4-Nitro-1,2-Xylene: Commentary on Its Development, Properties, and Prospects
Historical Development
In the early days of aromatic compound chemistry, researchers turned to methylated benzenes as a way to expand both dye and pharmaceutical synthesis. The discovery of nitration routes for compounds like 1,2-xylene made it possible to produce a family of downstream chemicals, including 4-nitro-1,2-xylene. Nitration technology moved forward through the nineteenth century, mainly thanks to pioneers in European chemical industries who worked to transform abundant coal-tar derivatives into useful products. The ease with which aromatic rings accept substituents made xylene derivatives one of the backbones of industrial chemistry. As wartime needs increased demand for explosives, chemists closely studied nitroaromatics for their energy content and chemical reactivity, laying the groundwork for a generation of technical and commercial processes centered around nitro-substituted benzenes and xylenes.
Product Overview
4-Nitro-1,2-xylene stands out as a light yellow crystalline solid, mainly valued as an intermediate in fine chemical manufacture. Its distinct molecular arrangement, with two methyl groups at the ortho positions and a nitro group at the para position, results from precise control of reaction conditions during nitration. Specialty chemical suppliers often reference it by multiple synonyms, and researchers have spent decades developing selective preparation methods to maximize the para-nitro isomer yield over other possible nitroxylenes. Laboratories and manufacturers use it both as a building block and as a research staple to access a wide array of functionalized aromatics.
Physical & Chemical Properties
The physico-chemical profile of 4-nitro-1,2-xylene includes a melting point around 48–51°C and a boiling point in the 295–297°C range. Its modest solubility in water, higher solubility in organic solvents, and pale-yellow coloration are direct consequences of its aromatic and nitro functionalities. Unlike its parent xylene, the nitro group decreases electron density in the ring, modifying both the reactivity and the toxicity profile of the compound. This feature becomes important in handling and downstream transformations, as different reagents interact with the electron-poor aromatic system more selectively than with electron-rich methylbenzenes.
Technical Specifications & Labeling
Chemical suppliers frequently specify assay, melting range, and impurity profile to guarantee product reproducibility. Typical assay values exceed 97%, while allowable levels of 1,2-xylene, other nitroxylenes, and trace water are tightly controlled through routine gas chromatography and Karl Fischer titration. Labels include standard hazard pictograms for flammable solids, a precaution that reflects vapors generated at elevated process temperatures. Adequate labeling and material safety data sheets are not mere bureaucratic hurdles—anyone who’s spilled even a small amount in a poorly vented lab remembers the nose-stinging effect and yellow stains that linger in porous materials.
Preparation Method
Industrial-scale synthesis relies on nitrating 1,2-xylene using a mixture of concentrated nitric and sulfuric acids. Older protocols led to isomer mixtures, but advances in temperature control, acid ratios, and post-reaction separation techniques have allowed commercial plants to boost para-selectivity. Careful extraction and crystallization from non-polar solvents yield the desired 4-nitro isomer in purities fit for further transformation. Neither academic synthesis nor large-scale manufacture escapes the need for meticulous waste acid neutralization and handling, given that nitrogen oxides and spent acid mixtures present both environmental and workplace hazards.
Chemical Reactions & Modifications
Chemists often reduce 4-nitro-1,2-xylene to access 4-amino-1,2-xylene or transform it through nucleophilic aromatic substitution due to the activating effect of the nitro group. These derivatives serve as stepping stones for heterocycle formation, agrochemical development, and advanced pharmaceutical synthesis. The methyl positions, activated by the electron-withdrawing effect of the nitro group, also allow for selective bromination or sulfonation, expanding the library of downstream chemicals available from a single parent structure. Reactivity studies report that substitutions preferentially occur at sites determined by both resonance and inductive effects, insights that continue to inform process improvements today.
Synonyms & Product Names
Industry and regulation reference this compound under several names, such as 4-nitro-o-xylene and 1,2-dimethyl-4-nitrobenzene. These synonyms, along with various registry numbers (including those used by regulatory agencies and safety compliance databases), support clear communication across borders and enable buyers, regulators, and emergency responders to identify risks and properties without confusion. Mistakes in chemical nomenclature once led to costly errors—mislabeling the nitro position or confusing isomers can result in failed syntheses, cross-contamination, or violations of chemical safety law.
