4-Nitro-N,N-Diethylaniline: An In-Depth Commentary
Historical Development
The roots of 4-Nitro-N,N-Diethylaniline stretch back to the era of expanding synthetic chemistry in the late 19th and early 20th centuries. Back then, researchers frequently experimented with aromatic amines and nitro groups, following the footsteps of pioneers in dye and pharmaceutical chemistry. For decades, substitutes on the aniline molecule shaped the properties of resulting compounds and set new standards in dye manufacture. Production ramped up with the emergence of larger chemical companies, who realized the power of dialkylated nitroanilines for colorants and intermediates. Today’s applications grow from this base of trial, error, and drive for improved performance in color chemistry and analytics.
Product Overview
This compound shows up mostly as a pale yellow solid or crystalline powder, easy to recognize for anyone who has worked around nitroanilines. The structure consists of an aniline core, dialkylated at the nitrogen—with two ethyl groups—while the para position holds a nitro group. Both modifications significantly alter the basic aniline’s properties, making it far more hydrophobic than unsubstituted aniline, and change its reactivity in both synthesis and practical application. Chemists favor it for its ability to transfer electronic effects across the ring, which supports its demand in dye synthesis and spectrum-based assays.
Physical & Chemical Properties
Bulk 4-Nitro-N,N-Diethylaniline comes with a melting point just above ambient laboratory temperatures, usually clustering between 69 and 72 degrees Celsius. Unlike more volatile amines, it carries a decent boiling range (often close to 170-180 degrees Celsius at reduced pressure) and little water solubility. In solvents like acetone, ethanol, and ether, it dissolves easily, which reflects its organic backbone. Light sensitivity is a critical dimension—exposure tends to degrade its nitro group, flavoring its use toward careful storage. Bright yellow color betrays strong absorption in the visible spectrum; this property lies at the heart of its application in dye chemistry and spectrophotometry.
Technical Specifications & Labeling
Chemically savvy suppliers take pride in listing purity levels, usually above 98%, because research and industry rely on consistent feedstock quality. Storage recommendations reflect its sensitivity: well-sealed containers, kept in cool, dry conditions, shielded from light. Labels carry hazard warnings—mainly for irritation, toxicity, and environmental risk—underscoring respect due to nitroaromatic compounds. The compound falls under UN transport codes for hazardous materials, which shapes both purchasing decisions and protocols within the shipping chain. Safety Data Sheets list exposure limits, handling tips, and proper disposal channels, all drafted in line with REACH and GHS standards.
Preparation Method
Production usually starts with N,N-diethylaniline, a chemical staple. Chemists introduce the nitro group using a nitration reaction, commonly mixing nitric and sulfuric acids under controlled temperature. This synthesis isn’t just about pouring and mixing—careful control over temperature and timing decides the product ratio and purity. A side path may generate ortho- or meta- isomers, but most processes favor the para-product through sophisticated process design. After completion, the usual workup involves quenching, extraction, and recrystallization, turning an often messy reaction into a saleable solid. The skills required for clean separation and purification highlight chemical craftsmanship shaped by both tradition and innovation.
Chemical Reactions & Modifications
Organic chemists value 4-Nitro-N,N-Diethylaniline mainly as a precursor. The nitro group stands out as a handle for further reactions—reduction leads to the corresponding amine, which unlocks new classes of dyes or pharmaceuticals. Nucleophilic aromatic substitution leverages the electron-withdrawing nature of the nitro group, creating options for more intricate synthesis. Under basic conditions, chemists tweak the ethyl groups for functionalization or perform coupling reactions useful in dye production. These manipulation routes depend on decades of research into reactivity patterns among aromatic amines, and each pathway finds favor depending on end use.
Synonyms & Product Names
Over the years, the compound gathered a set of alternative names. Many chemists trade under “p-Nitro-N,N-Diethylaniline” or “4-Nitro-N,N-Diethylbenzenamine,” reflecting IUPAC naming conventions. Some manufacturers roll with legacy industry terms, offering it as “C.I. 10365 Intermediate” or other region-specific codes. Specific catalogs add batch numbers and internal references, but the nitro-diethylaniline motif stays consistent, guiding specialists toward the correct molecule every time, regardless of origin or supplier.
