4-Nitro-N,N-Dimethylaniline: An In-Depth Commentary
Historical Development
There’s a good bit of history tied up in 4-Nitro-N,N-Dimethylaniline, a molecule that tracks right back to the roots of modern organic chemistry. In the late nineteenth century, as chemical dyes pushed forward the Industrial Revolution, researchers hunted for ways to synthesize brighter, more resilient colors for fabrics and art materials. 4-Nitro-N,N-Dimethylaniline entered commercial consciousness as a key intermediate in azo dye manufacture. Chemists discovered that placing a nitro group onto the aromatic ring of N,N-dimethylaniline not only changed the color profile of resulting dyes; it shaped the whole performance spectrum of textile dyes, helping build the colorfast clothing and printed materials we all rely on. This early research laid groundwork for modern synthetic chemistry, which leans heavily on aromatic substitutions and stepwise functional group manipulations.
Product Overview
Move into any lab or production line where colorants matter and you’ll find 4-Nitro-N,N-Dimethylaniline as one of the standout building blocks. Companies buy it in bulk for its predictable performance and for its wider compatibility with nitro and amine chemistry. Because of its sturdy molecular backbone, this chemical stands up to extended storage and transport, which helps on both the practical and economic fronts. Beyond dye work, producers turn to this compound for making certain pharmaceuticals, and it finds occasional use as a reference standard in analytical chemistry. Each batch sold on the market comes with careful documentation, because this isn’t a compound that folks treat lightly—there’s too much at stake in terms of safety and quality.
Physical & Chemical Properties
If you pour 4-Nitro-N,N-Dimethylaniline onto a sheet, you’ll see a bright yellow to orange crystalline powder. Its molecular formula, C8H10N2O2, tells a simple story on paper, but the real-world behavior keeps chemists on their toes. It melts at about 74-77°C and boils at over 300°C, which gives it a stable presence under ordinary working conditions. The compound doesn’t dissolve well in water, showing hydrophobic character common to nitroaromatics, but mixes easily with organic solvents like ether or ethanol. Under the hood, the nitro group at the para position activates the ring for further substitutions, while those methyls on the amine crank up the electron density—providing real options for anyone planning a synthetic route.
Technical Specifications & Labeling
Every reputable vendor slaps a clear label on 4-Nitro-N,N-Dimethylaniline drums. The best suppliers will quote purity at over 98%, usually measured by gas chromatography or high-performance liquid chromatography. Labels list the precise melting range, possible trace contaminants, lot number, and storage recommendations. Product files record the molecular formula, CAS number (99-59-2), UN shipping classification, and emergency handling guidelines. Clear labeling helps workers avoid confusion with similar nitroaniline derivatives, which can carry very different hazard profiles. These specifications aren’t just box-ticking—they keep operations efficient and above all, safe, especially in regulated domains like pharmaceuticals and food dyes.
Preparation Method
The classic way to make 4-Nitro-N,N-Dimethylaniline runs through a controlled nitration of N,N-dimethylaniline. Pouring ice-cold sulfuric acid into a reaction flask, workers add N,N-dimethylaniline under stirring. Dropping in concentrated nitric acid, they keep things slow—temperature control is crucial, as nitration reactions run hot and can slip into runaway if not checked. After several hours, the batch goes through neutralization with sodium carbonate. Extraction with ether or another organic solvent pulls the product out. Drying and crystallization finish the job. This procedure requires skill, experience, and a full suite of safety gear. Some modern plants might swap in more sustainable nitrating conditions, aiming to cut down on waste and acid runoff, which helps both worker health and the surrounding environment.
