4-Nitro-2-Methoxyaniline: Beyond the Laboratory Bench
Historical Development
In the early days of synthetic chemistry, curiosity about aromatic nitro compounds led researchers to experiment with different substituents on a benzene ring. One standout discovery was 4-Nitro-2-Methoxyaniline. Chemists gravitated toward benzene derivatives, not just for academic curiosity, but driven by the growing dye industry and the pharmaceutical movement during the 19th and 20th centuries. Back then, many of the techniques relied on direct nitration and subsequent amination, a process that took plenty of trial and error. Over time, practical adjustments surfaced, like optimizing the timing of methoxylation to keep yields stable and impurities low. By the 1960s, as organic synthesis matured, production of 4-Nitro-2-Methoxyaniline became more reliable, and the compound found a place in both industrial and academic settings.
Product Overview
This compound sits among substituted anilines, recognizable for its pale yellow to orange powder. Chemists value it for that dual substitution pattern—one side offers the nucleophilicity of an aniline, the other brings the strongly electron-withdrawing touch of a nitro group, and right next to it, the electron-donating methoxy group. These features make 4-Nitro-2-Methoxyaniline handy in all sorts of reactions, whether the job calls for reduction, oxidation, or further functionalization. For anyone working in dye chemistry, the combination spells out possibilities, driving both color formation and stability.
Physical & Chemical Properties
4-Nitro-2-Methoxyaniline typically appears as small crystalline solids, occasionally clumping if exposed to humidity. Its melting point usually hovers around 104–106°C. It smells faintly chemical but doesn’t pack the pungency of some other nitroanilines. In organic solvents like ethanol, ether, or chloroform, it dissolves well, but water offers little solubility due to the balance between polar functional groups and the aromatic backbone. This compound resists moderate heat, but it doesn’t fare well near flames or strong oxidizers. Its molecular formula, C7H8N2O3, lays the foundation for further chemical manipulation in downstream products.
Technical Specifications & Labeling
Labs and manufacturers usually set purity benchmarks above 98%, and the color must be consistent, often measured by spectrophotometric analysis. Impurities—especially related nitro or methoxy compounds—can throw off batch performance, especially in dye work. Standard labeling calls for CAS Number 6627-16-9, hazard class information, and proper UN identification if shipping in bulk. Documentation trails every shipment, and safety data sheets spell out not just chemical makeup but fire, spill, and medical response info, too. Closed containers shield against both light and moisture, prolonging shelf life, while warning pictograms alert handlers to the risks.
Preparation Method
Producing 4-Nitro-2-Methoxyaniline means lining up reactions with good precision. The common route starts with 2-Methoxyaniline, which undergoes controlled nitration—concentrated nitric and sulfuric acids handled under chilled conditions. The goal is to land the nitro group on the para position; too little cooling, or excess acid, and byproduct formation jumps. After nitration, neutralization and washing strip out acids; then, the product crystallizes as the temperature rises. Skilled processors watch for premature precipitation or over-nitration. Good batch practice stresses slow acid addition and thorough mixing to avoid localized overheating, both safety and yield considerations.
Chemical Reactions & Modifications
This compound enters a wide stage of organic chemistry, most notably in reduction reactions. Using reducing agents like iron filings with acid or catalytic hydrogenation, chemists turn the nitro group into an amine, opening up fresh building blocks for pharmaceuticals or specialty dyes. Electrophilic aromatic substitution joins the list, with methoxy and amino groups influencing substitution patterns. Under basic or acidic conditions, 4-Nitro-2-Methoxyaniline can take on further alkyl or acyl groups, producing derivatives tailored for research or industry. Some labs experiment with coupling reactions, linking this molecule to diazonium salts for azo dye synthesis—a classic strategy for generating vibrant organic colors.
Synonyms & Product Names
Across the chemical literature, labels for 4-Nitro-2-Methoxyaniline vary. Some catalogues call it p-Nitro-o-Anisidine; others list 2-Methoxy-4-Nitroaniline or simply use its systematic name. Chemical databases keep things clear with the CAS number, but in practice, product names depend on context—dye industry, pharmaceutical work, or academic research. Correct naming ensures that researchers order the right compound without risking mix-ups, especially given close relatives like 2-Nitro-4-Methoxyaniline or 3-nitro substitutes.
