4-Ethylpyridine: Past, Present, and Future
Historical Development
4-Ethylpyridine, a molecule with a long chemical tradition, entered scientific literature in the early 20th century. Chemists first recognized pyridine structure in coal tar, and soon substitutions like the ethyl group at the “4” position caught interest for their distinctive behaviors. Back in the 1940s and 50s, synthetic organic chemistry relied on trial-and-error and limited analytical tools. Pyridine derivatives, especially those with small alkyl groups like ethyl, challenged research groups because of their volatility and sometimes stubborn impurity profiles. The drive for improved synthesis sharpened focus through decades, especially as new instrumentation—NMR, GC-MS—opened up the field. From lab curiosity to vital intermediate, 4-ethylpyridine grew up alongside industrial expansion, supporting flavor chemistry, specialty polymers, and spectrum analysis. Laboratories in Europe and the United States ran countless distillations, and with each iteration, understanding of production and application grew.
Product Overview
Few specialty chemicals thread themselves into diverse industries like 4-ethylpyridine. It’s a pale yellow liquid, with a strong, somewhat disagreeable odor that typifies lower alkyl pyridines. You’re more likely to find it sold in grades specializing for chemical synthesis or analytical standards than as an off-the-shelf consumer item. Within chemical catalogs, it shows up next to related pyridines, often separated by purity and intended use. Rarely used as a flavoring itself, it provides vital flavor markers in whisky and tobacco analysis, along with use in pharmaceutical intermediates and next-generation organic electronic materials. Those chemical catalogs, with decades of fine-tuning, carefully lay out its place in the chemical food chain.
Physical & Chemical Properties
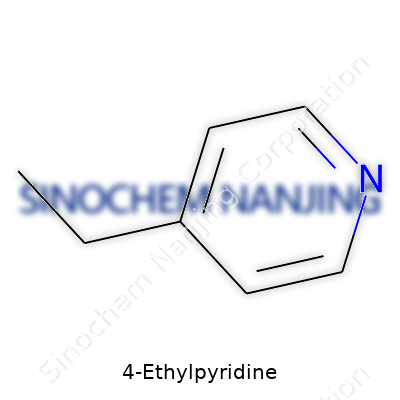
Chemists recognize 4-ethylpyridine by the molecular formula C7H9N, a mass of just over 107 g/mol. It boils just above 150°C, reflecting a relatively modest intermolecular interaction, thanks to the small ethyl group and pyridine ring. Liquids like this call for tight storage—exposure to air or light gradually sparks oxidation or evaporation, so properly sealed amber glass often sits in lab inventories. The molecule offers moderate solubility in water but dissolves freely in organic solvents—ether, benzene, acetone—an asset for complex synthesis steps. Its basicity, milder than ammonia but enough to bind acids, suits it to catalysis and extraction alike. Simple in appearance, it packs reactive nitrogen and a handle for further transformation. Looking at its reactivity, the character differs distinctly from unsubstituted pyridine—subtle shifts tweak acid-base balance, nucleophilicity, and resonance stabilization.
Technical Specifications & Labeling
Chemical suppliers don’t take shortcuts with their labels or specifications for 4-ethylpyridine. Purity often exceeds 98%, with data from gas chromatography or HPLC printed on certificates of analysis. Labels always include clear warnings: flammable, toxic if swallowed or inhaled, risk of environmental harm. Regulatory numbers like CAS (536-78-7) help with ordering and tracking. Shelf-life, typically two years with proper storage, comes from both experience and stability testing. Labs need transparent evidence—no one wants unexpected impurities in their reactions. Specific gravity, boiling and melting points, and trace impurity limits get cataloged, especially for analytical applications. Everything ties back to trust: accuracy in labeling and reliable documentation keeps research and manufacturing safe and compliant.
Preparation Method
Classic organic synthesis routes for 4-ethylpyridine start from pyridine ring formation. Chemists often rely on condensation of acetaldehyde with ammonia and an oxidizing agent, then careful tuning with catalysts and heat. Modern methods streamline this, using Friedländer or Chichibabin approaches with ethylating agents on pyridine or pyridine N-oxides. Some routes even use metal-catalyzed cross-coupling, where ethyl halides and pyridines unite under palladium or nickel catalysts. Waste minimization and greener solvents shape more recent work, reflecting both regulatory and economic pressures. Continuous-flow processes pop up in large-scale manufacture, maximizing yield and safety. For smaller quantities, bench chemists still lean on classical glassware, fractionation, and painstaking purification by distillation or chromatography to achieve research-grade product.
