4-Hydroxypyridine: Looking at a Core Compound Through Chemistry, Industry, and Research
Historical Development
Few compounds have made as steady a mark in both research and manufacturing as 4-hydroxypyridine. Chemists first explored pyridine derivatives in the nineteenth century, recognizing a pattern among aromatic nitrogen-containing rings. Labs found that swapping out a hydrogen atom for a hydroxyl group on the fourth carbon changed the molecule just enough to open the door for new types of reactions. Over decades, experiments across Europe and North America steadily built up our understanding. By the mid-twentieth century, the spotlight on 4-hydroxypyridine grew, with researchers harnessing it as a starting material for drugs, dyes, and agrochemicals, and as a standard intermediate for further synthesis.
Product Overview
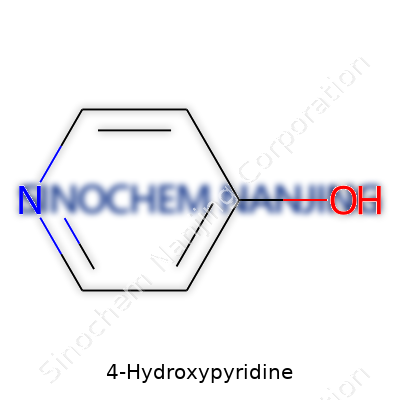
4-Hydroxypyridine belongs to the family of substituted pyridines. The structure looks simple on paper—a six-membered ring, a nitrogen at position one, and a hydroxyl group bonded to position four. In practice, that simplicity translates to versatility. Industrial labs use it in the preparation of corrosion inhibitors, pharmaceuticals, agrichemicals, and complex ligands for metal-catalyzed reactions. It's become a go-to building block for organic synthesis, often serving as a bridge between raw materials and high-value end products.
Physical & Chemical Properties
The compound usually appears as a white or off-white crystalline powder. It dissolves in water and many organic solvents, including alcohol and ether, which speaks to its flexible nature in both lab and plant settings. The melting point sits near 131-133°C, offering some stability during standard heating and drying. Chemists appreciate its subtle aromatic odor—a reminder of its pyridine backbone. The molecule forms strong hydrogen bonds due to the hydroxyl group, allowing for unique reactivity and solubility that makes it attractive for more than just basic synthesis.
Technical Specifications & Labeling
Purity has always driven value for 4-hydroxypyridine. Most commercial preparations offer assay values above 98%, with trace-level control for moisture and related impurities. The chemical follows clear hazard labeling, set by GHS. Labels point out the risk of skin and eye irritation, and warn about potential toxicity if swallowed. Storage recommendations emphasize sealed, cool, and dry conditions to avoid unwanted oxidation or degradation, reinforcing standard chemical hygiene.
Preparation Method
Lots of pathways exist for making 4-hydroxypyridine, but two stand out: direct hydroxylation of pyridine, and cyclization routes starting from aldehyde or ammonium precursors. Catalytic oxidation grabs the most attention in industry, partly because copper and iron salts boost selectivity for the para position of the ring. This streamlines purification and keeps costs under control. Some research groups have pursued milder and greener methods—enzymatic transformation and flow chemistry setups promise reduced waste and safer working conditions, responding to pressure for sustainability.
Chemical Reactions & Modifications
The molecule is as much a platform as a product. The hydroxyl group at the fourth carbon acts as both a weak acid and a nucleophile. Standard reactions include etherification, sulfonation, and alkylation, which open up ways to bolt on new functional groups. Chemists also use 4-hydroxypyridine as a ligand in organometallic chemistry, forming stable complexes with transition metals. Its reactivity with acylating agents has led to the design of new pharmaceuticals, crop protection agents, and materials with tuned electronic properties. In each case, the core ring resists breakdown, enabling chemists to tinker at the edges without destroying the scaffold.
Synonyms & Product Names
In chemical catalogs, the compound goes by several handles. Some call it para-hydroxypyridine, echoing the four-position of the hydroxyl. Others refer to it as pyridin-4-ol. Some patents and older literature use names like 4-Pyridinol. While those names fill textbooks and labels, any seasoned chemist knows that context and application often matter more than nomenclature details.
