4-Nitro-2-Toluidine: Exploring a Complex Chemical
Historical Development
Chemicals like 4-Nitro-2-Toluidine have been part of the industrial world far longer than many realize. Around the late nineteenth century, the dye industry’s expanding appetite for synthetic colorants sparked innovative minds in Europe to explore new nitro-aromatic compounds. Chemists needed resilient, vibrant pigments for cotton and wool, and aromatic amines helped make that possible. Crafting 4-Nitro-2-Toluidine was never about perfection but about meeting the needs of fabrics, paper, and plastics. Over time, refinements in nitration processes and purification allowed the compound to become a standard raw material, particularly in the demanding world of azo dye and pigment production. In the early years, the lack of robust safety standards led to various mishaps, and that historical record shaped both how we synthesize and handle it today.
Product Overview
With a yellow to orange crystalline appearance and a faint, peculiar odor, 4-Nitro-2-Toluidine finds its way into industrial storerooms as an essential building block. It doesn’t make headlines, yet its value in providing the backbone for a vast range of dyes and intermediates can’t be dismissed. One batch may turn up in bright red pigments for artists’ paints, another in the coloring of specialty plastics. Each grain carries with it the expectation of consistency, as many manufacturing processes depend on its reaction profile and purity.
Physical and Chemical Properties
4-Nitro-2-Toluidine has a molecular formula of C7H8N2O2, so it contains a methyl group, a nitro group, and an amino group hanging off a benzene ring. It tends to melt between 70 and 74 degrees Celsius, which means it will soften in a hot warehouse if not stored correctly. Its density lands at about 1.3 grams per cubic centimeter, and it is not particularly soluble in water—nature tends to drive aromatic organics toward organic solvents such as ether or ethanol. A strong odor warns users of its presence. Its reactivity comes from both the electron-donating amino group and the electron-withdrawing nitro group; they’re set up to make this molecule both reactive and somewhat tricky to handle. Unusually, it holds up relatively well under standard light and air conditions, which suits bulk storage.
Technical Specifications and Labeling
Anyone who’s worked in a chemical warehouse knows labeling isn’t a suggestion. For 4-Nitro-2-Toluidine, the key numbers matter: melting point 70-74°C, purity above 98% for most commercial uses, and maximum allowed water or ash content tightly regulated. Every drum or sack comes with hazard symbols calling out health and environmental risks, and every pallet carries transport codes—often UN 1662—showing this is a regulated, hazardous compound. All Safety Data Sheets (SDS) are exhaustive, listing known irritant and toxicological risks, and labeling complies with global harmonized standards. Careless handling can lead to real consequences, so firms do not cut corners on this front.
Preparation Method
Nobody prepares 4-Nitro-2-Toluidine on a whim; the process invites respect. Most routes start with 2-toluidine, which undergoes nitration in acid—the classic one-two punch of concentrated nitric and sulfuric acids does the trick. Long before computers tracked batch parameters, plant workers watched temperature and timing to avoid dangerous runaways. The nitro group can slip onto the aromatic ring in a few places, yet careful control ensures it lands in the right spot. Each step—acid mixing, stirring at low temperatures, controlled heating—requires real vigilance. Following nitration, skilled technicians extract product into water and then organic solvents to purify out unwanted isomers or side products. Yield improvements emerged gradually, spurred by improvements in agitation, temperature control, and analytical verification. Waste treatment protocols have evolved, driven by increasingly strict environmental norms.
Chemical Reactions and Modifications
This compound acts as a sturdy, versatile precursor. Chemists routinely reduce that nitro group to create splendid amines, or they fold it into azo linkages to create high-stability dyes. The toluene moiety is tough, but under certain conditions you can oxidize the methyl group or manipulate the ring for advanced intermediates. In specialty labs, researchers transform it into sulfonic acids or halogenate it for even more complex molecules. Each reaction is honed for selectivity, with process safety and environmental stewardship taking top priority. Careless handling—especially at large scale—can produce toxic byproducts or runaway exotherms, so experienced hands run these operations.
