4-Methoxy-4-Methyl-2-Pentanone: Where Science Meets Everyday Chemistry
Looking Back: The Road to Discovery
Long before digital research databases and streamlined synthesis processes, chemists set out to understand and shape the molecules around them. 4-Methoxy-4-Methyl-2-Pentanone, sometimes called Methyl Isobutyl Ketone Ether, comes out of that tradition of curiosity and dogged experimentation. Its story fits right alongside the evolution of organic chemistry in the twentieth century, a time when solvents branched out into uncharted territory and folks in labs searched for ketones and ethers that could solve new problems in industry and research. From the perspective of a working chemist, discovering ways to tweak existing compounds and inventing fresh ones wasn’t only about novelty—it was about finding better, safer, or more efficient solutions for real-world challenges.
Understanding This Compound
Some hear the name 4-Methoxy-4-Methyl-2-Pentanone and think exotic; others link it with core laboratory work. This compound falls into the family of ketones and features a methoxy group sticking out from the fourth carbon in the chain, with a methyl group keeping it company. That arrangement matters since the structure shapes how it behaves, how it smells, and even whether it dissolves easily in certain solvents. A technical breakdown points to its colorless appearance and moderate volatility. Compared to more famous chemicals, it hardly gets noticed outside of professional circles, but in practice, it brings subtlety and specificity to many molecular tasks.
Properties That Mean Something in Practice
At room temperature, 4-Methoxy-4-Methyl-2-Pentanone stays liquid—an automatic plus in systems needing reliable handling. It offers a lower boiling point than some related ketones and shows a respectable resistance to water, echoing traits useful for separating layers in extractions or speeding up reactions without gumming up apparatuses. Its mild odor, nowhere near as harsh as acetone, has drawn attention in labs that value comfort and air quality, especially with growing safety concerns and pressure from workplace regulations. Fewer folks want to spend hours near noxious solvent fumes, and chemists learn to appreciate every compound that leans toward a safer sensory experience.
Labels and Specs: What Matters on the Bench
Working in the lab, you care about more than the chemical formula. Labels need to share concentrations, list common contaminants, and nail down limits on moisture and impurities. For 4-Methoxy-4-Methyl-2-Pentanone, clear labeling matters. Users look for any sign of peroxides, along with the percent of active substance, since small shifts can hurt a whole day’s work. Proper bottle handling and storage — away from open flames, sunlight, and oxygen-rich environments — cuts down risk, and training techs and researchers on these points grows more important as expectations for safety get tougher. Pure substances with established batch records and testing data, not just mysterious liquid in an unlabeled jar, should be the norm, but shortcuts still tempt some users, risking both safety and results.
The Art of Making It
Synthesizing 4-Methoxy-4-Methyl-2-Pentanone starts with common chemical building blocks, but the route takes some ingenuity. Alkylation reactions, where simple molecules like methyl or methoxy donors merge with a suitable ketone backbone, demand precise control. Catalysts, temperature settings, and solvent choices all influence yield and purity. Having spent long hours in synthetic chemistry, I’ve seen how tricky it gets to avoid side reactions, particularly with small modifications that can create unwanted byproducts. Scaling up from milligram research batches to kilogram supplies for industry adds another layer—what works in a glass beaker throws curveballs in a stainless-steel reactor. Recipes in journals help, but every team ends up refining their steps based on real-world quirks of equipment and raw materials.
What Happens to It in a Reaction
The structure of 4-Methoxy-4-Methyl-2-Pentanone means it weaves well into a variety of chemical reactions. The ketone group responds to reductions and condensations, while the methoxy side attracts tests of nucleophilicity. People use this compound to make other molecules, serve as a solvent for blends, or test selective reactivity. In fact, its modest reactivity compared with plain methyl ketones can be a plus when you want more control and fewer surprises. Research journals mention a range of modifications, from classic Grignard reactions to attempts at breaking or shifting the ether group. These pathways link it with larger families of fine chemicals and intermediate compounds, serving the worlds of pharmaceuticals, specialty coatings, and even flavor or fragrance work.
Say It Another Way: Synonyms in the Wild
If you cut through the chemistry jargon, you’ll come across names like Methyl Isobutyl Ketone Methyl Ether or just MIBK Methoxy Ether. Having multiple names can frustrate anyone ordering supplies or combing the literature for new applications. I’ve run into shipments delayed over naming confusion, highlighting the real-world impact of what looks like an academic quirk. Cross-referencing synonyms on Safety Data Sheets and in technical catalogs cuts confusion and can make or break the smooth delivery of research projects or industrial runs.