Safety & Operational Standards
Handling any pyridine derivative calls for respect; 4-hydroxypyridine fits that rule. Gloves, goggles, and good ventilation matter most in the lab. Direct contact can irritate skin and eyes, so spills and splashes get quick cleanup. Inhalation rarely occurs under good practice, but keeping dust levels down stays on every protocol. Local and international chemical safety bodies have weighed in, and MSDS documentation lays out responses for exposure and fires. Over the years, I’ve seen that even with lower-hazard compounds like this, accidents usually result from lapses in attention, never from the chemistry itself. Training and repetition close that gap.
Application Area
Use cases for 4-hydroxypyridine keep expanding. Medicinal chemists value it as a precursor for antibacterials and vasodilators. Agrochemical firms build it into herbicides that fight pests while breaking down in soil more safely than older options. Semiconductor researchers experiment with 4-hydroxypyridine-based ligands to tweak catalytic properties for greener electronics. Environmental labs run analyses using the compound as a marker, tracing transformation pathways in soil and water. In each of these roles, cost and reliability filter all choices—nobody pays for chemistry that wastes time or money on a plant floor or clinical trial. From my own work in pharmaceutical development, versatility mattered just as much as purity. A compound that combines both gets a fast lane to real-world adoption.
Research & Development
R&D teams focus on two angles: better production and smarter applications. Green chemistry approaches seek cleaner routes with less solvent waste, using catalysts that last longer and produce fewer byproducts. Others run screens for new biological activity, aiming to find untapped uses in medicine and agriculture. From a bench scientist’s perspective, the incremental improvements matter just as much as the headline breakthroughs. Each tweak in method or property supports more robust and affordable manufacturing, which sets off a chain reaction toward more products and bigger impact.
Toxicity Research
Data on the toxicity of 4-hydroxypyridine indicate relatively low acute danger, but experience tells us to dig deeper. Animal studies show some irritation and mild systemic effects at high doses, yet standard handling and storage mean those risks rarely come into play for trained personnel. Regulatory agencies require regular review, ensuring long-term studies catch any environmental or chronic health effects that could turn up as production scales. Taking cues from the last decade’s safety reviews, manufacturers have increased both the frequency and depth of toxicity testing. That hasn’t only improved worker protection; it's also helped downstream users in pharmaceutical and agricultural settings make more informed choices.
Future Prospects
Many see more opportunity ahead for 4-hydroxypyridine. Electronic materials research hints at a future where modified versions serve as charge carriers or molecular wires in advanced devices. Pharmaceuticals tend to favor molecules that scale up cleanly, which fits the bill here. As green chemistry pressure grows, production and downstream use will need to keep up, probably steering toward more efficient syntheses and stricter sourcing of raw materials. Researchers likely will keep probing the core structure for new reactivity, especially as analytical tools get faster and more sensitive. Broadly speaking, the story of 4-hydroxypyridine tracks the larger story of specialty chemicals—fine-tuned, old enough to trust, yet always ready for a new chapter.
What is 4-Hydroxypyridine used for?
Getting to Know 4-Hydroxypyridine
4-Hydroxypyridine stands out as a simple molecule at first glance, but underneath, its science shapes real change for medicine and industry. I first came across it researching specialty chemicals in pharma labs, and what caught my attention was the way tiny tweaks to a molecule can ripple into huge differences in real-world applications. This one, in particular, paves the way toward new drugs, dyes, and sometimes even metal-recovery tools.
Pharmaceuticals: More Than Just a Building Block
Most conversations about 4-Hydroxypyridine drift toward pharmaceuticals. Researchers reach for it because it easily slides into the backbone of more complex molecules. It has a knack for fitting into active drug compounds that tackle everything from bacterial infections to inflammation. I’ve seen medicinal chemists start with a 4-hydroxypyridine core, then tweak the rings and add groups, until the result shows real promise in lab tests. For example, derivatives from this compound help scientists design antifungal medications and new types of antibiotics—urgently needed as bug resistance rises.