Safety & Operational Standards
Dealing with nitro-aromatic compounds comes with real-world challenges. Short-term exposure brings skin and eye irritation while inhaling dust can upset airways. Folks in industrial labs don gloves, goggles, and lab coats, and respect restricted-ventilation handling areas. The nitro group also brings environmental worries—accidental spills and improper disposal contaminate soil and water, pushing responsible labs and factories to tightly manage waste. Fire risk comes mainly from accompanying organic solvents rather than the compound itself, yet all safety plans call for proper fire extinguishers, spill kits, and emergency showers nearby. In the regulatory world, many regions follow OSHA guidelines for exposure and storage, and long-standing experience in chemical manufacturing shows the wisdom of strict adherence on safety protocols.
Application Area
Industries value 4-Nitro-N,N-Diethylaniline as a workhorse intermediate. The dye sector leans on it to produce yellow, orange, and red azo dyes—often for textiles, plastics, or paper—where it bridges raw chemical input and vibrant end products. Analytical chemists use it as a colorometric reagent, especially for quantifying trace elements by forming clear, measurable complexes. That bright yellow hue, which once limited its use, forms the backbone of its spectroscopic role. Some laboratories reach for it during the setup of calibration standards, counting on reliable molar absorptivity. In the environmental field, workers rely on it to support water analysis, helping measure small pollutant concentrations. In R&D settings, the compound supports medicinal chemistry, engaging in exploration of novel pharmacophores and bioactive analogs, pushing boundaries on known therapeutic strategies.
Research & Development
R&D teams constantly seek ways to adapt classic intermediates for contemporary needs. With 4-Nitro-N,N-Diethylaniline, the eye turns to cleaner synthesis, waste minimization, and process efficiency. Green chemistry movements encourage using less hazardous reagents, recycling solvents, and lowering overall waste. The search for alternative nitrating agents runs strong, with pilot facilities experimenting on catalytic and phase-transfer methods. In analytical chemistry, research teams innovate with new sensor designs utilizing the compound’s unique optical absorption, hoping to push the boundaries of portable testing or high-throughput screening. Every generation, novel derivatives or application profiles emerge, usually riding on advances in understanding electron transfer, solubility, and photolysis.
Toxicity Research
Toxicologists pay close attention to aromatic nitro compounds due to their historic role in occupational and environmental health. Lab studies flag both acute and chronic hazards: skin contact can cause dermatitis, while animal studies point toward risks from repeated exposure, such as anemia or liver effects. Breakdown products, sometimes formed during waste processing, present persistent dangers in soil and groundwater. Over time, data from workplace surveillance and ecological monitoring build the case for stringent exposure controls and advanced remediation technology. Environmental toxicology also focuses on aquatic impacts, as many nitroaromatics cause significant stress to aquatic life at low concentrations. It all comes back to a chemistry community united by a desire to innovate and improve safety across the life cycle—from synthesis to end-use and disposal.
Future Prospects
The market for smart analytical reagents and high-performance dyes grows year-on-year, increasing pressure on molecules like 4-Nitro-N,N-Diethylaniline. Tech advances drive demand for purer, more predictable intermediates that guarantee performance while lowering byproduct levels. Sustainability commands a new respect as regulators and customers demand lower environmental footprints and safer chemical processes. Researchers keep digging into photolytic degradation, bioremediation, and alternative synthetic routes, hoping to marry efficiency with reduced toxicity. As new application sectors grow—ranging from optoelectronic devices to advanced diagnostics—the compound’s legacy and promise depend on the open collaboration between industrial know-how, academic insight, and regulatory guidance. Real-world experience shows that every challenge carries potential lessons for the next generation of chemical innovation.
What is 4-Nitro-N,N-Diethylaniline used for?
Why This Compound Gets Attention
4-Nitro-N,N-Diethylaniline doesn’t come up in daily conversation unless you spend your days around dye factories or chemistry labs. At first glance, its name looks like something from a textbook, but the uses behind this compound reach well beyond the classroom. This chemical serves as a building block in making many dyes and pigments. The textile industry, for one, can’t function without sturdy and reliable chemicals that help turn plain fabric into something sold on hangers and shelves all over the world. Anyone who’s worn brightly colored clothing or worked in textile printing has likely seen its effects, even without knowing the name.
