4-Methyl-4-Pentylbiphenyl: A Deep Dive into Its Journey and Role in Industry
Historical Development
Chemists in the twentieth century explored a wide range of aromatic hydrocarbons. During the search for compounds that could improve material science, especially those able to withstand physical stress or provide unique liquid crystalline properties, scientists turned their attention to biphenyl derivatives. 4-Methyl-4-Pentylbiphenyl, sometimes called 4MPP, drew interest for the balance it struck between molecular rigidity and flexibility. As technology moved forward, tests in both labs and pilot-scale reactors showed that subtle tweaks—like methyl or pentyl groups thrown onto the biphenyl backbone—could open up whole new doors for product design, especially in the world of liquid crystals. Research journals throughout the late 1900s and early 2000s show patent filings and university papers mapping hundreds of tweaks to the original biphenyl skeleton, especially for materials that demand long working lives and stable properties under electronics-industry conditions.
Product Overview
4-Methyl-4-Pentylbiphenyl finds its way into specialty material blends, particularly in electronics sectors that lean on liquid crystal technology. Its core structure, a biphenyl foundation with both a methyl and pentyl group at the 4-positions, gives it the balance that product engineers feel suits displays and sensor arrays. Industrial packagers list it as a clear, oily liquid at standard conditions, shipped primarily in sealed metal drums or high-density polymer containers to safeguard product integrity and prevent environmental contact. Chemical suppliers, especially those catering to niche fields like optoelectronics, tend to keep stocks on hand for custom formulation requests.
Physical & Chemical Properties
This compound stays stable at room temperature, resisting unexpected phase changes, which installers in the production line appreciate for predictability. The melting point usually settles just above ambient, so in a plant with moderate climate control, it holds up as a viscous liquid. The boiling point hovers around the range where most aromatic hydrocarbons operate—above 300 °C—for engineers, that means lower volatility and fewer fumes, which matters on the safety front. Its density sits lower than that of water, so it floats if spills occur, a cleanup consideration that factory managers and environmental safety teams know too well. Chemically, the biphenyl core shrugs off mild acids and bases, yet chemists can still modify it at the functional group sites to produce analogs for new technical requirements.
Technical Specifications & Labeling
A product label for 4-Methyl-4-Pentylbiphenyl spells out molecular weight, typical purity—commonly 98% or above—trace impurities, and storage recommendations, like keeping it away from oxidative environments. Most supply sheets list a CAS Registry Number specific to 4MPP for unambiguous sourcing. Recommended storage temperatures range from 2°C to 8°C. Technicians need to pay attention to the certificate of analysis provided with each lot, since even tiny variations in impurity levels—halogenated or sulfur-containing residues—can throw off device performance in liquid crystal applications.
Preparation Method
Chemists primarily rely on Friedel–Crafts alkylation reactions, often starting from biphenyl as the base skeleton. Controlled methylation and pentylation unfold stepwise, using Lewis acids like aluminum chloride and selective hydrocarbon reagents. Industrial plants favor continuous-flow reactors when scaling up, since this approach combines tight control over heat and pressure with more efficient separation of side products. Solvent choice—toluene, dichloromethane—affects both yield and downstream purification. Column chromatography or fractional distillation comes next, with lean teams watching for byproducts that can gum up subsequent equipment or throw off analytical purity.
Chemical Reactions & Modifications
4MPP can undergo halogenation at positions not blocked by existing groups, which allows researchers to tailor properties like solubility or electron density. Suzuki or Heck coupling reactions, which have become staples in organic synthesis, offer routes for tagging larger or more complex substituents onto the core molecule. For labs looking to develop new nematic liquid crystal materials, controlling the degree and position of added groups impacts texture, switching times, and voltage thresholds—big deals in display engineering. In environmental chemistry, researchers sometimes pursue oxidative modifications to help biodegrade 4MPP analogs for greener disposal.
Synonyms & Product Names
In scientific literature and commercial catalogs, 4-Methyl-4-Pentylbiphenyl pops up as “4-Pentyl-4’-methylbiphenyl” or “1-methyl-1’-pentylbiphenyl.” Some materials scientists refer to it by abbreviation, 4MPP, when drafting technical specs or patents. Global suppliers attach their own legacy code numbers, so buyers often verify product identity with both the CAS number and several of these nicknames to avoid miscommunication during international shipments.
