The Story and Substance of 4-Methylisoquinoline
Tracing the Journey of 4-Methylisoquinoline
4-Methylisoquinoline might not sound familiar to most people, but it traces its roots deep into the evolution of organic chemistry. Its parent structure, isoquinoline, popped up in the research journals of the late 1800s, as scientists raced to crack the mysteries of coal tar derivatives and plant alkaloids. For years, chemists dug into these tangled molecules, not for kicks or curiosity alone, but to untangle nature’s blueprints. When researchers first began tweaking isoquinoline rings—nudging on a methyl group here and there—they weren’t simply hunting for clever names. They were searching for changes in chemical character, differences that could turn a generic ring system into something valuable, whether as a chemical intermediate, a pharmaceutical scaffold, or a building block for dyes. As organic chemistry tools grew sharper, so did people’s abilities to fine-tune these little structures, and 4-Methylisoquinoline found itself under the spotlight. Not because it was flashy, but because it offered something new: manageable reactivity, a hint of biological intrigue, and—importantly—the kind of reliability synthetic chemists look for.
Learning the Basics of 4-Methylisoquinoline
Pick up a vial of 4-Methylisoquinoline and you’ll deal with a faintly yellowish liquid, a little heavier than water, with a sharp, often described as “amine-like” odor. Its molecular formula, C10H9N, fits neatly in the books, and this minor alteration from the mother compound changes the landscape in ways that matter to chemists. Pop that methyl group on the fourth carbon, and you suddenly shift reaction rates, alter solubility, and, occasionally, mess with toxicity. I’ve handled this material in the lab myself, and the first thing I learned is you don’t want it anywhere near your skin or nose for long. It slips through gloves if you aren’t careful, reminding you why reading up on every new compound in advance is a part of chemistry life.
Digging into Physical and Chemical Properties
With a boiling point hovering near 260°C, 4-Methylisoquinoline endures some serious heat. It dissolves well in most organic solvents, but doesn’t want much to do with water. This speaks to why chemists like to use it in non-aqueous synthesis—reactions run smoother, side products stay out of the way. You’ll spot its UV absorbance around 250-270 nm, classic for aromatic compounds. The ring acts as a base, which means you can count on protonation reactions if you’re building salts. Toss in that extra methyl group, and you’ll see a subtle shift in electron density, making its reactivity a bit different than its parent isoquinoline. This small variation means it becomes a tool for specific transformations when standard isoquinoline just won’t cut it.
Technical Details and Preparation
Labels on the reagent can feel intimidating, and not without reason. Purity claims for research-grade material should stretch north of 98%, and contamination with other methylisoquinolines will ruin your day if you’re chasing high precision results. Most producers ship it in amber bottles to fend off light degradation, with warnings about keeping it away from open flames. You don’t want accidental ignition—vapors can catch with little provocation. The classic method to make it in the lab starts by methylating isoquinoline under acidic controls, but plenty of folks have played with variations, including Pd-catalyzed cross-couplings that deliver clean 4-position products. Some chemists reach for the Bischler-Napieralski reaction, tweaking phenethylamines to close the ring and add that methyl with finesse.
Reacting and Modifying 4-Methylisoquinoline
The presence of both the aromatic ring and the nitrogen atom opens up options for chemical modifications. Electrophilic aromatic substitution usually stays selective, letting you decorate the molecule at positions that’d otherwise be tough to reach. I’ve used this trick to introduce halogens or nitro groups under carefully controlled conditions, turning basic methylisoquinolines into more elaborate intermediates for targets like antihypertensive compounds. Reductive alkylation, nucleophilic addition, and catalysis all become a bit different, thanks to the methyl group. Its stability under a range of conditions is a plus when mapping out synthetic routes—for those trying to build up complexity without watching their starting material disappear to the side reactions.
What Else Is It Called?
Chemists rarely use a single label for one compound, and 4-Methylisoquinoline is no exception. Older papers toss around “4-Methyl-quinoline” or simply “4-Methyl-isoquinoline” as alternative tags. Casually, some labs refer to it just as “MeIqn-4” or shorthand that only the initiated will follow. Its chemical registry numbers tie it down for the databases, but I’ve never met a synthetic chemist who walks into a storeroom asking for anything but “4-Methylisoquinoline.”
