4-Methylaniline: Deep Roots and Modern Hurdles
Historical Development
Chemistry owes a lot to the stubborn ambition of early industrialists and researchers. 4-Methylaniline, known as para-toluidine in many labs, came onto the scene during the blossoming years of coal tar chemistry in the late 1800s. People turned aromatic hydrocarbons scraped from coal processing into a rainbow of chemicals, and methylated anilines soon found places as essential building blocks for dyes. A century ago, the colorants that shaped textiles, inks, and leather often could be traced back to simple anilines, and when someone tweaked a hydrogen for a methyl group at the right spot on the benzene ring, 4-Methylaniline was born. The world barely blinked at this colorless to pale yellow liquid, but as I’ve seen in countless industry stories, quiet chemicals often mask decades of impact and sometimes trouble.
Product Overview
Anyone who’s worked near synthesis labs or dye halls knows the sharp, almondlike smell of anilines. 4-Methylaniline isn’t flashy, but it seeds reactions that touch medicine, paints, pesticides, and, especially in the past, textile dyes. Its raw, faintly sweet aroma lingers at low thresholds—so much so that its presence in a factory always seemed obvious. The nitty-gritty is this: 4-Methylaniline isn’t sold to the home hobbyist. Chemical suppliers send it to companies and researchers who blend raw ingredients into complex molecules—mostly away from public view.
Physical & Chemical Properties
Handling 4-Methylaniline, you first spot its oily, faintly colored form. It melts at 43°C, boils near 200°C, and dissolves modestly in water but prefers organics. With both a methyl group and an amine hanging off its benzene backbone, it lands chemical punches in two directions: basicity from the amine, and a subtle tweak in electron density from the methyl. Old textbooks note it burns with a smoky flame and reacts eagerly with acids to form salts. Producers store it in solid or liquid form depending on the plant's local temperature, always with ventilation and care to avoid unwanted reactions.
Technical Specifications & Labeling
Buying or shipping pure 4-Methylaniline usually means a label stamped with a CAS number (106-49-0), purity percentages—sometimes above 99% for research labs—and warnings for acute and chronic toxicity. Labels spell out dangers in clear terms. Long before REACH, GHS, and other modern regimes, mishaps pushed chemical safety regulations into daily conversation. Repeated incidents, especially in cramped dye factories, taught generations of workers to respect those labels and keep unknown dusts and vapors away from skin or lungs.
Preparation Method
Older textbooks hint at the classic toluidine synthesis: start with toluene, hit it with nitration conditions to get nitrotoluene, then reduce the nitro group using iron filings and hydrochloric acid. A lot of steam, clanging drums, and a fair bit of worry about runaway exotherms mark those processes—almost every industrial chemist I know has a story about piloting a synthesis plant or cleaning up a spill. Today’s refinements use catalytic hydrogenation, higher selectivity, process controls, and stricter isolation, all steps brought about to waste less and expose people to toxins less often.
Chemical Reactions & Modifications
4-Methylaniline interacts readily with acid chlorides to form amides, with aldehydes to yield Schiff bases, and heads off into azo coupling in dye synthesis—the bread and butter for early color chemists. I remember lab days spent tuning substituents, nudging the reactivity of that amine, aiming for colors that stuck better to cotton or faded less under sunlight. Chemical reactivity translates into both opportunity and risk. The same electron-rich ring that makes para-toluidine a handy synthon can generate sticky, mutagenic byproducts if not tightly controlled.
Synonyms & Product Names
Outside academia, you see 4-Methylaniline called para-toluidine, p-toluidine, and 1-amino-4-methylbenzene. Bulk shipments travel under these names across continents. The jargon may sound arcane, but in finding SDS documents or casualty lists, names like p-toluidine signal the right hazards—even before someone checks the chemical structure.
Safety & Operational Standards
Work with 4-Methylaniline demands close eye on safety measures. With toxicity levels on par with many aromatic amines, it sinks through skin and gets swiftly into the bloodstream. Symptoms from high vapor exposure or skin contact aren’t subtle—headaches, methemoglobinemia, and long-term damage have haunted those who didn’t respect lab coats, goggles, or extractor fans. OSHA, EU-OSHA, and other regulators hammered out benchmarks for exposure, personal protection, emergency shower locations, and spill responses. My own first encounter with stained gloves following a broken bottle taught an unforgettable lesson. Training and vigilance are constant companions to anyone using aromatic amines.