Clinical journals highlight how modifications based on this molecule create selective enzyme inhibitors. These target bacterial enzymes, leaving human cells alone. Drugs based on these structures can be gentle on patients while tough on infections. This approach lines up with best practices: only using safe, proven compounds and thoroughly vetting any new medicine before use.
Dyes, Pigments, and Beyond
Beyond medicine, 4-hydroxypyridine carves out a niche in dye and pigment manufacturing. Its chemical backbone links into dye structures that must survive harsh sunlight and chemical washes. I worked with synthetic fibers, and those products relied on small tweaks in chemical ingredients, including this molecule, to keep colors bold and stable after months of outdoor exposure or machine washing.
Companies lean on 4-hydroxypyridine because it brings stability and intensity. A single batch of pigment or dye might end up in everything from designer dresses to coated wires or public benches. This saves resources and cuts down on waste, a practical need as industries look to reduce their environmental load. And since manufacturers can trace their chemicals back to trusted suppliers, they can keep product safety and environmental impact in check, which more consumers now demand.
Specialty Chemicals, Metal Extraction, and Research
4-Hydroxypyridine appears in other places less obvious to the public. It sometimes shows up in research labs testing catalysts for industrial processes, or in extractants designed to pull metals like copper or nickel out from ore. This isn’t about large-scale mining—it’s precision chemistry, where a dash of the molecule can make separation sharper and purer.
Responsible Use, Looking Forward
The challenge comes with making sure these chemicals deliver benefits without bringing harm. Factories need strict quality controls. Researchers check that every molecule made follows published safety standards, and regulations make sure nobody turns a blind eye to environmental risks.
With proper stewardship, 4-hydroxypyridine carries a lot of promise. It fuels research, supports new drugs, and keeps clothing looking sharp. Beyond the lab, it’s all about knowing the impact each chemical has and questioning whether it earns a place in the final formula—not just for performance, but for people and the planet as well.
What are the chemical properties of 4-Hydroxypyridine?
Structure and Basic Characteristics
4-Hydroxypyridine catches the eye with its simple, six-membered aromatic ring, holding a nitrogen atom and a hydroxyl group in a dance across from each other. This setup pushes the molecule into the spotlight for chemists. In everyday language, we’re talking about a ring with one spot attracting water and the other acting a bit basic. The presence of both nitrogen and oxygen atoms means it likes to form hydrogen bonds—a huge reason this chemical turns up in biological systems and industrial research labs.
Behavior in Water and Other Solvents
Drop 4-hydroxypyridine in water and it dissolves fairly well. That comes down to the hydroxyl group, which encourages interaction with water’s own hydrogen bonding dance. I remember during my college years, this made it easy to handle for basic reaction testing. The molecule does not stay stuck to itself, which means chemists can use it as a starting point for more complex compounds. Stick it in ethanol or methanol, the story stays much the same. Both the nitrogen and oxygen create a sweet spot between water-loving and oil-loving behavior, making it a regular on lab benches.
Acidity and Basicity
The pH story for 4-hydroxypyridine gives us some valuable clues. The nitrogen in the ring pulls electron density, nudging the hydroxyl group to act more acidic than you’d expect from a simple alcohol. The pKa value lands around 9. This detail turns important for chemists who want to swap that hydrogen with something else in a reaction. In basic conditions, the molecule loses a proton easily and takes on a negative charge, ready to form salts or act as a catalyst in reactions.
Reactivity and Uses in Synthesis
Labs use 4-hydroxypyridine for its lively nature. The lone pairs on both the nitrogen and oxygen give chemists flexibility. I’ve seen the hydroxyl site become a launch pad for attaching new groups, with acylation and alkylation reactions opening the door to new pharmaceutical compounds or catalysts. The nitrogen atom’s basicity sometimes lures electrophiles, letting it help out in more complex synthesis projects. The aromatic system in the ring, helped along by the electron-donating nature of the hydroxyl group, means the ring itself can take part in substitution reactions, expanding its use in dye and pigment production.