Following Safety and Operational Practices
I’ve made mistakes with aromatic amines before, and the lesson sticks every time: gloves, goggles, and a fume hood are non-negotiable. 4-Methylisoquinoline can burn the eyes and mucous membranes, and like most small aromatic heterocycles, it doesn’t play nice with chronic exposure. It demands regular monitoring of air quality, tight container closures, and attention when weighing out solids that could aerosolize. Regulatory agencies flag it for standard chemical handling, but the real risk sits in handling volume and frequency. Larger scales or more adventurous chemistry may require respirators and splash protection. The most sustainable labs run regular hazard audits, keeping everyone sharp and accidents rare.
Where Do You See It Used?
In the world of organic synthesis, 4-Methylisoquinoline slips into more reactions than you’d guess at first glance. Pharmaceutical researchers chase its backbone in the search for new drugs, partly because the isoquinoline ring pops up in natural products with antibacterial, anticancer, or neurological effects. Once that methyl group is in place, the profile of the molecule ticks in a way that can unlock new binding affinities. Outside pharma, its story continues in the dye and pigment industry, where specific tweaks to aromatic structure change color and stability. Some polymer scientists look at it as a way to introduce controlled defects into carbon-based frameworks, particularly for materials that demand tailored electrical properties. The push for greener chemistry opens doors for more selective, low-waste transformations based on scaffolds like 4-Methylisoquinoline.
Pushing Research Forward
Search the literature and you’ll find work on methylisoquinolines stretching back decades. Research goes cyclical: one decade the interest cools, the next, someone unearths a new reaction or biological effect that triggers a rush of fresh studies. Recent years brought computational chemistry tools to the scene—mapping how tiny tweaks change electron distribution or pharmacological profiles. In drug discovery, researchers dig into libraries of small molecules, testing each one against disease targets with automated screening that 19th-century chemists could only dream about. This new data sometimes points back to simple structures with methyl tweaks, showing once again that modest changes can disrupt stale narratives in drug and materials design.
What Does the Toxicity Data Show?
Data on 4-Methylisoquinoline’s human health effects grows slowly, partly because it isn’t a household chemical or a large-scale industrial ingredient. Animal testing highlights the usual dose-dependent tricks—high doses impact the liver and kidneys, just as you expect for many aromatic amines. Some structural relatives raise red flags for genotoxicity or carcinogenicity, setting a cautious tone for anybody testing new derivatives. There’s no sense in downplaying the risk or pretending all aromatic heterocycles behave gently in the body. Every new application comes with a phase of careful toxicological scrutiny, as should be the norm for everything heading toward the pharmaceutical or consumer marketplace.
Looking Ahead
The story of 4-Methylisoquinoline keeps evolving along with the fields it serves. The hunger for new, reliable building blocks in organic synthesis won’t end soon. Demand from pharmaceutical and materials research is likely to push refinements in both preparation and application. Advances in green chemistry may prompt streamlined syntheses that cut down hazardous waste, and novel catalytic protocols could unlock even more selective functionalizations. The digital era transforms how chemists design molecules—software now proposes modifications that labs then test, combining efficiency and creativity in ways that no earlier generation could have managed. Wherever this chemistry goes, lessons from the past echo forward: never underestimate what a small change in structure can bring, and always treat new variants with respect, both scientifically and from a safety standpoint.
What is 4-Methylisoquinoline used for?
Digging into 4-Methylisoquinoline
4-Methylisoquinoline is a chemical that doesn’t pop up in daily conversation, but it supports some key work in chemistry and medicine behind the scenes. It belongs to the family of isoquinolines, which chemists value for their complex structure and unique properties. This molecule gives scientists a building block for developing many different kinds of compounds, including pharmaceuticals, agricultural chemicals, and dyes.
Pharmaceutical Applications and Research
For drug researchers, 4-Methylisoquinoline is more than a quiet spectator. Its structure helps scientists create new medicines, especially for conditions needing molecules that target the nervous system or certain cancers. Some cancer drugs trace their core skeleton to this very molecule. Chemists craft 4-Methylisoquinoline into more elaborate compounds, chasing benefits like pain relief or improved treatment for rare diseases. The story here reflects years of benchwork and countless hours of trial and error.