Safety & Operational Standards
Routine handling of 4-Nitro-2-Methoxyaniline keeps safety at the forefront. Both nitro and amino substituents come with concerns—skin and respiratory irritation, and over long exposure, possible mutagenic risks. Gloves, goggles, and local ventilation are standard in both labs and factories. Dust collection saves the lungs of workers and the integrity of other products. Disposal doesn’t head straight to the drain; methods tie into current local laws covering nitroaromatic waste, usually involving incineration with scrubbers or solvent extraction for recycling. Training refreshers and emergency spill drills work as real-world insurance for those who work with the compound every day.
Application Area
If you check into dye chemistry or pigment formulation, traces of 4-Nitro-2-Methoxyaniline surface in many recipes, especially for producing red and orange azo dyes. Specialty ink manufacturers prize its reactivity, helping produce colors that last through sunlight and repeated washings. Specialty chemical firms take advantage of the molecule’s framework for further functionalization, especially in creating intermediates for pharmaceuticals, antioxidants, or imaging agents. In organic labs, instructors and students use it in teaching reduction and substitution chemistry—its success or failure providing quick feedback on technique.
Research & Development
R&D pushes the boundaries of what this molecule can do. Newer efforts dig into selective catalytic reductions, hoping for cleaner conversions to diamines or other target molecules. Analytical chemists spend hours tracking minor impurities with HPLC or mass spectrometry, their goal to raise yields and bring consistency from batch to batch. Academia explores alternative nitration methods to curb hazardous waste, such as using ionic liquids or recyclable acids—less runoff, less environmental damage. Pharmaceutical groups screen new derivatives for antimicrobial or anticancer activities, sometimes finding surprising results from tweaking the methoxy or nitro positioning.
Toxicity Research
Toxicity remains a hard reality with many nitroaromatic compounds, and 4-Nitro-2-Methoxyaniline isn’t exempt. Animal studies highlight risks at moderate or high doses: liver stress, possible hemolytic activity, and in some cases, changes in blood chemistry. Regulatory reviews point out eye and skin irritation for handlers, so workplace limits stick close to minimal contact and strong PPE use. Not much makes it into the environment—disposal rules see to that—but chronic exposure often becomes an issue in production facilities more than downstream use. Over the years, researchers have developed more sensitive detection in water, air, and soil, expanding our knowledge of both acute and chronic exposure consequences.
Future Prospects
The chemical sector pushes for safer handling, greener synthesis, and broader applications all at once. Demand for vibrant, sustainable dyes grows alongside the call for less toxic alternatives, so research explores if modifications to the aniline skeleton produce brighter pigments without sacrificing safety. Advances in catalysis point toward less wasteful routes, sometimes even replacing sulfuric acid with milder or recyclable acids. As analytical tools catch up, manufacturers anticipate tighter product specifications and more comprehensive tracking. At the same time, computational chemists and pharmaceutical researchers peek at possible bioactive roles, like enzyme inhibition or drug precursor potential. Whether change arrives from environmental policy, lab innovation, or synthetic necessity, this molecule will keep evolving, shaping what comes next in both chemistry and the industries that depend on it.
What are the main applications of 4-Nitro-2-Methoxyaniline?
On the Front Lines of Dye Manufacturing
Anybody who’s ever watched textiles shift from drab white to punchy red knows the magic rests in chemical building blocks. 4-Nitro-2-methoxyaniline stands out as one of those behind-the-scenes agents. Chemical companies lean on this compound when developing azo dyes. These are the workhorses coloring everything from your favorite red T-shirt to certain plastics. The process starts with coupling—chemists link 4-Nitro-2-methoxyaniline’s amine group to an aromatic ring, laying the groundwork for a dramatic, stable color. This efficiency in producing vibrant, long-lasting shades keeps the compound in high demand at dye plants around the world.
Supporting Pharmaceutical Research
In drug development, flexibility matters. 4-Nitro-2-methoxyaniline offers a versatile toolkit for medicinal chemists. The nitro and methoxy groups invite selective reactions, making it easier to build complex compounds stage by stage. Take the hunt for anti-infective agents: researchers have used this molecule to design intermediates for antibiotics and antifungal agents. Its structure lets scientists tweak molecules to improve their medical properties or minimize toxic side effects. That chance to modify a building block, instead of starting from scratch, saves time and cuts costs in pharmaceutical pipelines.
