4-Nitro-L-Phenylalanine: Looking Behind the Molecule
Historical Development
Before researchers started to see the value in tweaking amino acids, the lineup always looked traditional: glycine, alanine, tryptophan, and so on, doing their jobs in biological systems. The idea of adding a nitro group to phenylalanine seemed odd decades ago. Scientists in the early 20th century played with aromatic substitutions, but the concrete synthesis of 4-nitro-L-phenylalanine didn't hit the journals until improvements in organic synthesis, like nitration under controlled temperature, allowed for careful selection of para positions. Biochemists chased new building blocks for proteins, and the introduction of the nitro group into the phenyl ring proved interesting. Over time, this simple idea fueled a lot of research about how structure changes function, with labs competing in the latter half of the 20th century to prove what functions proteins could gain just by sneaking this molecule into their backbone.
Product Overview
Today, 4-nitro-L-phenylalanine shows up as a yellowish crystalline powder, marked by the nitro group sticking out at the para position on the phenyl side chain. It doesn't pop up in nature; it’s a synthetic analog. The phenylalanine skeleton stays the same, but this one sports an extra electron-withdrawing group, giving new behavior to otherwise familiar biochemistry. Labs often keep this product for building unusual peptides, studying enzyme mechanisms, or creating molecular probes that play tricks on protein structure. The package usually lands with a high purity percentage, suitable for direct application in research, especially where modifications in protein chemistry pay off in new data.
Physical & Chemical Properties
The pure solid usually appears yellow or pale orange, a sign the nitro group changes the chromophore nature of the phenyl ring. This molecular tweak makes a difference—not just in color, but in melting point and solubility, too. Unlike plain phenylalanine, the nitro derivative resists dissolving easily in water, thanks to that bulky, electron-hungry group, but it’s more cooperative in organic solvents like DMSO or DMF. The molecular weight lands at 226.19 g/mol. In terms of stability, the nitro group can draw the attention of reduction chemistry, but under standard storage—dry, cool, out of direct sunlight—the compound keeps for months. A bit of acidity or extreme heat can send it breaking down or turning colors, reminding researchers that even stable derivatives need respect.
Technical Specifications & Labeling
Suppliers mark 4-nitro-L-phenylalanine with clarity, listing purity over 98%, and giving details about batch number, manufacturing date, and sources of raw input. A proper label does more than check legal boxes—it reassures teams who need traceability, especially when running experiments with tight tolerances. The product arrives with identity confirmation, often backed by NMR and IR data. Researchers care about stereoisomerism, so a true L-enantiomer gets its rotation checked—measurements reported in degrees—and any sign of racemization leads to protocol reviews. Proper labeling gives peace of mind and supports the reproducibility so many journals demand now.
Preparation Method
I remember first seeing the nitration of L-phenylalanine in a synthetic chemistry class, and the professor hammered home the point: control is everything. Start with L-phenylalanine and introduce an electrophilic nitration agent, like a chilled mix of concentrated nitric and sulfuric acid. Temperature drops below 10°C so side reactions don’t sneak through. Stirring, patience, and quenching with ice water stops things from running wild. Crude product precipitates, gets filtered, and endures recrystallization—frequently from dilute ethanol-based solvents. The nitro group settles into the para position because that’s where the reactivity lines up. Modern improvements swap out harsh acids for milder reagents and enzyme-mediated reactions—greener, but not always scalable. Afterward, a solid round of purification with HPLC and checks for optical purity make sure no D-isomer contaminates the batch.
Chemical Reactions & Modifications
No chemist sees 4-nitro-L-phenylalanine as a final destination. The nitro group opens up redox chemistry possibilities, letting you reduce it to an amino group and then play with further substitutions. In peptide chemistry, it’s used to introduce new cross-linking sites. I’ve seen labs attach fluorescent tags through nucleophilic aromatic substitution—perfect for tracking proteins inside cells. Enzymatic modifications become more interesting, too, since enzymes sometimes don’t recognize the nitro group and slow down or stop, revealing hidden control points in biosynthesis or folding. Even simple catalytic hydrogenation of the nitro group leads to para-amino-L-phenylalanine, which belongs to another expansive world of protein engineering.