Application Area
Dyes and pigment precursors still claim the lion’s share of 4-Methylaniline demand, but its chemistry plays out beyond colors. Pharma manufacturers see it as a side chain or a scaffold; crop protection chases new fungicides built off the ring. In specialty chemicals, it seeds resins, rubber accelerators, and monomers. As companies explore alternatives, 4-Methylaniline remains enshrined in older patents and legacy products—testament to its central role in generations of manufacturing.
Research & Development
Academic and corporate R&D have nudged the usage and modification of 4-Methylaniline with each decade. Green chemistry icons steer major research into greener synthetic routes, safer alternatives, and better waste management. Chromatographers chase improved purity profiles while toxicologists dissect every impurity, always worried about carcinogenic findings from animal models. Sensors, catalysts, and even battery research occasionally revive interest in its molecular tweaks. The chemical’s straightforward structure belies a surprising complexity in usage and development.
Toxicity Research
Almost every list of hazardous aromatic amines gives 4-Methylaniline a grim mention. Documentation links chronic exposure to toxicity in blood and organs, hinting at lingering risks for workers and end-users. Studies across Europe and Asia reported subtle and not-so-subtle genotoxic effects. In many conversations with industrial hygienists, concerns stretched from acute methemoglobinemia to long-term cancer risks. Most modern chemical firms routinely test air and surfaces, log exposures, and swap out old equipment to limit leaks and hand contact—lessons written in painful, sometimes fatal, mishaps.
Future Prospects
Many traditional industries look to phase out hazardous intermediates whenever substitutes or alternative methods permit. In the case of 4-Methylaniline, growing regulatory scrutiny and evolving green synthesis methods are nudging manufacturers toward less toxic substitutes and closed-loop processes. From lab to plant, every improvement helps contain exposure and minimize persistent pollutants. As markets shift toward safer colorants and newer functional materials, some commercial roles for 4-Methylaniline may fade, but its legacy as a chemical workhorse isn’t going away quietly. I see labs still exploring new uses, but with every fresh regulatory framework and cleaner synthesis, the story edges away from routine handling and toward a more thoughtful role in specialty chemistry.
What is 4-Methylaniline used for?
Understanding the Role of 4-Methylaniline
As a writer who keeps an eye on industrial chemicals, I’ve noticed that 4-Methylaniline often pops up in manufacturing conversations. It’s not something you hear about over dinner, but it finds a place in a surprising range of products. Its main job comes from its use as a building block for dyes, pesticides, and pharmaceuticals. In my own research, I’ve found that many large textile mills count on this compound to produce colors that don’t fade after a couple of washes. Bright reds and blues owe much of their punch to this substance.
Dyes and Pigments: Why 4-Methylaniline Matters
Makers in the dye business count on 4-Methylaniline for its ability to bring vibrant shades to fabrics and plastics. Growing up, my family had a textile shop where good color was the sign of quality. I remember seeing customers run their hands over cloth, checking for evenness in each hue. The demand for bold, lasting colors hasn’t changed. Chemical bases like 4-Methylaniline make it possible for companies to create richly colored products. This isn’t just about looks; color consistency holds value for branding and consumer trust. Studies show that global dye production relies heavily on anilines, especially for items that reach markets worldwide.
Pesticides and Agriculture
Agriculture brings another major use for this compound. Pesticides often require chemical intermediates to target pests without harming crops. Farmers lean on these substances to protect their harvests from insects and fungus. During a visit to a citrus farm, I saw the difference effective crop protection can make. Chemicals derived from 4-Methylaniline go into the production of various herbicides and insecticides. The benefit: stronger yields and less loss for growers. At the same time, health and environmental groups worry about possible runoff and bioaccumulation, urging better management and alternative approaches.