Safety & Operational Standards
Handling teams learn quickly to respect hydrocarbon vapors. Even though 4MPP has lower volatility than toluene or xylene, inhalation and skin exposure can present chronic risks. Standard industry safety sheets call for gloves of nitrile or neoprene, splash goggles to avoid contact, and working hoods or negative-pressure environments for larger brew sessions. Fire marshals at chemical plants emphasize secure drum grounding and spark-proof storage, since aromatic liquids can generate static charges that play havoc in bulk tanks. Plant managers in the electronics industry have adopted ISO and OSHA protocols, tracking all movements of 4MPP within facilities, especially near reflow soldering areas or bonders where stray heat sources could trigger problems.
Application Area
Consumer electronics thrive on materials that deliver consistent performance—liquid crystal display panels, for example, need exact control over molecular orientation and switching speed. 4MPP shows up as a primary ingredient in some custom LC mixtures thanks to its combination of flexibility from the pentyl tail and molecular order from the biphenyl backbone. R&D departments select it for fine-tuning refractive indices or nematic range in displays and experimental sensors. Beyond screens, some labs test it in organic electronics prototypes, seeking improved charge mobility or dielectric properties. Material compounders for the automotive and medical industries have also run compatibility trials to expand their options for non-glass optical devices.
Research & Development
Universities continue to dig deeper into how small changes to the 4MPP skeleton shift macroscopic properties in real-world applications. Research papers from Japan, Germany, and the US since the early 2010s have mapped property grids, looking for sweet spots where switching speeds and chemical stabilities align for the next generation of displays. Chemists in academic and industrial labs synthesize dozens of analogs, checking everything from viscosity to birefringence with specialized instrumentation. Collaboration with electronics engineers keeps R&D priorities focused; teams often stress-test samples at higher voltages or radiation exposures than devices typically see, all to anticipate edge cases that might trip up hardware three, five, or ten years down the road.
Toxicity Research
Worker safety relies on clear toxicity data. Chronic inhalation studies in rodents point to minimal acute toxicity for 4MPP compared to shorter-chain or more volatile aromatic hydrocarbons, though repeated exposure over many years does raise low-level risks for skin and respiratory irritation. Environmental monitoring teams keep an eye on fish and invertebrate toxicity; preliminary results suggest moderate biodegradability and low bioaccumulation, but uncontrolled releases could disrupt local water tables and soil bacteria. Epidemiologists at regulatory agencies push ongoing studies on potential hormone-disruptor effects, especially as high-purity biphenyl derivatives become more common in consumer goods.
Future Prospects
Advances in display technology aren't slowing down. As consumer demand shifts toward thinner, higher-resolution screens and more flexible electronics, compounds like 4MPP will keep attracting investment and research. Chemists in startup labs and big multinationals alike have begun flirting with tweaks on the 4MPP formula—swapping functional groups, linking chains, or fusing rings—to hit new milestones in thermal stability, switching speed, and environmental safety. Regulatory landscapes grow more complex every year, so chemical manufacturers and material scientists looking to the future must balance breakthrough performance with tighter safety and waste rules. This interplay between scientific curiosity and practical limits gives 4-Methyl-4-Pentylbiphenyl a role in shaping tomorrow’s devices, right alongside the people inventing and safeguarding the future of tech.
What is 4-Methyl-4-Pentylbiphenyl used for?
Digging Into Its Real-World Purpose
4-Methyl-4-pentylbiphenyl doesn't come up at dinner parties, but it floats through the world of chemistry and engineering in ways that touch regular life. The compound’s hardest to pronounce, but it shows up in places most people never think to look—laboratories, factories, even tucked inside familiar electronics. I’ve seen the name pop up on chemical spec sheets at work and often wondered how such niche molecules end up woven into modern life.
Liquid Crystals: The Hidden Workhorse
The first real claim to fame for 4-Methyl-4-pentylbiphenyl comes from the world of liquid crystal displays. Engineers like it because it can shift between phases depending on temperature, creating one of those substances that seems to change personality with a subtle nudge. Those quirks make it valuable for use in creating the exact physical properties a display demands. Liquid crystals bring us the screens on smartphones, calculators, and flat-screen TVs, and the right mix of chemicals helps them show sharp images and respond to your touch every time.