Chemical Reactions & Modifications
If you put 4-Nitro-N,N-Dimethylaniline in front of a research chemist, chances are they’ll see not just one compound, but a platform for further invention. The nitro group’s electron-withdrawing effect changes the ring’s reactivity, setting up routes for reductions, substitutions, and condensations. On a lab bench, you might see the nitro group getting reduced to an amino group, spinning out other important intermediates like 4-amino-N,N-dimethylaniline. Cross-coupling reactions, especially those heading toward azo dyes, rely on the clean para orientation to bring two aromatic systems together. This compound tolerates mild acid and base without decomposing, but under strong conditions, you better watch out for side reactions—those methyls can get cleaved off or oxidized.
Synonyms & Product Names
Trade paperwork, regulatory filings, and international customs declarations don’t always speak the same language, so it pays to know alternate names. Alongside “4-Nitro-N,N-Dimethylaniline,” you’ll see listings for p-Nitro-N,N-dimethylaniline, 4-Dimethylamino-1-nitrobenzene, or even its archive-era names like Ehrlich’s Reagent Component. Sometimes suppliers abbreviate as NDMA or 4-NDMA, but such shorthands drift in and out of fashion. Knowing all these identifiers helps buyers, safety inspectors, and anyone handling inventory avoid costly—or dangerous—mistakes.
Safety & Operational Standards
Handling 4-Nitro-N,N-Dimethylaniline takes discipline. Material Safety Data Sheets list the compound as harmful if swallowed, inhaled, or absorbed through skin. Workers wear gloves, splash-proof goggles, and masks, even when measuring out a scoop in a fume hood. The powdered dust irritates eyes and upper airways—good engineering controls like local exhaust and sealed containers help limit exposure. Spills mean grab the absorbents, not a broom. Workers go through regular training to understand the symptoms of acute exposure and how to respond, including eye-washing and emergency decontamination. Laboratories often restrict use to dedicated areas, with secure waste protocols and regular audits. In factory settings, automated handling equipment, sealed reactors, and air monitoring keep risks to a minimum.
Application Area
The dye industry draws on this molecule for its reliability. In batch after batch, chemists anchor azo couplings or push toward pigments that light up clothing, plastics, and inks. Some manufacturers use 4-Nitro-N,N-Dimethylaniline in making photographic chemicals or as a stabilizer for lubricants and polymers, which depend on well-behaved aromatic intermediates. Pharmaceutical giants sometimes slot it in as a precursor for developing active compounds, especially where a substituted aniline structure improves bioactivity. Laboratory testing businesses buy up smaller quantities because its distinctive properties help calibrate or benchmark chemical analyses. The reach spreads pretty wide for such a modest-looking yellow powder.
Research & Development
Academic research groups keep digging into the chemistry of substituted nitroanilines. With their toolkit, they chase new dye classes, especially ones that break beyond traditional reds and yellows, heading toward infrared pigments and biological staining agents. Synthetic chemists keep tweaking the process to lower byproduct waste, enhance selectivity, or move toward greener, solvent-free reactions. Patents in recent years propose new catalysts and flow reactors that raise efficiency. In drug discovery, medicinal chemists build off the dimethylaniline scaffold to probe for antiviral or anticancer properties, testing structure-activity relationships. Even as industries embrace smart polymers and advanced sensing materials, the groundwork laid by this molecule keeps shaping pathways forward.
Toxicity Research
Toxicologists study 4-Nitro-N,N-Dimethylaniline closely. Chronic exposure connects with methemoglobinemia, where the body struggles to carry oxygen—a risk that can show up in factory or lab accidents if workers breathe dust or fumes without proper protection. Animal tests and environmental screenings show that the compound persists in soil and water, breaking down only slowly. Regulators flag it as a substance of concern, subject to strict handling, labeling, and disposal controls. Industry best practices now favor containment, detailed record-keeping, and regular medical surveillance for at-risk workers. With environmental groups pushing for tighter controls on nitroaromatic runoffs, pressure keeps rising to trap or degrade waste streams before they can migrate.