Synonyms & Product Names
Diving into vendor catalogs, this compound picks up a few names. Commonly, it’s sold under the official title 4-nitro-L-phenylalanine, but you might spot p-nitro-L-phenylalanine, or even L-2-amino-3-(4-nitrophenyl)propanoic acid. Some academic literature trims it down to “4-NO2-L-Phe.” Each label follows conventions of organic nomenclature, underscoring where the nitro group sits. It’s wise to cross-check synonyms in any procurement process, as catalog numbers and purity grades sometimes trek across slight naming differences.
Safety & Operational Standards
No one wants trouble in the lab, so handling 4-nitro-L-phenylalanine comes with basic protocols. Gloves keep potential skin contact at bay; the nitro group can cause irritation. Ventilation matters—dust from powders can drift, and while this isn’t as volatile as industrial nitro compounds, it’s better to use a fume hood for weighing and transfers. Clean workspaces, closed storage, and easy access to safety data sheets help. Separation from reducing agents and strong bases avoids accidental decomposition. Any accidental ingestion, inhalation, or extensive skin contact needs prompt medical evaluation. Spills get scooped up with damp disposable towels—not swept dry to avoid dust clouds. Waste management stays strict, using designated organic chemical waste disposal routed through environmental health offices.
Application Area
My encounters with 4-nitro-L-phenylalanine have always circled back to protein engineering projects or enzyme mechanism studies. Researchers put this molecule into synthetic peptides to ask pointed questions about electron distribution, hydrophobicity, or reactivity in active sites. The nitro group blocks sites for potential hydrogen bonding and pushes the aromatic ring’s electronics, serving as a reporter molecule when paired with spectroscopy. Pharmaceutical teams use this derivative in drug metabolism studies, mapping how bulky aromatic substitutions alter absorption or metabolic breakdown. In cell biology, this non-canonical amino acid helps design sensors—the unique absorbance and fluorescence footprint lets people shine light on protein location and folding. Chemical biologists turn to it for defining protein-protein interactions that depend on subtle changes, which only non-natural amino acids can unlock.
Research & Development
Plenty of innovation flows into expanding the toolkit around 4-nitro-L-phenylalanine. Early stage research looks at how ribosomes and synthetases handle incorporation of such modified residues—early work showed stumbling blocks, but recent directed evolution creates tRNA-synthetase systems picking up this analog like it’s no big deal. This unlocks living cell incorporation, not just test tube chemistry. Analytical chemists dig into the compound’s spectroscopic signatures, crafting new peptide-based sensors that respond to reducing environments in real time. Some teams in drug development incorporate this amino acid into therapeutic peptides, aiming for better specificity or resistance to breakdown by proteases. The field keeps trying novel modifications—reducing, methylating, or further substituting the aromatic ring after insertion—to turn simple yellowish powder into probes for mapping biological landscapes.
Toxicity Research
Any time a nitroaromatic pops into the picture, toxicology questions follow. Most studies lump 4-nitro-L-phenylalanine in the “low risk, high caution” zone when handled in research settings. Oral, dermal, or inhalation toxicity shows up at far higher doses than typical lab use, but data from animal models underline the need for vigilance. The nitro group can trigger oxidative stress in cells, especially after metabolic reduction. Chronic exposure to nitro-substituted aromatics, especially in commercial environments, has been linked with hemotoxic effects in rodents—methemoglobinemia being a known outcome. For cell culture projects, teams always check for unexpected cytotoxicity—results vary with cell line and exposure time, so no one uses this compound on living systems without a pilot study. Gloves, goggles, waste separation—it’s less about overkill and more about never letting routine breed carelessness.