Playing by the Rules: Safety and Operations
No one wants to cut corners with chemicals, especially something volatile and organic. 4-Methoxy-4-Methyl-2-Pentanone needs the usual respect for its flammability and potential health effects. Direct skin or eye contact brings discomfort, and inhaling vapors can irritate lungs. Good ventilation—sometimes overlooked in smaller labs—makes a world of difference. I’ve worked with folks who downplayed the importance of gloves and safety goggles, only to regret it after minor accidents. Today’s safety standards expect airtight labeling, spill procedures, and storage in fire-safe cabinets. Routine training, spill kits, and good habits go a long way to preventing trouble and ensuring that working with this compound stays as safe as possible.
Where It Fits: Real-World Uses
In industry, 4-Methoxy-4-Methyl-2-Pentanone doesn’t draw much attention from the public, but technical circles know its value. As a solvent, it can dissolve and carry resins or polymers, making it useful in customized coatings. In laboratories, it steps in during syntheses where more reactive solvents might create headaches or extra waste. Some research even points to specialty uses in flavors and fragrances—tiny tweaks in structure can open doors to new smells or stabilization tricks. Its selectivity in chemical reactions helps in pharmaceutical research, where unwanted byproducts can gum up costly purification steps. From personal experience, choosing a solvent like this for a tricky crystallization can sometimes mean the difference between success and hours of troubleshooting.
Science on the Move: Looking Ahead in Research
Research into ketone ethers like 4-Methoxy-4-Methyl-2-Pentanone continues to evolve, especially as industries demand greener and safer chemicals. Teams test its performance in new synthetic strategies, or swap it into reaction schemes once dominated by older solvents. Journals report on its stability and performance under stress, as well as clever derivatizations. There’s a push to explore how modifications affect not only lab work but also waste streams and environmental impact. Universities and companies share knowledge at conferences about potential recycling or reuse approaches, hoping to keep costs in check and improve sustainability.
Digging into Toxicity and Regulation
Safety doesn’t take a back seat, especially as new data emerges around exposure risks. The available toxicity data, though not as comprehensive as for some industrial solvents, suggest it requires careful handling. Studies often target irritant effects and the potential for chronic issues with repeated exposure. In my experience, following strict regulatory guidelines protects both workers and the environment, and staying up to date on evolving recommendations is crucial. Government agencies and workplace health groups press for more thorough long-term studies, ensuring future generations won’t inherit the mistakes of the past.
What’s Next?
Future prospects for 4-Methoxy-4-Methyl-2-Pentanone depend on the intersection of regulation, sustainability goals, and the constant march of research. The move toward greener chemistry demands alternatives to hazardous solvents and insists that every compound offers not just utility but also a gentler profile for workers and ecosystems. Companies search for new uses in specialty coatings, pharmaceuticals, and custom synthesis, while researchers test tweaks that could reduce risk or boost utility. It’s an ongoing process—one that challenges chemists to weigh convenience against responsibility and keep searching for improvements that benefit not just science but society at large.
What is 4-Methoxy-4-Methyl-2-Pentanone used for?
Getting to Know This Compound
4-Methoxy-4-methyl-2-pentanone sounds just as complex as it is: a chemical with a specialized place in modern industry. The folks who handle everyday coatings, inks, specialty resins, or adhesives, often work with this compound, usually under the name methyl isobutyl methyl ketone (MIBK-methoxy). It isn’t some random chemical stashed on laboratory shelves. Over years of working in operations and quality labs, I came to recognize how essential certain solvents are—even if most people never cross paths with them.
In the Paint and Coatings World
Manufacturers dealing with paints and coatings look for predictable drying times, stability in their recipes, and a finish that meets demanding standards. 4-Methoxy-4-methyl-2-pentanone stands out as an efficient solvent for resin systems. Its unique structure provides a blend of volatility and solvency. Fast-drying solvents flash off too quickly for some jobs, while slow-evaporating ones can gum up the works. This compound offers a practical middle ground, helping resins dissolve and level smoothly without delays. The experience of working on an automotive refinishing line underscored just how finicky finishing can get—an effective solvent supports consistency and reduces costly defects.