Chemical Reactions & Modifications
4-Ethylpyridine acts both as a building block and a versatile reactant. The nitrogen lone pair opens doors to alkylation and acylation, making quaternary pyridinium salts for ionic liquids or phase transfer catalysts. Oxidation of the ethyl group to a carboxylic acid enables access to new heterocyclic structures. Electrophilic substitution favors the ring, with halogenation producing functionalized intermediates for pharmaceutical or agrochemical use. Coupling reactions—Suzuki, Heck, Sonogashira—bring further potential by exploiting that ethyl handle or the aromatic core. Synthetic strategies keep evolving, emphasizing atom economy and selectivity. Chemists shape 4-ethylpyridine towards targets—designer ligands, organic conductors, specialty additives—and keep searching for productive reaction channels.
Synonyms & Product Names
Within scientific texts, 4-ethylpyridine sometimes appears as alpha-picoline, 4-picoline, or 1-ethyl-4-pyridine. Non-systematic names stem from historical nomenclature and differences in translation. Commercial catalogs prefer IUPAC-correct terms to avoid legal or logistical confusion. Data sheets always cross-reference major synonyms to minimize risk of error during procurement. Sometimes, flavor chemistry literature refers obliquely to “ethyl-pyridine flavor marker,” especially in the context of tobacco smoke or spirit analysis. All these names signal awareness of how regional, historical, and functional language can blur boundaries in the chemical lexicon.
Safety & Operational Standards
Common sense and strict adherence to protocols shape lab work with 4-ethylpyridine. Its volatility and toxicity call for gloves, chemical goggles, and sometimes full-face respirators, depending on scale. Spills demand immediate attention, absorbent pads, and fume hood disposal. Safety data sheets recommend storage segregated from oxidizers and acids. Fire risk stays high: keep ignition sources away and use explosion-proof equipment for bulk storage or transfer. Waste streams require careful labeling and collection, avoiding sinks or open drains. Regulatory bodies—OSHA, REACH, EPA—lay down limits on exposure and mandates for spill response and incident reporting. Some labs rotate staff through chemical safety seminars, reviewing real-world incidents to maintain a strong safety culture. Mishaps with pyridines, whether inhalation or skin contact, motivate vigilance above all.
Application Area
4-Ethylpyridine turns up in sectors from analytical chemistry to pharmaceuticals. Analytical labs value it as a flavor marker in food and beverage profiling, especially whisky maturation profiling where subtle alkyl pyridines flag oak aging or fermentation conditions. It’s used as an intermediate in production of anti-malarial, anti-inflammatory, and anti-cancer drugs. In electronics, it serves as an additive in organic semiconductors, where small tweaks to electron density yield big changes in material performance. Chemical engineers sometimes use it in developing corrosion inhibitors, tapping its surface affinity and interaction with metals. The molecule punches above its weight as a research tool for studying reaction mechanisms on pyridine rings—kineticists praise its tractability in model systems. Its distinctive odor, a trace component in tobacco smoke, helps in forensic and regulatory assays for public health research.
Research & Development
Recent research pivots on novel reactivity and safer synthetic pathways. Academic and industrial groups target greener processes, eyeing transition-metal catalysts with high selectivity, recyclable media, and scaled-down environmental impact. Chemists dive into asymmetric synthesis, hoping to turn 4-ethylpyridine into chiral ligands for drug design or specialty catalysis. Analytical chemists study its trace presence in complex biological matrices, refining extraction and detection methods. Regulatory requirements, both from workplace safety and environmental emission, drive R&D funding towards closed-loop production and better personal protection gear. Some groups track 4-ethylpyridine derivatives as scaffolds for metal-organic frameworks or as phase-switch compounds, where reversible chemical switches draw commercial curiosity. Academic labs push for publication, but industry needs repeatability, scalability, and cost control; both sides need to keep innovation grounded in real-world practice.