Safety & Operational Standards
Handling 4-nitro-1,2-xylene demands strict adherence to chemical hygiene protocols. Personal experience says never skimp on protective gloves or splash-resistant goggles; despite a lack of acute volatility at room temperature, the substance stains skin and irritates mucous membranes. National and international guidelines recommend sealed systems for large-volume transfers, rigorous air monitoring, and controlled incineration or chemical neutralization for waste. Material safety data sheets, updated as toxicology evolves, inform chemists about thresholds for exposure, symptom recognition, and emergency steps. Lab-scale users bear special responsibility for proper storage, as both the nitro group and methyl substituents show higher reactivity under heating, strong oxidizers, or reduction, greatly increasing both fire and explosion risks in poorly ventilated spaces.
Application Area
Manufacturers feed this compound into organic synthesis pipelines, targeting processes that produce pigments, specialty polymers, and pharmacologically active molecules. The nitro group’s versatility opens doors to both reduction and displacement chemistry, supporting not only direct product formation but also multi-step syntheses where intermediates retain or modify the original nitro functionality. In certain dye and pigment workflows, this nitroaromatic offers a key color precursor. In research circles, synthetic chemists use it as a template in mechanistic studies when probing regioselectivity and substitution reactions on methylated aromatics.
Research & Development
Modern chemical research seeks greener and safer routes to nitroaromatic intermediates. Laboratories worldwide experiment with ionic liquids, alternative nitrating agents, and more benign catalysts—all to curb hazardous waste and improve selectivity. Computational chemists model reaction pathways for nitration and downstream modifications, providing valuable predictive power in scaling new processes. Pharmaceutical teams explore nitro aromatics not only for direct utility but as scaffolds for more complex biologically active architectures. Newer publications report on engineered enzymes and biocatalysts with capacity for regioselective nitration, which hold promise for much less polluting synthesis in the future.
Toxicity Research
Toxicologists consider 4-nitro-1,2-xylene a substance of moderate concern, based on animal studies and data from related nitrocompounds. Short-term exposure can cause skin and respiratory tract irritation, while higher exposures associate with methemoglobinemia, a blood disorder that compromises oxygen transport. Long-term studies in rodents have raised questions about possible carcinogenic effects, although definitive links remain unproven by regulatory agencies. Industry pays close attention to permissible exposure limits, with modern workplace air monitoring and medical surveillance programs designed to spot problems before symptoms arise. Responsibly managed, the compound rarely causes harm, but historical records of improperly vented production facilities highlight the need for constant vigilance.
Future Prospects
Sustainable chemistry and regulatory pressure will shape the landscape for nitroaromatic production. The push for process intensification, better waste stream control, and safer alternatives will drive innovation in the coming decade. I see increasing adoption of continuous flow reactors and digital process control, especially in markets where compliance costs keep climbing. Advanced analytical methods can help spot trace contaminants early, ensuring safer intermediates for downstream uses in pharma and specialty manufacturing. Looking beyond traditional nitration, enzyme engineering and photochemical nitration could change the way that 4-nitro-1,2-xylene and its siblings come to market. Today, the strong demand for highly controlled aromatic intermediates—for catalysts, fine chemicals, and advanced polymers—will keep this compound relevant, as long as the industry continues refining both process and safety.
What is 4-Nitro-1,2-Xylene used for?
A Look Inside a Chemical With a Big Impact
4-Nitro-1,2-xylene doesn’t come up in daily conversation. Still, this yellow crystalline compound matters more than most realize, especially in labs and factories pushing boundaries in science and production. Spotting it on a chemical shelf signals some heavy-duty work behind the scenes.
Key Role in Making Dyes and Pigments
My first hands-on experience with 4-nitro-1,2-xylene happened in a research lab focused on organic synthesis. We ran trials making new azo dyes. You can spot the importance of this molecule in the letters and fibers all around us—think art paper, fabrics, even printer ink. Chemists value this compound for its ability to help build color molecules that stick and last. With its dual methyl groups and the nitro group, 4-nitro-1,2-xylene takes part in reactions that create vibrant and durable hues. Industries still bank on this chemistry for coloring plastics, rubber, and textiles.
Stepping Stone in Pharmaceuticals
Some compounds get used straight out of the bottle, but not this one. 4-Nitro-1,2-xylene often plays the role of a building block, especially in the hunt for new medicines. Thanks to the nitro group’s chemistry, it’s a reliable candidate for reduction to corresponding amines—key ingredients in pain relief meds, antibiotics, and even some cancer treatments. Its structure allows researchers to explore a wide range of modifications, leading to new drug possibilities.
