic substitution holds up due to the combined directing effects of both the nitro and methoxy groups, making it a handy scaffold for more intricate syntheses. Such properties place it on the shelves of those who like to build new organic frameworks efficiently.
Synonyms & Product Names
You’ll spot 4-nitroanisole listed under names like para-nitroanisole, 1-methoxy-4-nitrobenzene, and sometimes as p-nitroanisole. Sellers often categorize it by its CAS number (100-17-4) or EC number (202-819-0) to avoid confusion with the ortho- or meta- substituted isomers. On safety sheets, trade catalogs, and regulatory documents, these synonyms spring up based on regional habits or labeling traditions. Having clear confirmation of synonyms helps researchers and procurement teams avoid ordering the wrong chemical, especially with similar nitro- or methoxy- substituted benzenes in circulation.
Safety & Operational Standards
Safety culture around 4-nitroanisole owes a debt to experience rather than abstract guidelines. Handling dry crystals or vapors without gloves, goggles, or a fume hood quickly leads to irritation. The dust isn’t benign and can prompt sneezing, skin redness, or worse if exposure drags on. Inhalation may leave workers feeling light-headed or nauseous, and chronic exposure carries risks not always spelled out in old literature. Modern labs use chemical splash goggles, nitrile gloves, and ensure good ventilation. Spill kits keep sodium bicarbonate handy to neutralize residue, and waste disposal follows local EPA or REACH guidelines for aromatic nitro compounds. Firms who care about staff keep updated MSDS sheets within arm’s reach and require regular hazard training for all technical staff.
Application Area
This molecule’s ability to serve as an intermediate remains the heart of its appeal. Dyes owe their structure and vividness to transformations involving 4-nitroanisole, especially dramatic yellow and red shades for textiles or inks. Pesticide makers value it as a point of entry to more potent actives. When it comes to custom organic syntheses, chemists prize it for functional group transformations, especially introducing amino, hydroxy, or other substituents on the benzene ring. Some pharmaceutical research teams adapt it as a precursor to test new pathways for drug development, hoping to uncover fresh leads in anti-cancer, anti-fungal, or anti-bacterial screens. Few chemicals in a synthetic chemist’s arsenal punch above their molecular weight quite like this one.
Research & Development
Academic and corporate labs invest well in learning more about nitroanisoles, with modern focus zooming in on green chemistry. Reaction pathways that cut waste or use milder solvents see a steady stream of journal entries and patent filings. There’s solid interest in catalytic processes, with particular energy given to metal-catalyzed reductions for safer, faster production of downstream amino compounds. Analytical chemists sharpen up detection techniques to spot trace contaminants in water or soil, especially near production facilities. R&D teams in large chemical companies enroll fresh graduates to streamline older preparation routes, aiming for fewer byproducts and lower environmental burdens.
Toxicity Research
Environmental and occupational researchers have not ignored the potential risks. Chronic exposure studies in rodents and cell cultures highlight risks to liver and kidney function. The nitro group, especially after metabolic reduction, has shown potential to form refractory toxic metabolites. There’s enough epidemiological evidence to keep regulatory bodies interested in usage restrictions and possible carcinogenicity. Water and soil testing at sites near production plants shows minor persistence, though it tends to degrade under sunlight or in the presence of strong oxidants. Toxicology testing now includes aquatic organisms and soil bacteria to assess wider ecosystem impacts. Experts urge strong ventilation, personal protection, and regular workplace monitoring, recognizing that even common bench chemicals deserve respect.
Future Prospects
In industry and research, interest in 4-nitroanisole will likely stick around as demand for custom dyes, advanced materials, and pharmaceutical building blocks grows. There’s a steady shift toward greener nitration processes and smarter recycling of waste streams. Startups and academic labs tinker with enzymatic or photocatalytic methods, hoping to cut energy use and waste. Digital synthesis planning and AI-driven route optimization promise to squeeze even more efficiency from aging methods. With regulatory eyes focusing on environmental impact and potential toxicity, forward-thinking labs place a premium on testing safer derivatives and mapping out degradation products. In a changing chemical landscape, those who know how to work safely with nuanced molecules like 4-nitroanisole will have a practical edge.
What is 4-Nitroanisole used for?
Digging into 4-Nitroanisole
Every so often, a chemical names loses the battle for popularity outside science circles, but still quietly shapes everyday products. 4-Nitroanisole fits into that category. You won’t stumble on it in a supermarket, but if you trace it through manufacturing, you’ll find it nestled inside dyes, pigments, and even pharmaceuticals.