Future Prospects
The demand for 4-Nitro-N,N-Dimethylaniline won’t vanish, though changes in the regulatory and technological landscape could shift where it’s used most. Customers push suppliers for chemicals made under greener processes—less waste, better energy use, and fewer toxics released during manufacturing. Biotech companies explore pathways to swap petrochemical inputs for more renewable feedstocks, looking to both offset cost and trim environmental footprint. Researchers keep searching for ways to recycle or degrade leftover nitroaniline more safely. Some dye companies place bets on alternative coloring agents, but the need for specialized aromatic intermediates stays strong, especially where performance and legacy processes demand consistency. Ongoing improvements in safety engineering, testing, and regulatory oversight will keep shaping how 4-Nitro-N,N-Dimethylaniline gets made, handled, and applied in coming decades.
What is 4-Nitro-N,N-Dimethylaniline used for?
Understanding 4-Nitro-N,N-Dimethylaniline
4-Nitro-N,N-dimethylaniline might sound like something plucked straight out of a dense chemistry textbook, but it plays a big part in industries that often slip under the radar. The compound stands out as a building block for dyes, especially those that give color to everyday items from clothing to printing inks. Synthetic dyes often start with careful manipulation of aromatic rings like the one in this molecule, and the nitro group attached to the aniline structure gives manufacturers a lot of flexibility to produce vibrant, lasting colors.
The Role in Dye Manufacturing
I used to walk through colorant manufacturing plants and the air buzzed with energy and risk. Workers wore heavy-duty gloves, and the smell sometimes clung to your clothes for days. 4-Nitro-N,N-dimethylaniline finds a home in the ‘azo dye’ process. This reaction forms the backbone of many textile and ink dyes. The compound’s nitro and dimethylamino groups provide essential chemical properties, allowing for the development of diverse dye shades.
My experience tells me that consistency in color means a lot to factories, especially those working on batch after batch of fabric for major clothing brands. If a dye made from this compound varies just a bit from one batch to the next, entire runs of fabric can end up as rejects. Physically, workers often keep close tabs on the chemical’s purity and how it handles the high temperatures and pressures in the reactors. This sort of vigilance keeps finished products looking sharp on store shelves.
Research and Laboratory Use
This chemical serves as a reference standard in organic laboratories due to its defined structure and reactivity. Chemists preparing new molecules use it as a model for studying electron-withdrawing and donating effects on aromatic rings. These insights lay the groundwork for breakthroughs in pharmaceuticals, crop protection products, and new classes of colorants. More than a decade ago, I watched grad students pull apart these molecules with advanced NMR equipment, hunting for physical patterns that might improve the next generation of synthetic materials.
Risks and Regulatory Scrutiny
There’s a side to this story that deserves respect: safety. The bright side of this chemistry comes with real risk. Nitro aromatic compounds, including 4-Nitro-N,N-dimethylaniline, have a reputation for toxicity and possible links to cancer. Reports from the International Agency for Research on Cancer flag similar chemicals for strict workplace exposure rules. Handling these compounds without proper training or gear can lead to health problems for workers, especially if exposure becomes frequent or careless.
Improving Practices and Outcomes
Practical steps make a difference. Labs lean on double-sealed containers, well-ventilated hoods, and regular environment monitoring to cut down the chance of spills or fumes. Dye factories shift toward automated dosing of raw materials, reducing human contact. Wastewater from dye plants gets filtered and treated with advanced oxidation to strip out any stubborn organics before the water flows back to city systems. New tools like chemical alternative assessments help manufacturers identify safer substitutes or tweak their processes to minimize hazards while keeping products vivid and reliable.
As regulations continue to shift and new research emerges, the use of chemicals like 4-Nitro-N,N-dimethylaniline demands attention and responsibility. Every batch of dye or bottle of lab reagent ties back to balance: supporting useful products while keeping people and the environment out of harm’s way.
What are the physical and chemical properties of 4-Nitro-N,N-Dimethylaniline?