Teams in pharmaceutical labs keep their eye out for molecular structures that promise new treatments. I remember talking with colleagues who pore over chemical libraries looking for candidates. Isoquinolines, with their fused-ring structures, offer real promise for building drugs that interact strongly with enzymes or faulty cells. Documents from the National Institutes of Health show how derivatives influence the development of agents against infections and neurological disorders. The process isn’t fast — each tweak to the molecule’s design might move research a step closer to safety and real-world benefit, but one miscalculation can set teams back by months. In this way, 4-Methylisoquinoline acts as a kind of starting line for important races in drug discovery.
Role in Chemical Synthesis
4-Methylisoquinoline plays a practical part in the world of organic synthesis. Chemists who need to build bigger, more complicated molecules sometimes rely on this compound as a stepping-stone. When making specialty chemicals, manufacturers use 4-Methylisoquinoline in finely controlled reactions, shaping it into dyes that color textiles or additives for advanced polymers. Its structure lets it act as both a target and a tool, depending on the type of reaction underway.
Researchers from academic labs to commercial outfits cross paths with this molecule. At universities, students learn to handle it because it opens doors to deeper sections of synthetic chemistry. I recall how careful we had to be using it in graduate school—one wrong move meant starting from scratch. The molecule’s availability and adaptability keep it on the shelves of academic labs around the world.
Challenges and Safety
Though 4-Methylisoquinoline brings clear benefits to research, working with it brings risks. Like many organic compounds, it needs careful handling and proper ventilation. The safety data sheets flag issues around inhalation, skin contact, and long-term exposure. Health organizations provide recommendations to keep everyone in the lab safe, urging gloves and fume hoods whenever people handle it. This doesn’t stop the progress, but reminds us that science balances advancement against safety every day.
Future Potential
A strong future possibility sits in designing new medicines that build on its chemical backbone. More research could uncover eco-friendly uses or smarter ways to recycle materials derived from 4-Methylisoquinoline. Some teams look into converting waste from its production into safer chemicals or useful byproducts. By keeping an eye on cleanup, the chemistry community can help steer industry and society toward safer and cleaner solutions.
What is the chemical structure and formula of 4-Methylisoquinoline?
Unpacking the Structure
Chemistry often feels like learning a new language. The names, the shapes, the quirks of each compound — these details matter when work steers into research, innovative medicine, or even creating new materials. 4-Methylisoquinoline sits in a class that has seen years of study, both in academic labs and in industries needing specialized chemical building blocks.
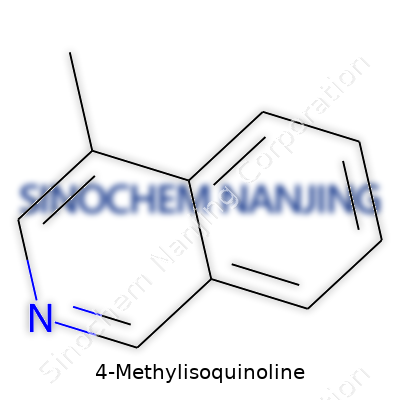
Isoquinoline forms the base structure, which means this compound looks like two aromatic rings fused together, a bit like naphthalene but with one nitrogen atom swapped in. Methyl groups are common substituents and here, the methyl (-CH3) goes to the fourth carbon atom on the isoquinoline scaffold. Even for those who don't live in a chemistry lab, seeing how this arrangement changes the physical and chemical behavior can be surprising. Tiny changes, such as sticking a methyl group on a ring, often tune boiling points, solubility, or how the molecule interacts with others in a reaction.
Getting Technical: Formula and Structure Representation
The molecular formula for 4-Methylisoquinoline reads as C10H9N. Staring at this formula, it’s natural to picture how these atoms lock together. Beyond the shorthand, chemists draw out structures using aromatic rings connected side by side, showing a six-membered benzene fused to a six-membered ring containing a nitrogen. Put the methyl at position 4 and you have the skeletal shape in mind: aromaticity intact, nitrogen in the correct place, methyl tacked on.
Many in the lab sketch this as:
- A fused bicyclic system with nitrogen at the first position
- A methyl group sticking off the fourth carbon of the isoquinoline skeleton
Small details like these matter for researchers designing drugs or tweaking catalysts. Fused rings open doors to new reactivity, help drugs fit neatly into enzyme pockets, or give off optical properties for materials science.
Why Structure Shapes Use and Discovery
Grasping the structure and formula isn’t academic nitpicking. When a pharmaceutical team looks at isoquinoline derivatives, the position of a methyl group can drive whether a molecule hits the right biological target. Drug development, for instance, pays close attention to substitutions on the aromatic rings, since even a single methyl group changes how the body recognizes or metabolizes a compound. Patents often hinge on slight molecular reshuffles because even a minor modification can create a novel drug.