Handling and Safety
People working in labs need to think about safe handling. Organic compounds with small size like this sometimes evaporate quickly and release fumes, which may irritate the skin or lungs. Glove use and good airflow go a long way. Regulators focus on exposure limits, and academic research stresses checking for toxicity in aquatic and human cells.
Industry and Future Research
Scientists keep turning to 4-hydroxypyridine for its role in new drug development. The ease with which it reacts opens up pathways for making molecules that treat infections, inflammation, or even cancer. Environmental chemists also study this compound’s breakdown in wastewater, looking for safe ways to dispose of it post-synthesis. Its wide range of reactions makes it a favorite among researchers, always looking for better, greener methods to use it and eventually clean up after it.
How should 4-Hydroxypyridine be stored?
Respecting the Chemistry
4-Hydroxypyridine isn’t a household name, but anyone working with this compound knows the risks don’t take a day off. It’s a building block for pharmaceuticals and lab work, and getting storage just right avoids countless headaches. Every time I’ve seen chemical storage get sloppy, it didn’t end well—either a near-miss or expensive spoiled material. Keeping a level of respect for what the bottle holds makes all the difference.
Environmental Control Keeps the Powder Dry
Moisture can turn 4-Hydroxypyridine into a clumpy mess or make it break down. From my experience, chemicals like this want a dry, cool, and dark place. A tightly sealed glass container with a robust screw cap protects from air and humidity. Storing it alongside silica gel packets offers extra insurance against dampness sneaking in during humidity spikes. Anyone who trails into a lab during summer or the monsoon season knows how quickly things deteriorate under the wrong atmospheric conditions.
Why Temperature Matters
Keeping 4-Hydroxypyridine away from heat sources—never near radiators or close to sunny windows—slows down any chance of degradation. It sits best at room temperature or a bit below, maybe in a special chemical refrigerator if available. Fluctuating temperatures invite condensation, a real risk for powders that don’t tolerate water. I’ve seen storerooms that cycle between sweltering and cold turn perfectly fine supplies into questionable material.
Equal Emphasis on Light and Labeling
Bright lights aren’t a friend to many chemicals, and 4-Hydroxypyridine follows this rule. Ultraviolet light from overhead lights or sunlight pushes unwanted reactions. Amber glass keeps out most UV rays, so anyone restocking should look for this feature on their bottles. The right container earns its keep, sparing labs from nasty surprises and extra costs. Labels should be crisp and clear—date received, date opened, the name, and clear hazard symbols. I’ve met enough half-labeled bottles on back shelves to know missing this step is an accident waiting to happen.
Handling and Access: A Culture of Care
Control access to 4-Hydroxypyridine. People with the proper training are less likely to improvise or ignore protocols. Small, designated portions in the main lab, backed up by a larger, untouched stockroom supply, limit exposure and waste. Safety goggles, gloves, and a lab coat play their protective part, with spills sitting just a quick reach away from the right cleanup materials. I once saw a glove-less student fumbling a bottle; a harsh lesson but also a valuable reminder about building a culture around correct storage and handling.
Solutions that Work
Seeking guidance from safety data sheets bridges gaps in knowledge—these papers don’t just sit in a binder for audits. Regular checks of containers for leaks, signs of clumping, or mislabeling go much further than a hands-off approach. Teach new staff the specifics as part of onboarding. Audits and prompt responses to expired inventory stop accidents before they get traction. Simple, deliberate steps go much further for the safety of those around and the shelf life of what’s inside the jar. After years in labs, I find that routines built from small, consistent details keep work running smooth and safe.
Is 4-Hydroxypyridine hazardous or toxic?
Why We Should Care What’s In Our Chemicals
There’s no shortage of chemicals in the modern world. Each has its own risks and quirks. Take 4-Hydroxypyridine. If you haven’t worked in a lab or dabbled in pharmaceuticals, the name might sound like something best kept behind locked doors. It’s natural to feel uneasy with names you can’t pronounce. Let’s dig in and see what the story really is with this one.