Pharmaceuticals and Industrial Use
Beyond dyes and agriculture, the pharmaceutical industry has carved out uses for 4-Methylaniline during drug synthesis. It serves as a starting material for certain pain relievers and local anesthetics. Working with a pharmacy technician a few years ago, I watched as complex molecules were assembled in clean rooms. Compounds like 4-Methylaniline make it into the process early on, long before hitting the pharmacy shelf. Chemical manufacturers value its ability to serve as a flexible intermediate, allowing for efficiency in multi-step syntheses.
Health and Safety Concerns
Handling and exposure form a critical topic wherever this chemical is present. Both the U.S. Environmental Protection Agency and European agencies list 4-Methylaniline as a hazardous substance. I’ve seen firsthand how strict industrial protocols keep exposure in check—protective gear, ventilation, and constant air monitoring remain standard for workers. Long-term inhalation can lead to health problems ranging from skin irritation to more serious concerns, so rules aim to lower these risks wherever possible.
Striking a Balance: Solutions and Future Directions
Industrial growth demands safe and sustainable solutions. Manufacturers have invested in closed-loop systems to stop leaks and spills. Some companies research less hazardous substitutes for dyes and pesticide synthesis. More education—both for workers and communities—could lower risks as well. I’ve walked through more than one facility where training makes the real difference. The future of 4-Methylaniline lies in balancing its valuable contributions with a responsibility for health and the environment.
What are the safety precautions for handling 4-Methylaniline?
Understanding What You're Working With
4-Methylaniline, also known as p-toluidine, plays a role across chemical industries, from dye manufacturing to laboratory analysis. Every chemical I’ve worked with, no matter the purpose, demands respect for its hazards—4-Methylaniline sits high on that list. It carries acute health risks and long-term consequences, especially for those exposed without proper training or equipment.
Personal Protective Equipment: The Real Shield
Standard lab coats and cotton gloves don’t provide much protection against aromatic amines. When handling 4-Methylaniline, I reach for chemical-resistant gloves—nitrile or neoprene beat latex in resisting leaks. Safety goggles with side shields protect eyes from splashes, and a face shield gives better coverage during mixing or pouring. Closed, chemical-resistant shoes make a big difference if there’s ever a spill. Since skin absorption and inhalation can both lead to toxicity, a fitted lab coat and long pants block another path for this chemical to reach the body.
Ventilation and Air Quality
Most chemistry veterans have a story about fume hoods saving the day. For me, one memory stands out—a careless teacher let aromatic vapors escape, and headache plus nausea kicked in fast. 4-Methylaniline’s vapors go unnoticed until symptoms appear, so working outside a chemical fume hood is never worth it. Local exhaust ventilation cuts exposure, and good general room airflow adds a safety net.
Skin and Inhalation Exposure
Even brief skin contact with 4-Methylaniline triggers irritation, while chronic exposure risks blood disorders like methemoglobinemia. A single glove breach once left me with a tingling hand for an hour—a wake-up call to double-check for punctures and swap gloves at any sign of damage. If spills reach skin, immediate washing with plenty of water is critical. Inhaling dusts or vapor brings on respiratory distress, so a properly fitted respirator with an organic vapor cartridge makes sense during transfers or cleanup, especially in areas without strong hoods.
Handling, Storage, and Spill Readiness
Storing 4-Methylaniline away from oxidizers and acids in a well-ventilated, labeled cabinet helps prevent accidents. I use secondary containment trays, which catch drips or leaks and make cleaning simpler. Keeping only as much as needed on hand—never more—is a habit I stick with to cut risk. Every time I decant or weigh the compound, even small amounts, spill kits with absorbent materials and neutralizers stay close. Quick action during spills—absorb, bag, and ventilate—prevents vapors from building up.
Medical Response and Contamination Control
Supervisors and coworkers must know first aid basics. Immediate decontamination—like eyewash or shower—reduces chemical burns and systemic absorption significantly. Reporting symptoms right away and seeking medical advice beats trying to tough it out, especially given the possibility of delayed effects. Waste disposal in lined, marked containers closes the contamination loop, making it safer for others who handle laboratory trash.
Training and Culture Matter
Chemicals like 4-Methylaniline don’t leave room for shortcuts. Regular safety drills and training keep everyone fresh on procedures. Lessons stick best through shared stories and visible caution. Creating a culture where speaking up about unsafe practices is welcomed makes all the difference. In my own work, having a mentor who emphasized vigilance saved me—and others—more than once from exposure and long-term health issues.