Chemists working on organic synthesis face similar choices. Choosing 4-Methylisoquinoline as a starting material may streamline a total synthesis or help sidestep patent thickets. In practical lab work, knowing where to introduce a methyl group means selecting the right reagents, protecting groups, and understanding what impurities could form. Mistakes in the early steps often snowball into costly purification problems down the road.
Looking for Better Pathways
In the bigger picture, the quest for simpler, safer, or greener ways to produce compounds like 4-Methylisoquinoline keeps chemists busy. Some routes use palladium-catalyzed coupling, while others turn to more traditional Friedländer synthesis. Every process demands a balance—yield, safety, environmental impact, and scalability for industry. For those of us in the lab or working in industry, tracking down efficient synthetic paths helps reduce waste and keeps the process competitive, both from a financial and an environmental standpoint.
In daily work, having solid information on molecular structures and formulas shapes everything from the search for new materials to drug discovery. The little choices—like where a methyl group lands—echo through the science and steer what gets built tomorrow.
Is 4-Methylisoquinoline hazardous or toxic?
Understanding This Chemical
Ask folks who work in a lab or chemical plant, and they’ll often tell you about a long list of chemicals they encounter. 4-Methylisoquinoline might not be a household name, but it regularly shows up in chemical research and some industrial settings. Its formula, C10H9N, describes a structure close to compounds used in manufacturing and organic synthesis. Some chemists reach for it as a starting material for drugs, dyes, or other specialty chemicals.
The Straight Facts on Hazards
Stories about chemical safety always turn personal. One slip, one whiff too many, and you know how real the risks get. So what about 4-methylisoquinoline? The public databases point out its hazards clearly. The European Chemicals Agency and other regulatory groups tag it with warnings for skin and eye irritation. The compound’s vapors can bother the respiratory system, particularly for people who already deal with asthma or allergies.
Research on its toxicity doesn’t flag it as one of the nastiest chemicals out there. It’s not classified as a known carcinogen. Its acute toxicity for mammals lands in a range similar to other simple nitrogen-heterocycles—meaning, accidental mouthfuls or big spills could make a person very sick, but not on the level of cyanide or heavy metals. In my experience, any liquid with a sharp chemical odor deserves respect, gloves, and good ventilation. 4-methylisoquinoline definitely fits that mold.
No Room for Guesswork in the Workplace
Folks handling this chemical wear protective gear for a good reason. Even though it doesn’t top the list of most toxic compounds, it acts as an irritant that can quickly cause problems for skin, eyes, and lungs. In an industrial setting, safety data sheets lay out straightforward rules: avoid breathing vapors, keep it off bare skin, and never eat or drink around it. Forgetting those basics leads to hospital visits.
Spills happen, and cleanup should always include proper absorbent material and chemical waste procedures. Down the drain isn’t an option. Environmental laws frown on flushing organic chemicals, since they can hurt aquatic life. Besides, open drains risk fumes spreading to other parts of a building. Nobody wants a toxic surprise traveling through ventilation.
Gaps in Public Knowledge
A problem crops up with lesser-known chemicals: lack of detailed research on long-term effects. Decades ago, stories circulated about “safe” chemicals that only later showed serious dangers, like benzene or certain pesticides. While today’s protocols are far tighter, caution pays off. Regular air monitoring, quick smart responses to spills, and routine medical check-ups for workers all reduce the chance of long-term issues.
Knowing what a compound can do means more than just reading old papers. It means pushing for new studies and transparent updates from industry and government, especially as chemicals move from research labs into mainstream use. That’s part of building trust with people who may work or live near these materials.
Real-Life Solutions
Fining companies for lax safety doesn’t fix everything. Training for all staff, easy access to emergency eyewashes and showers, and clear reporting of minor exposures all help catch small problems before they escalate. Substituting safer chemicals, when possible, protects workers even more.
I’ve seen the difference a good safety culture makes: weekly walk-throughs, honest reporting, no shortcuts to save time. That’s what keeps dangerous chemicals from turning into big stories on the nightly news. No chemical, including 4-methylisoquinoline, deserves more attention than the people working around it.
How should 4-Methylisoquinoline be stored?