Toxicity Research
Careful toxicology matters for any pyridine derivative, and 4-ethylpyridine proves no exception. Acute exposure irritates the mucous membranes and respiratory tract—common sense dictates strong ventilation and limited exposure duration. Chronic effects get trickier: animal studies have shown liver enzyme elevation and mild neurotoxicity at sustained high doses, though levels well above typical lab exposure. Regulatory bodies flag it as potentially harmful to aquatic organisms, so discharge or spill management draws government scrutiny. Long-term occupational studies remain limited, but incident reports push for stricter exposure monitoring—blood or urine sampling for those working in production or formulation. Carcinogenicity remains unproven, but precautionary labeling endures. Clinical data lags behind lab modeling, so good practice means assuming worse-case risk rather than gambling on incomplete information. Chemical safety training and robust physical controls stay front-line defense against rare but serious exposure scenarios.
Future Prospects
Compound demand ties closely to economic trends in pharmaceuticals, flavors, and advanced materials. As emerging fields expand—wearable electronics, personalized medicine, green chemistry—4-ethylpyridine finds new space. Smaller, more agile companies look to niche uses in rapid diagnostics and specialty catalyst design, while global chemical firms push for cost-cutting amid tighter regulations. The ongoing march toward greener manufacturing promises lower emission and waste standards, pushing process chemistry into the sustainability spotlight. Automation and machine learning might optimize both yield and safety, with remote sensors alerting for spills or leaks before risk grows. Research teams see value in mapping biological impacts at the molecular level, exploring how structure tweaks control toxicity or reactivity. For many, 4-ethylpyridine will keep serving as a backbone—a dependable platform for transformative chemistry and technology, so long as vigilance and creativity build hand-in-hand.
What is 4-Ethylpyridine used for?
What Is 4-Ethylpyridine?
Digging through the names of chemical compounds can feel like getting lost in a library with all the books out of order. Still, some of these compounds, like 4-ethylpyridine, play bigger roles than the name suggests. It’s an organic molecule with a strong, penetrating smell found in tobacco smoke, roasted foods, and even in some industrial processes.
Relevance in Research and Industry
4-Ethylpyridine rides under the radar in daily headlines, yet researchers and chemists know it well. Labs tap into it as a marker compound when analyzing flavor and aroma development. I spent some time in a flavor chemistry lab, and we constantly ran gas chromatography equipment, looking for 4-ethylpyridine among hundreds of volatile molecules in roasted coffee or baked goods. It helps analysts understand why some foods taste inviting or off-putting. For example, higher levels appear in some dark-roasted coffee beans, so you have to keep an eye on it to keep flavors balanced.
This molecule also becomes an ally for the tobacco industry’s quality teams. 4-Ethylpyridine pops up when they analyze cigarettes or cigars, especially when setting standards for product consistency and meeting regulatory limits. The U.S. Food and Drug Administration includes it in their list of harmful constituents found in tobacco smoke, not because it’s the most dangerous, but because it shows up every time you burn tobacco. Having tracked these chemicals in public health research, I’ve seen how important it is to know not just how much nicotine someone gets but also how much of these byproducts they breathe in with every puff. Authorities lean on data around 4-ethylpyridine for both regulatory checks and scientific studies on long-term exposure risks.
Building Blocks for Bigger Chemistry
Chemists see 4-ethylpyridine as more than a flavor molecule—it’s a handy piece for building larger, more complex materials. Some pharmaceutical projects use it when piecing together molecular scaffolds for experimental drug candidates. It brings a key connection spot that can help give a drug its unique shape and properties. I’ve seen it pop up in journals as a starting material for synthesizing specialty chemicals, helping chemists tweak molecules until they have just the right action in the lab or factory.
Public Health and Environmental Questions
Research teams, both academic and regulatory, have flagged 4-ethylpyridine as a concern in indoor air, tobacco smoke, and foods cooked at high heat. Its sharp odor even turns up in complaints about air quality near roasting plants or tobacco processing facilities. These complaints sometimes spark local health studies and calls for better ventilation in workspaces. I’ve helped analyze air quality complaints where molecules like this jumped above background levels and made life tough for workers or nearby neighborhoods. Simple solutions don’t always exist, but upgrading exhaust systems and monitoring emissions can make a difference.