Intermediary for Agrochemicals
Farmers depend on dependable products to protect their crops. Many agrochemical companies make use of 4-nitro-1,2-xylene during the manufacture of pesticides and herbicides. This isn’t always obvious to the people buying bottles of spray for their fields, but the journey of safe, effective plant protection often begins in a lab with this compound. With world food demand rising and the threat of crop diseases growing, innovative new agrochemicals built on strong chemical intermediates remain crucial.
Industry Challenges and What Can Help
The use of 4-nitro-1,2-xylene brings up real-world issues. Toxicity can’t be brushed aside: it can irritate the skin, mess with the respiratory system, and, like many nitroaromatics, its byproducts need careful handling. Getting rid of or recycling hazardous waste stays expensive and tricky. My time in the chemical industry underlined just how much strict handling protocols matter—the cost of mistakes adds up fast, whether in lost product or community health risks.
Factories looking to cut risks and lower waste are experimenting with greener processes—catalysts that make cleaner reactions or closed-loop systems that capture leftover chemicals instead of venting them. Routine employee safety training works best when paired with up-to-date air filtration and emergency response plans. Good ideas travel: sharing workable green chemistry learnings helps all players reduce the hazards linked to production.
Looking Forward
4-Nitro-1,2-xylene stands as a key tool, not just as a note in a catalog but in the products lining store shelves and homes. As demand for sustainable materials grows, pressure mounts to balance performance, efficiency, and stewardship. With enough determination and innovation, the chemical industry has a shot at protecting both consumers and the environment, even while relying on quirky building blocks like 4-nitro-1,2-xylene.
What are the safety precautions when handling 4-Nitro-1,2-Xylene?
What Makes 4-Nitro-1,2-Xylene Worth Extra Caution
Few things get my attention in a lab like a nitro compound. 4-Nitro-1,2-Xylene joins that list pretty fast. The stuff has a reputation for being more than just a simple chemical. Its structure puts it closer to the zone of unstable and potentially hazardous materials. Breathing in the vapors or letting the powder touch the skin isn’t a risk I’m ready to take. I’ve seen colleagues develop headaches and dizziness from accidental exposure – and cleanup takes a lot more than a paper towel and soap.
Working with 4-Nitro-1,2-Xylene: The Basics
In my time at the bench, I found that the best personal protective equipment (PPE) doesn’t seem optional after looking up safety data on this compound. Splash goggles handle accidental squirts much better than glasses, especially if a reaction vessel tips or a transfer goes sideways. You want gloves that stand up to organics – think nitrile, double-gloving if you’ve got a hefty amount or plan to do anything outside a closed system. Wearing a lab coat with fitted sleeves stops unexpected skin contact.
Ventilation secures a close second in importance. Fume hoods are where all pipetting, weighing, and transfer should happen. The smell may not always warn you; small-scale spills or dust in the air drift quietly unless air is being pulled away from your face. I remember a grad student getting a mild cough after working outside the hood, not realizing a fine mist had formed as she weighed out the powder.
Dealing with Spills and Accidents
Small spills call for immediate attention, not tomorrow’s cleanup crew. I reach for absorbent pads made for chemical spills rather than paper towels. For solids, scoop up the bulk into a labeled waste container. Then, wet, not dry-wipe, to avoid dust going airborne. After handling, I always ditch the gloves and wash hands, even if I think I didn’t touch anything. Larger spills or ones on clothing usually mean a call to hazardous response. Walking into those situations without backup equipment or training brings more harm than help.
Storing 4-Nitro-1,2-Xylene the Right Way
Simple plastic bins can’t contain everything safely. I use bins marked for hazardous organics, sealed up tight and kept away from sunlight and heat. Storing it near acids or strong bases never made sense to me; accidental mixing during a shelf collapse or a broken bottle gets ugly. I’ve seen small leaks corrode shelving and leave odors that linger for months. Regular checks and good labeling cut down on surprises for anyone else sharing storage.
Waste Disposal is Everyone’s Problem
Pouring leftovers down the sink is a recipe for trouble and pipe damage. I set up a separate waste jar marked for nitro-aromatics in every lab space I use. Waste gets handed off to licenced disposal teams with the right paperwork. Old containers, even if empty, wind up in hazardous waste bins, not regular trash. There’s a case for each type of chemical waste, not just mixing everything together for convenience – enforcement fines are steep, but the real cost is exposure to janitors and others who don’t expect these hazards.