Getting Real about Chemical Storage
Walk into any small lab or industrial site and you’ll probably recognize a certain glass bottle, covered with fingerprints, shoved in the back of a dark cupboard. Sometimes this is where 4-Methylisoquinoline ends up—sometimes it leaks a little, sometimes it “disappears.” Storing this compound isn’t just about following textbook rules or ticking boxes on a checklist. Practical experience counts. Keeping this compound safe means looking after both people and property. Every step matters, from the warehouse shelf to the fume hood.
Flammability and Stability: No Room for Guesswork
4-Methylisoquinoline acts like many organic chemicals—it evaporates quickly and catches fire easily. Nobody wants to open a storage cabinet and smell a faint hint of something sweet and musty. If you can smell it, exposure already happened and that’s never good news. Vapors can irritate the nose and eyes, and every spilled drop raises the risk of fire. So storage practices shape both short- and long-term health outcomes.
Glass containers with tight, reliable seals stand out as the safest bet. Polyethylene or polypropylene might work for other chemicals, but this one can soften plastics over time. Moisture spoils the purity, so a dry cabinet protects both the chemical and future experiments. Folks storing it near windows or heat vents invite trouble—sunlight and warmth chip away at its stability and let contamination sneak in. Many of us have learned the hard way that one hot summer spell in a stuffy storeroom can render an entire batch useless.
Label It Like You Mean It
Mislabeled bottles turn even a careful lab into a minefield. Clear, permanent labels and hazard warnings protect the next person—maybe a new technician, maybe yourself on a tired Monday morning. Storing 4-Methylisoquinoline without labeling every bottle, vial, and secondary container asks for accidents. Using color-coded stickers for flammables or a separate shelf helps even experienced workers move fast and safe in an emergency.
Locked Cabinets, Real Security
Access matters just as much as the right jar. A locked, ventilated chemical cabinet stops both curious passersby and casual theft. Chemical spills happen, but restricted access keeps surprises to a minimum. I once watched a valuable supply walk out the door because someone left the cabinet unlocked during a lunch break. That sort of mistake strains budgets and trust—simple locks make a difference where insurance and policy cannot.
Temperature: Keeping It Cool, Not Cold
A cool, dry space extends shelf life and boosts safety. Refrigerators work, but only the models designed for flammables. Do not stash flammable organics in a kitchen fridge. Sparks, poor airflow, and wide temperature swings can turn a routine check into a fire drill. If your lab can’t afford a flammable-storage fridge, push for one in next year’s budget meetings—or share resources with a neighbor. Sharing costs saves money and makes sure no lone bottle gets forgotten on a shelf just out of sight.
Training and Shared Responsibility
Proper storage means everyone on the team knows the drill. Training shouldn’t end with one safety meeting. Checking the storage area for leaks, drift, or strange smells finds problems before they escalate. Keeping record of inventory and expiration dates helps labs plan ahead. Smart labs hold seasonal safety reviews, always with real examples of both good and bad storage choices. Sometimes the best lessons come from sharing close calls, not just rulebooks. Taking chemical safety seriously protects futures, not just lab benches.
What are the common methods of synthesizing 4-Methylisoquinoline?
Methods That Actually Work
Chemists searching for 4-Methylisoquinoline rarely take one universal shortcut. Synthetic methods depend on what works in the real world—availability of materials, scalability, and practicality. In pharmaceutical chemistry, this compound often gets attention for its presence in biologically active molecules. The common approaches reflect years of creative problem-solving in the lab.
The Bischler-Napieralski Route
The Bischler-Napieralski cyclization stands out as an old reliable. Starting with a β-phenylethylamine and an acyl chloride, chemists make a cyclized imine using a Lewis acid like POCl3 or P2O5. To land on 4-Methylisoquinoline, you’d use a methylated phenylethylamine. Once the intermediate cyclizes, a reduction step with something like Pd/C and hydrogen strips out the imine, giving the final heterocycle.
This method makes sense for decent yields and clear pathways. It also tolerates a range of substituents, so researchers adapt it for different analogues. Fewer side products means less time spent purifying afterwards—always appreciated when every hour in the fume hood counts.
Pomeranz-Fritsch Synthesis Gives Flexibility
Another respected tool, the Pomeranz-Fritsch synthesis, builds isoquinoline rings from benzaldehyde derivatives and aminoacetaldehyde diethyl acetal. Introduce a methyl on the aromatic ring before reaction, and the synthesis drops a methyl group into the four-position. After acid catalysis to push ring closure, the imine forms and aromatizes, giving that 4-methyl flavor.