Synonyms and Product Names
Over the years, this compound collected quite a few aliases. Old-timers might know it as "2-Methyl-4-nitroaniline," while some warehouse receipts still record "2-Amino-4-nitrotoluene." Suppliers that trade globally may label their drums with "C.I. 37120" or "C.I. 2-Nitro-p-toluidine" to avoid confusion across borders. Some research papers lean into the IUPAC name "4-nitro-2-methylaniline." Despite the name shuffle, the core structure stays the same—a nod to the stubborn consistency of chemistry.
Safety and Operational Standards
Nobody who’s ever had direct contact with aromatic amines underestimates their risk. 4-Nitro-2-Toluidine causes skin and eye irritation, and repeated handling contributes to chronic health conditions. Toxic dust can irritate the airways, and there are questions about long-term carcinogenicity. Safety rules demand gloves, goggles, and excellent ventilation at a minimum. Spill procedures exist for a reason, as does proper fume extraction. Storage spaces stay sealed, cool, and dry, far from oxidizers and open flames. In busy production lines, personal protective equipment becomes second nature, and regular medical monitoring is routine. Modern factories that cut safety budgets pay later in injuries, fines, or long-term health claims.
Application Area
Mostly, industrial users turn to 4-Nitro-2-Toluidine for dye production—reds, oranges, and violets owe their shade to this intermediate. Commercial textiles, printing inks, and even some plastics owe much of their vibrancy to careful chemical blending starting here. Its utility also stretches into specialty chemicals, agricultural research, pharmaceuticals, and laboratory reference materials. In the research world, its well-defined reactivity makes it a reliable testbed for synthetic experiments or mechanistic study. Although most don’t recognize it, anyone who’s ever worn colored fabric or seen safety markings in construction likely interacted with a dye made from this molecule.
Research and Development
Academic interest in 4-Nitro-2-Toluidine never fades completely. Teams probe its behavior in new coupling reactions and uncover ways to drive greener, higher-yield syntheses. Slashing hazardous waste tops many wish lists, and some labs pursue catalytic nitration or biotransformation. Others dig deeper, examining potential applications in next-generation organic materials for electronics or as scaffolds for drug discovery. Analytical chemists continuously refine tools for detecting trace impurities, supporting both regulatory compliance and advanced research. Innovation marches on as universities and companies chase better yields, cleaner conversions, and higher stability products.
Toxicity Research
Worker health remains front and center with this compound. Researchers dig into both acute and chronic impacts, looking for evidence of mutagenic or carcinogenic pathways. Animal studies reveal harmful effects with high exposures—liver, kidney, and skin damage surface in the literature. Epidemiologists track workers exposed over decades, linking some cases to increased cancer risk. Based on these results, regulatory bodies place strict exposure limits and require ongoing hygiene monitoring in plants. Bioaccumulation appears low, but the drive to substitute safer alternatives in consumer-facing products continues.
Future Prospects
The landscape for 4-Nitro-2-Toluidine will change as environmental, workplace, and consumer safety standards tighten. Demand for environmentally friendly colorants pressures manufacturers to consider alternatives, redesign processes, or improve waste handling. Technologies for greener synthesis gather momentum—biocatalysis and solvent minimization stand out as promising research avenues. Scrutiny from non-governmental organizations and regulatory agencies won’t relax either. As society pays closer attention to worker health and ecological footprint, the future for this molecule likely tracks toward safer processing, sharper compliance, and creative chemistry to reduce hazardous residues—all while industry seeks new frontiers in pigment science, material chemistry, and advanced industrial manufacturing.
What is 4-Nitro-2-Toluidine used for?
Understanding Its Role
4-Nitro-2-Toluidine doesn’t get much attention outside industrial circles, but its influence stretches far into everyday products. As someone who’s followed the progress of chemical research and regulation, I see how its role makes it both valuable and concerning. Chemically, it’s a crystalline substance with a light yellow color. Workers often encounter it in dye manufacturing plants. Unlike consumer-familiar dyes and pigments, this chemical usually shows up behind the scenes in factories that supply paints, textiles, and plastics with vibrant colors that last.
The Dye Industry’s Dependence
Plants that make colorants often lean on 4-Nitro-2-Toluidine as an intermediate step in dye synthesis. In practice, this compound reacts with other chemicals to create azo dyes, many of which go into coloring fabrics and plastics. These dyes tend to resist fading, which helps keep clothes and colored plastics looking sharp through sunlight and washing. Decades-old reports, like those from the National Toxicology Program, confirm its recurring presence on the factory floor. It rarely shows up in finished consumer goods in significant amounts, but even small traces raise eyebrows.
