Commentary on 4-Nitro-2-Aminophenol: Insights, Uses, and the Road Ahead
Historical Development
Organic chemists began exploring compounds like 4-Nitro-2-Aminophenol during the late nineteenth century, as the pharmaceutical and dye industries searched for novel building blocks. The nitroaniline family caught early interest because their ease of synthesis and vibrant chromatic properties drove both laboratory experimentation and eventual factory-scale adoption. These efforts helped fuel Europe’s fine chemicals revolution and shaped the foundation for modern medicinal chemistry. Getting this far, however, took plenty of trial and error—synthetic strategies improved incrementally, with small research teams digging into what worked at the bench and in manufacturing. Those involved learned quickly that safety, consistency, and scalability could not take a back seat—even minor mishaps carried risks that put entire projects on hold.
Product Overview
The reputation of 4-Nitro-2-Aminophenol now stretches from academia to industry. Researchers in pigment chemistry and pharmaceutical sciences choose this compound not just for its chemical versatility, but also for its reliability in scaled processes. It carries the appeal of a relatively low-cost intermediate, with production methods established well enough for predictable quality across suppliers. This reliability is essential; it means that chemists and manufacturing engineers can focus on innovation instead of troubleshooting purity or consistency issues with each new batch.
Physical & Chemical Properties
On the bench, 4-Nitro-2-Aminophenol shows up as a yellow-to-brown crystalline powder with a melting point typically in the 138–142°C range. It doesn’t dissolve easily in water, but goes into organic solvents such as ethanol and acetone, which lets users tailor their processes without battling solubility headaches. The molecule’s functional groups—a nitro group opposite an amine and hydroxyl group on a benzene ring—create interesting reactivity. Oxidative conditions transform it, and reductions can access valuable amine derivatives. It’s worth noting that the nitro group, while valuable, brings with it a potential for sensitization, so protective gloves and ventilation become critical in labs and plants handling decent quantities.
Technical Specifications & Labeling
Producers label 4-Nitro-2-Aminophenol with clear technical specifications: assay (typically >98%), melting point, loss on drying, and presence of residual solvents find their way onto certificates of analysis. Purity tracking isn’t just a formal exercise—it reduces batch-to-batch guesswork and improves process validation. Proper labeling helps handlers stay informed about hazard classifications. With increasing scrutiny from regulators, GHS-compliant labels, safety data, and best-practice storage instructions keep operations running smoothly, minimizing both operational mistakes and regulatory fines.
Preparation Method
Lab-scale syntheses of 4-Nitro-2-Aminophenol have been boiled down to well-documented routes, with the nitration of 2-aminophenol standing out as a workhorse method. The process typically uses concentrated nitric and sulfuric acid, followed by careful temperature control to avoid over-nitration or degradation. Batch operators monitor for exotherm and gas release, since mistakes can escalate quickly in these conditions. Larger synthesis campaigns sometimes adopt reduction strategies, converting 4-nitro-2-nitrophenol by catalytic hydrogenation. At any scale, the message remains clear: good technique equals good outcomes, and a haphazard approach risks both yield and safety.
Chemical Reactions & Modifications
Reactivity is where 4-Nitro-2-Aminophenol shines for chemists. Its amine and hydroxyl groups open routes to acylation, diazotization, and coupling reactions, writing it into dye synthesis protocols as fast as into pharmaceutical intermediates. Reductive chemistry transforms it into various aminophenol isomers, each carrying potential downstream value. For instance, applying diazotization enables the synthesis of azo dyes, which remain staple materials for textile coloration. In medicinal chemistry labs, modifications of the basic scaffold generate leads against bacterial infections or enzyme inhibitors. By approaching reactions with the right reagents and handling, researchers can coax this molecule into new frameworks without constantly battling side reactions or excessive byproducts.
Synonyms & Product Names
No small molecule reaches the shelves without picking up a variety of aliases. In various catalogs and academic articles, 4-Nitro-2-Aminophenol turns up as 2-Amino-4-nitrophenol, C.I. 37105, or Para-Nitro-O-Aminophenol. These names reflect both the commercial domains using the compound—textiles, specialty pigments, pharmaceuticals—and the shifting language conventions of chemical cataloguing. For buyers and handlers, the trick is to check CAS numbers as a way of confirming the order matches the intended structure; confusion between regioisomers or differently substituted aminophenols could set back an entire line of R&D.