The Path from Laboratory to Industry
4-Nitroanisole shows up mainly as a building block. Chemical companies start with this compound to craft more complex molecules. The main gig: turning it into azo dyes. For decades, these dyes have been giving rich color to textiles, inks, and plastics. People love their vibrant blues and reds, and 4-Nitroanisole helps make those possible.
There’s also a role for it in pharmaceutical manufacturing. It works as an intermediate for certain drugs—think of it like the middle piece of a jigsaw puzzle, connecting where things start with where they need to be. Researchers figured out how to transform 4-Nitroanisole through various reactions, shaping active ingredients for medicines. The chemical itself won’t cure a headache, but it helps lay the foundation.
Weighing the Risks
Working around chemicals always brings a question about safety and health. 4-Nitroanisole is no different. Laboratory tests flagged it years ago for possible toxic effects, including cancer risk in animals. The evidence isn’t airtight for humans, but the prudent move is extra caution. Factories using this stuff set up protocols: protective gear, closed systems, regular air monitoring. It’s a headache for managers, but nobody wants a preventable incident showing up in the news.
Contamination also deserves attention. Any volatile chemical can escape, especially if storage tanks age or transport rules get ignored. This isn’t a substance most folks will ever touch, but stories of chemical spills remind everyone that leaks can ripple through drinking water, air, and soil. Cleanup means more than scrubbing a lab bench—sometimes entire communities have to chip in.
Building Better Systems
Having spent time in manufacturing plants, shortcuts never pay off with chemicals like this. Companies serious about worker safety keep a close eye on training and equipment. Sensors and double-layered containers cost money upfront, but the alternatives—lawsuits, sick employees, lost product—hit a lot harder later on.
Smart businesses also look for substitutions. Whenever chemists develop dyes or ingredients from safer starting materials, the pressure lifts off compounds with long side effect lists. That doesn't erase the need for 4-Nitroanisole today, but it creates a goal line for tomorrow's manufacturing.
Making Choices with Eyes Open
The world doesn’t run on chemicals like 4-Nitroanisole alone, but those behind-the-scenes compounds make a difference. From the vivid colors on T-shirts to ingredients inside a medicine bottle, this chemical leaves its mark. Honest attention to safety, sprinkling in innovation, and keeping the public informed—that’s the way forward. Society benefits not by avoiding these substances entirely, but by respecting their power and managing their risks.
What are the safety precautions for handling 4-Nitroanisole?
What Makes 4-Nitroanisole Tricky?
4-Nitroanisole shows up as a pale-yellow powder. People use it in labs and for chemical synthesis, especially when making dyes or pharmaceuticals. My own time in academic labs showed me how even a mild mistake around organic nitro compounds creates more problems than it ever solves. 4-Nitroanisole carries toxicity risks through inhalation, skin contact, and accidental ingestion. Overexposure can lead to dizziness, headaches, or skin and eye irritation.
Some old-timers used to handle such powders with their bare hands. Many of them later dealt with chronic coughs or unexplained headaches. Science now backs up what experience warned: nitro compounds shouldn’t touch your skin or get near your mouth and nose.
Personal Protective Equipment: Not Optional
PPE keeps trouble away. Latex or nitrile gloves and safety goggles matter here. I’ve seen gloves eaten through by careless exposure; always double-check chemical compatibility before trusting common lab gloves. Lab coats or chemical aprons stop accidental splashes from making things much worse. Clothes should cover your legs and arms. Never trust open-toed shoes in this environment.
Air Quality and Ventilation
Once, a colleague used 4-Nitroanisole outside a fume hood and triggered a fire alarm with the fumes. Good ventilation prevents this. Always transfer, weigh, or pour this powder under a certified fume hood. Local exhaust keeps concentrations low. I always check airflow with a simple tissue-paper test—if it doesn’t move, neither should the chemical.
Storage Wisdom: Loose Lids Don’t Cut It
Moisture sometimes turns a safe powder into a clumpy mess. 4-Nitroanisole works best when kept in tightly sealed, labeled containers away from incompatible chemicals like strong oxidizers or acids. I’ve seen cabinet corrosion from careless storage. Stash it in a cool, dry spot designed for organic chemicals. No storing near heat sources or sunlight. The best chemists I learned from kept obsessive inventory records—the right label meant no confusion after a long shift.