Building Habits for the Long Run
Focusing on simple habits—wearing the right gear, using ventilation, storing properly, and practicing spills—protects workers and labs alike. Familiarity can lead to complacency with hazardous materials, so revisiting safety procedures joins the regular rhythm of any workspace. A little attention every time goes further than treating safety as a checklist, and it keeps accidents rare.
What is the molecular formula of 4-Methylaniline?
A Closer Look at What 4-Methylaniline Really Means
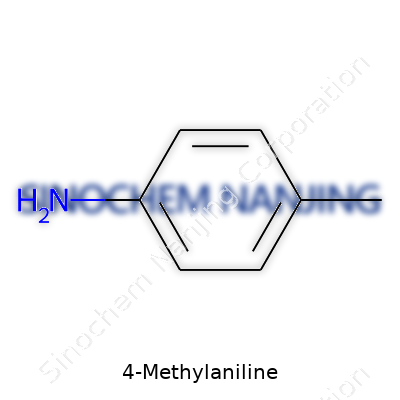
4-Methylaniline pops up in organic chemistry discussions, especially for those interested in industrial dyes and pharmaceuticals. Its molecular formula, C7H9N, captures a lot more than you might guess by just staring at a sequence of elements. For anyone who’s handled aniline compounds in the lab or leaned over chemical process manuals searching for raw materials, 4-Methylaniline brings a sense of familiarity. That methyl group attached to the fourth position changes more than just a number on paper; it influences the physical properties, reactivity, and applications in real products—sometimes in subtle, sometimes in eye-opening ways.
Understanding the Structure: Why the Numbers Matter
I remember trying to memorize the benzene derivatives in college, getting tripped up not by complex names, but by where to put those extra CH3 or NH2 groups. Here, the amino group (-NH2) sits on a benzene ring, and with a methyl (-CH3) group at the para position, the formula unfolds simply as C7H9N. Even so, just rearranging that methyl to a new spot brings a different compound. That’s how precise organic chemistry forces us to be.
Careful placement in molecular structure unlocks new uses or raises fresh safety concerns. The para position throws off fewer electrons towards the amino group, often making 4-Methylaniline slightly less reactive in certain syntheses compared to other isomers. Recognizing that, chemists use this data to tweak reactions, minimize waste, create better pharmaceuticals, and improve industrial yields—all things that ripple through to product safety and cost, and ultimately to patients and consumers.
Real-World Impact and Health Considerations
In my own work, safety briefings around aromatic amines like 4-Methylaniline have never been mere formality. Exposure risks exist—these compounds can be toxic, and even low concentrations warrant a careful approach. The fact that just a single methyl group changes its behavior in biological systems underscores how small differences can have big outcomes. Regulatory bodies classify anilines as potentially hazardous; thus, proper handling protocols must remain a regular conversation in academic labs and manufacturing facilities alike. Strong evidence links improper exposure to serious health effects, so gloves, good ventilation, and rigorous protocol are essential every single time.
Supporting Innovation While Addressing Safety
Modern research keeps finding ways to substitute or safely process materials like 4-Methylaniline. Green chemistry pushes for new catalysts or alternative processes to reduce hazardous byproducts, especially during scale-up in manufacturing. Cooling open containers in a fume hood, using personal protective equipment, or developing substitutes altogether—each step safeguards workers and the end user, while still delivering the performance that industries rely on.
Industry experts and research scientists have a responsibility to relay clear, accurate data about substances like 4-Methylaniline all the way down the supply chain. Sharing clear structure-activity relationships and enforcing transparency in labeling helps prevent incidents and protects everyone, not just chemistry insiders.
Learning from the Formula
Getting a grip on something as focused as the molecular formula of 4-Methylaniline means looking at the bigger context. Those nine hydrogens, seven carbons, and one nitrogen don’t work in isolation. They’re pieces of a puzzle, influencing how we produce dyes, pharmaceuticals, and specialty chemicals used every day.
How should 4-Methylaniline be stored?