Future Prospects
Synthetic amino acids, especially those with aromatic side chain modifications, drive a fresh wave of biological exploration. A few decades ago, no one thought proteins could pack in non-standard building blocks without falling apart. With 4-nitro-L-phenylalanine, researchers tap into new protein chemistries—biosensors, therapeutic scaffolds, and advanced imaging agents. As site-specific incorporation technologies mature, this analog will keep finding new homes in proteins engineered for next-level diagnostics or tailored functions, perhaps even as biocatalysts for industrial transformation. Environmental concerns may drive greener synthesis and tighter safety monitoring, but the demand for unique electronic and chemical properties will keep interest alive. Universities and startups both aim to harness non-natural amino acids, using compounds like this to answer questions ordinary biochemistry leaves hanging.
What is 4-Nitro-L-Phenylalanine used for?
What Drives Interest in 4-Nitro-L-Phenylalanine?
In the world of scientific research, small changes in a single molecule can open new paths to bigger discoveries. 4-Nitro-L-Phenylalanine isn’t a household name, yet for chemists and biologists, it means a new level of control over proteins. For years, researchers turned to this amino acid for more than just another ingredient in a lab—it helps them see, measure, and even tweak how life’s machines work at the tiniest level.
Helping Proteins Tell Their Story
Think of proteins as the tools and machines running everything inside your cells. Tracking what they do, where they go, and how they change can get tricky. That’s where 4-Nitro-L-Phenylalanine stands out. Its biggest value shows up in the way it sticks out: the nitro group on its ring absorbs light and changes how the protein behaves. Because of this, scientists use it to ‘label’ proteins. With this chemical marker, experiments get clearer results. Instead of guessing, labs can follow the journey of a protein in a living cell or watch what happens during a reaction as it unfolds.
This matters for drug development. Medicines work best when they hit the right protein at the right time. By slotting 4-Nitro-L-Phenylalanine into a protein, researchers can watch interactions unfold under microscopes that snap hundreds of pictures per minute. Mistakes from the past—like drugs that failed because their targets weren't clear—have taught laboratories to lean on these markers.
Peeling Back the Mystery with Precision Tools
My own run-ins with synthetic amino acids showed how useful they get in day-to-day lab work. It’s not just tagging—4-Nitro-L-Phenylalanine tweaks the local chemical environment. In practice, this means scientists can push proteins to do things they rarely do in nature. Sometimes, it makes a protein absorb light differently, leading right into what’s called “spectroscopic” studies. Here, a researcher can shine a specific color of light and read off real-time signals that tell if the protein is folded correctly or tangled up in knots—key information for anyone working on diseases caused by misfolded proteins, like Alzheimer’s or cystic fibrosis.
Proteins mixed up with 4-Nitro-L-Phenylalanine also help in mapping out where tiny changes lead to big differences. Picture a team changing out parts in a car engine, one piece at a time—they’d quickly notice which tweak leads to a smoother ride or a shuddering stop. Biologists do the same but at a molecular level. This method helps cut down wasted time and funding when chasing what truly matters in therapeutic research.
Tackling the Roadblocks
No tool comes without trade-offs. Introducing an unnatural amino acid like this one asks for careful planning. Sometimes, the protein won’t work as expected, or it might refuse to accept the new part. Teams must check results against trusted controls. That’s where experience in the lab pays off. Mistakes teach that regular validation and adjusting the approach keep things honest and accurate. Rushing or skipping these steps leads to misleading results and wasted resources.
Addressing scarcity is next. Not every lab has access to a constant supply, which limits what some groups can try. Universities and suppliers can build networks to pool stock and share resources, opening the door so smaller outfits get the same chance as the bigger labs. Crowdsourcing rare chemicals through academic consortia already makes a difference for research communities.