Spill Responses: Clean It Like You Mean It
Even careful hands drop things. If you spill the powder, don’t blow or sweep it. Dampen it down with a wet paper towel or proper absorbent, scoop it up, and never forget your gloves and goggles. Dispose of clean-up waste in hazardous bins—the risk isn’t worth cutting corners. I always reported a spill to the supervisor, no matter the size; hiding mistakes usually multiplies them.
Fire Safety: Don’t Underestimate Risks
While 4-Nitroanisole isn’t classed as highly flammable, organic powders can surprise you. Once, a spark from static electricity by a wool sweater set off a minor puff of smoke. Keep fire extinguishers—preferably CO₂ or dry chemical—near your work area. Avoid sources of ignition, unplug any chargers, and ban hotplates unless they’re essential.
Training and Documentation: Responsibility Goes Beyond the Individual
No one wants to be the person who leaves a mess for someone else. Safety data sheets must stay accessible, and everyone using the chemical should understand the health and reactivity risks. In my experience, regular reminders—short Toolbox Talks before starting a project—save more time and injuries than any warning label. Ongoing training protects both you and everyone who works in the same space.
Looking Ahead: Shortcuts Don’t Pay
Working with 4-Nitroanisole rewards straightforward caution. No respected lab or experienced worker trusts luck. From gloves to clean-air practices to sober respect for documentation, every habit shapes the next day’s outcome. Experience shows that a little attention today prevents a lot of pain tomorrow—and that’s a lesson that holds up in any serious laboratory.
What is the chemical formula of 4-Nitroanisole?
Understanding 4-Nitroanisole at the Molecular Level
4-Nitroanisole comes with the formula C7H7NO3. Anyone who took high school chemistry might remember seeing this structure drawn out: a benzene ring carrying a nitro group (–NO2) at the fourth position and a methoxy group (–OCH3), which makes it an anisole derivative. It’s a small detail on paper, but formulas like this aren’t just for exams. They are used every day in labs, research, and even industry.
Real-World Relevance
4-Nitroanisole isn’t just an academic curiosity. This compound pops up in the production of dyes and as a chemical intermediate. Chemists look at C7H7NO3 and know instantly it will react in predictable ways. The presence of the nitro group makes the ring less reactive for certain types of substitutions, but it opens doors for reductions and other transformations. Having worked in synthetic chemistry, the straightforward insight a correct formula provides speeds up any process, from planning experiments to troubleshooting why a reaction fizzled out.
Why Details Like a Chemical Formula Matter
Accuracy matters, especially when safety is on the line. Missing or swapping an atom can mean using the wrong chemical — and in a commercial setting, that could mean wasted money or, worse, a safety risk. Even a basic formula like C7H7NO3 acts as a universal shorthand, cutting across language barriers. In my own experience, tracking down a mislabeled chemical set back a project by a week. With hazardous compounds, the right formula can literally be the difference between business as usual and a hazmat team arriving.
Data Integrity and Research
Anyone writing scientific articles or patents must make every chemical formula count. Regulatory agencies hold companies to strict standards, and errors can lead researchers down the wrong path. I’ve seen grant applications flagged over a single typo in a compound name. This costs researchers time and credibility. A correct formula isn’t just a technicality — it is a foundation.
Improving Practices in Chemistry
Mistakes slip in where there’s a rush or when people cut corners on standard operating procedures. Simple habits, like double-checking reference materials or using digital tools that flag errors, reduce those risks. For students just starting out, learning to write and check chemical formulas precisely builds a foundation they’ll rely on for years. In the lab, reviewing safety data sheets and making sure what’s on the bottle matches what’s on the formula sheet keeps everyone safer and more efficient.
From Formula to Application
4-Nitroanisole, with its formula C7H7NO3, might not sound glamorous, but chemistry depends on this kind of precision. Real people use this data to make decisions — sometimes fast, sometimes with a big investment on the line. A solid grasp of these building blocks helps avoid expensive mistakes and keeps new research moving ahead. Every compound tells a story, and it all starts with getting the basics right.
How should 4-Nitroanisole be stored?
Recognizing the Hazards
Anyone who has worked with lab chemicals long enough knows each bottle has its own story and quirks. 4-Nitroanisole, while not as infamous as some reagents, brings its own list of concerns. With chemical safety, past experience shapes habits: I once opened a poorly sorted storage cupboard, and the confusion wasn’t just annoying—it made for real hazards.