Room to Grow in Knowledge and Safety
New studies keep pushing to clarify what long-term exposure means for workers and consumers who bump into 4-ethylpyridine through the air, food, or consumer products. Scientists need funding and access to up-to-date equipment for this work. Companies have a part to play, too, by tracking and reporting what they release and listening to the communities living or working nearby. Strong regulation, solid public health research, and transparency keep everyone safer, so more folks understand not just what’s in their product, but why every ingredient got there in the first place.
What is the molecular formula of 4-Ethylpyridine?
Looking Closer at 4-Ethylpyridine
Walking through the world of organic chemistry has taught me to appreciate the way small changes in structure lead to big shifts in properties. Take pyridine, a compound you find in everything from vitamins to industrial solvents. Add an ethyl group to the fourth carbon in the ring, and you get 4-ethylpyridine—an aromatic heterocycle with a distinctive profile.
Breaking Down the Formula
4-Ethylpyridine operates on a six-membered ring mostly made up of carbon atoms, with a single nitrogen thrown in the mix. If you’ve ever studied aromatic rings, you know their unique stability comes from the resonance found in the structure—in pyridine’s case, five carbons and one nitrogen form the backbone. Attach an ethyl group (two carbons and five hydrogens) at the fourth spot, and now you’ve built 4-ethylpyridine.
The puzzle comes together piece by piece: the base ring, pyridine, holds the formula C5H5N. Slide in the ethyl group, and you tack on C2H5. Combine both, and you arrive at C7H9N. Nothing fancy, just careful counting—two more carbons, four more hydrogens atop the base ring.
Importance in Research and Industry
People might shrug off minor modifications like that small ethyl group and its impact. In my experience, these “minor” details shape everything from reactivity and solubility to the way a molecule interacts with biological systems. 4-Ethylpyridine appears in flavor chemistry, analytical standards, and sometimes turns up as an impurity during certain fermentation processes in the spirits industry.
For researchers, a clear handle on molecular formulas goes beyond numbers on paper. Incorrect data can lead to wasted time, confusion in data sheets, and even flawed experimental outcomes—problems that cost money and credibility. Labs routinely rely on molecular formulas for stoichiometry, chemical ordering, and hazard assessments. Precision in that area keeps projects on track.
Why Accuracy and Transparency Matter
Science has always demanded rigor, but with the spread of misinformation online, trusted sources matter more. Resources like PubChem and NIST Chemistry Webbook offer precise references—both list the molecular formula for 4-ethylpyridine as C7H9N. Peer-reviewed databases prevent costly errors, something I learned by watching a mentor double-check everything before signing off.
This attention to accuracy flows into public trust. When industries publish their results or submit filings, auditors expect the basics to be right. Getting a formula wrong can start a cascade of issues, including compromised safety data and rejected patents. As global supply chains connect chemists everywhere, using standardized, clear nomenclature ensures teams remain on the same page.
Building Forward: Awareness and Education
Just memorizing formulas does little. Active learning, visualizing molecular structures, and tracing their origins in real work builds lasting understanding. Higher education has moved forward by mixing hands-on experiments with digital tools. Chemistry educators encourage students to question, verify, and reference credible sources. I often steered new lab workers toward open databases before ordering a gram of any new compound.
As careers branch out, lessons in accuracy and responsibility echo. Anyone working around chemicals, whether in university labs, medical research, or manufacturing, carries the shared duty of making sure every detail—down to a molecule’s formula—checks out. Mistakes at this level don’t just waste time, they can carry real consequences for safety and success.
Is 4-Ethylpyridine hazardous or toxic?
Understanding 4-Ethylpyridine in Everyday Terms
4-Ethylpyridine doesn’t often make headlines, but it has found a place in labs, industry, and even food science. Drawn from the world of organic chemicals, this compound shows up in research and manufacturing. Some people might recognize its name from tobacco smoke studies, which led to questions about its safety.