Looking at the Essentials
4-Nitro-N,N-Dimethylaniline shows up as a bright yellow solid, catching the eye far more than the typical pale powders scattered around lab benches. If you walk through any synthetic dye house or an undergraduate organic chemistry lab, this chemical’s color makes it easy to spot. It weighs in at 166.18 g/mol, which lands it among the lighter specialty organics. Its structure features a benzene ring with a nitro group resting on the fourth carbon, and two methyl groups attached to the nitrogen. These small structural changes make a big difference to how molecules behave.
Melting, Boiling, and Solubility
Pop a sample of 4-Nitro-N,N-Dimethylaniline into a melting point apparatus, and it starts to soften around 76–78°C. That temperature keeps it solid at room conditions, so you won’t see it as a liquid unless you really heat things up. If you keep heating, it boils somewhere near 282°C. That temperature puts it far above boiling water or acetone, so it won’t turn to vapor in day-to-day handling.
If you tip this compound into water, not much happens. A quick swirl and you’ll see most of it settle to the bottom rather than dissolve. That’s what organic chemists expect from compounds that pack benzene rings and methyl groups. On the other hand, toss it into ethanol, diethyl ether, or chloroform, and it dissolves readily, so working up a reaction or separating it from other organics becomes much easier.
Chemical Behavior and Practical Hazards
People working with aromatic nitro compounds notice the strong and, honestly, somewhat harsh odor these chemicals put out. That smell hints at their chemical reactivity: the nitro group sits ready to join redox reactions or attach to other compounds. The nitrogen in the dimethylamino group opens even more possibilities, letting this molecule take part in nucleophilic substitutions or serve as a starting point for complex dyes. In my own hands, I’ve seen students misjudge the redox power of nitro aromatics—one slip-up with a reducing agent, and suddenly the reaction flask changes color or gives off strange fumes.
These same active groups bring safety questions. If inhaled or left on the skin too long, aromatic nitro compounds can cause headaches or more severe symptoms, since the body sometimes struggles to process them. While there aren’t stories about dramatic explosions from pure 4-Nitro-N,N-Dimethylaniline, it pays to avoid sparks and open flames, since similar compounds pick up reactivity when heated fast or exposed to strong acids and bases. Gloves and fume hoods become more than formality here; they keep headaches and skin rashes out of the picture when you work long hours with the powder.
Importance in Industrial and Laboratory Settings
This chemical finds its main use in dye manufacture. Those bright yellows and reds in textiles and inks often owe their color to compounds like this one. Having tried separating dyes with students, I’ve seen firsthand how a small tweak to the molecule makes a noticeable shift in color. Beyond dyes, researchers use this compound to craft other organic molecules—think of it as a building block for more complex pharmaceuticals or lab reagents.
Regulations often require storing 4-Nitro-N,N-Dimethylaniline away from sunlight and strong acids or oxidizers. I still remember a colleague dealing with clumped solid from moisture exposure, learning quickly that humidity ruins purity and future reactions. Keeping this compound dry and sealed ensures reliable results in both small research labs and big factories.
Looking Ahead: Practical Steps
Anyone handling 4-Nitro-N,N-Dimethylaniline in large amounts should use good ventilation, sturdy gloves, and sealed containers. Simple chemical hygiene habits, like washing hands after use or labeling every bottle, protect users and anyone else in the workspace. Exploring alternative less-toxic dye intermediates for industrial purposes offers one path to reducing health risks, though the color strength and efficiency of nitro aromatics mean they won’t disappear overnight. Education, respect for the hazards, and careful storage can keep accidents rare and creativity thriving in the lab.
Is 4-Nitro-N,N-Dimethylaniline hazardous or toxic?
The Real Risks Behind the Chemical Name
Chemicals with long names tend to scare people, and 4-Nitro-N,N-Dimethylaniline is no exception. This compound sometimes pops up in labs, especially in the making of dyes or as an intermediate in certain chemical syntheses. Most of us never see it in everyday life, but the risks spill over into the workers handling it and the folks living near facilities that use it.