Why Cutting Corners Just Isn’t Worth It
Safety routines can feel like a drag, especially if experiments run late or supplies run low. I watched one co-worker skip gloves on a busy day and land in the student health center for a quick checkup when he noticed tingling in his fingers later on. The best labs I’ve worked in keep extra supplies visible, training recent, and emergency numbers posted at every entrance.
Stepping Up to the Plate
Working with 4-nitro-1,2-xylene demands respect and good habits. Industry guidance points to the same steps that have worked in my experience. Proper gear, good ventilation, targeted cleanup, smart storage, and serious waste management form the backbone of smart lab work. Asking questions, double-checking labels, and looking after coworkers always round out the recipe for a space nobody regrets walking into on a Monday morning.
What is the chemical structure of 4-Nitro-1,2-Xylene?
A Closer Look at Molecular Details
Diving into organic chemistry, you come across names like 4-Nitro-1,2-xylene, and it can feel like a puzzle you need to solve. I remember sitting at a cluttered desk in university, staring down at aromatic rings with tiny sticky notes pointing to what each group meant. Xylene brings you a pair of methyl groups on a benzene ring. The 1,2 part tells you these methyl groups sit right next to each other—what the textbooks call the ortho position. Drop a nitro group on the fourth carbon, and things get interesting.
Visualizing the Skeleton: Structure Unpacked
Think of benzene as a sturdy six-carbon ring, the backbone of many industrial and pharmaceutical compounds. For 1,2-xylene, you attach methyl groups at carbon 1 and 2. Add a nitro (NO2) group at the fourth carbon—carbon 4 counting around the ring from one of the methyl groups. You end up with a formula of C8H9NO2.
Here's how it looks:
- Methyl group at carbon 1 (–CH3)
- Methyl group at carbon 2 (–CH3 right next to the first one)
- Nitro group (–NO2) at carbon 4, straight across from the first methyl
Why This Structure Matters
Chemists pay close attention to the position of each group. You move a nitro or methyl even one carbon over, and you change how that compound reacts. I remember working on a lab project that swapped nitro groups from position four to three on a similar backbone. Suddenly, our reactions went from slow and steady to explosive—no joke, you hear a little pop when too much heat hit the flask.
The 4-Nitro-1,2-xylene arrangement changes its reactivity compared to other isomers. That’s not just a curiosity for scientists in a lab; it impacts how safe something is for storage or transport, and whether it plays nice in chemical manufacturing. For instance, nitro-aromatics sometimes act as intermediates in drug or pesticide production. If you don’t keep an eye on which isomer you have, the downstream effects can include byproducts you never intended.
Challenges and Better Practices
Getting the right isomer can challenge even skilled chemists. Techniques like recrystallization or chromatography help with purification. Analytical methods—NMR, mass spectrometry, and IR spectroscopy—let researchers double-check what they’ve made. Once, we discovered a batch labeled as 4-nitro-1,2-xylene actually had a small impurity from its 3-nitro sibling, just because someone skipped part of the process. Even tiny mistakes can cause big safety or quality problems.
Improving labeling, ensuring good ventilation, and investing in quality assurance save time and, honestly, keep people safe. Training new researchers to recognize these subtle differences doesn’t just build good science—it builds a safer lab, factory, or warehouse.
Looking Ahead
As technology advances, synthesizing well-defined aromatic compounds like 4-Nitro-1,2-xylene gets easier, but you still rely on fundamentals. Understanding that structure—where each group lands on the ring—means fewer surprises, smoother production, and more innovative uses down the road.
How should 4-Nitro-1,2-Xylene be stored?
Understanding the Risks
Working around chemicals like 4-Nitro-1,2-Xylene brings a sense of responsibility that can’t be shrugged off. This compound, with its yellow tint and sharp smell, often ends up on lab shelves, used as an intermediate in chemical synthesis or research. It’s not something the average person encounters, but for those of us in labs or industrial settings, it’s more common than some folks might think.
Looking at Safety Basics
Experience teaches caution long before a formal safety briefing. With 4-Nitro-1,2-Xylene, opening the container without proper ventilation or gloves invites real trouble. The fumes irritate eyes and lungs, and direct skin contact leads to rashes or worse over repeated exposure. It’s not just about possible discomfort; this compound could pose both short-term and long-term health risks if ignored.