Safety & Operational Standards
Daily handling of 4-Nitro-2-Aminophenol requires more than compliance paperwork—it calls for a culture of active attention. The compound classifies as harmful if swallowed and potentially irritating to skin, eyes, and respiratory systems. GHS pictograms and precautionary statements aren’t decorative; in a practical sense, they help teams avoid complacency. Glove standards, local exhaust ventilation, and spill protocols address both chronic and acute exposures. On the plant floor, fire risk from fine powders and solvents factors into both process design and emergency preparedness. Regular training makes the difference between a safe workplace and a facility where mistakes pile up. From experience, nothing beats having teams who know both the black-and-white policies and the practical skills needed to respond when a drum leaks or a flask cracks.
Application Area
Industry has put 4-Nitro-2-Aminophenol to good use primarily in the world of dyes and colorants. Its structure lends itself to the formation of various azo and anthraquinone-based pigments, which hold up well in textiles and coatings. Research outfits leverage its bifunctional reactivity to build libraries of small molecules, ranging from antimicrobial agents to process intermediates for active pharmaceutical ingredients. Some specialty hair colorants count on the unique property of this molecule to create specific shades that won’t bleach out easily, while others have tested it for use in photographic developers. With every use case, demands on purity and reactivity have refined both marketplace quality expectations and the capabilities of chemical distributors. Having handled pigments applications, I’ve seen how even minor tweaks to feedstock quality can shift the tone or durability of end products, which circles back to the importance of tight technical controls throughout the supply chain.
Research & Development
Teams are not content to treat 4-Nitro-2-Aminophenol as a finished story. Ongoing research explores greener synthesis methods, aiming to replace nitric acid with milder, lower-impact reagents. New catalytic systems promise less waste and better selectivity, and early results look promising for scale-up. Analytical chemists push for ever-tighter control of trace impurities, using HPLC and mass spectrometry to pick out contaminants that old methods missed. There’s also interest in modifying the molecule to tune its photochemical and electronic properties, opening potential for novel electronic materials and organic sensors. For those at the research coalface, the work remains challenging and rewarding; iterating on reaction routes and product testing may not make headlines, but it slowly shifts both science and industry forward.
Toxicity Research
Evaluating the safety profile of 4-Nitro-2-Aminophenol takes long-term vigilance. Acute toxicity tests in animals have pointed out moderate toxicity via oral exposure, while longer-term studies suggest risks to liver, kidneys, and blood-forming organs when doses climb. Regulatory data recognize it as an irritant, and repeated exposure can trigger sensitization in some people. Environmental researchers worry about persistence and possible impact on aquatic life, since nitroaromatic compounds can resist natural breakdown. Better analytical methods let environmental labs monitor water supplies and effluents for trace contaminants, but society benefits most from a blend of clear labeling, adequate safety gear, and process engineering that keeps exposure minimal both in the plant and downstream.
Future Prospects
Looking ahead, 4-Nitro-2-Aminophenol stands at an interesting crossroads. The growing demand for sustainable manufacturing pressures suppliers to develop pathways that curb waste, cut solvent use, and ramp up safety even further. Synthetic biology may offer breakthroughs, using enzyme-based transformations to handle previously harsh chemical steps under mild and water-based conditions. With advances in automation and digital process control, precision manufacturing can keep quality levels high and incidents low. For applied scientists, new domains may emerge as organic electronics and sensor technologies reach market maturity. The future will depend both on technical innovation and on the kind of unglamorous, detail-focused work that has always kept the chemical supply chain safe, efficient, and ready for whatever tomorrow’s demand brings.
What is 4-Nitro-2-Aminophenol used for?
What’s Behind This Chemical?
People seldom talk about 4-Nitro-2-aminophenol, even though it’s found in plenty of everyday products. This compound moves quietly through industries. Its structure makes it valuable in chemical manufacturing, but it’s not the sort of thing most would recognize on a list of ingredients. Yet, a closer look at its role in dyes and colorants exposes a far-reaching impact many overlook.
Industrial Value and Everyday use
Hair dye manufacturers rely on 4-Nitro-2-aminophenol for its rich color effects. Professionals in the cosmetic world chase after shades that last through washes and sunlight—this chemical offers that depth. You’ll find it helping create deep reds and purples in hair colors, made to satisfy folks who want their look to stick. Textile factories see similar value; workers dyeing fabrics need chemicals like this to bind pigment tightly to fibers, creating shades that catch the eye and don’t bleed after laundry days.