4-Nitroanisole, known for its pale-yellow crystals and its use in research and dye production, gives off toxic fumes if heated or left to break down. It isn’t especially unstable, but it stands on the list of substances you can’t forget about. Over time, containers that aren’t sealed right invite trouble—inhalable dust, unpredictable chemical reactions, or leaks. Safety data sheets mark 4-Nitroanisole as harmful if swallowed, inhaled, or absorbed through the skin. Inhalation and prolonged exposure can damage organs.
Storing with Safety, Not Just Routine
Nobody should treat storing chemicals as bureaucracy. I remember a graduate student who thought an ordinary plastic tub would make a good home for substances labeled as “not very hazardous.” The tub cracked from a previous spill—what leaked out after a week needed a lot of time to clean. 4-Nitroanisole deserves a lot more respect.
The right place for this compound is in a cool, dry, well-ventilated cabinet. Shelving needs strength and chemical resistance—metal with a corrosion-proof finish or heavy-duty plastic, not plywood likely to trap moisture. Sunlight and strong room lights steadily break down many chemicals; opaque or tinted glass containers cut exposure. Never store chemicals near a sink, heat, or food.
Store 4-Nitroanisole in clearly labeled, tightly capped bottles. If the label fades or falls off, confusion quickly follows—nobody needs to play chemical detective after an accident. I’ve seen the cost of neglect here. It’s tempting to skip secondary containers, but they limit spreading if leaks occur. Relying on household shelving is a shortcut that often fails chemical workers; a chemical-resistant cabinet with a lip at the front stops bottles from sliding and breaking during earthquakes or accidents.
Good Habits Protect People
Using shared lab spaces or crowded chemical stores raises another issue: cross-contamination. 4-Nitroanisole belongs with organics, away from acids, oxidizers, and flammables. Mixing up storage groups leads to fire risks. If a spill happens, absorb with sand or inert material and wear gloves, goggles, and long sleeves. Open bottles in a fume hood, not on an open bench.
Regular inspections catch problems before they grow. If you spot crusty crystals or an odd odor, review your material. Unfortunately, many labs ignore expiration dates, but chemical age makes a difference. Old, unstable compounds produce unexpected products—there’s a reason some places choose to carry only small quantities and dispose of old stock quickly.
Solving Common Storage Mistakes
Education fixes more problems than expensive cabinets. Training for new researchers, regular room walkthroughs, and peer reminders create safer habits. Transparent, simple signage—something you can read from across the room—reduces errors. Institutions that keep inspection reports public often see fewer accidents. If the budget allows, upgraded cabinets with automated monitoring catch humidity or temperature swings before damage happens.
Storing 4-Nitroanisole safely is good chemistry practice, but more than that, it’s respect for everyone in the building. Mistakes cost time, health, and trust. Treat every bottle as something that deserves care, and the rest of the safety routine doesn’t feel like a chore.
Is 4-Nitroanisole hazardous to health or the environment?
Navigating Chemical Hazards in the Everyday World
People working with chemicals like 4-nitroanisole often have safety on their minds. This compound, usually found as a pale-yellow solid, pops up in the production of dyes and organic chemicals. I’ve spent time in labs and factories where the air fills with unfamiliar scents—4-nitroanisole blends right in until the headaches or nausea set in. Over the years, I’ve seen what can happen when basic safety edges off the priority list.
Human Health Under the Microscope
Inhaling even low amounts of 4-nitroanisole powder or dust won’t go unnoticed. Short-term exposure to the skin, eyes, or airways brings irritation—hands itching or red eyes become daily visitors for workers without gloves or goggles. A bigger concern comes from research linking this chemical to more troubling effects. The National Toxicology Program and several journals have flagged it as a possible carcinogen. Animal studies show liver and bladder tumors cropping up after repeated doses, a risk that follows people home after long shifts. The International Agency for Research on Cancer hasn’t yet given its final word, but health agencies recommend treating 4-nitroanisole with caution.
As someone who’s watched health and safety meetings slide into the background when deadlines get tight, I can't forget that cutting corners or skipping proper ventilation invites trouble. Workers breathing unfiltered air or picking up contaminated equipment put themselves at risk without ever realizing what hit them. Skin absorption is a serious route for this compound, and it will not always leave symptoms until significant harm begins. The Mayo Clinic highlights that chemical exposures are among the most common causes of occupational illness in some sectors, which makes this threat personal for many families relying on industrial jobs.
Environmental Fallout
Once 4-nitroanisole slips into wastewater or seeps into soil, problems multiply. The compound doesn't break down easily—soil and water samples have shown residues months after initial contact. Aquatic life faces real dangers, with toxicity affecting fish and smaller organisms, tipping the ecosystem out of balance. I’ve walked along creeks near production sites, where slicks and dead fish say more than any report could.