Industry reports and safety data sheets hammer home the importance of keeping this chemical away from open flames and sparks. The nitro group in its structure likes to react given the slightest provocation, which adds an explosive edge to its profile. One slip—one afternoon of inattention—can change everything. The 2022 review published in Journal of Chemical Health found that nearly 40% of solvent-related accidents in small labs linked back to bad storage habits or faulty labeling, pointing directly at human error, not faulty products.
Storing Smart and Safe
So what’s the right way to store 4-Nitro-1,2-Xylene? In practice, this means finding a cool, well-ventilated spot away from any heat source. Flammable storage cabinets make sense, not only for compliance, but because fire wants to find this chemical. I’ve seen labs where a careless bottle of solvent was parked next to a space heater “just for a day”—and later everyone wondered why the air stung their eyes.
Acidic or oxidizing chemicals kept in the same place spell extra risk. Mixing them by mistake, even through fumes, can trigger unexpected reactions. I keep a habit: label everything clearly, never leave an open container, and check the shelf—both the location on the shelf and the temperature of the room—at the end of every week. Peer-reviewed guides, like those from the American Chemical Society, recommend secondary containment trays for every bottle, so minor spills become nuisance, not disaster.
Accountability and Training
Supervisors and team leads bear non-stop pressure to keep everyone honest and safe. Training doesn’t stop after day one. Regular refreshers about storage protocols, emergency showers, gloves, and goggles actually cut down on near-misses. In an experiment at a teaching university, slipping once on protocol led to EMTs in the hallway. After that, they rewrote storage procedures, and guess what? No repeat accidents the next three years. Real-life stories stick longer than dry lectures.
Potential for Improvement
Technology makes safer labs possible. Automated sensors can alert staff about high vapor concentrations or accidental temperature rises. Locks and tech badges now control who enters chemical storage rooms. These upgrades cost money, but the tab for a cleanup or injury runs higher still.
No perfect fix exists for every lab or warehouse, but sharing stories and hard facts can save hours of headache and keep people healthy. Responsibility in storage starts with the individual and grows with the team—making every shelf and every label count for something.
What are the physical and chemical properties of 4-Nitro-1,2-Xylene?
Getting to Know 4-Nitro-1,2-Xylene
4-Nitro-1,2-xylene offers an interesting look into both the physical and chemical features of nitroaromatic compounds. It grabs attention in chemical circles because of how its structure shapes its properties—a benzene ring holds two methyl groups and a nitro group, which all play a part in its behavior.
Physical Traits That Matter
Anyone who's handled nitroaromatics knows many don’t play nicely; 4-Nitro-1,2-xylene is no exception. This compound forms yellowish crystals under normal room conditions. It doesn’t dissolve in water—pouring it into a beaker with just H2O means watching it collect at the bottom. Try heating it up, and the crystals start melting around the 76°C mark, though this can shift a little depending on impurities.
Put it in organic solvents instead, and you’ll see a big difference. Toluene, chloroform, and ether break it down with ease. This solubility makes it more useful for those in organic synthesis labs. For folks working with it, inhaling the powder isn't a good idea. That yellow dust can irritate eyes and noses or worse over time, so a good set of gloves and a hood are in order.
Chemical Properties: Reactivity Built In
Adding a nitro group to a xylene ring changes everything. That -NO2 doesn’t just make it yellow; it throws a wrench into how the molecule reacts. For ordinary xylene, the ring tends to react quickly with electrophiles. Stick a nitro group on, and the electron cloud around the ring gets pulled away, slowing down a lot of those fast reactions. Sulfonation, halogenation, and some types of oxidation become trickier—less push on the electron-rich spots means fewer fireworks.
On the flip side, the nitro group can come off under the right conditions, producing different amines. Plenty of dye chemistry and pharmaceuticals take advantage of this step. Laboratories working on reduction of nitro compounds use reagents like iron and hydrochloric acid to swap out -NO2 for -NH2. In that sense, 4-nitro-1,2-xylene steps up as an intermediate for things well beyond its simple form.
Risks and Challenges: Health, Safety, and Environment
Using this compound comes with a responsibility to safety. Nitroaromatics haven't earned a great reputation among occupational health experts. Overexposure, especially through skin or respiratory tract, can cause long-term issues. Like a lot of organic solvents, disposal becomes a headache. Dumping nitroaromatics down the drain spells trouble for water systems; they don’t break down quickly, and in some cases, they poison aquatic life. Waste treatment that destroys the nitro group is the best bet, either by catalytic hydrogenation or incineration at high heat. For smaller labs, collection and professional chemical disposal prevent big environmental risks down the road.