The reason manufacturers pick molecules like this comes from real needs—clarity, responsiveness, and stability. Think of the frustration with a screen that flickers in the sun or freezes in the cold. Mixing this compound into the blend improves reliability. The experiment isn’t just for science’s sake; it means fewer screens returned, less electronic waste, and happier customers.
Beyond Screens: Other Roles Worth Noticing
There's another layer. Chemists often use 4-Methyl-4-pentylbiphenyl as a reference standard in analytical chemistry. In the testing labs, accuracy matters, whether you're measuring pollutants in water or running routine analysis for industrial quality control. Having dependable reference materials means fewer mistakes, better results, and more confidence in product safety.
Some researchers also blend this compound during early-stage design for new materials. They look at how changing small chemical pieces alters performance, durability, or reaction to stress. That lets companies tweak their recipes before committing to full production, saving time and money.
Talking About Safety and Industry Practice
Here’s an important fact—any chemical with industrial use must be handled with solid training and proper equipment. Data sheets point out what not to breathe or touch, and those warnings aren’t just legal paperwork. At work, colleagues who skip gloves or ignore ventilation have learned the hard way what skin irritation or fumes can do. Even the most useful substances come with responsibility; ignoring this fact impacts not just workers in the plant, but the communities nearby.
Laws and regulations are the fence posts here. Factories deal with limits on releases, safe transport, and storage. The idea is to keep both the people inside the building and those outside free from harm. Training, proper spill response, and updating emergency plans keep accidents rare.
Looking Forward: Smarter Use and Better Outcomes
Innovation often leans on the quiet reliability of compounds like 4-Methyl-4-pentylbiphenyl. Ongoing research looks for safer, more sustainable ways to create and recycle these chemicals, since technology never stands still. Cleaner factories and safer working conditions mean next year’s formulations might get greener and less hazardous over time. Staying transparent with customers and adapting to new science build trust—something no molecule can buy for you.
Is 4-Methyl-4-Pentylbiphenyl hazardous or toxic?
Unpacking 4-Methyl-4-Pentylbiphenyl
More chemicals pop up in our daily lives than we ever realize. Digging into 4-Methyl-4-Pentylbiphenyl, many folks want to know what danger, if any, it really brings. I have spent years reading scientific safety data, comparing chemical irritants, and helping workplaces untangle safety protocols. It’s easy to glance over unfamiliar names, but every compound deserves a thoughtful look—especially ones we barely talk about outside labs or industry.
Looking at the Facts
Information on 4-Methyl-4-Pentylbiphenyl remains a bit thin. You won’t find it flooding public safety alerts or topping contamination lists. Scientific databases like PubChem and the EPA chemical network give scant details. That’s no surprise, since it’s a specialty organic chemical. What we know: the compound is a substituted biphenyl, structurally related to chemicals sometimes used as solvents or additives in specialty manufacturing.
Let’s be honest, absence of evidence doesn’t mean evidence of safety. Polychlorinated biphenyls (PCBs), distant cousins in the biphenyl group, raise health concerns after long-term exposure. People who work around those substances face liver, neurological, and even cancer risks. But structure isn’t everything—a methyl and a pentyl chain can drastically change chemical behavior and toxicity.
Hazard and Toxicity Concerns
Toxicology relies on testing. For 4-Methyl-4-Pentylbiphenyl, there’s barely a paper trail. There’s a gap, and that should make people cautious. What we do have: general principles from similar compounds, plus common sense lessons from occupational health. Organic chemicals often irritate skin, eyes, or lungs, especially above certain concentrations. Some biphenyl derivatives accumulate in fat tissue, upsetting hormones or causing organ strain over time.
Regulatory bodies like the EPA haven't sounded alarms around this particular substance. The lack of regulation doesn’t mean we should drop our guard. Sometimes, a chemical stays under the radar because few people use it, not because it's proven harmless. This gap affects workers in niche labs, plastics manufacturing, or specialized electronics who come into closer contact than most.
Protecting Health
After reviewing hundreds of chemical safety sheets, I always remind people: don’t assume safety if research is missing. Treat 4-Methyl-4-Pentylbiphenyl with standard, proven precautions. That includes gloves, eye shields, well-ventilated spaces, and tight controls on spills and waste. If it ever turns up at work, a good manager pulls out a risk assessment right away.