Ink Formulations and Printing
Printers care about more than color and design. Whether making lotions for packaging or catalog graphics, inkmakers need chemicals that promote flow, prevent blockages, and dry at the right rate. 4-Methoxy-4-methyl-2-pentanone fits into this world as a co-solvent. Inks using this compound spread evenly and dry without clogging presses or leaving residues. Having sat through enough frustrating production runs where dried ink built up on rollers and caused downtime, I learned that the right solvents often mean the difference between a clean shift and a stressful one.
Specialty Adhesives and Cleaners
Some adhesives demand precise control over stickiness, set time, and spread. Traditional solvents can leave residue or make fumes unbearable for workers. 4-Methoxy-4-methyl-2-pentanone becomes valued because it dissolves certain polymers that don’t play well with more caustic alternatives. In my years of overseeing workplace safety, I noticed that choices like this can affect both quality and worker well-being.
Beyond adhesives, companies use this compound in cleaning agents for industrial equipment and electronics. Its chemical profile helps lift tough residues without attacking sensitive components or releasing harsh odors. Operators appreciate products that don’t leave them reaching for ventilation masks or scrubbing for hours.
Safety, Supply, and the Road Ahead
Every chemical used in the shop or plant comes with its own risks. 4-Methoxy-4-methyl-2-pentanone shows low acute toxicity compared to more notorious solvents, but that doesn’t mean anyone can be reckless with storage or disposal. Spills, fumes, and waste streams demand robust procedures and training. Greener alternatives continue to surface, but practical, affordable replacements often take years to meet regulatory and performance hurdles.
Tracking supply chains also matters. Many specialty solvents depend on a handful of global manufacturers. Any hiccup—from war to trade embargoes to natural disaster—ripples out fast. Building supplier relationships and updating chemical management systems remain key tasks for anyone depending on solvents like this. Some companies begin to reformulate around safer, more renewable blends, but each change requires real-world testing—not just a quick literature search.
4-Methoxy-4-methyl-2-pentanone doesn’t make headlines, but it keeps industrial wheels turning. Its ongoing use reminds us how seldom the building blocks of modern manufacturing get any glory, even as they shape our day-to-day world.
What are the safety precautions when handling 4-Methoxy-4-Methyl-2-Pentanone?
Understanding the Risks
4-Methoxy-4-Methyl-2-Pentanone serves as a solvent in paint, coatings, and other chemical processes. This chemical brings a unique profile but also comes with some hazards. In my years spent working in lab environments, I’ve seen many talented people cut corners or overlook personal safety until an incident forces a change. Many solvents in labs give off harmful vapors, and this one belongs to that club. It can irritate skin, eyes, and lungs and may cause headaches or worse after long exposure.
Preparation: Dressing for Protection
Before opening that container, think about what you wear. Ordinary clothes and thin gloves give a false sense of security. Nitrile gloves hold up to most organics, so I always kept boxes of them within reach. Goggles, not glasses, block splashes from the sides. Lab coats catch drips—plus, a good pair of chemical-resistant aprons and closed shoes keep you safer.
Ventilation Makes All the Difference
Over the years, I learned not to trust your nose—by the time you smell vapors, you may have breathed too much. Fume hoods or well-ventilated workspaces help remove solvent fumes before they reach your lungs. Portable exhaust fans work, but nothing beats a proper hood. Indoor air quality often gets ignored since fumes spread quickly and linger. Even if a chemical smells sweet or mild, that doesn't mean it's harmless.
Safe Handling Practices
Spills happen, even to careful folks. I stored solvents in labeled, sealed containers with secure caps. Transfer liquids using pumps or pipettes instead of pouring straight from a bottle. It’s easy to get overconfident and skip steps, but splashes regularly surprise people—especially when a bottle slips. If you spill, wipe it up with absorbent material designed for chemicals, followed by a washdown of the area. Don’t grab the closest rag, since many materials break down and spread the mess further.
Know What to Do in an Emergency
In the lab, knowing where the nearest eyewash or emergency shower stands makes a huge difference. I saw one person rush for water in a panic, trip, and nearly add a broken arm to their troubles. Practice walking through exits, showers, and eyewash stations before starting real work. If something splashes onto your skin or into your eyes, wash with cool running water for at least 15 minutes—every second counts. Inhalation issues require fresh air fast.
