4-Hydroxy-4-Methyl-2-Pentanone: Digging Into a Workhorse Compound
Historical Development
A lot of stories in chemistry rarely get the attention they deserve, and 4-Hydroxy-4-methyl-2-pentanone stands as one of those unsung contributors. Back in the late 1800s, both organic synthesis and industrial chemistry were gaining steam. Laboratories began to see the value in tweaking simple molecules for broader uses. This compound came to light during an era when folks were refining methods of oxidation, condensation, and reduction to make what industry and academia needed. Chemists realized pretty early that the structure—featuring both a ketone and an alcohol group—opened doors. Over decades, it became a staple in the production of various solvents and intermediates. Through several generations of refinement, recipes became safer and more straightforward, showing how practical need drives innovation.
Product Overview
At its core, 4-Hydroxy-4-methyl-2-pentanone presents as a colorless liquid, appreciated mainly for how easily it dissolves in water and a wide array of organic solvents. In manufacturing, its biggest spotlight comes from being a versatile intermediate for making other chemicals. Think of it as a handy building block, able to morph into different molecules with a few strategic modifications. Many industries—from paints to pharmaceuticals—rely on its reliable behavior. Because of its dual functional groups, it connects well with a larger family of chemicals, carving out roles in adhesive formulation, resins, and even agrochemical products. In the day-to-day, most people will never encounter the pure material but benefit from products made possible by it.
Physical and Chemical Properties
Looking at its molecular makeup, 4-Hydroxy-4-methyl-2-pentanone sits at C6H12O2. This gives it a molecular weight hovering around 116 grams per mole. The physical state stays liquid under ordinary temperatures and pressures. Its boiling point sits above that of water, which actually helps during certain purification tasks. With a relatively low viscosity and distinct odor, the compound mixes well with many alcohols and ethers. The hydroxyl group invites hydrogen bonding, giving it a sturdy solubility profile in polar and nonpolar mixtures. Chemical reactivity comes from both the tertiary alcohol and the ketone group—meaning folks in labs can access a range of downstream reactions by adjusting conditions.
Technical Specifications and Labeling
Clarity about purity stands out as a top priority. Suppliers offer this compound with varying grades. For tighter applications—like pharmaceuticals or electronics—labs look for analytical-grade material. Lower grades serve industries willing to accept minor impurities, such as paints or construction. Labeling follows regulatory frameworks guiding the safe handling of ketones and alcohols, so anyone using it must stay sharp about compatibility and risks. Bottle and drum labels usually highlight concentration, hazard warnings, and recommended storage temperatures. These details, while sometimes overlooked, keep operations safe and reliable.
Preparation Method
Preparation most commonly starts from acetone. Through self-condensation in the presence of a base, acetone forms diacetone alcohol, which is another name for 4-Hydroxy-4-methyl-2-pentanone. This base-catalyzed aldol reaction has become a favorite in teaching labs because it’s a textbook example of how new carbon-carbon bonds form. On a larger scale, plant chemists adjust pH, temperature, and solvent ratio to keep yields high and byproducts minimal. Safety protocols ramp up during scale-up, given how exothermic things can get and the flammable nature of starting materials. This method has evolved to include improvements for energy efficiency and waste reduction, especially in regions with stricter environmental controls.
Chemical Reactions and Modifications
The compound offers plenty of reaction possibilities. The alcohol group can form esters through reactions with acids, while oxidation can take it further to diketones if needed. The ketone part reacts with amines, hydrazines, and other nucleophiles, so chemists use it in the synthesis of larger, more complex organic scaffolds. Hydrogenation and reduction open new paths, while dehydration can turn it into mesityl oxide, which is useful on its own in the chemical industry. Plenty of folks in fine chemical and pharmaceutical labs use its reactivity to develop process routes they can scale up. These choices reflect a balance between efficiency, safety, and the type of bonds they want in the end product.
Synonyms and Product Names
The naming game in chemistry can cause some headaches. 4-Hydroxy-4-methyl-2-pentanone mostly goes by diacetone alcohol. Over time, product labels picked up alternate descriptors, like DAA or DAAK. In older literature, terms like 4-Methyl-4-hydroxypentan-2-one sometimes crop up. This jumble underscores the need for discipline in record keeping, especially as chemicals cross paths with new regulatory environments. For anyone working internationally, knowing these aliases prevents mix-ups and helps with import or export licensing.