Looking to the Future
What gets me excited isn’t just the chemical’s power in one experiment, but in where it points for the next generation of research. 4-Nitro-L-Phenylalanine acts as a signpost, urging science to dive deeper, track precise changes, and avoid once-common guesswork. Progress picks up speed each time these precise measurements tell a story about health, life, or medicine that we couldn’t have written before.
Is 4-Nitro-L-Phenylalanine safe for laboratory use?
Understanding What’s Involved
Working in any lab always teaches you to respect the bench. Over time, you start to notice how every substance has its quirks. 4-Nitro-L-Phenylalanine brings its own set, from handling requirements to its spot on a lab shelf. As an amino acid derivative, it's been around for specialized chemistry, protein design, and some pretty innovative experiments in molecular biology. Still, lining up with the bottles and powders you use daily, it’s smart to look up safety data and keep your habits sharp.
Weighing the Hazards
This nitro-substituted amino acid has a yellowish tint and, like many aromatic compounds, needs careful handling. Safety data tells us to keep it away from strong reducers and other reactive agents. Its dust or fine powder can be irritating to lungs and eyes, so nobody wants a cloud of it floating around. Chemical suppliers recommend gloves, goggles, and a solid lab coat. Pulling from my own days at the bench, any powder like this had me reaching for a mask and working inside a fume hood, no questions asked.
MSDS sheets lay out concerns—avoid ingestion, keep off skin, don’t breathe it in, flush with a lot of water if there’s contact. Nitrated aromatics sometimes draw extra scrutiny because of their potential toxicity. No major incidents linked to this specific compound show up in the literature, but its close relatives, like nitrobenzene, aren't exactly gentle. That pushes things firmly toward “respectful caution.”
Fact-Checking Safety Claims
Trusted chemical suppliers keep data sheets fresh. Sigma-Aldrich points out moderate hazards at worst, with nothing explosive or acutely poisonous about 4-Nitro-L-Phenylalanine in typical lab settings. That lines up with what’s reported in chemical hazard databases and published case studies—no signals suggesting this compound stands out as a danger if basic protocols are respected. Studies with protein engineering in E. coli used it without accident, just following standard safety routines.
Eyes should always land on the shelf life and how to store it. Moisture, light, or mixing with strong acids or bases can change its chemistry or increase risks. Container labels from reliable suppliers spell out the precautions, and most of what you’ll find falls under regular chemical hygiene guidelines.
Safety in Daily Use
I’ve been in labs that range from state-of-the-art facilities to teaching classrooms. Every place runs best with routines: label everything, wear the right gear, know where the eyewash sits, keep the MSDS handy. Reviewing safety before using a new chemical never feels wasted. Accidents happen most often when someone gets too casual or tries to save time. A healthy respect for safety culture, not just one-off warnings, keeps people out of trouble far better than rules written in a binder.
How to Handle and Reduce Risk
Safe handling starts with gear—well-fitted gloves, lab coat, eye protection, and working with powders inside a ventilated hood. Training helps spot trouble before it happens, and every new teammate benefits from seeing these habits modeled in real time. Wasting time looking for a data sheet during a spill will just raise stress and risk. Spills or exposure need quick, clear action—wash the area, alert others, and dispose of waste following local rules. Fire extinguishers and chemical spill kits belong ready, not buried in a cabinet.
Many teams hold short refreshers and walk-throughs. These may sound simple, but they keep memories fresh and prioritize people’s safety where it matters most.
Looking Ahead
Treating 4-Nitro-L-Phenylalanine with respect helps build good habits. Labs win by sticking to clear habits, keeping training up-to-date, and checking data from solid sources. Experience teaches that care taken with “routine” compounds keeps surprises at bay. Staying curious and cautious has always delivered the best days in science.
What is the purity level of 4-Nitro-L-Phenylalanine?