Storage and Waste Disposal
Always keep chemicals out of sunlight and store them away from heat sources. 4-Methoxy-4-Methyl-2-Pentanone has been known to react with strong oxidizers, so check storage compatibility charts before mixing different chemicals on the same shelf. Collect solvent waste in closed, labeled containers and follow your facility’s disposal procedures. Pouring solvent down a drain doesn’t just hurt the environment—it often breaks local laws and can get everybody in trouble.
Building a Culture of Safety
Everyone working with this solvent, or any similar material, benefits from staying updated on the latest safety guidelines. Training and refreshed knowledge save lives. Unsafe shortcuts may speed up a job for a moment but can lead to months of health problems.
What is the chemical formula and structure of 4-Methoxy-4-Methyl-2-Pentanone?
Taking a Closer Look at 4-Methoxy-4-Methyl-2-Pentanone
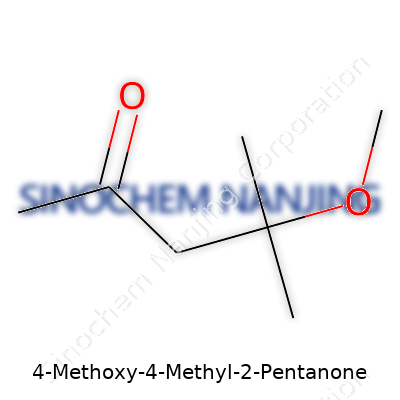
Chemistry gives names like 4-Methoxy-4-Methyl-2-Pentanone to keep things precise, but every name tracks back to a specific arrangement of atoms. With this compound, the story off the bat is in the formula: C7H14O2. Every bit of that formula connects to how the molecule behaves, where it ends up, and why chemists find it useful.
In practice, you get seven carbon atoms, fourteen hydrogens, and two oxygens. Folks with a little experience in organic chemistry can spot that the backbone stretches out over five carbons (pentanone), with a ketone group on the second carbon. The methyl group (a one-carbon, three-hydrogen arm) and a methoxy group (an oxygen with a methyl attached) crowd onto the fourth carbon. This setup means there are two notable "branches" sticking out from that chain, each changing the properties of the molecule.
Drawing the Structure — Why Position Matters
I’ve drawn 4-Methoxy-4-Methyl-2-Pentanone on a lab notebook more than once. It helps to lay out:
- The straight five-carbon spine (pentanone part)
- An oxygen double-bonded to the second carbon (the ketone functional group)
- Both a methoxy group (–OCH3) and a methyl group (–CH3) attached to the fourth carbon
This type of structure makes it clear why it has a unique boiling point and why it dissolves better in some solvents than others. The branching on the fourth carbon kicks up some steric hindrance, crowding the molecule and affecting how it fits together with others. The methoxy group isn’t just a detail — its presence can change chemical reactivity and, in some cases, introduces a whole new layer of polarity.
Relevance in Everyday Practice and Industry
Beyond naming and drawing, chemists lean on 4-Methoxy-4-Methyl-2-Pentanone as an intermediate. I’ve seen it listed in syntheses aiming for specialty chemicals, flavors, and pharmaceutical applications. That methoxy group stands out since it can be swapped out or built upon, giving chemists another handle to manipulate the molecule. Processes that depend on precise control over solubility, boiling points, or selective reactivity can take advantage of these structural tweaks.
Companies want chemicals that offer both stability and flexibility. With this backbone, researchers can introduce new side groups or tweak the molecule for a specific profile — higher solubility, different melting points, or better stability during manufacturing. Its controlled reactivity helps minimize byproducts and simplifies purification, which makes lab techs and engineers a little happier at the end of the day.
Addressing Hazard and Environmental Concerns
No conversation about organic chemicals feels right without talking about health and safety. The presence of the ketone and methoxy groups puts this molecule on the watchlist for potential inhalation or dermal risks in unventilated spaces. Solvent handling safety matters here, both in academic labs and in industry. I remember an incident during a late-night lab session where a small spill taught us the sting of not double-checking gloves and ventilation — lessons worth sharing.
With stricter waste disposal rules, environmentally responsible labs track compounds like this from purchase to disposal. Teaching new lab workers about safe handling and low-impact disposal routes pays off in reduced spills and less toxic load on the environment. Guidelines don’t always jump out as thrilling reading, but careful adherence keeps both chemists and neighbors safer down the line.