Stepping Into Agrochemical Science
Farmers rely on chemicals to protect crops and boost yield, and companies compete to develop compounds that hit pests hard but break down quickly. Here, 4-Nitro-2-methoxyaniline plays a supporting role. It enters synthetic pathways toward herbicides and pesticides, especially where the goal is targeted function—like killing weeds but not harming corn. Researchers building new classes of crop protection agents prize its reactivity and structural features, enabling targeted modifications that enhance potency or selectivity. Its record in this field keeps expanding as companies respond to evolving regulatory and environmental challenges.
Challenges With Use and Safety
Everything useful carries some caution. 4-Nitro-2-methoxyaniline shows toxicity at certain exposures, which makes strict workplace safety essential. Experienced lab staff know to handle the substance in well-ventilated hoods and wear proper protection, because skin contact or inhalation can cause harm. Disposing of waste safely also takes planning, with local guidelines shaping every step. Industry’s responsibility depends not just on good chemistry, but on limits that safeguard both workers and water supplies. Year by year, better standards turn up—driven by real incidents and smart policies forced by experience.
Moving Toward Greener Chemistry
Many researchers want a future where brilliant dyes and effective drugs no longer burden rivers or workers. Green chemistry holds promise here. Chemistry teams look for alternative solvents that cut environmental impact, or reactions that skip harsh metals and high temperatures. Companies invest in processing methods that reclaim solvents and minimize any dangerous byproducts. These solutions don’t just deliver cleaner production—they shield communities and ecosystems that bear the brunt of chemical run-off and accidents. 4-Nitro-2-methoxyaniline offers industry genuine power, yet the push for safer processes and cleaner outputs has never been more relevant.
What is the chemical formula and molecular weight of 4-Nitro-2-Methoxyaniline?
Straight to the Science: What’s in the Bottle?
Ask anyone who’s had their hands on 4-Nitro-2-Methoxyaniline during a late-night lab session, and you’ll hear about the potent yellow powder with a precise fingerprint. The chemical formula stands clear: C7H8N2O3. That’s seven carbons, eight hydrogens, two nitrogens, and three oxygens joined in a tightly packed structure. Each of those atoms carries its weight, literally. Add them all together, and the molecular weight clocks in at 168.15 g/mol.
The Real Weight of a Number
This number isn’t just for show on a bottle. During synthesis, a few tenths of a gram off-target throws off an entire batch. Anyone scaling reactions for educational or industrial purposes watches these numbers closely. Even tiny differences impact melting points, reactivity, and, most importantly, safety. In one university synthesis project, we ran short of info, and trusting that “roundabout mass” led our reaction to flop. It cost us both time and money in wasted chemicals. It takes accuracy to avoid repeating mistakes, and that value ripples out into research outcomes and even product pricing later down the line.
Where Facts Turn Practical
Chemists, process engineers, and anyone invested in dyes or pharmaceuticals work under tight regulatory expectations. 4-Nitro-2-Methoxyaniline often appears in dye formulations. The blueprint—the formula and exact molecular weight—guides crucial calculations for mixing, quality control, and hazard assessment. An incorrect entry on a batch record doesn’t just slow things down. It could mean compliance issues, shipment rejection, or worse, unsafe material slipping through.
Health risks also ride on these details. The nitro group on this molecule can spur toxic effects if mishandled. Fact-based chemical identification is part of safe usage—no one wants uncertainty around compounds carrying risk. Safety data sheets reference formula and weight for this reason. Technicians use these facts to calibrate detection equipment, design neutralization processes, and limit exposure. There’s peace of mind knowing every milligram came from the right calculation.
Tackling the Ongoing Challenges
Errors rarely come from the math. They creep in through missed updates or vague documentation. Updates to chemical inventories, online databases, and safety protocols tend to lag or provide conflicting info. Lab teams and educators should insist on authoritative sources, such as the American Chemical Society or trusted materials handbooks. Nobody’s ever complained about too much clarity when safety and quality are on the line.
In one instance, access to fresh, accurate data helped a colleague avoid buying a mislabeled reagent. They caught an out-of-date entry in a supplier catalog, flagged it, and received corrected documentation. Collaborative vigilance—double-checking sources, keeping up with reference materials, and pushing suppliers for updated literature—pays off. I’ve made it routine to review chemical profiles at the start of each project, not waiting for a red flag mid-reaction.