Health and Environmental Concerns
Drawing from my experience covering chemical safety, the health risks drive most of the controversy around this substance. Research links exposure to 4-Nitro-2-Toluidine with skin irritation, and, on a deeper level, possible carcinogenic effects. A 2010 review by the World Health Organization listed it as possibly carcinogenic based on animal studies. That’s not enough to panic, but I know workers handle it with gloves and masks, especially in countries where safety rules have teeth. Long-term exposure in poorly ventilated spaces stands out as a real hazard.
Contamination doesn’t stick only to factory air and worker safety. Residue in wastewater from dye manufacturing can travel far, affecting aquatic environments. Studies show breakdown byproducts that don’t disappear easily, posing problems for fish and local communities who live downstream. European Union guidelines now put extra rules on discharge and handling. Companies in places like India and China, which produce most of the world’s dyes, sometimes struggle to follow these rules, either from cost pressures or lack of oversight. The result sometimes ends up in water that nobody wants to swim in or drink from.
Moving Toward Solutions
No one really expects the dye industry to quit overnight, but efforts are growing to cut risks. Factories switching to closed-loop systems can lower the odds of leaks and spills. Those who follow textile supply chains push for stricter audits, even outside the West. An eco-label or “green chemistry” certification often comes from treating or replacing hazardous intermediates like 4-Nitro-2-Toluidine. It won’t solve every problem, but alternatives do exist. For example, some manufacturers use vegetable-based or less toxic aromatic amines, though color quality sometimes suffers or production costs rise.
Public access to safety data improves every year. In my view, making this information available helps keep pressure on both producers and regulators. Most people I meet don’t realize the link between cheap, brightly colored clothing and the more complex realities behind chemical sourcing. Open discussion and toughened policies help strike a balance between the benefits of durable dyes and the risks of harmful intermediates. Only with honest acknowledgment can the textile and plastics industries shape a safer production path forward.
What are the safety precautions when handling 4-Nitro-2-Toluidine?
What’s in Front of Us
4-Nitro-2-Toluidine isn’t just another chemical in the storage closet. Working with it, you run into risks you can’t put on the back burner. You’re talking about a yellow crystalline powder used in dye, pigments, and sometimes research labs. In my graduate research, I saw firsthand that familiarity breeds carelessness in labs. People put on gloves and goggles, but half the time didn’t know why. Understanding the risks means people actually follow through—not just check boxes.
Why Safety Isn’t Optional
The facts are straight. This substance is toxic if inhaled, swallowed, or absorbed through skin. The nitro group makes it more hazardous—exposure can irritate eyes, skin, and lungs, but long-term you need to worry about its cancer risk. OSHA lists aromatic amines and nitro compounds as occupational hazards. If you don’t respect the volatile nature of these compounds, you put your health, your team, and the environment at risk.
Gear That Keeps You Breathing Easy
I’ve learned the hard way that good intentions don’t stop chemical burns. Double up on nitrile gloves—latex can break down with aromatic chemicals. Always grab safety goggles, not just regular glasses. Go old school with a lab coat that covers you up to your wrists. If you have facial hair, make sure your respirator still seals. Fumes sneak through the smallest gaps. I watched a lab mate shrug off a properly fitted mask, and he spent hours coughing in the emergency room.
Ventilation: Your Invisible Shield
People underestimate the power of a well-set-up fume hood. Don’t just open a window—always work inside a certified fume hood. It pulls toxic vapors away before you get hit. Inspect airflow indicators every time. I once found out our hood had clogged filters, and it took a near miss for facilities to fix it. A working ventilation system saves lungs in the short term, and it lowers the invisible risks that spiral out over years of lab work.
Clean-Up Counts for Everything
Spills happen, but panic doesn’t help. Have a spill kit—don’t rely on makeshift materials. Baking soda and paper towels don’t cut it for something this nasty. Absorbent pads, neutralizers, and proper disposal bags make all the difference. Label every waste container with chemical names, concentrations, and date. I once saw an unmarked bottle tossed out as regular trash. That puts janitors and waste handlers at risk, too.