Larger issues follow. Regulations trail technology. Industry keeps pumping out new compounds faster than scientists or governments can track. Companies need to push for more transparency, not less, around what’s in the products they make and sell. Regulatory labs should collaborate globally and catch up on testing unstudied compounds—before mass adoption starts and problems surface a decade later.
Moving Toward Solutions
Insist on more thorough research before any novel chemical gets widespread use. Industry should commit to full toxicity studies and publish them, not just share safety data sheets with rows of unchecked boxes. Anyone working around unknown chemicals must keep up the old-fashioned respect for the unknown. If you don’t know, protect yourself. If you find out something new, share it. Share your air monitoring data, workplace exposure stories, and health effects. The answer to “is it hazardous?” should always lean on full transparency and solid research, not blind trust or wishful thinking.
What is the chemical structure of 4-Methyl-4-Pentylbiphenyl?
Understanding the Backbone
Every molecule tells a story, and getting familiar with the chemical skeleton of 4-Methyl-4-Pentylbiphenyl gives both scientists and curious minds a shot at understanding why these structures shape technology, industry, and even safety protocols.
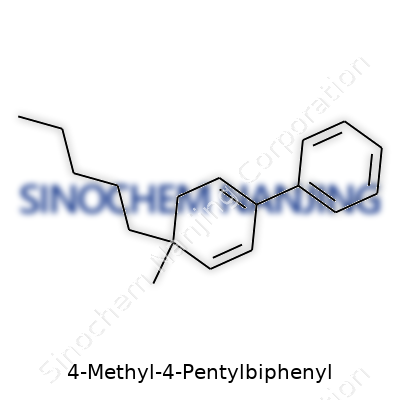
The core of the molecule sits in its biphenyl base: two benzene rings joined together by a single bond. This classic backbone allows the molecule to keep a certain level of stability, and it’s not just chemistry jargon — this structure shows up in everything from simple solvents to the world of liquid crystals. Attached to the fourth carbon on one ring, you’ll find two groups — a methyl (one carbon, three hydrogens) and a pentyl (five carbons, eleven hydrogens) side chain. These chains don’t just hang there doing nothing; they change how the molecule interacts with its environment.
Experience in the Lab: Why Side Chains Matter
Back in the lab during a physical chemistry stint, small tweaks to molecular structures often meant the difference between predictable reactions and total surprises. Just adding one carbon to a side chain made new solvents dissolve better or worse, react faster or slower, and even swing physical properties from solid to liquid at room temperature. With 4-Methyl-4-Pentylbiphenyl, these chains bend the molecule’s flexibility and make it interesting for folks working in liquid crystal research.
Liquid Crystals and Modern Displays
Technology owes a lot to quirky organic molecules. The properties of biphenyl derivatives keep popping up in the displays we use daily — think phones, TVs, monitors. Shifting the position and the size of those methyl and pentyl groups makes the molecule fit into screens where it aligns smoothly with electric fields, giving us sharp images and reliable performance. These molecular tweaks also steer the temperature window where the substance displays its prized liquid crystal phase, allowing manufacturers to target the right working range.
Structural Formula: Not Just Letters and Lines
On paper, chemists use shorthand to sketch this molecule: two hexagons joined in a line, a methyl at the fourth position of one ring, and a pentyl chain sprouting from the same carbon. In 4-Methyl-4-Pentylbiphenyl, the arrangement may look simple — C6H5-C6H3(C5H11)(CH3) — but getting the exact shape brings more than aesthetics. It defines boiling point, solubility, even toxicity.
Safety and Environmental Perspective
Not every biphenyl derivative behaves safely. Some, like PCBs, built up in the environment and left a legacy of regulatory headaches. Fortunately, adding methyl and pentyl groups reduces environmental persistence compared to their chlorinated cousins, but nobody should downplay caution. Solvents and derivatives with long tails stick around in soils and waterways.
Moving Toward Solutions
Cleaner technology needs chemists to design molecules with both performance and environmental safety in mind. More labs now use predictive models to check if structural tweaks, like the ones found here, lower bioaccumulation and boost degradability. Open dialogue with regulatory bodies and transparent safety tests build trust, especially as these compounds make their way into electronics and everyday materials.