Pushing Toward Safer and Smarter Solutions
Innovation matters. Solvents and synthesis methods have moved forward because chemists keep searching for alternatives with lower toxicity and better safety profiles. For instance, green solvent systems sometimes let researchers work with nitro compounds without the fumes and hazards of older approaches. More predictive modeling in toxicity assessment also helps structure new regulations or best practices for nitroaromatics, protecting workers and ecosystems alike.
Experience and Perspective
Working in a lab, the differences between textbook data and real-world behavior always stand out. Melting points might vary, and handling safety can mean the difference between a productive day and a call to the fire department. Relying on data sheets isn't enough; personal experience with 4-nitro-1,2-xylene has shown me that its impact stretches well beyond its molecular formula. Each step from its creation to disposal ties into broader questions about chemical responsibility. We owe it to ourselves and the next generation to keep pushing for better science and smarter practices every time we use materials like these.
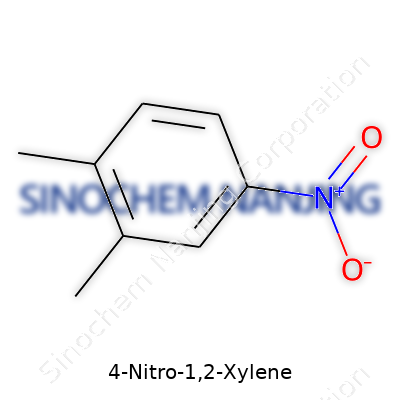
| Names | |
| Preferred IUPAC name | 1-methyl-4-nitro-2-methylbenzene |
| Other names |
4-Nitro-1,2-xylene 1,2-Dimethyl-4-nitrobenzene 4-Nitro-o-xylene |
| Pronunciation | /ˈfɔːr ˈnaɪtroʊ waɪn tuː ˈzaɪliːn/ |
| Identifiers | |
| CAS Number | 89-87-2 |
| 3D model (JSmol) | `JSmol.loadInline('data:model/x-pdb;base64,C1=CC(=C(C=C1C)[N+](=O)[O-])C')` |
| Beilstein Reference | **136358** |
| ChEBI | CHEBI:76257 |
| ChEMBL | CHEMBL2131431 |
| ChemSpider | 68218 |
| DrugBank | DB08347 |
| ECHA InfoCard | 100.016.431 |
| EC Number | 202-342-9 |
| Gmelin Reference | 104412 |
| KEGG | C14522 |
| MeSH | D009601 |
| PubChem CID | 86424 |
| RTECS number | ZE2450000 |
| UNII | SRX7K8E9B6 |
| UN number | UN1663 |
| Properties | |
| Chemical formula | C8H9NO2 |
| Molar mass | 150.16 g/mol |
| Appearance | Yellow crystalline powder |
| Odor | sweetish |
| Density | 1.21 g/cm³ |
| Solubility in water | Insoluble |
| log P | 1.98 |
| Vapor pressure | 0.00204 mmHg at 25°C |
| Acidity (pKa) | 4.31 |
| Basicity (pKb) | 9.88 |
| Magnetic susceptibility (χ) | -61.0 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.595 |
| Viscosity | 1.221 cP (20°C) |
| Dipole moment | 3.90 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 229.4 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -8.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4913.7 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause respiratory irritation, may cause damage to organs through prolonged or repeated exposure. |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS02,GHS07,GHS09 |
| Signal word | Danger |
| Hazard statements | H302 + H315 + H319 + H332 + H335 |
| Precautionary statements | P210, P261, P280, P301+P312, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 3-2-1-* |
| Flash point | 82 °C (180 °F; 355 K) |
| Autoignition temperature | 650°C |
| Lethal dose or concentration | LD50 oral rat 1800 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1596 mg/kg (rat, oral) |
| NIOSH | NIOSH: TT3850000 |
| PEL (Permissible) | PEL (Permissible): Not established |
| REL (Recommended) | 0.5 mg/m3 |
| IDLH (Immediate danger) | IDLH: 50 ppm |
| Related compounds | |
| Related compounds |
1,2-Dinitrobenzene 2-Nitrotoluene 4-Nitro-o-xylene 4-Nitro-m-xylene 4-Nitro-p-xylene 4-Nitroanisole |