What Research Shows About Toxicity
Looking at the available research, 4-Nitro-N,N-Dimethylaniline doesn’t play nice with the human body. It can irritate eyes and skin, but the bigger issue comes from inhaling or swallowing the stuff. Cases reported in chemical databases show that, after exposure, people developed methemoglobinemia, a condition where blood can’t carry oxygen as well. Symptoms include shortness of breath and a blue tint to lips and skin—never a good sign. In animal studies, exposure led to liver damage and negative effects on the blood system. The U.S. Environmental Protection Agency notes that nitroaniline compounds like this often show toxicity in animal models at relatively low doses.
Worker Safety and Exposure Limits
I’ve handled plenty of chemicals during late nights in underfunded college labs, and the strongest lesson came from the quiet reminders taped to fume hoods. Gloves, goggles, and keeping those chemicals under the hood aren’t just for show. For 4-Nitro-N,N-Dimethylaniline, personal protective equipment makes a difference. No major environmental or occupational group has published a definitive safe exposure level, but companies dealing with this compound use strict controls—think enclosed systems, local exhaust, and good gloves. Lax standards or skipping safety culture multiplies the chance for health problems. In one documented case, someone developed cyanosis—a telling sign—not from a single splash but from chronic, small-scale exposure over time.
Environmental Impact Matters
Chemicals don’t stay where you put them. Once spilled or dumped, they can make their way into soil or groundwater. Some nitroaniline derivatives have turned up in river water near industrial sites, raising alarms for both aquatic life and the people living downstream. Long-term buildup means risk doesn’t disappear once the lab door locks for the night. Effective waste treatment and responsible disposal matter much more than after-the-fact cleanup. No one wants to watch a local stream turn into a dead zone for fish.
Setting Safer Standards
Hazards linked to 4-Nitro-N,N-Dimethylaniline aren’t a distant issue only for researchers. The bottom line is that companies and regulators have to set better tracking and tighter exposure limits, backed up by regular inspections. Local communities deserve to know what chemicals travel through their neighborhoods. Clear, confident labeling, robust spill training, and real-time air and water monitoring bring a safer work and living space. If a facility uses the compound, folks should ask questions and demand transparency. Chemicals keep the modern world humming, but safety and respect for health shape smarter decisions.
What are the proper storage and handling conditions for 4-Nitro-N,N-Dimethylaniline?
A Chemical That Deserves Respect
4-Nitro-N,N-Dimethylaniline doesn’t make its way into regular conversation, but among chemists and lab staff, it’s a familiar name. This compound, with its bright yellow color, isn’t just a streak of pigment—it’s a strong reagent, and it comes with a list of hazards you don’t ignore. Improper storage or careless handling kicks up genuine risks, from skin irritation to far more serious consequences if vapors build up or spills slip past your attention.
Simple Steps That Protect in Real Labs
There’s a temptation to toss every reagent on the same shelf and assume the annual safety talk covers all bases. From real-world lab experience, that shortcut never works—especially with compounds like this. Safety rules get written in sweat and sometimes in regret. My first time working with aromatic nitro compounds drove this home: treat each bottle with intention, or problems compound fast.
The best place for 4-Nitro-N,N-Dimethylaniline sits in a cool, dry cabinet. Heat and humidity don’t just mess with stability—they threaten the container’s seal and the safety of everyone near the space. I remember one partner sweating through a flask seal during a summer outage; the smell alone sent us running for airflow and gloves. That’s the lesson: good airflow, shade from sunlight, and zero moisture keep this substance quiet and stable.
Keep It Sealed and Separate
It’s not just about temperature. The containers have to close tight every time. This isn’t negotiable. Once air sneaks in, contaminants can trigger reactions you don’t want—mixing with strong acids or bases, or with accidental splashes of incompatible chemicals, could start a real mess. My university’s aging lab had ancient stoppers that leaked fumes, turning a short task into an afternoon of ventilation and paperwork. Use glass or high-density plastic that stands up to organic solvents, check the seal, and store it away from oxidizers and acids.