Safety and Operational Standards
People handling this compound need to treat it with respect. Like many solvents and intermediates, 4-Hydroxy-4-methyl-2-pentanone brings both irritant and flammability risks. Proper ventilation in labs and factories keeps airborne concentrations in check. Glove and goggle use becomes second nature, and people trained in spill management and first aid best practices see fewer accidents. Storage guidelines point to keeping containers in well-ventilated places away from open flames and oxidizers. Regulations vary across regions, but organizations like OSHA in the USA and ECHA in Europe outline protocols for exposure and emergency response. Consistent training proves critical, especially for less-experienced staff and seasonal workers.
Application Area
This compound shows up in quite a range of industries. Paint and coating manufacturers use it as a solvent—its strong solvency cuts through resins and pigments, improving finish and drying time. Adhesive makers blend it in to modify viscosity and tack. It helps in the preparation of herbicide and pesticide products, not only as a solvent but as an intermediate for active ingredients. In the world of inks, its volatility and solvency make for sharp, even prints on challenging surfaces. Some cleaning formulations call on it to dissolve greasy residues quickly and cleanly. The overlap with pharmaceutical and cosmetic manufacturing lies in its role as a synthesis building block—though not as a direct additive.
Research and Development
Researchers constantly seek safer, greener pathways to make and modify this compound. Some focus on catalyst alternatives that reduce energy demand or isolate purer product. Others take aim at bio-based acetone as a starting point, hoping to reduce reliance on petrochemicals. In academic circles, diacetone alcohol often serves as a model substrate in studies on catalysis, reaction kinetics, and green chemistry. These projects sharpen understanding of solvent effects and reactivity, improving future synthesis. Emerging research sometimes targets applications in advanced materials, where its unique mix of properties can lead to new formulations or functions not currently available.
Toxicity Research
4-Hydroxy-4-methyl-2-pentanone does not tend to rank among the most hazardous organics, but safety studies reveal more detail. Animal studies show central nervous system effects at high concentrations, similar to other short-chain ketones. Skin and eye irritation remains the biggest occupational concern. Researchers stress the cumulative nature of exposure—focusing on ventilation, PPE, and limited contact. Long-term chronic toxicity studies are limited, keeping room for more work on metabolic byproducts and biodegradability. Waste treatment also sits under the microscope, as improper disposal can threaten water systems. Regulating agencies keep updates rolling as new data emerge.
Future Prospects
Markets for this compound look steady, backed by the growth of construction, coatings, and adhesive industries in developing regions. Demand for environmentally friendlier synthesis ramps up as chemical companies chase sustainability goals. There is solid potential for bio-based routes, possibly leveraging fermentation-derived acetone, which could change the energy footprint of industrial processes. Research pursues new uses in specialty formulations and advanced composite materials. Regulators and watchdogs push for tighter emission controls and exposure limits, which challenge manufacturers to innovate cleaner production technologies. Whether for established applications or emerging niches, the role of 4-Hydroxy-4-methyl-2-pentanone grows with creativity and vigilance.
What are the main applications of 4-Hydroxy-4-Methyl-2-Pentanone?
Solvent Powerhouse in Manufacturing
Factories rely on chemicals that pull their weight without fuss, and 4-Hydroxy-4-Methyl-2-Pentanone (often called diacetone alcohol) fits the bill. Walk into a paint shop, and chances are, it helps keep that paint smooth and easy to spread on surfaces. Strong solvents like this one grab hold of substances others leave behind. Artists and automotive workers value its contribution to a good flow and reduced brush marks or streaks. In my own bit of home renovation work, I noticed paints that include this solvent are less likely to separate in the can and cover better in fewer coats.
Big Impact in Printing Inks
Printers need inks that dry just right—quick enough to stay sharp, but slow enough not to clog expensive equipment. This chemical helps printers deliver those results, especially with solvent-based inks used in packaging, labels, and posters. The ability to keep the ink wet long enough for proper application, then vanish without a trace, gives companies more control during high-speed runs. Commercial printers swear by it because fewer print runs get ruined by sticky messes or poor drying.
Essential in Pharmaceuticals
Medicine-makers trust chemicals that can dissolve active ingredients without leaving behind unwanted flavors or smells. Diacetone alcohol plays a role behind the scenes here. As a coupling agent, it links ingredients that otherwise would not mix. This property helps pharmaceutical companies keep drugs stable in liquid suspensions and syrups. I once talked with a pharmacist who said that consistency in creamy ointments often rests on high-quality solvents like this one.