Dye Manufacturing and Colorants
Walk into any store, and almost every item of clothing owes its shade to a careful cocktail of dye molecules. 4-Nitro-N,N-Diethylaniline gives dye-makers a foundation for pushing bright yellow and orange colors onto textiles and plastics. Once mixed or treated with other chemicals, it helps create stable colors that stick to fabric and resist fading from washing or sunlight. In my experience working in a textile research facility, we’d constantly check for colorfastness. Compounds like this one saved us grief by holding up better than alternatives in our tests. That's value measured in less wasted product and fewer complaints down the road.
Applications in Organic Synthesis
Beyond dyes, this compound finds its way into other chemical syntheses. Companies and research groups rely on it as a starting material to build more complicated molecules, sometimes for pharmaceuticals and sometimes for specialty chemicals used in electronics or ink formulations. In the process, a team of chemists can tweak the structure and use 4-Nitro-N,N-Diethylaniline as a launchpad for new inventions. I’ve watched teams in industrial labs invest weeks working out the kinks for a process, all hinging on dependable access to stable starting materials like this one.
Health and Safety: A Practical Concern
Reading a safety data sheet for 4-Nitro-N,N-Diethylaniline will make anyone think twice before handling it without gloves or proper gear. The compound isn’t what you’d want to spill on your skin or inhale day after day. Long-term exposure brings health risks, so plants using it have strict controls and insist on good ventilation. During a factory tour, I saw firsthand the layers of safety measures in place, and I couldn’t help but respect the diligence from both management and workers. Handling chemicals safely isn’t just a rule, it’s a practice that keeps livelihoods and lives protected.
What Can Improve?
There’s no escaping the need for strong chemicals in manufacturing, but there’s always room to innovate. Green chemistry continues to push for safer alternatives or improved processes that cut down on waste and pollution. Recently, I worked with a group focused on cutting the solvent use in dye production, which had side benefits—less chemical runoff and happier neighbors. Efforts like these nudge the industry forward. Investment in training, updated equipment, and ongoing monitoring can raise safety standards and shrink the environmental footprint of plants that use 4-Nitro-N,N-Diethylaniline. These practical steps lead to real change, not just improved public perception.
What is the chemical structure and formula of 4-Nitro-N,N-Diethylaniline?
Chemical Structure and Formula
4-Nitro-N,N-diethylaniline doesn’t just catch chemists’ attention with a long name. In practical terms, this compound brings together both a nitro group and an aromatic amine backbone. Structurally, you find a benzene ring as the base. A nitro group (-NO2) attaches directly to the fourth carbon, counted from the amine substituent. The core amine isn’t left in its simplest form – it’s N,N-diethylated, which means the nitrogen on the aniline carries two separate ethyl groups. The chemical formula lines up as C10H14N2O2. Written out, that means ten carbons, fourteen hydrogens, two nitrogens, and two oxygens. The molecular structure echoes both aromatic chemistry and the modifications made to functionalize simple aniline for broader applications.
What’s Special About This Structure?
This isn’t just theory on paper – 4-Nitro-N,N-diethylaniline matters due to how these groups interact. The nitro group, by pulling electrons, changes the character of the aniline ring, dampening some of the reactivity of the aromatic amine while helping stabilize certain products in reactions. The diethyl groups protect the nitrogen, reducing the nucleophilicity a good bit. What you get is a molecule less prone to aggressive oxidative breakdown than unsubstituted anilines. In labs, this tweaking lets chemists control outcomes in important syntheses.
Uses and Real-World Impact
I’ve run across this molecule in settings where color matters a lot. It shows up as an intermediate in dye production, brightening textiles and sometimes powering color chemistry in analytical methods. The structure drives color development in reactions used by dye makers, and folks also turn to it as a reference standard for certain measurements, like assessing solvent purity. Its stability under regular lab conditions cuts down on mess and shortens prep timelines when compared with more reactive relatives.
Why Staying Cautious Makes Sense
Handling aromatic nitro compounds calls for a steady hand and some respect for their hazards. A nitro group raises flags because of potential toxicity, not to mention flammability if things go wrong. Safety data highlight risks like skin or respiratory irritation and longer-term side effects tied to certain nitroaromatics. Proper ventilation, gloves, and goggles become non-negotiable. Training lab staff or anyone working in industrial settings about handling and storage keeps both people and facilities far from harm.
Dealing With Environmental and Health Risks
People ask about greener alternatives, since persistence in the environment raises questions about breakdown and long-term toxicity. Pushing for more robust wastewater treatment, minimizing waste generation, and recycling solvents or intermediates can make a difference. Research crews keep searching for less hazardous analogs or new routes that skip over risky intermediates while delivering similar performance. Regulators and industry teams have learned from past oversights—modern manufacturing standards prioritize containment, clean disposal, and ongoing exposure monitoring.