Pharmaceutical developers sometimes use this compound to build more complex molecules for medications. The path from raw material to finished drug draws on chemicals like this one, letting researchers tweak molecular arrangements for better treatments. Anyone taking medication shaped through these labs relies, even indirectly, on specialty chemicals playing small but critical parts.
Environmental and Health Questions
No point dancing around concerns, either. Factories using 4-Nitro-2-aminophenol need to keep waste management tight. The byproducts of dye production can harm water supplies if not managed. Years back, some dye plant towns reported higher pollution levels, and scientists tied runoff to local river damage and sickened fish populations.
Workers handling this chemical face their own risks. Extended skin contact often irritates, and without masks, powder or fumes cause trouble in the lungs over time. Health and safety regulators developed guidelines that stress good ventilation and protective gear, but smaller operations sometimes cut corners. Stories crop up of workers in textile hubs dealing with rashes or coughing, usually in places where gloves and respirators get ignored.
Fact-Based Steps Toward Safer Use
Switching to greener chemistry in dyes and cosmetics makes sense. Research groups keep experimenting with plant-based alternatives, and some cosmetic brands have phased out the more hazardous synthetic dyes. Success here depends on new tech that offers the same durability and vibrance without the toxic baggage. It’s tough, since customers expect hair color to last for weeks or clothes to hold color for years.
Stronger safety culture changes things, too. Factories providing fresh gloves and keeping exhaust fans in top shape make a real difference. Regular testing of local waterways around chemical plants helps catch trouble before it escalates. Clean-up tech—like filtration systems or chemical neutralizers—acts as a first responder when spills happen. I’ve seen labs transform after adding simple spill response kits, reducing medical complaints instantly.
4-Nitro-2-aminophenol isn’t going away overnight. It keeps factories running, products colorful, and research ticking forward. Smart use—rooted in experience, tough regulations, and honest follow-up—creates a future where people can enjoy bold colors and strong medicines without gambling on safety. It’s a story I see echoed in science, industry, and neighborhoods alike.
What are the safety precautions for handling 4-Nitro-2-Aminophenol?
A Closer Look at Risks
4-Nitro-2-Aminophenol doesn’t sit on most people’s shopping lists, but anyone in a chemical lab or industrial setting might run into it. This yellowish powder, often used in dyes or lab tests, does pack a punch when it comes to health risks. Skin stains that won’t scrub off and fumes that sting the eyes are the easy signs it’s best to show some respect.
What Direct Contact Feels Like
My years around small-scale research labs taught me no one forgets the first chemical splash that tingles—or worse, burns—on bare skin. Gloves matter as much as a good lab partner. With 4-Nitro-2-Aminophenol, full nitrile gloves work best, since latex can break down with some solvents. A chemical-resistant lab coat and splash-proof goggles aren’t overkill—they stop the burns and avoid that miserable dry, red skin. Open wounds or cracked knuckles? An unprotected patch of skin works like a sponge for this powder, so applying hand lotion for a barrier might save trouble.
The Air We Breathe
Volatile compounds in a closed room leave a smell you remember long after a shift ends. Respirators aren’t just for show. An N95 mask sometimes seals out dust, but if you see fine particles billow, a full chemical cartridge mask works better. Fume hoods, the humming machines with glass windows, make mistakes less likely. A fume hood draws fumes away from your face; I’ve watched colleagues cough their way through days because they skipped this step. Clean, strong ventilation doesn’t need to be fancy, just reliable.
Preventing Accidents with Simple Routines
Spills seem rare—until the day a bag rips or a scale gets knocked off the bench. Keeping absorbent powder and a step-by-step guide posted near lab benches reduces panic. Immediate containment with spill pads and scooping up with plastic, not metal, shovels keeps static sparks out of the equation. Double-bagging waste, followed by labeling it for hazardous pickup, saves headaches later.
Don't Eat Where You Work
Lunch in the lab once seemed like a way to save time. One quick sandwich later, my stomach twisted for hours. Food, drinks, and lab benches never mix. Even the most harmless-looking powders can find their way onto bread or fruit. No water bottles close by, unless sealed. Washing hands with soap—it’s a small price for peace of mind.