The stepwise nature helps with controlling regioselectivity, which matters when the market demands a specific isomer. Labs with limited resources can use this reaction since the starting materials show up in standard catalogs. Despite its old-school status, this method hasn’t faded from the playbook.
Recent Moves: Transition Metal Catalysis and Cross-Coupling
As the cost of palladium has dropped relative to the early 2000s, labs shifted some routes to cross-coupling chemistry. Suzuki or Heck reactions come into play for constructing substituted isoquinolines, with methyl boronic acids helping guide methyl group placement on the intermediate. You see conditions like Pd(PPh3)4 in toluene with K2CO3 as base, combining building blocks in a more modular way. This method offers access to unusual methyl patterns, not just the four-position, and it scales up without much drama.
This approach cuts down on tedious multi-step procedures. Anyone who’s run one reaction and watched it fail at scale appreciates the reliability found here. You end up with clean products, high yields, and fewer headaches getting rid of trace metals via workup protocols.
The Why That Matters
I remember pounding out a research project on isoquinoline analogues and chasing after better synthetic shortcuts. The hustle for cleaner routes isn’t just about academic one-upmanship. Drug companies, agrochemical firms, and even flavor chemists care about minimizing impurities and lowering costs. Using established methods like Bischler-Napieralski or branching out to modern metal-catalyzed couplings lets scientists hit both targets.
Finding better catalysts, safer reagents, and one-pot sequences still stands as a frontier. Every new avenue—whether an electrochemical dehydrogenation or a biocatalytic ring closure—offers another chance to clean up the process and reduce environmental impact. The push for greener chemistry comes from both regulation and a sense of responsibility felt across the industry.
| Names | |
| Preferred IUPAC name | 4-Methylisoquinoline |
| Other names |
4-Methylisoquinoline 4-Methyl-isoquinoline 4-MIQ |
| Pronunciation | /ˌfɔːrˌmɛθɪlˌaɪsəʊˈkwɪnəliːn/ |
| Identifiers | |
| CAS Number | 6866-98-0 |
| 3D model (JSmol) | `4-Methylisoquinoline | JSmol 3D model string:` `C1=CC=C2C(=C1)C=NC=C2C` |
| Beilstein Reference | **1204784** |
| ChEBI | CHEBI:51530 |
| ChEMBL | CHEMBL146834 |
| ChemSpider | 120481 |
| DrugBank | DB04118 |
| ECHA InfoCard | 100.054.379 |
| EC Number | 211-438-0 |
| Gmelin Reference | Gmelin 20877 |
| KEGG | C16336 |
| MeSH | D06INP0F1K |
| PubChem CID | 70183 |
| RTECS number | NL3675000 |
| UNII | R24O0I31Z2 |
| UN number | UN3436 |
| CompTox Dashboard (EPA) | DJ8IJQ8O4X |
| Properties | |
| Chemical formula | C10H9N |
| Molar mass | 143.19 g/mol |
| Appearance | Colorless to light yellow liquid |
| Odor | amine-like |
| Density | 1.06 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 1.97 |
| Vapor pressure | 0.0786 mmHg (25°C) |
| Acidity (pKa) | 5.41 |
| Basicity (pKb) | 9.68 |
| Magnetic susceptibility (χ) | -81.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.613 |
| Viscosity | 2.1 cP (20°C) |
| Dipole moment | 1.98 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 212.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 141.4 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3752.6 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P261, P280, P302+P352, P305+P351+P338, P332+P313, P337+P313 |
| NFPA 704 (fire diamond) | 2-2-0 |
| Flash point | Flash point: 93°C |
| Autoignition temperature | 860°F (460°C) |
| Explosive limits | Explosive limits: 1.1–7.1% (estimated) |
| Lethal dose or concentration | LD50 (oral, rat) > 2000 mg/kg |
| LD50 (median dose) | LD50 (median dose): 400 mg/kg (mouse, intraperitoneal) |
| NIOSH | UQ8225000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 100 mg |
| Related compounds | |
| Related compounds |
Isoquinoline 4-Methylquinoline 2-Methylisoquinoline 5-Methylisoquinoline 6-Methylisoquinoline 7-Methylisoquinoline 8-Methylisoquinoline |