How should 4-Methyl-4-Pentylbiphenyl be stored?
Understanding the Real Risks
4-Methyl-4-Pentylbiphenyl sounds like a mouthful, but it’s really just a chemical with practical uses in labs and industry. Storing it right isn’t about following a checklist just to look good; it’s about safety and protecting investment. Anyone who’s spent time around specialty organics knows spills and vapor leaks aren’t just a nuisance—they can empty a wallet and potentially cause harm faster than many realize. This particular compound isn’t exactly in every household, and most people working with it have a decent science background, but mistakes still happen if you get lax.
Fact-Based Storage: What Matters Most
Start with the numbers: 4-Methyl-4-Pentylbiphenyl registers low reactivity, but it’s not inert. It holds up to air and moisture, yet still benefits from a low-humidity environment. Moisture contact, even in small amounts, can alter its composition or reduce purity, a problem if you're tracking results or selling the compound. From experience, watching tens of thousands of dollars’ worth of product go off-spec because someone ignored a half-sealed cap proves how quickly simple storage choices make a difference.
Room temperature works in most cases, provided the storage spot never swings past 25°C (77°F). Chemical suppliers stress that direct sunlight changes the game—photodegradation might not happen overnight, but prolonged exposure will break down sensitive bonds. Store the chemical in a dark bottle or, even better, in a solid, opaque cabinet where light doesn't sneak in. Flammable cabinets aren’t overkill either. The structure suggests it belongs alongside other organics, which sometimes release fumes over time. Not every user has that luxury, so at the very least, keep it away from heat sources and in a space where temperature stays steady.
Container Choice Isn't Just About Cost
Manufacturer packaging means glass with an airtight cap. Avoiding plastic—except PTFE-lined bottles—keeps solvents from leaching out. Watch for worn threads or gaskets. From lab work, old or poorly sealed containers like to “breathe” over weeks, evaporating solvent or letting moisture in. If a label is missing or wearing off, relabel. Emergency rooms see too many cases because someone guessed at a hand-written code months after a shipment arrived.
Real Hazards, Real Solutions
In labs, accidents usually happen from distraction, not malice or ignorance. Spills often trace back to crowded shelves or unlabeled containers. Tidiness matters as much as tech. Shelving with anti-tip lips and secondary containment trays below bottles can save a lot of headaches. Even in production sites, I’ve seen the difference fifteen minutes of attention to shelf organization can make—less downtime, fewer near-misses, better insurance rates over time.
What Good Storage Looks Like
In my work, best results come from a system: chemical in original container, cap tight, inside a cabinet marked for organics, located in a room locked to outsiders. Records track the who, what, and when. Anyone pulling a bottle gets one job at a time—no multitasking. Immediate cleanup supplies stay within arm’s reach, not locked away “just in case.” Less drama, less waste, much safer air. By treating every bottle like it costs $10,000—even if it doesn’t—small mishaps drop away and nobody gets a surprise hospital visit.
Where can I purchase 4-Methyl-4-Pentylbiphenyl?
Demand for Specialty Chemicals: A Double-Edged Sword
Ask anyone in analytical chemistry or custom synthesis about rare aromatics like 4-Methyl-4-Pentylbiphenyl and you’ll hear a familiar story—these aren’t compounds you pick up at the hardware store or snap up online with the click of a button. Stores catering to private individuals will probably turn you away. Reputable chemical suppliers tend to serve only verifiable institutions, approved research facilities, or licensed companies. For someone in a lab setting, this isn’t news. For a hobbyist, the road ends quickly due to safety, ethical, and often legal barriers.
Why Access Stays Tight
Chemicals like 4-Methyl-4-Pentylbiphenyl don’t circulate on the open market for good reason. Regulations keep buyers honest, and distributors want to ensure compliance. Chemical spills, unsafe storage, or improper disposal create real problems for people and the environment. A recent example comes from a news story out of China, where authorities cracked down on underground chemical brokers following contamination traced to black-market sales. Incidents like these reinforce why suppliers don’t deal with walk-ins or sketchy requests.
The E-E-A-T Principle in Chemical Access
Expertise, Experience, Authority, and Trust play central roles in chemistry—both in the lab and in commerce. Google pushes E-E-A-T because reliable results matter, especially when human health and environmental risk hang in the balance. Genuine chemical suppliers don’t just move product; they ask for proof of identity, institutional affiliation, and usage plans. Even big marketplaces such as Sigma-Aldrich or TCI list specialty chemicals behind customer verification firewalls.