What Exactly Is 4-Hydroxypyridine?
4-Hydroxypyridine sounds intimidating, but it’s basically a chemical building block—a compound chemists use to make more complex molecules. You find it behind the scenes every time you look at dyes, pharmaceuticals, or certain agricultural chemicals. Its biggest claim to fame comes from its role in medical compounds designed to help the human body.
The Real Dangers You Need to Know
Every chemical deserves a close look when it comes to health and safety. According to reliable sources such as PubChem and the European Chemicals Agency (ECHA), 4-Hydroxypyridine requires basic lab safety: goggles, gloves, good ventilation. Studies so far have not placed it in the category of chemicals with established cancer risks. Inhalation or excessive skin exposure may cause irritation, but, as someone with a bit of lab experience, I would say that’s not unlike checkered floors filled with cleaning solutions. Most risks show up from mishandling or bigger doses than you’d get at home or in the field.
Weighing Toxicity: Facts Over Fear
The question of toxicity often comes down to amount and exposure time. Most documented hazards with 4-Hydroxypyridine happen at high concentrations—situations you don’t run into outside a laboratory or factory. The chemical doesn’t build up in the body like more notorious compounds. Its rapid breakdown lessens concern about long-term harm with routine handling. The U.S. National Institutes of Health doesn’t list major warning flags for cancer, genetic risk, or reproductive problems.
An example: accidental skin contact causes mild discomfort, a bit like what you might expect if you handle strong soap or cleaning supplies without gloves for too long. Longtime chemists know that mishaps tend to teach more about proper technique than scare tactics ever could. Safe habits trump fearmongering every time.
A Culture of Safety: How To Deal With It
In work settings, everyone benefits from clear guidance and preparation. The best labs always have eyewash stations, clear signage, and a culture where speaking up about spills or unsafe habits is encouraged. 4-Hydroxypyridine serves as just another reminder that respect for process outweighs panic. I’ve seen new lab workers worry at first, but confidence grows as people realize the structure behind chemical use—training, oversight, and personal responsibility.
Handling 4-Hydroxypyridine with basic personal protection—gloves, goggles, fume hood—completely removes most concerns. Any chemical has the potential to harm if treated carelessly, but context matters. There’s no substitute for real guidance, real supervision, and clear labeling.
Moving Forward With Clarity
Misinformation about chemical hazards fuels doubt, not good science. Solid information shows 4-Hydroxypyridine carries risks that can be controlled with common protocols in industry and research. Anyone using it should educate themselves, stick to the rules, and trust in established science. If all workplaces held each other to that standard, both chemistry and public trust would be better off.
What is the molecular formula and weight of 4-Hydroxypyridine?
Getting Familiar With 4-Hydroxypyridine
4-Hydroxypyridine sports the molecular formula C5H5NO. Its molecular weight reaches 95.10 g/mol, based on the combination of carbon, hydrogen, nitrogen, and oxygen atoms. On paper, these numbers tell a basic story. Real value surfaces once the conversation shifts to what this compound brings to the table for chemists, researchers, and folks working in health sciences.
Why This Molecule Gets Talked About
Sometimes, the smallest structural tweaks lead to new opportunities. Swapping just one hydrogen with a hydroxyl group on the pyridine ring opens doors to a world of new reactivities. In labs and industry, researchers often turn to 4-hydroxypyridine as an intermediate. Pharmaceuticals, dyes, and pesticides all use derivatives from this relatively simple molecule. It serves as a scaffold that scientists can tweak, and those tweaks often end up in things we rely on for treating disease or improving crops.
During a past stint in a synthetic chemistry lab, projects involving pyridine derivatives popped up again and again. Most routine? Adjusting the functional groups. Each small change taught me how differently these molecules could react or interact with biological systems. I saw publications multiply around this compound and its analogs, not only in journals but also in pharmaceutical patent filings.