Training Goes Beyond Paperwork
Reading a safety data sheet once a year never made anyone safe. Live training, drills, and real spill scenarios stick with people. In my first semester, we did practice drills with water and colored dye. That visual reminder stayed with me longer than any lecture. Mock emergencies get people moving with muscle memory—not just after the fact regrets.
Pushing for Better Practices
Advocating for updated Standard Operating Procedures (SOPs) makes an impact. I’ve pushed for QR code signs on storage shelves with instant access to handling tips right on a phone. Institutional memory fades—systems help people act fast, even if they’re new.
Staying safe around 4-Nitro-2-Toluidine isn’t just about ticking boxes. It’s about taking every step like it matters—because for anyone in the lab, it does.
What is the chemical formula of 4-Nitro-2-Toluidine?
Getting the Basics Right
4-Nitro-2-toluidine, often found in dye synthesis and pharmaceutical manufacturing, carries the formula C7H8N2O2. The structure looks simple on the surface—a benzene ring, a methyl group at position two, and a nitro group at position four. In a chemistry textbook, the formula gives a quick answer. In the real world, knowing this structure opens doors to understanding why the molecule earns attention and where risks can lurk.
Why Chemists and Factories Care
Each atom in C7H8N2O2 carries a purpose. In labs where dyes or pigments come to life, the position of that nitro group transforms an ordinary aromatic amine into a starting point for many reactions. Small tweaks—like shifting a group just one spot—can mean the difference between a medicine and a toxin. Research published in the Journal of Hazardous Materials highlights that simple compounds with nitro and amine groups can show both innovation and harm, depending on how they interact with living systems.
Risks Lurk in Everyday Use
Firefighters respond to accidents involving chemicals like 4-nitro-2-toluidine around the world. During post-accident debriefings, I've learned how overlooked safety data sheets and weak knowledge about formulas can endanger lives. Too often, small manufacturers skip the training on how precise labeling and structure relate to hazard. C7H8N2O2 doesn’t just sit on a label—it offers a warning. Many ignore that nitroaromatic compounds, if mishandled, trigger toxic fumes under heat or fire. It's easy to look at a formula and forget the stories behind chemical burns or respiratory struggles in under-equipped facilities.
Environmental Echoes and Regulatory Oversight
The impact of 4-nitro-2-toluidine shows up long after use. I’ve seen news reports about river pollution, with dyes leaking from textile plants. Even a small pipeline crack can push those nitro compounds into water, where their persistence causes trouble for aquatic life. The Environmental Protection Agency (EPA) and World Health Organization (WHO) both track the long-term fate of aromatic amines like this one, pointing out how breakdown products stick around and accumulate.
Smart Handling and Industry Responsibility
Factories making or storing these materials must train teams for safe storage, swift cleanup, and emergency response. Chemical fume hoods and proper ventilation lower the odds of releasing toxins. From personal experience in safety workshops, it’s clear that interactive risk drills beat paper drills. Keeping emergency phone numbers posted and showing staff the chemical formula in context—explaining not just the structure but the reactivity—made a difference in how seriously everyone took their roles. Technology helps too. Smart sensors to catch leaks, and tighter inspection schedules, keep processes safer.
Moving Toward Safer Choices
Every user, from small lab techs to leaders of global manufacturing plants, faces choices on what substitutes or protective procedures to use. The arrival of green chemistry pushes for alternatives with fewer risks but similar benefits. Staying updated with professional organizations and scientific journals exposes new, less hazardous compounds for similar applications.
Beyond the Formula—Living with the Molecule
The string, C7H8N2O2, sums up a world of reactions, health impacts, and environmental consequences. Paying respect to chemistry as more than numbers keeps standards high and communities safer.
How should 4-Nitro-2-Toluidine be stored?
Beyond Labels: Why Proper Storage Should Matter
Most people reading a chemical label probably ask themselves, “What’s the worst that could happen?” With 4-Nitro-2-Toluidine, it’s not just about checking off compliance boxes—there are big risks if the details get skipped. Years spent working next to chemical storage areas drove the point home for me: a little attention saves a lot of trouble later. Once, we found a container left under direct sunlight. A split lid and strong, nose-burning odor was all it took for the safety officer to shut down the lab for an hour. These mistakes are common when corners get cut.