Keeping Facts Front and Center
Chemical science moves forward on accurate, accessible information. The basics—a molecular formula and a molecular weight—carry substantial value. They link safe lab practice with regulatory compliance and operational success. By trusting reliable sources and paying attention to detail, everyone involved in working with specialty chemicals like 4-Nitro-2-Methoxyaniline keeps that work grounded in fact and safety.
What are the storage and handling precautions for 4-Nitro-2-Methoxyaniline?
Why 4-Nitro-2-Methoxyaniline Deserves Respect in the Lab
Plenty of folks who spend their days in labs already know the unwritten rules about handling chemical powders, especially the aromatic amines, but 4-Nitro-2-Methoxyaniline brings its own quirks. This compound, used in making dyes and pharmaceuticals, feels pretty straightforward on the surface—a yellowish powder with a slightly sweet smell—but a little complacency opens the door for accidents.
Simple Steps Make Big Differences
Small actions like controlling humidity keep stock fresh, and this one goes double in a humid climate. Anyone keeping an open jar on a bench for hours learns this lesson quickly. Leaving the jar exposed lets the powder clump up, and you experience a different level of frustration at your next batch. Sealed glass containers kept dry and cool, ideally below 25°C, save a lot of headaches. If you use a desiccator, silica gel packets work wonders at stopping the powder from taking on moisture. Light can cause slow degradation, so a cabinet out of direct light, or at least amber glass, cuts down on surprises.
Think Clean, Think Containment
Spills sneak up if you cut corners with transferring. Anyone who's worked with anilines knows how stains linger on hands and benches long after cleanup, and this one is no exception. Gloves offer real comfort—nitrile stands up well to the powder. Safety glasses stop dust from getting in your eyes and a dust mask in case you stir up a cloud. For larger weighing jobs, a balance enclosure or fume hood is worth the hassle. Breathing in even a little dust after a long day gets uncomfortable, especially as this chemical could cause irritation or have more lasting effects.
Why Labeling and Tracking Matter
Keeping a dated log of when you got each bottle and the state it's in pays off later. I’ve seen mix-ups lead to wasted experiments and lots of head-scratching. Clear labeling helps track age, and you can spot bottles starting to show strange odors or color changes. Even the sharpest chemists slip up if the shelf gets cluttered. In a shared lab, a simple logbook helps everyone avoid repeating mistakes or using spoiled material.
What to Do with Waste and Spills
Every seasoned lab tech eventually deals with a spill, sometimes when things get late or after a rushed transfer. Wiping the powder with bare hands isn’t wise. I find disposable wipes and immediate plastic bagging the safest way to handle contaminated material. Used gloves and wipes go in hazardous waste bins—not regular trash—as per local rules. Even small spills near sinks can enter water systems, so treating every cleanup as hazardous keeps the environment clean.
Health Hazards: Memories and Prevention
It’s easy to forget that repeated skin contact leads to rashes, or that breathing dust can irritate airways. I know folks who ended up with mild symptoms after underestimating the powder. Quick soap-and-water washes after handling, especially before eating or touching your face, staves off issues. Anyone feeling dizzy, lightheaded, or itchy needs to step outside and notify others. Quick response avoids big trouble.
Final Thoughts on Safe, Effective Handling
In my years around the bench, staying a little paranoid keeps you out of trouble. 4-Nitro-2-Methoxyaniline rewards those who treat every step—from delivery to disposal—as something that matters. Even tired eyes can spot risks when the storage and handling habits never slip.
Is 4-Nitro-2-Methoxyaniline hazardous or toxic?
Chemicals in Everyday Life
Most of us won’t ever see a bottle labeled “4-Nitro-2-Methoxyaniline” on a shelf at home, but it quietly supports several industries in the background. This substance isn’t a household name, yet it pops up in research labs, some dye work, and even pharmaceutical development. The important thing to keep in mind—no matter how obscure a chemical might sound—is safety.
Understanding Toxicity
Let’s not sugarcoat it: 4-Nitro-2-Methoxyaniline carries toxic risks. Data from the European Chemicals Agency and research articles indicate there’s solid evidence it can pose health hazards if not handled right. Skin contact, inhalation, or swallowing can make a person sick. Some studies note possible damage to blood, liver, and kidneys, along with other organ systems—especially if exposure happens often or in bigger doses than regulations advise.