Getting to the Root of Purity in Research Chemicals
In lab work, nobody takes chances with the chemicals on the bench. 4-Nitro-L-Phenylalanine often pops up in studies related to protein engineering and enzyme function. The story always ties back to purity. There’s no hiding poor quality behind big talk — results skew, experiments fail, grants dry up. Most suppliers target 98% to 99% purity for this amino acid. That means only a trace of anything else, like residual solvents or unreacted starting material, tags along in a batch that weighs one gram.
Packing More Science Into Every Gram
Some years ago, my team once hit a dead end because our sample of 4-Nitro-L-Phenylalanine clocked in at 95% instead of the advertised 99%. All it took was a few impurities interfering with protein folding, causing the reactions to drift. The funny part — it isn’t just about what you see on paper. Even a 1% sliver of impurity can mess up downstream data, especially during sensitive kinetics studies or crystallization work. LC-MS and HPLC reports don’t just collect dust. Researchers habitually cross-check these to confirm purity before a single pipette goes near the compound.
Why Regulators and Funders Look at Purity
Funding agencies and compliance auditors keep an eye on chemical specs. Grants often require documentation showing purity at 98% and above. Lower numbers kick up red flags — both in pharmaceuticals and advanced biochemical studies. Safety comes into play as well. Impurities can introduce unpredictable hazards or cloud toxicological results. One batch that missed specs led to unnecessary risk in a neighboring lab. Later, documentation showed how a supplier quietly dropped specs. Everybody paid the price.
Facts on Synthetic Amino Acid Quality
Most commercial 4-Nitro-L-Phenylalanine travels with a certificate of analysis. Reliable vendors use spectroscopic and chromatographic methods to back up their claims. A scan through Sigma-Aldrich, TCI, or Alfa Aesar catalogs reveals typical purity claims of 98% or slightly more. Quality differences are most obvious where the supply chain slides off track. During the early pandemic, delayed shipments and rapid sourcing left several colleagues with “lab-grade” product below 95%. Unreliable raw material triggers more than wasted money — it snowballs delays across graduate projects and published results.
What Can Lift Purity and Trust?
Building quality starts at trusted suppliers and never skips quality checks. Labs asking for bulk should push for updated batch analytics and ask about storage and handling. Some opt for recrystallization or further purification using preparative HPLC. That eats budget and time, but if a thesis, patent, or key paper rides on the line, nobody regrets it. Keep raw data from every new batch of chemicals. Cross-check vials against certificates and test on the most crucial experiment first, not after a month of hard work. Communication with chemical suppliers can shape quality, too. Feedback — positive and negative — pushes the industry to aim higher, close loopholes, and cut back on disappointing surprises.
How should 4-Nitro-L-Phenylalanine be stored?
What Experience Teaches About Storing Uncommon Amino Acids
Working with specialty amino acids like 4-Nitro-L-Phenylalanine brings its own lessons. On my lab bench, we always give extra care to chemicals decorated with functional groups like nitro on the aromatic ring. This compound doesn’t share the relaxed approach of standard household ingredients — it demands respect for its quirks and sensitivities.
Temperature control makes a difference. 4-Nitro-L-Phenylalanine, a yellowish powder, stays in much better condition if it remains away from summer heat or direct light. We designate a shelf in a refrigerator (typically 2-8°C), separate from open air and sudden temperature swings. This slows down unwanted reactions that happen with extended warmth or careless exposure.
Factors That Matter: Moisture, Air, and Light
The nitro group brings risk. Exposure to high humidity or water lets the structure break down or clump, affecting both research and production results. Here’s a real pain point: I once forgot to properly seal a bottle, and the next month the powder looked more like a useless cake than the free-flowing compound I started with. Silica gel packets or desiccators in storage make this situation less likely. Airtight glass bottles seal out the trouble caused by damp air.
Air can cause oxidation. Oxygen naturally moves in and can change sensitive molecules, making the original compound less pure or effective for its intended purpose. For practical safety, removing as much oxygen exposure as possible works—sealing bottles tightly, flushing with an inert gas like nitrogen for particularly sensitive chemicals, and limiting the time spent open on a busy bench all matter.