What the Numbers Actually Mean in Practice
A formula like C5H5NO translates to a predictable set of properties. This helps chemists plan syntheses and forecast outcomes. Four hydrogen atoms sit on the ring—a single one gets replaced with a hydroxyl group at the para-position, and a nitrogen sits in the six-membered ring. It sounds almost trivial, but that group dangles possibilities. Better water solubility, more reactivity under mild conditions, more options to link with other chemical entities. This versatility lets researchers attach other groups onto the molecule, aiming for drug candidates or advanced materials.
Molecular weight matters for more than lab calculations. When drugs keep their total size on the smaller side—like 4-hydroxypyridine and its kin—they often travel better through the body and can even cross certain biological barriers more easily. Medicinal chemists pay close attention to these numbers because they hint at how a molecule might behave in real life, not just in a beaker.
Challenges and Paths Forward
One common snag pops up during large-scale production. Controlled handling proves necessary, since nitrogenous heterocycles may show toxicity or environmental concerns. I remember facilities needing stricter waste treatment plans when processes required these types of molecules. Teams can tackle some of these issues by using greener, less hazardous synthesis routes. Employing catalytic reactions and recycling solvents already offers a path toward safer, cleaner chemistry.
Another important factor involves the data behind these molecules. Researchers, regulators, and manufacturers need reliable, open information about compounds like 4-hydroxypyridine. Comprehensive data ensures that science builds forward without unnecessary surprises—and lets society tap into the benefits while minimizing unintended consequences. Consistent investment in database infrastructure, knowledge-sharing protocols, and awareness on best handling practices pays off for industries, labs, and end users alike.
| Names | |
| Preferred IUPAC name | pyridin-4-ol |
| Other names |
4-Pyridinol Pyridin-4-ol gamma-Hydroxypyridine |
| Pronunciation | /ˈfɔːr haɪˌdrɒks.i.pɪˈrɪd.iːn/ |
| Identifiers | |
| CAS Number | 626-64-2 |
| 3D model (JSmol) | `4-Hydroxypyridine|JSmol` |
| Beilstein Reference | 120922 |
| ChEBI | CHEBI:16227 |
| ChEMBL | CHEMBL12114 |
| ChemSpider | 10497 |
| DrugBank | DB03941 |
| ECHA InfoCard | ECHA InfoCard: 100.007.345 |
| EC Number | 200-242-5 |
| Gmelin Reference | 64133 |
| KEGG | C06598 |
| MeSH | D017901 |
| PubChem CID | 967 |
| RTECS number | UY8225000 |
| UNII | KG16O84ME3 |
| UN number | UN2811 |
| CompTox Dashboard (EPA) | DTXSID2038447 |
| Properties | |
| Chemical formula | C5H5NO |
| Molar mass | 95.11 g/mol |
| Appearance | White to light yellow powder |
| Odor | weak pyridine-like |
| Density | 1.142 g/mL at 25 °C (lit.) |
| Solubility in water | soluble |
| log P | 0.06 |
| Vapor pressure | 1.98E-2 mmHg at 25 °C |
| Acidity (pKa) | 8.8 |
| Basicity (pKb) | 5.69 |
| Magnetic susceptibility (χ) | -41.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.505 |
| Viscosity | 1.63 mPa·s at 25 °C |
| Dipole moment | 2.07 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 109.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −38.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1994 kJ/mol |
| Pharmacology | |
| ATC code | N07XX02 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes serious eye irritation. Causes skin irritation. May cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302: Harmful if swallowed. |
| Precautionary statements | P264, P270, P301+P312, P330, P501 |
| NFPA 704 (fire diamond) | 2-1-0 |
| Flash point | 113°C |
| Autoignition temperature | 545°C |
| Lethal dose or concentration | LD50 (oral, rat): 2000 mg/kg |
| LD50 (median dose) | LD50 (median dose) of 4-Hydroxypyridine: Rat oral 1800 mg/kg |
| NIOSH | RN8480000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 20~25°C |
| Related compounds | |
| Related compounds |
2-Hydroxypyridine 3-Hydroxypyridine Pyridine Nicotinic acid Isonicotinic acid |