During my own training in chemical safety, instructors always drove home that a material doesn’t have to be notorious to be dangerous. I recall a peer getting a mild rash after contact with a similar nitroaromatic compound; it threw a wrench in the day and led to more careful procedures. These compounds can sneak into the bloodstream through the skin or lungs faster than you might think.
Workplace Safety
The place where most trouble arises isn’t at home, but in a workplace that overlooks the details. Jobs that deal with dyes, pigments, or intermediates could bring workers close to 4-Nitro-2-Methoxyaniline. Regulations—like those from OSHA and Europe’s REACH—recommend gloves, goggles, and proper ventilation. Spill response plans become essential, not just good practice. It’s not an overreaction. Chronic exposure brings real health risks, including effects on blood (sometimes methemoglobinemia, a condition that affects the blood’s oxygen-carrying ability), and animal tests raise questions about cancer risk. Even if human evidence isn’t strong yet, waiting for more victims before acting seems irresponsible.
Environmental Concerns
Waste management stands out as another challenge. Improper disposal can let these chemicals seep into water or soil, with potential harm for plants, animals, and communities downstream. Manufactured chemicals like 4-Nitro-2-Methoxyaniline don’t just vanish after pouring them down a drain. Regulatory programs push for neutralization and secure storage of byproducts, though mistakes still happen—usually from shortcuts or ignorance, not malice.
Responsible Handling
It always comes down to responsibility. Training workers makes a real difference, so does clear labeling and regular safety audits. From my time around research labs, the best run facilities use every opportunity to remind staff how to respect even the “uncommon” chemicals. Anything less risks health, lawsuits, and trust.
Substitution should be on the table as well. Chemists constantly search for alternatives that meet the same need but carry fewer health or environmental risks. Where that can’t happen, tighter safety measures deserve a bigger push. Manufacturers do best by investing in containment systems, monitoring tools, and backup plans for emergencies.
Looking Ahead
Toxic compounds like 4-Nitro-2-Methoxyaniline highlight how hidden threats can slip into supply chains or labs. Keeping workers safe, paying attention to disposal, and staying alert for better alternatives isn’t just about ticking boxes. It protects lives and prevents headaches. As we keep inventing new materials, respect for the chemistry—and the people exposed to it—can’t be an afterthought.
What is the purity grade and available packaging sizes for 4-Nitro-2-Methoxyaniline?
Understanding Purity Standards
Anyone working with specialty chemicals knows that purity isn’t just a marketing claim—it’s a real concern that affects the reliability of every experiment and process. 4-Nitro-2-Methoxyaniline, with its role as an intermediate in the synthesis of dyes, pharmaceuticals, and agrochemicals, gets a lot of attention for purity. From my own lab days, nothing derails a project faster than an unexpected impurity. For 4-Nitro-2-Methoxyaniline, you typically see a purity grade of at least 98%. This figure isn't plucked out of thin air—manufacturers base it on chromatographic and spectroscopic analysis, usually through HPLC and NMR. High purity ensures researchers avoid side reactions and get predictable results, both on a small scale in academia and in the controlled chaos of manufacturing plants.
Historically, chemical suppliers didn’t always provide detailed impurity profiles. But increased demand for transparency, along with regulatory scrutiny, pushed companies towards better documentation. It’s not just about ticking a box—the presence of trace contaminants can badly affect final product safety, efficacy, and even compliance with regional laws. Labs that stick to chemicals above 98% for intermediate synthesis tend to report fewer issues scaling up and lower rates of process failures down the line.
Available Packaging Sizes
Every chemist I know jokes about buying “just enough” for the project, but that sweet spot between cost and convenience comes down to packaging size. 4-Nitro-2-Methoxyaniline is sold in bottles as small as 5 grams up to bulk drums that hold 25 kilograms. Academic and research labs lean towards 5g, 10g, or 25g vials—no point in spending on material you’ll never use. Commercial operations, especially in dye or drug manufacturing, go for 1kg, 5kg, or 25kg drums.
I remember more than one project going off budget because someone thought a 100g bottle would last forever. Smaller sizes offer convenience and help manage shelf life, since degradation can creep in over time even with proper storage. On the flip side, bulk buyers negotiate larger packs both to keep prices down and limit the handling of multiple containers. Less handling means less risk of contamination and spillage—two words that drive lab managers up the wall.