Cleaner Production for Cleaners
Look at the ingredients on a bottle of industrial cleaner, and you may spot 4-Hydroxy-4-Methyl-2-Pentanone. Its grease-cutting strength means industrial workers can scrub machinery and shop floors faster without repeated rinsing. Cleaning crews in factories and big kitchens use products with this solvent so food equipment and engines come out sparkling. Using less water and elbow grease, especially for heavy oil and ink stains, solves headaches for large-scale maintenance crews.
Glue and Resin Production
Adhesive makers prize this chemical for how it fine-tunes glue. By keeping compounds in an even mixture, it stops adhesives from getting lumpy or separating on the shelf. The stuff helps create resin coatings that stick better, last longer, and cure without bubbles or gaps. Woodworkers and assemblers get more reliable bonds between surfaces, which matters when putting together furniture, flooring, or intricate tools.
Real-World Challenges and Safer Solutions
Every useful chemical brings some risk. 4-Hydroxy-4-Methyl-2-Pentanone, in large quantities, can irritate eyes, skin, and lungs. Worker safety means good ventilation and safety gear. Smart companies look for tech that traps solvent fumes or recycles them to keep indoor air healthier. Governments set exposure limits and call for proper storage practices, which keeps both workers and the environment safer.Sustainability drives the search for less harmful alternatives, yet so far, few green replacements match this solvent's versatility and effectiveness. Ongoing research may lead to bio-based versions that cut down on environmental impact without losing those key benefits in paints, inks, and adhesives.
What are the safety and handling precautions for 4-Hydroxy-4-Methyl-2-Pentanone?
Why You Can’t Afford to Be Careless
Most folks in labs or workshops come across chemicals every day. There’s a common one with a mouthful of a name—4-Hydroxy-4-Methyl-2-Pentanone, sometimes called diacetone alcohol. It doesn’t look flashy: clear, sometimes with a slightly sweet odor. To the untrained eye, maybe it seems harmless. It isn’t. Handling this chemical is serious business. Health and safety rules count for something, not because someone said so, but because mistakes here carry real risks.
What’s the Hazard?
Diacetone alcohol can irritate the eyes, nose, and throat. Some people brush off occasional minor stinging or sneezing, thinking it’s just part of the job. That attitude short-changes the risks. A big enough whiff or skin splash, and you’re reaching for the eyewash station or, worse, heading for medical attention. Chronic exposure—breathing it day in and day out—can drag down your lung health over time, and that damage doesn’t always bounce back.
Protecting Yourself: Lessons from the Floor
Most advice about personal protective equipment isn’t rocket science. I learned early on: don’t cut corners on gloves or goggles. Regular nitrile gloves stand up to diacetone alcohol, but thin vinyl or bare hands won’t. Lab coats and closed shoes—not a luxury, but bare minimum. Eye protection matters even if you’ve never had a splash. I once watched a co-worker rub his eyes in a hurry after cleaning up a spill. Minutes later, his run to the eyewash station became everyone’s wakeup call.
Keep the Fumes at Bay
Ventilation ranks high on the list of priorities. Working in a fume hood stops harmful vapors from sneaking up on you. I’ve seen the difference between a crowded, stuffy space and a well-ventilated bench, and nobody goes home happy from a lab that reeks. People who think cracking a window works soon learn that isn’t enough. Good airflow isn’t just a comfort—it's a health standard you can measure with detection badges or regular monitoring.
Storage: Don’t Just Shove It Anywhere
It’s tempting to toss bottles on any empty shelf when you’re in a hurry. Diacetone alcohol rewards that kind of carelessness with spills or leaks. Strong containers with secure lids are a must, always labeled, and never mixed with incompatible stuff—no acids, oxidizers, or amines nearby. Even in small operations, clear signage and secondary containment trays help prevent dumb mistakes. I once saw a bottle left open overnight corrode the shelf underneath, teaching everyone the value of double-checking at lockup.
What To Do If Things Go Wrong
Reacting fast can change the outcome after an accident. A spill kit stocked with absorbent pads and neutralizing agents comes in handy more often than anyone hopes. An emergency shower and eyewash close by, trained hands that know to flush for 15 minutes, and a clear path to the exit make all the difference. Regular drills keep those responses sharp—no fumbling for the phone or scrambling for the right number. Real-life accidents prove it’s not overkill.