Supporting Advancement with Chemistry Facts and Practical Solutions
Understanding the structure of C7H14O2 helps anyone working with 4-Methoxy-4-Methyl-2-Pentanone choose the right protocols, plan safe handling procedures, and invent solutions that keep pace with shifting regulations. Sharing chemical know-how, whether in the classroom or on the factory floor, makes the hurdles of daily lab life a little easier to clear.
How should 4-Methoxy-4-Methyl-2-Pentanone be stored?
Think Beyond a Basic Shelf
Working in a lab brings its share of risks, but smart handling of chemicals makes a big difference. 4-Methoxy-4-Methyl-2-Pentanone looks harmless enough in a simple drum or bottle, but there’s a reason for strict chemical storage rules. This solvent isn’t just another bottle you label and toss on the shelf. I remember my first week running a storeroom, hearing about a near-miss after someone stored a similar compound beside acids. The strong odor still lingers with me, reminding me that shortcuts don’t pay off.
Physical Properties Make a Difference
Storing 4-Methoxy-4-Methyl-2-Pentanone without considering its flash point and volatility is asking for trouble. This liquid falls into the flammable category, with its flash point around 48°C (118°F). Even a warm summer day could nudge those bottles closer to danger if you leave them near a heat source. Unlike household cleaners, a single spark or an old lightbulb can turn a casual mistake into a headline-worthy accident.
Keep It Cool and Ventilated
Experience has shown that keeping flammable solvents cool is non-negotiable. Dedicated flammable storage cabinets shield containers from temperature swings. Heat builds up fast even in closed rooms, especially if you store chemicals near south-facing windows or old radiators. I’ve seen what happens when someone stashes a can in a stuffy supply closet; the air grows heavy, headaches start, and before long, people are wheezing. Good airflow is a friend here, making sure fumes don’t build up.
Original Containers and Clear Labels Matter
Repackaging might seem harmless, but it opens the door to leaks, guessing games, and dangerous mix-ups. Manufacturers ship 4-Methoxy-4-Methyl-2-Pentanone in sturdy, chemical-resistant bottles for a reason. Keeping these labels visible means anyone who grabs a bottle can check hazard warnings right away, and this avoids costly confusion. During inspections, unlabeled containers draw unwanted attention from regulators, and they’re right to ask tough questions about safety.
Keep Away From Reactives and Water
Combining solvents with oxidizers, acids, or even water can spell disaster. Cigarette breaks outside a lab don’t pose as much trouble as a wet shelf or a drip from bad plumbing above a storage rack. In my former workspace, a leaky pipe once dripped onto the top shelf, and only a quick mop-up spared us from mixing unknown substances. Keep 4-Methoxy-4-Methyl-2-Pentanone well away from things like hydrogen peroxide or bleach to avoid violent reactions and fires.
PPE and Emergency Planning
Even with careful storage, accidents happen. Having goggles and gloves nearby protects against splashes, and spill kits tame minor leaks. Training pays off—I remember an exercise where a rookie grabbed the wrong kit and spread flammable dust. Having clear steps written out and running drills keeps the response sharp and avoids panic.
Regulatory Compliance Isn’t Just Red Tape
Rules aren’t just there to slow work down. The Occupational Safety and Health Administration (OSHA) and Environmental Protection Agency (EPA) both dig into chemical storage. Their standards reflect lessons learned the hard way by plenty of labs and manufacturers. Keeping records tidy and storage practices updated keeps fines off your back and, more importantly, everyone safe.
Finding Solutions in Routine
Storing 4-Methoxy-4-Methyl-2-Pentanone safely isn’t about fancy tech or big budgets. Consistent handling, working with the right containers, and sticking to routine checks cut down risk. The best results come from treating every bottle as if it might surprise you—because sometimes it does. As the old lab manager used to say, “Respect the chemical, and it’ll respect you right back.”
What are the potential health effects of exposure to 4-Methoxy-4-Methyl-2-Pentanone?
What Happens If You Breathe or Touch It?
Many chemicals in modern industry end up in air, water, or straight onto skin because of spills or daily work. 4-Methoxy-4-Methyl-2-Pentanone sometimes goes by the trade name ‘MIBK acetate.’ Factories making paints, cleaning agents, and coatings use it for its solvent power. Not everyone on the shop floor knows about its risks, and that’s a problem.