Why These Details Matter
Lab safety officers and quality assurance teams keep a close eye on these specifications for a reason. Pure chemicals limit variables in both R&D and finished product batches, so companies avoid costly recalls. Regulatory agencies, including FDA and EMA, expect traceability back to source materials, so records on purity grades and packaging sizes are part of standard audits. I’ve seen companies develop longer-lasting supplier relationships by choosing sources that disclose both impurity profiles and flexible packaging.
Shaping a Smarter Market
The chemical marketplace has shifted away from one-size-fits-all. Researchers, buyers, and manufacturers have grown more vocal about their needs for high purity and variety in packaging. Direct conversations with suppliers help clarify whether a lot matches their standards, and some even ask for a full impurity breakdown before committing. Labs that keep good records end up saving time and money, because they can quickly retrace any issues or adjust orders based on real consumption data.
The bottom line: buying 4-Nitro-2-Methoxyaniline means paying close attention to both purity and packaging. These aren’t minor details—they shape project timelines, quality, safety, and, ultimately, trust in a supplier.
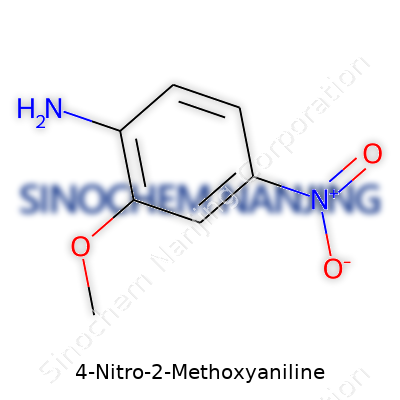
| Names | |
| Preferred IUPAC name | 4-nitro-2-methoxybenzenamine |
| Other names |
2-Methoxy-4-nitroaniline 4-Nitro-o-anisidine 2-Anisidino-4-nitro 2-Methoxy-p-nitroaniline |
| Pronunciation | /ˈfɔːr ˈnaɪtroʊ tuː mɛˈθɒksi əˈnɪliːn/ |
| Identifiers | |
| CAS Number | 698-96-4 |
| Beilstein Reference | 1209504 |
| ChEBI | CHEBI:21325 |
| ChEMBL | CHEMBL3185391 |
| ChemSpider | 128626 |
| DrugBank | DB08347 |
| ECHA InfoCard | ECHA InfoCard: 100.007.428 |
| EC Number | 612-207-00-5 |
| Gmelin Reference | 108481 |
| KEGG | C14361 |
| MeSH | D000077229 |
| PubChem CID | 145111 |
| RTECS number | BY9600000 |
| UNII | WF7R60G3K8 |
| UN number | UN1661 |
| CompTox Dashboard (EPA) | DTXSID3049156 |
| Properties | |
| Chemical formula | C7H8N2O3 |
| Molar mass | 153.14 g/mol |
| Appearance | Yellow powder |
| Odor | Odorless |
| Density | 1.33 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 1.08 |
| Vapor pressure | 2.55E-4 mmHg at 25°C |
| Acidity (pKa) | pKa = 2.30 |
| Basicity (pKb) | 10.24 |
| Magnetic susceptibility (χ) | -52.2×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.658 |
| Dipole moment | 3.98 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 156.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -55.2 kJ mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -1784 kJ·mol⁻¹ |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause respiratory irritation |
| GHS labelling | GHS02, GHS07, GHS09 |
| Pictograms | GHS07, GHS09 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H317, H334, H341, H351, H372 |
| Precautionary statements | P261, P264, P270, P271, P272, P273, P280, P301+P317, P302+P352, P304+P340, P308+P311, P330, P363, P405, P501 |
| NFPA 704 (fire diamond) | 1-2-1-🌟 |
| Flash point | 113°C |
| Lethal dose or concentration | LD50 (oral, rat): 288 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 315 mg/kg |
| NIOSH | MW3675000 |
| PEL (Permissible) | PEL (Permissible): Not established |
| REL (Recommended) | 10 mg/m3 |
| Related compounds | |
| Related compounds |
2-Methoxyaniline 4-Nitroaniline o-Anisidine p-Nitroanisole 2-Methoxy-4-nitrophenol |