Moving Science Forward
Chemists keep exploring why small tweaks to molecules like 4-Nitro-N,N-diethylaniline change their behavior so much. Each new discovery says something about safe design, cleaner production, and where the future of color chemistry or specialty synthesis might be heading. With practical know-how, smart safeguards, and solid research, industries can use the power of these molecules without repeating old mistakes.
What are the safety precautions and hazards associated with 4-Nitro-N,N-Diethylaniline?
Hazards Linked to 4-Nitro-N,N-Diethylaniline
Anyone spending time in the lab with aromatic amines like 4-Nitro-N,N-Diethylaniline quickly learns that safety isn’t just a formality. The yellowish crystalline powder and its vapors can spell trouble if you don’t respect them. Skin picks it up fast since it can absorb straight through. Inhalation isn’t much better—even a whiff of dust can irritate lungs and eyes, sometimes more.
Here’s something that hits home for most chemists: this compound doesn’t just sting a bit, it brings real toxicity concerns. Animal studies point to potential for harmful effects on blood and organs. Over time, long or repeated exposure eats away at one’s health, even if there’s no accident. Chemicals with nitro groups often mess with hemoglobin (methemoglobinemia can creep up). Handling powders always risks spills and fine particulates, and 4-Nitro-N,N-Diethylaniline floats easily in the air. Its vapors can hang around in still air if proper ventilation slackens.
Fire is another worry. Organic nitro compounds often catch fire quicker than you’d hope—this one won’t set a room ablaze like nitroglycerin, but a stray spark or static discharge finds a ready target. A lot of us have seen the color change and smell shift when fire or strong oxidizers hit it. Add heat, and you could see toxic fumes like nitrogen oxides and other breakdown products escape.
Preventing Accidents
In my experience, safety gear comes before speed or convenience. Nitrile gloves, goggles, and a snug lab coat won’t win fashion contests, but they keep dangerous dust off skin. For bigger setups with dry powders, a well-fitted mask or even a respirator keeps lungs safe. I never count on fume hoods alone—proper venting means double-checking airflow before every use.
Spills happen—quick clean-up matters more than scolding. I keep absorbent pads on hand. Sweeping powders dry may raise dust, so damp materials do the job best. Washing hands right after working makes a big difference, even with gloves on.
Proper storage sits right near the top of the list. This compound belongs in tightly sealed containers, away from sunlight, strong acids, bases, or oxidizers. I tag everything with clear hazard warnings so nobody grabs the wrong jar by mistake. Storing below room temperature helps slow decomposition.
Managing Waste and Emergency Situations
Disposal is a chore, but one that nobody wants to skimp on—waste containing 4-Nitro-N,N-Diethylaniline shouldn’t hit the regular trash. I label waste bottles for collection as hazardous organic material and check local guidelines for incineration or special disposal.
Eye wash stations and emergency showers get checked every week in our lab. Fast access cuts the worst from exposure, as delays bring bigger problems. For significant spills, evacuation and professional cleanup fit the bill, not improvising.
Better Habits, Safer Labs
The most important lesson I’ve picked up is that knowledge works better than luck. Training staff on what symptoms to watch for—headaches, odd skin color, fast breathing—saves lives. Safety culture grows stronger when everyone understands why the rules exist and looks after each other. Staying informed, speaking up about unsafe setups, and regularly auditing procedures all keep small hazards from turning into real harm.
Every hazardous compound deserves respect, but 4-Nitro-N,N-Diethylaniline pushes that lesson home for anyone who works with it. Preparation, attention, and open discussion keep people safe.
What are the storage and handling recommendations for 4-Nitro-N,N-Diethylaniline?
Understanding What You’re Dealing With
Most people don’t walk into work thinking about 4-Nitro-N,N-Diethylaniline. For chemists and industry workers though, it’s that yellow-to-brown powder that gets used in dyes and as an intermediate in certain syntheses. A quick look at its MSDS sheet tells you it’s not a kitchen-table chemical. Anyone who’s ever cracked open a laboratory safety manual knows that nitro compounds and anilines both have long lists of hazards. This stuff stands out for its potential toxicity and its risk to both the environment and human health.