What 4-Methylaniline Brings to the Table
4-Methylaniline, also called p-toluidine, finds its way into plenty of industrial routines. It shows up in dye-making, pharmaceuticals, and even some lab research. Just because you won't see it in a grocery store aisle doesn't mean you can turn a blind eye to how it gets stored. Talking with chemists and safety specialists, I've learned that people often focus on using chemicals without paying enough attention to where and how they live in storage. That shortcut leads to headaches—or worse—down the line.
The Risks You Can't Ignore
This chemical gives off a strong smell. It tends to catch fire a lot more easily than you might expect. A single spark or a bump in temperature above room-level might spell trouble. Breathing in vapors or letting it sit on bare skin can mess up your health. OSHA and the CDC warn about these risks; burn accidents and chemical exposures caused by sloppy storage do not just happen in faraway factories—they pop up in university labs and local workshops alike.
Real-World Solutions for Safe Handling
I once visited a site where someone kept 4-Methylaniline on a wooden shelf next to boxes of old paperwork. That setup felt like a recipe for disaster. Fire codes and common sense guide us otherwise. Keeping this stuff in tightly sealed, labeled containers cuts down on leaks. Glass or certain plastics work, but metal might react under some conditions. Anyone working with it regularly knows that cool, dry, and well-ventilated spaces make a difference. Sticking containers inside explosion-proof cabinets, away from sunlight and direct heat, doesn't just tick a training checklist—it prevents costly mistakes.
One mistake I saw in a small lab: storing solvents and acids mixed with organics like 4-Methylaniline on the same shelf. It made cleaning simple, but those chemicals don’t get along well. Corrosive fumes or accidental mixing can spark fires or toxic spills. A better approach is to split up incompatible groups and lock up anything that evaporates easily. Even a single fume hood for the dangerous jars pays off over time.
Training and Labeling: Not Just Formalities
Getting people to read safety data sheets (SDS) before carrying a bottle of 4-Methylaniline around the shop might feel like a waste of time, but it pays in safety. Proper label-making (clear chemical name, date received, expiration date) means anyone can walk up and know the score. Older workers sometimes roll their eyes at frequent training sessions, yet those reminders keep stories of burns and exposures in everyone’s heads.
Disposal and Accident Preparedness
Nobody likes to talk about barrels of hazardous waste, but safe disposal matters as much as storage. Municipal guidelines don’t always map neatly onto the realities of a factory or lab. Partnering with professionals for waste pickup or following EPA guidelines keeps harmful chemicals out of the regular trash stream and protects everyone. Emergency wash stations and spill kits close at hand mean employees won’t scramble during accidents.
Shared Responsibility
Safe use of 4-Methylaniline boils down to treating every bottle with respect. Storage choices affect the whole team and the neighborhood outside. Paying attention costs little in money but carries real weight in personal safety and peace of mind. The right cabinets, labels, and training turn something risky into just another tool that helps us get the good work done.
What are the health hazards associated with 4-Methylaniline exposure?
Worry Behind the Name
4-Methylaniline, sometimes called p-toluidine, comes up in textiles, dyes, rubber, and certain pesticides. The chemical smell from its pale-yellow liquid stays with anyone who’s handled it. Many ignore these smaller everyday exposures, but experience teaches us they're not harmless.
Short-Term Effects Many Ignore
Working near 4-Methylaniline, the first thing you notice: your eyes start to burn, headache inches in, nose tingles, throat feels scratchy. Touching it without gloves causes redness and irritation. Lungs work harder if you breathe too much vapor or mist. It doesn't stop at the surface. Nausea, dizziness, and sometimes confusion hit people who spend too long breathing it in or spill it on their hands. In extreme cases, high exposure can turn nails or lips bluish—a signal that oxygen isn’t carrying the way it should, a classic sign of methemoglobinemia poisoning.
Lasting Damage With Chronic Exposure
Ignoring symptoms over time or brushing off the smell can cost more than comfort. Kidney and liver damage show up quietly. Some workers in dye plants find themselves facing anemia after years on the job. I’ve walked factory floors and seen blue fingers and chronic coughs up close—symptoms no one should ever write off as just “part of the job.” Scientists tie regular exposure with an increased risk of bladder cancer, and the International Agency for Research on Cancer lists 4-Methylaniline as possibly carcinogenic to humans.