Don’t let bottles gather by the workbench. It pays to keep materials like this behind a safety barrier, labeled so nobody guesses at the contents. Worn-out tape and marker never cut it in an emergency. Legible, chemical-resistant labels avoid confusion if the bottle leaves the shelf in a hurry.
Emphasize Protective Equipment and Emergency Plans
The best gloves and goggles gather dust unless people put them on. 4-Nitro-N,N-Dimethylaniline can irritate eyes and skin. Lab coats, gloves (nitrile at a minimum), closed-toed shoes, and goggles come out every time. One careless classmate spent a week swapping between ice packs and steroid cream after ignoring glove protocol. Not worth it. Keep a spill kit in reach. Paper towels and soapy water won’t help here—use purpose-built absorbents and ventilate if you ever encounter a spill.
Follow Protocols, Not Shortcuts
People make mistakes when tired or rushed, and hazardous chemicals punish shortcuts. Take a minute to review the safety data sheet for details like flammability, chemical incompatibility, and first aid advice. Make reporting close calls a norm, not something to hide. Train new staff directly at the storage site, not in the conference room.
Better Habits Save Time and Trouble
Safe handling and storage aren’t about pushing paper or adding chores. These habits keep everyone working safely day after day—no mystery fumes, ruined experiments, or emergency evacuations. Spending the extra few minutes organizing chemicals, logging the opening and closing of containers, and checking seals means you deal with fewer surprises. That’s the value you learn on the job, not just from a binder on the shelf.
How can 4-Nitro-N,N-Dimethylaniline be synthesized or prepared?
Understanding the Need for 4-Nitro-N,N-Dimethylaniline
4-Nitro-N,N-dimethylaniline has found its place in the world of dyes, pigments, and organic research. Laboratories and the colorant industry lean on its structure for various reactions, especially where a nitro group’s electron-withdrawing punch matters. As someone who’s worked their fair share of hours at the bench, I’ve come to appreciate not just the chemistry behind such syntheses but the human element: safety, scale, and efficiency.
Paths to the Product: Going from Aniline Compounds to Nitro Derivatives
Many chemists start with N,N-dimethylaniline, a readily barked-out, oily-smelling amine you can order from standard suppliers. To introduce the nitro group, nitration with a mix of concentrated nitric and sulfuric acids usually fits the bill. This mix carries a fear factor for most students and researchers for a good reason. Vigorous exotherms can erupt if you toss the reagents around without care or cooling. Personally, I’ve found slow, ice-bath additions the key—no sense in racing through organic syntheses when acid burns and runaway reactions hover in the background.
In practice, you chill your N,N-dimethylaniline below room temperature, drizzle in cold nitrating solution, and keep stirring as pale to deep yellow color shows up—always a sign that the nitro group’s getting in. If you heat things up too much, you get byproducts or, if unlucky, a foamy disaster in your flask. After the action, lots of water dilutes the acid, and you extract your target with an organic solvent like ether. Washing, drying, then purifying by distillation or recrystallization brings out the golden product, which forms solid crystals on standing.
Why Order of Addition and Temperature Matters
Many beginner chemists trip up with order and temperature. There’s a lesson here: chemistry respects patience and planning more than bravado. Keeping your flask cold, adding nitric acid s-l-o-w-l-y to the amine, and keeping a thermometer handy make all the difference. Jumping in with both reagents at once or using a warm flask never goes well. Researchers keep an eye on safety not just for themselves but for the integrity of the final compound. Skipping these steps, I’ve seen reactions that boil over, killing yield and sometimes damaging glassware.
Byproducts and Purification: Facing Real-World Hurdles
Nitration often gives not just the 4-nitro isomer but also a 2-nitro version and sometimes dinitro compounds. Separating these calls for repeated crystallization or even column chromatography. Yields might drop, but careful purification pays off. There’s no shortcut—impurities can throw off both research results and industrial products. Having done column runs late at night more than once, I’ve learned the value of patience and good recordkeeping.