No-Nonsense Chemistry: Avoiding Trouble
You bring a container of 4-Nitro-N,N-Diethylaniline into your storage room. You don’t toss it on a shelf alongside flammables or acids. Experienced handlers always keep this chemical in a tightly-sealed bottle, preferably made of glass or compatible high-density polyethylene. Direct sunlight turns even the simplest bench chemical into a risk, so smart chemists store it in a cool, dry, and well-ventilated area. I remember an old incident in a poorly-managed lab in graduate school: the guy next to me learned the hard way about humidity and heat creeping into the storage cabinet. Result? Agglomeration and a near-miss when opening the flask—lesson learned.
Chemical compatibility charts matter. People sometimes make the mistake of stacking incompatible reagents together. Talking about nitroanilines, you steer clear of oxidizers, reducing agents, acids and bases in the same space. Separation is key—one slipup leads to hazardous vapors or even a fire. Most places give 4-Nitro-N,N-Diethylaniline its own secure spot, away from food and personal items.
Everyday Handling Practices That Actually Work
Experience shows personal protective equipment isn’t just for show. Gloves—nitrile, not just latex—long sleeves, and chemical splash goggles rank high on the must-have list. Fume hoods turn routine weighing or dissolving into a safer process. During my time assisting with undergraduate labs, I saw beginners try to work outside the hood, thinking a quick open and pour was harmless. Instant regret once a faint odor drifted up—headaches, watery eyes, and a safety officer giving them a stern talk.
Spill management matters more than some might think. Granular absorbents or vermiculite make cleaning up easier, and sealed disposal bags keep the substance in check until it goes to chemical waste disposal. Trying to sweep up with a broom or, even worse, using water, creates more problems than it solves.
Solutions for Safer Practices
Building a culture of chemical safety keeps people out of trouble. Routine safety drills, clear labeling, and easy-to-find safety data sheets lower the risk. I’ve seen plenty of labs where labeling slipped and unidentified powders sat around for weeks—a recipe for disaster. Involving all users in frequent audits and encouraging whistleblowing on poor practices helps avoid near-misses and real accidents.
Switching to less hazardous substitutes when possible drops the risk to everyone. If 4-Nitro-N,N-Diethylaniline is necessary, control access and track usage through logs. Good habits, not just rules, keep incidents low. Everyone from the rookie technician to the senior chemist has a stake in keeping the space safe, and it only takes one moment of forgetfulness to undo months of caution.
Where can I purchase 4-Nitro-N,N-Diethylaniline and what is its price?
Understanding What You’re Buying
There’s something odd about asking where to buy 4-Nitro-N,N-Diethylaniline—a chemical name that doesn’t roll off the tongue unless you spend time around chemistry labs or industrial supply companies. This compound, known for its use in dye manufacturing and certain niche reactions, lands squarely in the domain of professional buyers, researchers, and specialty manufacturers. If you’ve ever needed to work with organic synthesis or experimental analytical techniques, compounds like this one are more than just names; they’re puzzle pieces that fit into bigger scientific stories.
Legit and Legal—Sourcing Matters
Not every chemical is up for sale to the general public. Safety regulations protect everyone. Buying 4-Nitro-N,N-Diethylaniline means you’ll face checks: suppliers ask for your professional credentials, purpose of use, and sometimes more. In my own hunt for restricted reagents—especially those that pose risks if mishandled—every order turns into an exercise in due diligence. There’s paperwork and verification. It slows things down, but it’s there for a reason. Reputable companies won’t skip these steps, and it’s a red flag if they do.
Who Sells It? Finding Trusted Suppliers
Charles River, TCI America, Sigma-Aldrich, and Thermo Fisher fly to mind for rare or specialty chemicals. These suppliers maintain strict standards, which is a relief if you’ve had brushes with lower-quality products or sketchy online listings. In fact, most of the time you’ll need to work for an institution—universities, research centers, industrial facilities—and some suppliers simply won’t ship to home addresses or individuals. That’s part of their compliance game.
I remember trying to source a reagent outside of a university setting years ago. The process was frustrating—forms, safety training, and weeks of waiting. It forced me to acknowledge why these slowdowns matter. Chemicals like 4-Nitro-N,N-Diethylaniline demand respect. They don’t belong on eBay or unverified import sites, even if the price tag looks pretty low.