Health Risks and Exposure
Direct exposure to 4-ethylpyridine is rare for most folks. Still, scientific sources record it as an irritant, particularly to eyes, skin, and the respiratory system. Researchers in industrial hygiene agree it produces a strong, unpleasant smell, which hints at the need for caution. In the lab, the smell acts as an early warning signal for those handling it—higher concentrations in the air might follow a spill. Symptoms like headaches, nausea, and dizziness become likely at those levels.
One detailed toxicology report from the National Institutes of Health describes lab rats exposed to 4-ethylpyridine showing reduced activity and breathing trouble; higher doses led to more severe symptoms. This pattern matches what is seen with related chemicals in the pyridine family. In my years around chemical research, any compound that shows toxicity in animal studies makes me double-check safety sheets before use, no matter how common it seems in textbooks.
Environmental Impact
Beyond direct exposure, this chemical may cause harm in the environment. Scientists studying soil and water samples near industrial sites have flagged pyridine derivatives for their persistence. Some forms break down slowly, giving them time to move through soil or water—and this can threaten wildlife.
One real worry is aquatic life. Small studies found that fish exposed to high concentrations struggle to survive. Policy makers and environmental groups watch numbers like these. They often lobby for tighter controls, especially in regions close to chemical plants. Communities downstream from these sites have learned to ask tough questions about both wastewater treatment and air quality.
Occupational Safety and Regulations
Workplace rules matter here—OSHA and similar agencies outside the US keep tabs on chemicals like 4-ethylpyridine. Protective equipment, local exhaust ventilation, and limited work shifts all make a difference. The CDC’s National Institute for Occupational Safety and Health (NIOSH) updates guidelines for handling pyridine compounds often, and for good reason.
As someone who has dealt with hazardous chemicals, I pay close attention to updated Material Safety Data Sheets. Too many people overlook the importance of gloves and goggles until they come across a spill or chemical splash. Labs, workshops, and even university classrooms need clear rules and regular drills; the risks from accidental exposure don’t care if someone just started their job or has two decades of experience.
Looking for Safer Substitutes
Alternatives bring hope for safer workplaces and cleaner factories. Some researchers explore new synthesis routes or green chemistry protocols to limit or replace 4-ethylpyridine in finished products. These shifts take time, but working toward less hazardous chemicals pays off for everyone: employees, neighbors, and future generations.
My own experience echoes what many chemists say—prevention beats cure every time. Focusing on substitution, better engineering controls, and solid training keeps risks lower. As awareness grows, more companies and public labs look for technologies that turn away from reliance on compounds flagged for hazards.
What are the storage requirements for 4-Ethylpyridine?
Why Storage Conditions Matter for 4-Ethylpyridine
Anyone who has spent time handling chemicals understands how storage choices can shape safety. 4-Ethylpyridine isn’t an exception. With a sharp odor and a reputation for flammability, this compound carries risk if treated like something as harmless as table salt. Researchers handling pyridine derivatives will already know the headaches brought by volatile chemicals. Even a minor spill or leak means not just a mess, but safety hazards, health worries, and sometimes needing to evacuate a lab. Every chemist has run into the smell of pyridines—and nobody forgets it.
Basic Storage Notes
4-Ethylpyridine comes as a clear liquid. High volatility, low flash point—facts like these always push me to respect the bottle more than usual. Direct sunlight and open flames turn dangerous here. From my own experience in academic labs and the chemical industry, I’ve seen fire codes go out the window if storage becomes an afterthought. Older shelves and cluttered storage rooms help no one. This chemical deserves a space in a proper flammable materials cabinet, kept in a cool spot away from heat. Stories circulate among lab workers about bottles left near hot pipes or even on sunny windowsills, leading to problems no one wants to clean up.
Humidity and moisture may not rust the bottle, but they open doors to chemical degradation, and sometimes to more dangerous byproducts. Tight caps go a long way. Too often, expired or poorly sealed containers turn up, releasing fumes. The smell alone can close off a workroom for days. Local ventilation—fume hoods, or at least a fan—keeps daily work healthy and stops invisible vapors from sharing the air with everyone nearby. In shared spaces, label every bottle with clear, chemical-resistant ink. Quick identification saves time during rushes or emergencies.