Light degrades plenty of organic molecules. The nitro group in 4-Nitro-L-Phenylalanine increases its sensitivity. Amber glass containers or aluminum foil wrappings provide another layer of protection, especially if shelf lights or sunlight reach the storage area. Most scientists I know—myself included—don’t take chances here. An opaque bottle in a cool, dark cabinet saves headaches later.
Label, Inventory, and Safety Precautions
Missing or damaged labels spell disaster. Chemicals often look similar but act differently, and even the most experienced lab workers can make mistakes under pressure. I train colleagues to double-check labels, write purchase and open dates, and list storage conditions on every bottle. Simple discipline like this prevents misuse and ensures that old, unstable product gets removed on time.
Gloves and masks help prevent direct exposure, since many aromatic nitro compounds irritate skin or produce dust that lingers. I recommend storing 4-Nitro-L-Phenylalanine in areas with clear signage, away from acids, bases, and oxidizers. Keeping an updated inventory with hazard symbols and emergency contacts gives everyone in the lab or storage room certainty if something goes wrong.
Shortcuts Create Risk, Not Efficiency
Cutting corners with specialty chemicals costs time and money down the line. Proper organization, temperature control, airtight packaging, and clear labeling keep 4-Nitro-L-Phenylalanine not only effective, but also safe for those working around it.
Experience—and a few hard-learned lessons—convince me that investing in secure storage wins out over any shortcut, every single time.
What are the shipping conditions for 4-Nitro-L-Phenylalanine?
A Look at Chemical Shipping Realities
Shipping chemicals like 4-Nitro-L-Phenylalanine puts responsibility squarely on everyone's shoulders—from the lab technician to the logistics driver. This compound, often used in pharmaceutical research or advanced biochemistry labs, doesn't just get tossed in a box with a shipping label slapped on. Safe delivery means respecting the nature of the material, industry regulations, and the people down the line who handle it.
Physical Profile Shapes the Rules
From every experience handling specialty amino acids over the years, real safety starts with knowing what you’re dealing with. 4-Nitro-L-Phenylalanine comes as a crystalline powder, sensitive to moisture and sometimes light. Contact with water risks degradation, and improper sealing can shorten shelf life. Packaging in airtight, light-resistant containers provides a layer of security before the carton leaves the warehouse.
Regulation in the Real World
No one gets a pass on chemical regulations—certainly not with compounds that might be hazardous. 4-Nitro-L-Phenylalanine, while not explosive, demands respect because of nitro-group reactivity and toxicity if mishandled. Countries have their own rules about shipping substances across borders. These rules touch everything from labeling to customs declarations. Failing to follow those guidelines can bring shipments to a halt, trigger fines, or endanger handlers.
Temperature and Journey Concerns
Some compounds degrade in the heat or become ruined if frozen. Shipping 4-Nitro-L-Phenylalanine doesn’t usually require refrigeration, but direct sunlight or high humidity in a delivery truck could cause trouble. From warehouse experience, crew members usually add desiccants into packaging, keep storage away from heat sources, and pick overnight deliveries in summer to avoid sitting in a metal truck as the sun beats down.
Broken Links in the Chain
Mistakes often happen in the transition points. Shipments might move from well-controlled storage to basic mailrooms. Someone on the receiving end, perhaps in a busy college mailroom or a startup lab, can miss warning labels or set an important box in an area with temperature swings. Training staff to recognize hazard symbols and double-check delivery conditions makes a difference, reducing risks both to people and experiments relying on that compound.
Possible Improvements
Better communication closes gaps. Suppliers now use QR codes on packaging, so anyone scanning the label routes straight to all the handling instructions required. I’ve seen this help new lab assistants avoid mistakes on stressful days. Electronic tracking lets customers know exactly where the shipment stands, useful for compound stability and peace of mind. For international orders, working with partners experienced in chemical logistics reduces the odds of unexpected delays.