Groundwater carries traces far from factories, threatening wells and drinking water. These chemicals build up over time, so what seems like a small spill can end up poisoning a field or stream over several seasons. Regulatory bodies in North America and Europe set limits for 4-nitroanisole discharge, aiming to keep levels far below what would threaten people or wildlife. From my own recordkeeping, surprise inspections rarely catch everything—illegal dumping and poor containment still slip by.
What Can Be Done?
Prevention starts with education and real accountability on site. In my experience, airtight rules only work when supervisors listen to workers and treat reports of symptoms or spills seriously. Companies benefit from closed-loop systems that reuse water and trap hazardous dust, cutting down on escape routes for chemicals. Government inspections, harsher fines for violations, and rewards for cleaner operations all help keep the pressure on manufacturers. But the attitude on the shop floor counts at least as much—open lines for safety feedback, regular health checkups, and investing in better protective gear make a difference you can see and feel.
Anyone in communities near chemical plants deserves full access to data and to have a voice in environmental planning. Posting remediation plans and water testing results publicly, holding open forums, and funding local cleanup efforts show respect for the people most at risk. 4-nitroanisole proves that keeping toxic chemicals out of bodies and waterways calls for more than compliance—it demands building trust and using common sense day by day.
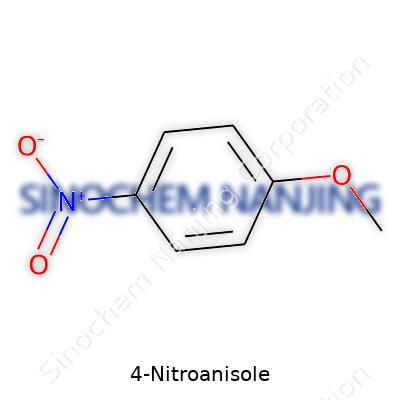
| Names | |
| Preferred IUPAC name | 1-methoxy-4-nitrobenzene |
| Other names |
p-Nitroanisole 4-Methoxynitrobenzene 1-Methoxy-4-nitrobenzene p-Methoxynitrobenzene |
| Pronunciation | /ˌfɔːrˌnaɪ.trəʊˈæn.ɪ.səʊl/ |
| Identifiers | |
| CAS Number | 100-17-4 |
| Beilstein Reference | 1208739 |
| ChEBI | CHEBI:77722 |
| ChEMBL | CHEMBL299126 |
| ChemSpider | 54639 |
| DrugBank | DB04219 |
| ECHA InfoCard | 100.014.337 |
| EC Number | 4.1.1.32 |
| Gmelin Reference | 613540 |
| KEGG | C14378 |
| MeSH | D009635 |
| PubChem CID | 7417 |
| RTECS number | OA9625000 |
| UNII | 0D0V5XKZB4 |
| UN number | 1667 |
| Properties | |
| Chemical formula | C7H7NO3 |
| Molar mass | 167.14 g/mol |
| Appearance | Light yellow crystalline powder |
| Odor | Odorless |
| Density | 1.209 g/cm3 |
| Solubility in water | slightly soluble |
| log P | 1.98 |
| Vapor pressure | 0.0033 mmHg (25 °C) |
| Acidity (pKa) | 18.9 |
| Basicity (pKb) | 11.02 |
| Magnetic susceptibility (χ) | -62.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.5890 |
| Viscosity | 2.27 mPa·s (at 60 °C) |
| Dipole moment | 3.96 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 243.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -50.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2455 kJ·mol⁻¹ |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause respiratory irritation, suspected of causing genetic defects. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07,GHS09 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P261, P264, P271, P280, P301+P312, P304+P340, P305+P351+P338, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-1-☴ |
| Flash point | Flash point: 113°C |
| Autoignition temperature | 420 °C |
| Explosive limits | Explosive limits: 0.75–5.2% |
| Lethal dose or concentration | LD50 oral rat 1750 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 3000 mg/kg |
| NIOSH | TT2625000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of 4-Nitroanisole: "Not established |
| REL (Recommended) | 50 mg/m³ |
| IDLH (Immediate danger) | IDLH: 200 mg/m3 |
| Related compounds | |
| Related compounds |
4-Nitroaniline 4-Nitrophenol 4-Nitrochlorobenzene Anisole Nitrobenzene |