Looking Ahead: Greener and Safer Approaches
Manufacturers and researchers keep searching for smarter, cleaner, or less hazardous methods. Alternatives like milder nitration agents or solid-acid catalysts intrigue many, especially where regulatory pressure and waste management costs loom large. Google Scholar and patent databases grow thicker each year with attempts to find that sweet-spot synthesis: high yield, low waste, safer for everyone involved.
Skill and care in organic synthesis don't just build better molecules—they build better chemists. Any route to 4-nitro-N,N-dimethylaniline calls for respect: for the chemistry, for the hazards, and for the colleagues and communities counting on a job well done.
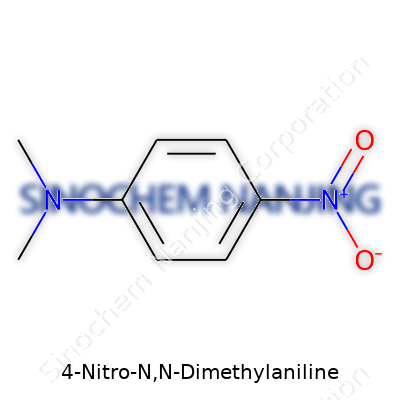
| Names | |
| Preferred IUPAC name | N,N-dimethyl-4-nitroaniline |
| Other names |
p-Nitro-N,N-dimethylaniline p-Nitrodimethylaniline 4-Nitro-N,N-dimethylbenzenamine N,N-Dimethyl-4-nitroaniline 4-Dimethylamino-1-nitrobenzene |
| Pronunciation | /ˈnaɪtroʊ ɛn ɛn daɪˈmɛθɪl.əˌnɪliːn/ |
| Identifiers | |
| CAS Number | 99-59-2 |
| Beilstein Reference | **1209247** |
| ChEBI | CHEBI:34568 |
| ChEMBL | CHEMBL21853 |
| ChemSpider | 13942 |
| DrugBank | DB08347 |
| ECHA InfoCard | 100.007.245 |
| EC Number | 202-203-2 |
| Gmelin Reference | 130369 |
| KEGG | C14419 |
| MeSH | D013698 |
| PubChem CID | 7226 |
| RTECS number | ZE9625000 |
| UNII | P9H61P20CM |
| UN number | UN2662 |
| Properties | |
| Chemical formula | C8H10N2O2 |
| Molar mass | 166.19 g/mol |
| Appearance | Yellow crystalline powder |
| Odor | Odorless |
| Density | 1.14 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 1.98 |
| Vapor pressure | 0.0187 mmHg (25°C) |
| Acidity (pKa) | 5.15 |
| Basicity (pKb) | pKb = 9.68 |
| Magnetic susceptibility (χ) | -38.2·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.613 |
| Viscosity | 1.39 cP |
| Dipole moment | 4.47 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 248.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −25.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2140 kJ/mol |
| Hazards | |
| Main hazards | Toxic if swallowed, in contact with skin or if inhaled; causes damage to organs; harmful to aquatic life. |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS02,GHS07,GHS09 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H332, H335, H351 |
| Precautionary statements | P261, P264, P271, P272, P273, P280, P302+P352, P304+P340, P305+P351+P338, P308+P313, P312, P321, P332+P313, P337+P313, P362+P364, P403+P233, P405, P501 |
| Flash point | 102 °C (216 °F; 375 K) |
| Autoignition temperature | Autoignition temperature: 626°C |
| Lethal dose or concentration | LD50 oral rat 282 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 830 mg/kg |
| NIOSH | JN8575000 |
| PEL (Permissible) | PEL: 5 mg/m3 |
| REL (Recommended) | REL (Recommended): 0.5 mg/m³ |
| IDLH (Immediate danger) | IDLH: 50 mg/m³ |
| Related compounds | |
| Related compounds |
4-Nitroaniline N,N-Dimethylaniline 4-Nitrodiphenylamine |