Training and Trust
Places that handle chemicals feel safer when everyone knows the rules and trusts each other to speak up. Regular, hands-on drills for spill response help even the most seasoned workers remember not to cut corners. I recall my supervisor stopping work mid-lab when someone rushed through cleanup. He valued people over production. This rubbed off on the team—nobody wanted to be the reason for the next accident report.
Building Layers of Protection
Gloves, goggles, a fume hood, and a clear mind—these layers catch what slips through. If supply chains threaten to run thin on safety supplies, management should find alternatives fast. Good chemistry can’t happen without safety. Each missed step in handling this powder risks expensive medical visits, lost work time, and damaged trust between coworkers. Careful handling, honest communication, and strong habits go further than any shiny procedure manual.
What is the chemical formula and molecular weight of 4-Nitro-2-Aminophenol?
Chemical Structure and Formula
4-Nitro-2-aminophenol has the chemical formula C6H6N2O3. It’s a benzene ring with a nitro group (-NO2) at the 4-position and an amino group (-NH2) at the 2-position, plus a hydroxy group (-OH) on the ring. These groups give the molecule its unique properties, both in the lab and in manufacturing. The molecular weight of this compound clocks in at 154.13 g/mol, a figure you’ll find referenced in analytical chemistry tables, safety documents, and lab catalogs everywhere.
Practical Importance and Everyday Impact
This compound isn’t some obscure reagent sitting on a dusty shelf. Chemists often turn to 4-nitro-2-aminophenol when they want to create dyes, especially those used in hair color formulations. Its functional groups make it react in predictable ways. That predictability opens up possibilities for creating vivid, lasting colorants. I remember colleagues in industrial chemistry labs spending hours tweaking formulas with this compound, hunting for that perfect balance between color strength and stability.
Safety and Handling Concerns
4-Nitro-2-aminophenol isn’t just about chemistry—handling it brings real-world safety concerns. As a nitro compound, it’s not especially volatile, but spills and contact demand attention. The nitro group and amino group can interact with skin, potentially causing irritation or allergic responses. Long-term exposure to aromatic amines and nitro compounds has raised flags in toxicology reports, so gloves, goggles, and proper ventilation make their way into any safety checklist.
Environmental and Health Ties
Compounds like these occasionally show up in discussions about water pollution and environmental persistence. Their stability in water and difficult-to-break-down nature means that careless disposal can create issues for local ecosystems. A few years back, a colleague worked with a small municipal lab testing water for dye contaminants. They found trace amounts of nitrogen-rich chemicals from hair dye factories in wastewater—a clear signal that better waste management is needed wherever these molecules turn up.
Solutions: Better Practices and Forward Thinking
It’s important not to look away from these challenges. Waste treatment innovation makes a big difference. Installing activated carbon filtration and advanced oxidation processes in manufacturing plants stops most of these compounds before they ever leave a facility. Labels on consumer products now often explain safe disposal, nudging users to keep chemicals out of drains.
Tighter regulation also helps. Agencies now ask for environmental impact data before approving new chemicals for large-scale use. Research groups focus on designing molecules that break down more safely once their job’s done. More funding for green chemistry programs, and greater industry transparency, can cut down on chemical pollution—even as products continue to improve. As someone who values both chemistry and clean water, seeing these shifts makes a difference.
Final Thoughts
4-Nitro-2-aminophenol, with formula C6H6N2O3 and weight 154.13 g/mol, carries both opportunity and responsibility. Understanding its chemistry lets us innovate, but awareness and careful stewardship keep communities and ecosystems safer. The science matters, but so does every decision along the way—from the bench to the environment outside.
How should 4-Nitro-2-Aminophenol be stored?
Why Storage Choices Matter
In any chemistry lab, safety habits can outlast a single experiment. A compound like 4-Nitro-2-Aminophenol can trip up even experienced hands if kept alongside the wrong chemicals, or forgotten in a damp drawer. I’ve seen a few storerooms where the right storage practices saved the day—no panic, no wasted time, no injuries. That stuff matters, especially with substances carrying real risks.