A Smarter Path Forward
Shipping demands don’t just spring from a rulebook. They exist due to real risks and sustainable practices. Investing the extra effort into controlled packaging, accurate documentation, and ongoing staff training doesn’t just protect one shipment. It builds trust in the system, from the supplier bench to the research lab. Anyone working with 4-Nitro-L-Phenylalanine, or any specialty chemical, learns quickly that care in shipping supports every step that follows.
| Names | |
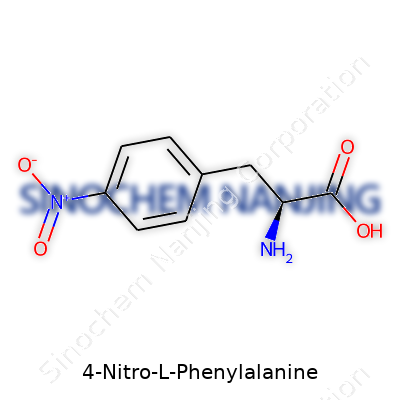
| Preferred IUPAC name | (2S)-2-amino-3-(4-nitrophenyl)propanoic acid |
| Other names |
(S)-2-Amino-3-(4-nitrophenyl)propanoic acid L-4-Nitrophenylalanine 4-NO2-L-Phenylalanine |
| Pronunciation | /ˈfɔːr ˈnaɪtroʊ ɛl fɪˌniːlˈæləniːn/ |
| Identifiers | |
| CAS Number | 37541-18-5 |
| Beilstein Reference | 1728443 |
| ChEBI | CHEBI:27661 |
| ChEMBL | CHEMBL504159 |
| ChemSpider | 26234710 |
| DrugBank | DB02120 |
| ECHA InfoCard | 82cd9105-8f20-4e2f-a812-9be8d7c90fd7 |
| EC Number | 4.1.1.43 |
| Gmelin Reference | Gmelin 83391 |
| KEGG | C02949 |
| MeSH | D017614 |
| PubChem CID | 44213609 |
| RTECS number | SY8575000 |
| UNII | D90GB75X3F |
| UN number | Not classified |
| CompTox Dashboard (EPA) | DTXSID0041818 |
| Properties | |
| Chemical formula | C9H10N2O4 |
| Molar mass | 226.19 g/mol |
| Appearance | Light yellow to yellow powder. |
| Odor | Odorless |
| Density | Density: 1.4 g/cm3 |
| Solubility in water | Soluble in water |
| log P | -2.08 |
| Acidity (pKa) | 4.2 |
| Basicity (pKb) | 2.29 |
| Magnetic susceptibility (χ) | -61.7·10^-6 cm³/mol |
| Dipole moment | 4.73 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 372.6 J/(mol·K) |
| Std enthalpy of combustion (ΔcH⦵298) | -1602.0 kJ/mol |
| Hazards | |
| Main hazards | H302 + H312 + H332: Harmful if swallowed, in contact with skin or if inhaled. |
| GHS labelling | GHS07, GHS09 |
| Pictograms | GHS07,GHS09 |
| Signal word | Warning |
| Hazard statements | H302: Harmful if swallowed. |
| Precautionary statements | Precautionary statements: P261, P305+P351+P338, P304+P340, P312 |
| NFPA 704 (fire diamond) | 2-3-0-OX |
| Flash point | Flash point: >110°C |
| LD50 (median dose) | LD50 (median dose): >5 g/kg (rat, oral) |
| PEL (Permissible) | Not established |
| REL (Recommended) | 10 mg/kg |
| Related compounds | |
| Related compounds |
Phenylalanine L-Tyrosine 4-Nitrophenylalanine methyl ester 3-Nitro-L-phenylalanine p-Nitroaniline Nitrotyrosine |