Separating Fact from Habit
4-Nitro-2-Toluidine carries a reputation for toxicity and fire risk. Many chemical accidents trace back to ignoring these basic facts. The compound can inflame respiratory tracts, irritate skin, and do worse if someone eats, drinks, or breathes it in over time. Improper storage also leads to dangerous fumes and reactions if mixed with the wrong substances. Data from the CDC and OSHA point to cancers and blood disorders for workers exposed without proper controls. So, good storage isn’t just about staying “official”—it’s about real health and safety.
Humidity, Heat, and a Recipe for Disaster
4-Nitro-2-Toluidine remains stable under normal circumstances but doesn’t tolerate heat or moisture. High temperatures speed up breakdown, leak odors, and sometimes trigger fire. Moisture causes clumping, which complicates safe handling and can encourage dangerous byproducts. I remember a neighbor at the plant leaving drums close to a boiler room for convenience. After a week of warm pipes humming, someone noticed a strong, acrid smell and stains spreading down the side of the drum. A trained eye caught it early, but that fix could have cost lives and money if ignored.
The Routine That Keeps Trouble Away
Store 4-Nitro-2-Toluidine in tightly sealed containers, away from sunlight and heat sources. Choose a cool, dry spot, well removed from acids, oxidizers, and food areas. Line shelves with firm, chemical-resistant material—nothing cheap or prone to flaking apart. Lock the doors, and post clear signs on entry. Ventilation deserves real attention: fans or air flow should move fumes outside, not into adjacent spaces. Rigorous labeling and monthly checks for leaks and rust catch small issues before they turn critical. Small spills need an emergency plan and absorbent materials on hand—nothing gets cleaned up “tomorrow.” Workers suit up with gloves, goggles, and approved masks before entering the room.
Commitment Makes a Difference
Solid routines come from seeing how much trouble one mistake can cause. The best employers run drill after drill, treat every routine check as a priority, and talk safety up and down the chain. Some companies reward workers who spot problems, creating a safety culture that stops accidents before they start. Outside audits, fresh signage, and spot training sessions turn smart plans into daily practice. The value isn’t just in avoiding fines. Proper storage keeps people healthy, keeps reputations intact, and saves money in the long run. Anyone who’s seen what a single chemical spill can do won’t ever take short cuts again.
Is 4-Nitro-2-Toluidine hazardous to health?
The Real Risks of 4-Nitro-2-Toluidine
Anyone who’s ever worked in labs or industries where pigments and dyes come into play has caught wind of quirky chemical names. 4-Nitro-2-Toluidine crops up often enough, mostly as a building block for certain colors in plastics, rubbers, and textiles. The question always pops up — just how risky is it?
No need to dress this up: yes, 4-Nitro-2-Toluidine brings along some serious health risks. Workplaces that use the stuff have material safety data sheets for good reason. Contact with skin or eyes sparks irritation. Breathing in dust or vapors will cause troubles for the respiratory system. There isn’t any reason for this stuff to get close to food or drink production.
Evidence From Science and Industry Experience
Diving into real health studies — the kind cited by health agencies like the US Environmental Protection Agency (EPA) and the European Chemicals Agency — workers exposed to 4-Nitro-2-Toluidine in higher concentrations over months or years have shown increased rates of bladder cancers and DNA changes. Researchers saw that the compound can trigger mutations in cells. Nobody wants to play dice with carcinogens just for colored plastic.
Most folks won’t run into this chemical outside workplaces. Still, accidents happen. Chemical suppliers flag it as potentially toxic to aquatic life. Folks on factory floors say that even brief skin contact without gloves causes red patches and stinging. Long-term contact sometimes leads to allergic reactions, and breathing too much dust sets off coughing or nosebleeds. Years back, I remember an old coworker who got a bad rash from handling pigment powder without realizing it had 4-Nitro-2-Toluidine in the blend. He wound up needing corticosteroid cream. Basic mistakes can become big problems fast.
Proper Handling and Safer Alternatives
Direct exposure to 4-Nitro-2-Toluidine can and does hurt people. No mystery there. The right approach: wear gloves, goggles, and serious masks where this chemical gets used. Showers and washing stations in the lab or shop are not just for show; they make a real difference when splashes happen. Companies using this material have a duty to train their teams about safe handling, spill response, and disposal: it’s not enough just to hand out a pamphlet.