Hazards Away From the Factory Floor
Drifting beyond factory work, 4-Methylaniline can show up in water sources or soil if disposal practices slip. Studies show this chemical doesn’t break down quickly in the environment, so leaks or spills linger. Neighbors to manufacturing plants tell stories about odd-smelling water, dead plants, and unexplained headaches. Drinking water with traces, or even living near where it’s used without proper safety steps, means risk spreads throughout a community. Children and older folks grab a bigger piece of that risk since their bodies don’t handle toxins as well.
Prevention Starts With Knowledge and Action
A handful of simple moves curb exposure. Gloves and respirators are worth every penny. Local exhaust ventilation takes vapors out of breathing space. Showering and changing clothes after a shift stop chemicals from following you home. Moves like these sound basic, but skipping steps or using shoddy gear means danger sneaks in. Training helps, but only if everyone knows why small exposures stack up over years. Regulators like OSHA and EPA set limits for workplace air, but nothing matches the power of open eyes and shared stories between workers. If you see sloppy spills or smell something “off,” that’s a red flag. Safe handling, strict waste disposal, and honest conversations can block long-term harm.
What Should Change
Companies need to share risks clearly, not bury them in technical jargon on a dusty data sheet. Everyday workers deserve real-world information—what to look for, when to worry, and how to stay safe. Good ventilation, fast cleanup, regular monitoring, and medical checkups matter more than glossy promises. Researchers keep digging for safer alternatives, but change takes honest voices, tough questions, and a community that refuses to let chemical risks get brushed aside. Transparency, vigilance, and advocacy save lives where silent dangers creep in.
| Names | |
| Preferred IUPAC name | 4-Methylaniline |
| Other names |
p-Toluidine 4-Aminotoluene p-Methylaniline p-Methylbenzenamine 1-Amino-4-methylbenzene |
| Pronunciation | /ˈfɔːr ˌmɛθ.ɪl.əˈnɪl/ |
| Identifiers | |
| CAS Number | p-106-49-0 |
| Beilstein Reference | 876080 |
| ChEBI | CHEBI:18219 |
| ChEMBL | CHEMBL1432 |
| ChemSpider | 7199 |
| DrugBank | DB14133 |
| ECHA InfoCard | 100.058.883 |
| EC Number | 202-440-0 |
| Gmelin Reference | 82129 |
| KEGG | C01426 |
| MeSH | D000438 |
| PubChem CID | 8659 |
| RTECS number | BX9275000 |
| UNII | 4E8Y382YNU |
| UN number | UN2291 |
| CompTox Dashboard (EPA) | DTXSID8020404 |
| Properties | |
| Chemical formula | C7H9N |
| Molar mass | 121.18 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | amine-like |
| Density | 1.008 g/cm3 |
| Solubility in water | slightly soluble |
| log P | 1.680 |
| Vapor pressure | 0.16 mmHg (25°C) |
| Acidity (pKa) | 4.46 |
| Basicity (pKb) | 9.37 |
| Magnetic susceptibility (χ) | -67.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.571 |
| Viscosity | 0.94 mPa·s (25 °C) |
| Dipole moment | 1.37 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 164.3 J⋅mol⁻¹⋅K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 86.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3485 kJ/mol |
| Hazards | |
| Main hazards | Toxic if swallowed, in contact with skin or if inhaled; causes damage to organs; suspected of causing cancer. |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H311, H331, H315, H319, H317, H351 |
| Precautionary statements | P261, P280, P304+P340, P312, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-2-E |
| Flash point | 137 °F |
| Autoignition temperature | 615°C |
| Explosive limits | Explosive limits: 1.2–7% |
| Lethal dose or concentration | LD50 oral rat 1750 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1750 mg/kg (oral, rat) |
| NIOSH | B016 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of 4-Methylaniline: "2 ppm (skin) |
| REL (Recommended) | REL: 2 ppm (8-hour) |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
Aniline 2-Methylaniline 3-Methylaniline N-Methylaniline N,N-Dimethylaniline |