The Price Tag: What to Expect
Pricing always runs the gamut. A compound as specialized as 4-Nitro-N,N-Diethylaniline draws premium costs from reputable catalogues. At time of writing, the price hovered around $150 for 25 grams on platforms like Sigma-Aldrich and TCI America. Bulk purchases drop the per-gram price, but not by much unless you’re placing industrial-sized orders. Import taxes, shipping fees (which jump if a chemical is classified as hazardous), and documentation charges often surprise new buyers.
Cut-price listings appear occasionally on lesser-known sites, typically overseas. Experience shows these bargains rarely hold up: purity claims fall short, batches vary, and customer service disappears the moment something goes wrong. Labs rely on consistency for reproducible results—cutting corners on source material can shatter weeks of careful work.
Risks and Responsibilities Are Real
Chemicals with energetic nitro groups (that yellow tinge isn’t for show) and toxicological quirks demand careful storage, knowledgeable handling, and respect for disposal rules. Labs invest in fume hoods and protective equipment because substitutes don’t exist for safety. Rules aren’t just red tape; they keep people alive and facilities open.
Seeking a Solution
For those outside traditional workplaces who genuinely need 4-Nitro-N,N-Diethylaniline, partnerships with universities or registered labs provide a safer, legal route. Teaming up with verified institutional buyers also opens access to regulatory guidance and keeps the paperwork neat. Relying on trusted suppliers means fewer unwanted surprises and a clear path if things go wrong. That kind of peace of mind proves worth the initial headache.
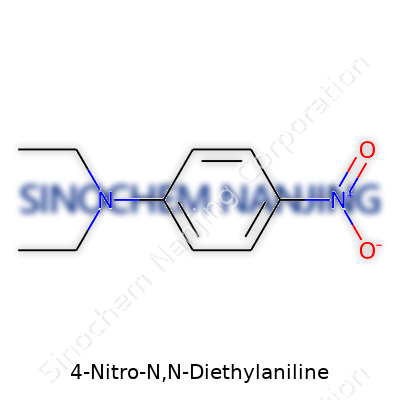
| Names | |
| Preferred IUPAC name | N,N-Diethyl-4-nitroaniline |
| Other names |
N,N-Diethyl-4-nitroaniline 4-Nitro-N,N-diethylaniline p-Nitro-N,N-diethylaniline Diethylaniline, p-nitro- N,N-Diethyl-p-nitroaniline |
| Pronunciation | /ˈfɔːr ˈnaɪtroʊ ˌɛn ˌɛn daɪˌɛθɪl.əˈnaɪliːn/ |
| Identifiers | |
| CAS Number | 99-57-0 |
| Beilstein Reference | 1209973 |
| ChEBI | CHEBI:54514 |
| ChEMBL | CHEMBL14203 |
| ChemSpider | 132946 |
| DrugBank | DB08312 |
| ECHA InfoCard | 03ef8384-820c-4256-ba3f-f0da5af8b4b7 |
| EC Number | 202-734-6 |
| Gmelin Reference | 80734 |
| KEGG | C19217 |
| MeSH | D017020 |
| PubChem CID | 7090 |
| RTECS number | SJ8225000 |
| UNII | Y8O9P25WCA |
| UN number | UN2811 |
| Properties | |
| Chemical formula | C10H14N2O2 |
| Molar mass | 194.24 g/mol |
| Appearance | Yellow to orange crystalline powder |
| Odor | amine-like |
| Density | 1.08 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 2.93 |
| Vapor pressure | 2.7E-3 mm Hg (25 °C) |
| Acidity (pKa) | 10.68 |
| Basicity (pKb) | pKb = 10.25 |
| Magnetic susceptibility (χ) | -61.2×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.5750 |
| Viscosity | 2.31 cP (20°C) |
| Dipole moment | 4.44 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 206.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -34.1 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2647.2 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07, GHS09 |
| Signal word | Danger |
| Hazard statements | H302 + H315 + H319 + H335 |
| Precautionary statements | Precautionary statements: P280, P301+P312, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 2-3-1 |
| Flash point | 142 °C |
| Autoignition temperature | 370 °C |
| Lethal dose or concentration | LD50 oral rat 1200 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1650 mg/kg (oral, rat) |
| NIOSH | SN 1750000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 4-Nitro-N,N-Diethylaniline: Not established |
| REL (Recommended) | 0.5 mg/m³ |
| Related compounds | |
| Related compounds |
4-Nitroaniline N,N-Diethylaniline N,N-Dimethylaniline p-Nitroanisole p-Nitrophenol |