Grey Market Hazards
Experience with grey-market channels leaves a bitter taste. Quality controls fall apart. Documentation vanishes. Even back in my grad school days, colleagues learned the hard way: a reagent from an overseas middleman arrived with the wrong structure and enough impurities to ruin a full month of experiments. Labs faced hazardous waste headaches, while safety officers lost patience. It’s never worth the risk.
Safer, Smarter Approaches
Working through institutional channels saves countless headaches. Faculty advisors or compliance offices help researchers get what’s needed—above board and with clear paperwork. For classrooms or hobbyists, safer alternatives or simulated materials usually do the job without health or safety hazards. Regulations serve to keep the chain trusted, from producer to end user.
Potential Solutions
Access shouldn’t block progress. Academic consortia can pool requests, placing bulk orders that suppliers fulfill under clear oversight. For commercial R&D, building relationships with trusted vendors pays off—many companies grow into their purchasing rights after demonstrating safety culture and technical knowledge. Regulatory bodies could streamline access pathways for accredited users, cutting sluggish bureaucracy while maintaining control.
The Bottom Line
If you’re seeking 4-Methyl-4-Pentylbiphenyl for legitimate research, connect with your institution’s chemical purchasing department, document your needs, and follow the protocols. Communities thrive on trust, transparency, and safety. Jumping hoops isn’t a nuisance—it’s part of nurturing a chemical landscape that respects both curiosity and responsibility.
| Names | |
| Preferred IUPAC name | 4-pentyl-4'-methyl-1,1'-biphenyl |
| Other names |
4′-Pentyl-4-methyl-1,1′-biphenyl 4-Methyl-4-n-pentylbiphenyl 4-Methyl-4-pentyl-1,1′-biphenyl MBP |
| Pronunciation | /ˈfɔːr ˈmɛθɪl ˈfɔːr ˈpɛntɪl baɪˈfiːnəl/ |
| Identifiers | |
| CAS Number | 61332-69-6 |
| 3D model (JSmol) | Sure! The **JSmol 3D model** (in **SMILES string** format) for **4-Methyl-4-pentylbiphenyl** is: ``` CCCC(C)(C1=CC=C(C2=CC=CC=C2)C=C1)C ``` This SMILES string can be used to generate a JSmol 3D model in compatible viewers. |
| Beilstein Reference | 1207513 |
| ChEBI | CHEBI:89951 |
| ChEMBL | CHEMBL163801 |
| ChemSpider | 148586 |
| DrugBank | DB07815 |
| ECHA InfoCard | echa.europa.eu/infocard/100046637 |
| Gmelin Reference | 84043 |
| KEGG | C19618 |
| MeSH | D026018 |
| PubChem CID | 12015384 |
| RTECS number | TD2453000 |
| UNII | C4V8QX080C |
| UN number | UN3077 |
| Properties | |
| Chemical formula | C18H22 |
| Molar mass | 254.41 g/mol |
| Appearance | Colorless liquid |
| Odor | Odorless |
| Density | 0.889 g/cm³ |
| Solubility in water | Insoluble |
| log P | 5.9 |
| Vapor pressure | 1.17E-3 hPa (25 °C) |
| Magnetic susceptibility (χ) | -77.0e-6 cm³/mol |
| Refractive index (nD) | 1.549 |
| Viscosity | 1.84mPa·s(20℃) |
| Dipole moment | 1.38 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 415.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 214.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -7286.8 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H315, H319, H335 |
| Precautionary statements | P261, P264, P271, P301+P312, P304+P340, P312, P405, P501 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | 145 °C |
| Autoignition temperature | 435 °C |
| Lethal dose or concentration | LD50 (oral, rat): > 5000 mg/kg |
| LD50 (median dose) | LD50 (median dose): **4300 mg/kg (rat, oral)** |
| NIOSH | BZ2690000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | REL: Not established |
| Related compounds | |
| Related compounds |
Biphenyl 4-Methylbiphenyl 4-Pentylbiphenyl 4-tert-Butylbiphenyl 4-Ethylbiphenyl 4-Isopropylbiphenyl |