Inhaling this stuff can lead to headaches, dizziness, or feeling faint. These symptoms come up because the chemical tampers with the nervous system. Breathing a lot of it at once, or spending too long in a poorly ventilated workshop, can leave someone lightheaded, sick, or even unconscious. Nose, throat, and lung irritation follows quickly, especially in people with asthma or chronic lung disease. People in those lines of work, like painters and cleaners, have to pay attention – their health has been put on the line before from similar solvents.
Touching the chemical with bare skin can bring a burning or stinging feeling. Washing up with soap and water right away keeps things from getting worse, but repeated exposures often end in dermatitis or cracked skin. Safety data sheets from chemical suppliers stress gloves and goggles for good reason. I’ve known workers who ignored this advice, calling it a pain, only to end up at the clinic later on. It all adds up to lost workdays, not to mention medical bills.
What Does the Research Say?
The National Institute for Occupational Safety and Health (NIOSH) warns about solvents like this for good reason. Chemicals in this family can cause central nervous system depression, and sometimes harm kidneys or liver with enough exposure over time. Animal studies show signs of organ damage from high amounts, though most day-to-day exposure in humans wouldn't climb that far—unless safety slips or equipment breaks.
Long-term health effects around this solvent haven’t been mapped out as well as bigger names like benzene or toluene. One study found workers exposed for years faced more headaches and memory trouble compared to those without exposure, though results weren’t conclusive. Research has gaps, but the scattered evidence doesn’t look comforting.
Building Safer Workplaces
Basic protection starts with smart ventilation. Open doors, hoods above tanks, and fans all make a difference. Respirators matter where fumes get strong, usually in small rooms or during spills. Gloves, coveralls, and splash-proof goggles are cheap insurance next to the cost of an emergency room visit.
Workplaces get better when they train folks on chemical hazards, rather than hiding them away in thick manuals nobody reads. Posters and brief safety talks every week keep risks fresh in people’s minds. I’ve watched places transform their safety records after every team member—management included—walked through spill drills together.
Looking Forward
Alternatives to harsh solvents are on the rise, pushed by both regulation and market demand. Water-based cleaning systems already knock out the need for chemicals like 4-Methoxy-4-Methyl-2-Pentanone in plenty of jobs. For now, anyone near the stuff ought to understand what they’re dealing with, and bosses should set up workplaces so nobody needs to get sick just for a paycheck.
| Names | |
| Preferred IUPAC name | 4-methoxy-4-methylpentan-2-one |
| Pronunciation | /ˈfɔːr mɛˈθɒksi fɔːr ˈmɛθɪl tuː ˈpɛntəˌnoʊn/ |
| Identifiers | |
| CAS Number | 821-55-6 |
| Beilstein Reference | 1209267 |
| ChEBI | CHEBI:89634 |
| ChEMBL | CHEMBL153044 |
| ChemSpider | 86504 |
| DrugBank | DB14641 |
| ECHA InfoCard | 19b54ce0-7e28-4624-ae03-dfcb444bb903 |
| Gmelin Reference | 86988 |
| KEGG | C19102 |
| MeSH | D051436 |
| PubChem CID | 10738 |
| RTECS number | SA7175000 |
| UNII | KG3H51IUII |
| UN number | UN number: "1265 |
| Properties | |
| Chemical formula | C7H14O2 |
| Molar mass | 116.16 g/mol |
| Appearance | Colorless liquid |
| Odor | mild, fruity |
| Density | 0.877 g/mL |
| Solubility in water | Insoluble |
| log P | 0.95 |
| Vapor pressure | 0.7 mmHg (20 °C) |
| Acidity (pKa) | pKa ≈ 19.5 |
| Basicity (pKb) | 4.10 |
| Magnetic susceptibility (χ) | -7.94×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.414 |
| Viscosity | 0.925 cP (25°C) |
| Dipole moment | 3.15 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 355.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -391.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | –3205.7 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | '' |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07,GHS08 |
| Signal word | Warning |
| Hazard statements | H319: Causes serious eye irritation. |
| Precautionary statements | P210, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | 64°C |
| Autoignition temperature | 235 °C |
| Explosive limits | Explosive limits: 1.0–7.0% |
| Lethal dose or concentration | LD50 Oral Rat 3690 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 3300 mg/kg |
| NIOSH | GV5950000 |
| PEL (Permissible) | 100 ppm (410 mg/m³) |
| REL (Recommended) | 50 ppm |
| IDLH (Immediate danger) | IDLH: 300 ppm |