Regulations try to keep airborne concentrations low for workplace safety. Still, not every small business has top-tier ventilation. I’ve seen shops swap to less-hazardous colorants over time, and the switch usually pays off. It lowers risk on the shop floor and earns trust from the workers. Some dye manufacturers phase out 4-Nitro-2-Toluidine altogether by moving toward pigments designed for safety and environmental friendliness. This change takes time and may cost more in the short term, but injuries always cost more in the end.
Moving Forward With Caution
Industry and regulators know the risks. They have hard numbers, personal stories, and a mountain of precautions. At the end of the day, it’s about respect for the material and the people using it. If the world can use something with less downside, it should. Until then, 4-Nitro-2-Toluidine deserves serious respect — and no excuses.
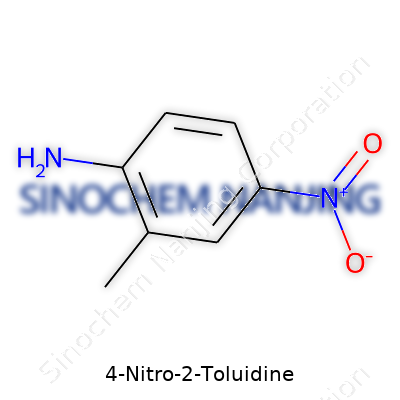
| Names | |
| Preferred IUPAC name | 4-Methyl-2-nitroaniline |
| Other names |
2-Amino-4-nitrotoluene 4-Nitro-o-toluidine 2-Methyl-4-nitroaniline |
| Pronunciation | /ˈfɔːr ˈnaɪtroʊ tuː təˈluːədiːn/ |
| Identifiers | |
| CAS Number | 99-51-4 |
| 3D model (JSmol) | `CC1=CC=C(C=C1[NH2])[N+](=O)[O-]` |
| Beilstein Reference | 1208892 |
| ChEBI | CHEBI:34630 |
| ChEMBL | CHEMBL109441 |
| ChemSpider | 2030690 |
| DrugBank | DB16948 |
| ECHA InfoCard | 200-207-1 |
| EC Number | 611-041-00-5 |
| Gmelin Reference | 82834 |
| KEGG | C14343 |
| MeSH | D014006 |
| PubChem CID | 7462 |
| RTECS number | XZ3850000 |
| UNII | 8E4G4U65S8 |
| UN number | 1663 |
| CompTox Dashboard (EPA) | DTXSID3047665 |
| Properties | |
| Chemical formula | C7H8N2O2 |
| Molar mass | 137.14 g/mol |
| Appearance | Orange powder |
| Odor | ammonia-like |
| Density | 1.28 g/cm3 |
| Solubility in water | slightly soluble |
| log P | 1.97 |
| Vapor pressure | 0.0000415 mmHg at 25°C |
| Acidity (pKa) | 4.68 |
| Basicity (pKb) | 9.64 |
| Magnetic susceptibility (χ) | -60.8·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.660 |
| Dipole moment | 3.61 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 150.2 J/mol·K |
| Std enthalpy of formation (ΔfH⦵298) | −29.9 kJ mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -1852.8 kJ/mol |
| Pharmacology | |
| ATC code | |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin irritation, causes serious eye irritation, may cause an allergic skin reaction, may cause cancer |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H335, H351 |
| Precautionary statements | Precautionary statements: "P261, P264, P270, P272, P273, P280, P302+P352, P308+P313, P321, P363, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-0 |
| Flash point | 142°C |
| Autoignition temperature | 540°C |
| Lethal dose or concentration | LD50 oral rat 1190 mg/kg |
| LD50 (median dose) | LD50 (median dose) of 4-Nitro-2-Toluidine: "2,480 mg/kg (oral, rat) |
| NIOSH | SN 21000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | REL: 1 mg/m³ |
| IDLH (Immediate danger) | 100 mg/m3 |
| Related compounds | |
| Related compounds |
2-Nitrotoluene 4-Nitrotoluene 2-Toluidine 4-Toluidine 2-Nitroaniline 4-Nitroaniline |