Understanding Hazards
4-Nitro-2-Aminophenol doesn’t hide its danger. With a sharp yellow hue, it stands out on the shelf. Skin contact can trigger irritation. It can spark harmful reactions with strong oxidizers, or release hazardous gases under the wrong conditions. Reliable sources like PubChem and the NIOSH Pocket Guide point to respiratory irritation and headaches from extended exposure. Handling safety depends less on elaborate protocols and more on attention to routine detail.
Setting Up a Safe Storage Area
Temperature makes a difference with this chemical. Store at room temperature, in a spot that stays cool and dry. I’ve worked in labs where a single air conditioning failure made an entire cabinet of sensitive compounds questionable. Humidity creeps in, and with it comes clumping, caking, or unwanted chemical changes. Moisture in the air can encourage 4-Nitro-2-Aminophenol to degrade or even react with other stuff, which is just asking for trouble. A climate-controlled stockroom can avoid these headaches, and closed, well-labeled containers ensure the compound doesn’t end up in the wrong hands.
Keeping Incompatibles Separate
This isn’t a compound to throw in with random supplies. Strong oxidizers, acids, and bases must never sit close by. A former coworker once stacked incompatible chemicals on a single shelf to save time—one spill later, and the whole lab needed an evacuation. That experience sticks with you. Segregating 4-Nitro-2-Aminophenol from anything reactive is not paranoia; it’s basic risk management. Flammable cabinets or those lined with inert materials do the trick here.
Leakproof, Labeled Containers
Leaky jars or half-labeled bottles send the wrong message about lab culture. I always use containers with tight lids made out of materials that show no fancy reactions. Polyethylene or glass bottles both earn trust—no soft plastics or metal cans that might corrode or crack. Every bottle needs a solid, chemical-resistant label with the full name, hazard pictograms, and the date it entered the storeroom. If something looks off, I’d rather toss it than gamble with a possible contaminant or degraded sample.
Access Control and Awareness
It helps to know who touches what. Centralize access for staff trained on the compound’s risks. Limit distractions in storage spaces. A brief log entry can mean the difference between easy tracking and a wild goose chase if something turns up missing or off-color. Bring this to life with regular audits. Build a space where every team member understands what safe storage looks like and correct habits become second nature, not afterthoughts. A couple of conversations per month beat a mountain of paperwork after an avoidable accident.
Disposal—Don’t Leave it for Later
Old or unneeded 4-Nitro-2-Aminophenol should not gather dust. I set up a system where out-of-date or excess material moves into hazardous waste routes without delay. This decision stops clutter and cuts the odds of accidental misuse.
Making It Stick
Smart storage for chemicals like 4-Nitro-2-Aminophenol doesn’t come from fear. It stems from a respect for the risks and respect for the people who might clean up after someone else. Every choice about containers, temperature, or audit rhythms matters. In this field, solid habits keep a team out of trouble—and that’s a lesson I won’t forget.
What is the CAS number of 4-Nitro-2-Aminophenol?
What Is the CAS Number for 4-Nitro-2-Aminophenol?
Every chemical substance you come across in a lab or on a product label comes with a unique identifier known as the Chemical Abstracts Service number, or CAS number. For 4-Nitro-2-Aminophenol, this sequence is 99-57-0.
CAS Numbers Build Trust in Science and Industry
In my years in research, checking a CAS number often saved me from buying the wrong chemical. CAS numbers cut through the noise of regional names, nicknames, and even sloppy handwriting. Nobody wants a scenario where a mislabeled chemical creates hazards or wrecks an expensive experiment. Accuracy matters—one digit off, and you’re dealing with a completely different compound.
Why Is 4-Nitro-2-Aminophenol Noteworthy?
This particular molecule finds its way into several processes, especially in the dye industry. Manufacturers rely on its intense color and stability when formulating products for textiles or hair colors. Environmental scientists also keep an eye on it, since it may show up as a contaminant in wastewater streams. Regulators searching for proper data point to CAS number 99-57-0 when shaping safety protocols or exposure limits. Confusion between similar-looking names leads to real-life mistakes, so the CAS system forms the backbone of chemical management.
Risks and Responsibility
Some older industrial plants still operate with paper logs or hand-typed spreadsheets. Missing a CAS number, especially with lookalike names, places workers in danger and increases corporate liabilities. Checks on the chemical’s exact identity let managers handle storage, transportation, and disposal more carefully. Non-compliance leads straight to fines, or worse, news headlines. As someone who has audited chemical inventories, one misplaced identifier turns routine safety checks into scavenger hunts.