Mental Shifts—More Than Just Rules
Working safely with chemicals like 4-Hydroxy-4-Methyl-2-Pentanone isn’t just about following a checklist. It grows from a personal sense of responsibility, a culture that values looking out for yourself and the next shift. There’s no shortcut here. Checking labels, wearing the right gear, and speaking up about hazards come from that place. Stories and mistakes stick with a crew much longer than laminated guidelines on a wall.
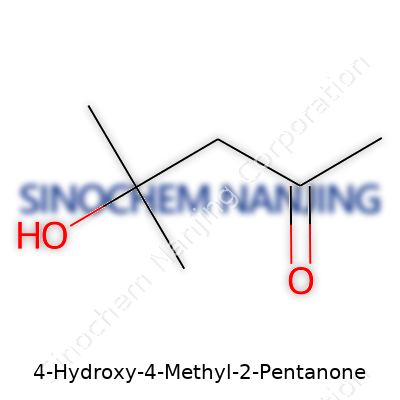
What is the chemical structure and molecular formula of 4-Hydroxy-4-Methyl-2-Pentanone?
Getting To Know the Molecule
4-Hydroxy-4-Methyl-2-Pentanone often goes by another name: diacetone alcohol. Its molecular formula is C6H12O2. Chemists sometimes call it DAA for short. This compound sits among the simplest examples of a beta-hydroxy ketone, a class of chemicals that supplies building blocks for solvents, resins, and coatings. For anyone who ever cleaned brush bristles with paint thinner, worked in a lab, or purchased household adhesives, this ketone might feel closer to daily life than expected.
Chemical Structure: A Closer Look
Visualizing the structure helps. The carbon skeleton shapes out as a five-carbon chain, punctuated by a methyl group and a hydroxy group at the fourth carbon, and a carbonyl group at the second. For those comfortable with chemistry diagrams, it is drawn like this: CH3COCH2C(OH)(CH3)2. The backbone runs: methyl group, carbonyl group (the “ketone” part), two more methylene groups, then at the fourth carbon, a hydroxy group and another methyl sprout out together. This spot creates a fork in the molecule, a trait that gives it unique chemical properties.
One practical thing about such structure, especially the hydroxy group next to a ketone, is the ability to engage in hydrogen bonding. In my own undergraduate days running reactions in a chemistry lab, I learned that molecules like DAA dissolve well in both water and organic solvents — not always a given for carbon-heavy compounds. DAA’s “both-worlds” ability comes from the polar hydroxy and nonpolar methyl ends living on the same carbon spine. This gets valuable when blending chemicals in formulations, allowing easier mixing and quicker diffusion.
Why Does This Structure Matter?
The unique arrangement brings more than just lab interest. Its chemical features make DAA useful for thinning coatings, cleaning electronics, and even acting as a component in specialty inks. Some industrial chemists turn to DAA for its ability to balance volatile strength and toxicity, lowering risks for workers handling large volumes. I once talked to a paint formulator who picked DAA over acetone because it dries a bit slower, which extends the “open time” for brushing or rolling without hard edge marks.
Factories depend on molecular details like the extra hydroxy group. This group helps create bonds with resins or catalysts, making finishes that last longer or resist stains better. In my own work on industrial product labeling, the difference between inks that run and those that stick often falls back on a solvent’s chemistry. DAA’s profile gets the nod because it won’t evaporate before you lay down a crisp mark, yet doesn’t linger for hours to gum up a production line.
Navigating Risks and Opportunities
Not all chemistry comes free of concern. Even versatile molecules like DAA need careful handling. The balance between usefulness and safety matters. Anyone using it in bulk should wear gloves, use fume hoods, and have spill kits ready; DAA can irritate skin, and like many solvents, vapors will harm your lungs with long exposure. Some states are reviewing emissions regulations as newer research links solvent fumes to health issues. Industry innovators have started developing closed-loop recovery systems that capture DAA vapor and recycle it, slashing both waste and risk.
There’s another opportunity: biobased synthesis. Traditional DAA production leans on petrochemical feedstocks. A growing group of green-leaning chemists now explores fermentation-based approaches, using yeast to build carbon chains. This could cut fossil input and reduce environmental footprint, meeting consumer and regulatory demands for safer, more responsible solvents in products most people touch each day.
How should 4-Hydroxy-4-Methyl-2-Pentanone be stored?
Getting Real About Chemical Safety
Working with chemicals every day teaches you one thing: small oversights lead to big problems. I’ve seen otherwise clear labs turn into disaster zones over a careless lid or a wrong shelf. 4-Hydroxy-4-Methyl-2-Pentanone, also known as diacetone alcohol, isn’t some wild outlier in the chemical world, but it deserves respect. People use it for coatings, inks, and cleaning, so you’ll spot it in both industry settings and research spaces. It’s colorless and has a mild, sweet smell—easy enough to forget as you move through rows of bottles, but you won’t forget the trouble if you get storage wrong.
One Wrong Move, One Bad Day
Safety data on this compound tells a clear story: heat and open flame turn this solvent from handy tool to hazard. It evaporates pretty fast and its vapors catch fire easily. During my years running inventory, I watched someone set a bottle near a heat gun. The room emptied out fast—smoke detectors yelled, the sprinklers started, and people shook their heads. Storing this chemical far from heat sources and direct sun isn’t just following the rules, it’s how you avoid big cleanup bills and potential injury.
Containers and Closets: Keep It Tight and Cool
Somebody once told me, “A chemical never escapes a good seal.” That’s stuck with me. 4-Hydroxy-4-Methyl-2-Pentanone can eat away at weak plastics. Factory bottles usually do fine, but I’ve seen people transfer it to old soda bottles. That’s a recipe for leaks and vapor build-up. Use tight, chemical-resistant containers, stick to the original stuff when you can, and label everything with clear writing. I don’t trust faded Sharpie labels in a rush, and you shouldn’t either.
Give this chemical its own ventilated spot on a shelf or in a flammable storage cabinet if you have one. A locked cabinet with exhaust means fewer fumes in the air and less risk that an untrained hand reaches in for the wrong bottle. At temperatures near 20°C (about room temp), you’re in the safe zone. Getting cooler is better, but don’t freeze it, since that messes with handling down the line.
Solving the Human Part of the Problem
Labels, records, and teamwork change everything. Training sessions grow stale, but bringing folks together to talk about near-misses helps. I remember a night shift worker telling us about a spill he nearly caused by stacking boxes too high around the storage cabinet. Simple fix: keep the area clear, post reminders, and check your storage spots once a week. Some places use digital logs and barcodes, others rely on clipboards. Pick what people actually use, not what looks neat in an audit.
Real World vs. Rule Books
There’s no magic in tidy sheets and checklists if no one cares enough to enforce them. It’s not just about fire inspectors or lab managers giving orders. Everyone, from shift workers to PhDs, has a role to play. If you see condensation inside a chemical bottle, or a loose cap, fix it on the spot instead of passing the buck. Accidents shrink when everyone treats storage like it’s personal.
In the end, storing 4-Hydroxy-4-Methyl-2-Pentanone safely isn’t mysterious work. Keep it sealed, cool, and away from heat. Use real containers, trust good habits, and build a team that learns from its own stories. Chemicals like this one become boring, everyday tools if you don’t cut corners—just the way any lab or workshop should want it.
What is the purity specification and available packaging for 4-Hydroxy-4-Methyl-2-Pentanone?
Purity Matters in Chemical Sourcing
In the chemical industry, purity isn’t just a percentage on a certificate—it shapes the way professionals approach each experiment, reaction, or product formulation. 4-Hydroxy-4-Methyl-2-Pentanone, known as diacetone alcohol, lands in a lot of labs and production floors because of its role as a solvent and intermediate. Purity specifications make or break the value of this material. Most buyers search for minimum purity levels of 99.0%. Lower grades often bring in trace water, residual acetone, and even traces of other alkanols. Even tiny impurities can foul up catalysts or create unwanted byproducts in fine chemical work.
Certainty matters; regulations keep getting tighter, and product recalls hit budgets and reputations hard. Laboratories and producers depend on solid certificates of analysis that support their documentation for audits and batch traceability. Experienced chemists look past headline purity and also review moisture content, acid values, and color numbers. Even a small blip on an assay or a raised water content—often flagged in a Karl Fischer titration—can affect final yields and storage stability.
Packaging Choices Have Real World Impact
Safe handling of materials hinges on thoughtful packaging. From personal experience, bulky or fragile containers often lead to spills, slowdowns, or worse, unnecessary exposure. Suppliers who pay attention to details—like tight-sealing drums, tamper-evident closures, and easy-pour jugs—take a lot of stress off workers.