Improving Identification Practices
Manufacturers and distributors moving chemicals across borders find the CAS system indispensable. It cuts down on translation hiccups and helps customs officials check for banned or restricted substances. Building a culture of accuracy starts at the education level. Training chemists and warehouse staff to check CAS numbers by habit lowers costs related to mix-ups and emergencies. Reliable digital inventories backed with barcode scanning or RFID tags make tracking easier than any handwritten list ever could.
Solutions for Reducing Chemical Confusion
Education stands out as the strongest solution. Training sessions for new hires about the importance of CAS numbers pay off quickly, no matter the scale of the operation. Software upgrades offer another boost. Digital systems flag discrepancies and prompt a second look before a chemical ships, mixes, or disposes. Labeling improvements play a practical role—large, clear CAS numbers on every container banish ambiguity from the shelf.
Conclusion
Looking at the case of 4-Nitro-2-Aminophenol, attention to detail shapes safe, efficient, and ethical work. A unique number like 99-57-0 makes all the difference, helping everyone from students to seasoned chemists avoid error and keep operations humming along smoothly.
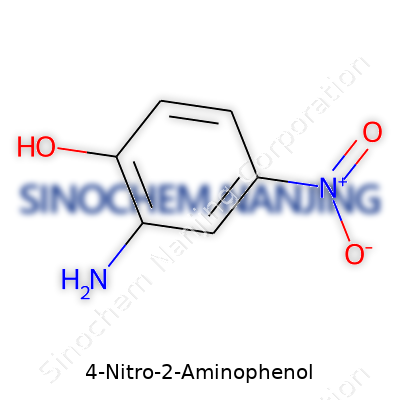
| Names | |
| Preferred IUPAC name | 4-nitro-2-aminophenol |
| Other names |
2-Amino-4-nitrophenol 4-Nitro-o-aminophenol p-Nitro-o-aminophenol 4-Nitro-2-hydroxyaniline 2-Hydroxy-5-nitroaniline |
| Pronunciation | /ˈfɔːr ˈnaɪtroʊ tuː əˈmiːnəʊfəˌnɒl/ |
| Identifiers | |
| CAS Number | 99-57-0 |
| Beilstein Reference | 1207930 |
| ChEBI | CHEBI:51947 |
| ChEMBL | CHEMBL16386 |
| ChemSpider | 15423 |
| DrugBank | DB03248 |
| ECHA InfoCard | DTXSID2041652 |
| EC Number | 226-285-3 |
| Gmelin Reference | 84113 |
| KEGG | C05608 |
| MeSH | D009633 |
| PubChem CID | 91481 |
| RTECS number | BY8400000 |
| UNII | 869S2Z6SXX |
| UN number | UN2662 |
| CompTox Dashboard (EPA) | DTXSID4021266 |
| Properties | |
| Chemical formula | C6H6N2O3 |
| Molar mass | 138.12 g/mol |
| Appearance | yellow to brown powder |
| Odor | Odorless |
| Density | 1.429 g/cm³ |
| Solubility in water | Slightly soluble in water |
| log P | 0.33 |
| Vapor pressure | 0.0000114 mmHg (25°C) |
| Acidity (pKa) | 7.2 |
| Basicity (pKb) | 9.38 |
| Magnetic susceptibility (χ) | -53.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.735 |
| Viscosity | Viscosity: 2.74 cP (50°C) |
| Dipole moment | 3.9094 Debye |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 110.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −68.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1944 kJ/mol |
| Pharmacology | |
| ATC code | D11AX11 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes serious eye damage, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07,GHS09 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | Precautionary statements: "P261, P280, P305+P351+P338, P337+P313, P501 |
| NFPA 704 (fire diamond) | 2-3-1-OX |
| Flash point | 174°C |
| Lethal dose or concentration | LD50 oral rat 282 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 282 mg/kg |
| NIOSH | NA052 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 4-Nitro-2-Aminophenol: Not established |
| REL (Recommended) | 0.1 mg/m³ |
| IDLH (Immediate danger) | NIOSH: Unknown |
| Related compounds | |
| Related compounds |
2-Nitro-4-aminophenol 2-Amino-5-nitrophenol 4-Nitrophenol 2-Aminophenol 4-Aminophenol 2-Nitrophenol |