Bulk industrial users usually go for steel or HDPE drums, often 200 liters in size, for big batch runs or ongoing plant processes. Some operations, such as those in pharmaceuticals or specialty coatings, work at smaller scales. Five-liter, twenty-liter, and fifty-liter cans or bottles cover those needs without leaving leftover material to degrade on the shelf. Handling costs for oversized packaging can strain budgets in smaller outfits, so having several options opens doors for more organizations to use the same high-purity material.
Manufacturers who commit to clear batch labeling and robust containers protect product value through transport and storage. Even a small dent or weak seal can trigger quality deviations. This attention to packaging not only avoids accidental waste but provides traceability if something goes wrong. For companies with zero-tolerance policies on contamination, sealed and inert-lined packaging makes all the difference.
Quality and Transparency Set the Tone
In the past, I’ve seen companies struggle with off-spec batches that traced back not to poor chemistry, but to residual solvents leeching from reused drums or unlisted water pickup. This drives home the importance of transparency—suppliers who volunteer GC traces, batch numbers, and storage condition advice help protect reputations and build trust. A detailed certificate of analysis builds bridges between simple purchasing and consistently successful application.
Feedback from end users often guides suppliers to tweak both purity specs and packaging. Some chemical plants might ask for drums with inner liners, while labs might need amber bottles to keep the material stable in light. Active collaboration between users and suppliers changes the standard from “good enough” to “best fit,” reducing waste and unexpected rework.
Stronger Standards Power Safer and More Reliable Outcomes
Whether dealing with bulk volumes or delicate, one-off syntheses, treating purity specification and packaging with respect pays dividends. Quality materials and packaging earned through shared industry feedback push operations closer to hassle-free days. Professionals who demand both high purity and practical packaging don’t just keep one batch running smoothly—they build a track record others trust.
| Names | |
| Preferred IUPAC name | 4-hydroxy-4-methylpentan-2-one |
| Other names |
Diacetone alcohol DAA 4-Hydroxy-4-methylpentan-2-one |
| Pronunciation | /ˈhaɪ.drɒk.si ˈmɛθ.əl tuː ˈpɛn.teɪ.nəʊn/ |
| Identifiers | |
| CAS Number | 123-42-2 |
| Beilstein Reference | 1907931 |
| ChEBI | CHEBI:38937 |
| ChEMBL | CHEMBL15636 |
| ChemSpider | 8077 |
| DrugBank | DB01942 |
| ECHA InfoCard | echa.europa.eu/substance-information/-/substanceinfo/100.003.420 |
| EC Number | 208-626-7 |
| Gmelin Reference | 1202096 |
| KEGG | C00712 |
| MeSH | D015209 |
| PubChem CID | 7905 |
| RTECS number | SA8575000 |
| UNII | OL7D0LC10A |
| UN number | UN1233 |
| Properties | |
| Chemical formula | C6H12O2 |
| Molar mass | Molar mass: 116.16 g/mol |
| Appearance | Colorless to slightly yellow liquid |
| Odor | mild fruity odor |
| Density | 0.921 g/mL at 25 °C (lit.) |
| Solubility in water | Miscible |
| log P | 0.38 |
| Vapor pressure | 0.67 mmHg (25°C) |
| Acidity (pKa) | pKa = 13.5 |
| Basicity (pKb) | pKb ≈ 5.2 |
| Magnetic susceptibility (χ) | -7.52 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.414 |
| Viscosity | 2.97 mPa·s (20 °C) |
| Dipole moment | 2.75 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 300.7 J⋅mol⁻¹⋅K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -348.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3218 kJ·mol⁻¹ |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H319, H335 |
| Precautionary statements | P210, P261, P271, P280, P301+P312, P303+P361+P353, P304+P340, P305+P351+P338, P312, P337+P313, P403+P233, P501 |
| NFPA 704 (fire diamond) | 1, 2, 0 |
| Flash point | 58 °C (closed cup) |
| Autoignition temperature | 285 °C (545 °F; 558 K) |
| Explosive limits | Explosive limits: 1.0% - 5.4% |
| Lethal dose or concentration | LD50 oral rat 1600 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 2080 mg/kg |
| NIOSH | MV3675000 |
| PEL (Permissible) | 50 ppm (180 mg/m3) |
| REL (Recommended) | 50 ppm |
| IDLH (Immediate danger) | 300 ppm |
| Related compounds | |
| Related compounds |
Diacetone alcohol Acetone Mesityl oxide Isophorone Methyl isobutyl ketone |