Personal and Environmental Health Concerns
Every year, hospital visits and workplace incidents emerge because someone didn’t think enough about safe chemical storage. 4-Ethylpyridine vapors create headaches, dizziness, or worse for those exposed. Contact with skin or eyes means possible irritation or even burns, and accidental ingestion or powerful inhalation should always lead straight to medical help. Ensuring personal protective gear—like gloves made from nitrile and fitted goggles—protects not just the handler but also others sharing benches or storage rooms. Never transfer chemicals to unmarked bottles. Too many near-misses and horror stories have started with a plain glass container and a lost label.
Beyond personal safety, the earth remembers sloppy handling. A spill sneaks into the drain, and local water becomes another risk point. Responsible storage setups prevent those “oops” moments: secondary containment trays, absorbent pads nearby, and spill kits ready help turn possible disasters into manageable incidents. Regular checks on inventory and expiration dates keep everything fresh, with less chance of hidden bottles going unnoticed.
Simple Shifts for Safer Labs
Chemical safety improves with shared knowledge and practice, not just regulations. Training newcomers and long-time workers builds habits: always return 4-Ethylpyridine to its safe spot, seal it tightly, update the logbook. Everyone benefits from a culture where double-checking and safety drills come naturally. Even if regulations change, old lessons—cool place, no flames, secured caps—never lose value. By focusing on these habits, every person in the room can help protect colleagues, the building, and the community outside the door.
What is the boiling point of 4-Ethylpyridine?
Why Boiling Points Matter in Chemistry and Industry
Talk to anyone working with chemicals, and the boiling point comes up as quickly as safety glasses. It’s not just a number in a textbook. It’s the difference between safe handling and dangerous fumes in the air. Boiling points speak to how a substance behaves under heat and pressure. One substance I’ve crossed paths with more than once—4-ethylpyridine—brings up interesting points about lab safety, research planning, and industrial pursuits.
Facts You Can’t Ignore: 4-Ethylpyridine’s Boiling Point
4-Ethylpyridine boils at around 162°C (323.6°F). People might skim past that figure, yet it packs real value. Laboratories rely on this kind of information for distillation, storage, and transport. If the boiling point were even a few degrees different, storage shelves, glassware, and safety equipment would change. In my time cataloging reagents, overlooking a boiling point meant broken equipment and frazzled nerves—not fun for anyone involved.
Practical Impact in Research and Manufacturing
4-Ethylpyridine serves as more than a trivia answer. This compound finds its way into advanced materials, catalyst development, and pharmaceutical research. Knowing the boiling point influences which solvents work best and which aren’t worth the risk. Incorrect storage or handling because of neglected data can damage projects and compromise safety. If you’ve ever been in a lab where an unexpected vapor cloud started forming, you know that these details are not minor.
Transporting materials like 4-ethylpyridine requires accurate temperature controls. Industrial sites use this knowledge when scaling up from flask to vat. I remember a pilot plant project where missing a key physical property set us back a month. Engineers had to retrofit equipment, wasting both time and money—resources always in short supply. Boiling points might look academic, but in reality, they mold production schedules and budgets.
Risks and Safety: Lessons from the Field
Compounds boiling above 150°C need special ventilation and containment. Improper airflow can let hazardous vapors drift, creating risks for anyone nearby. NIOSH and OSHA have published warnings about inhaling pyridine vapors. Exposure leads to headaches and, in higher doses, serious health problems. Data-driven protocols can’t get thrown out the window—real-life accidents have painful consequences.
Chemical suppliers provide boiling points not just for the sake of it, but to keep workers out of harm’s way. Training new staff in a lab, stressing the importance of checking boiling points ranked high on the list. Too many people assume they know a substance until unpredictable events prove otherwise.
Better Practices for Handling 4-Ethylpyridine
Mistakes offer lessons for the future. Keeping up-to-date labels, double-checking chemical databases, and using digital thermometers save labs and factories from costly errors. Temperature monitoring technology can track even minor changes, alerting teams before trouble brews. Consistent review of equipment against actual boiling points keeps projects humming along without violent surprises.
More facilities now mandate digital recordkeeping and automatic control systems. These tools turn the boiling point from a possibility to a practical parameter. Labs and businesses find that building a culture of accuracy around properties like boiling point pays off through reduced incidents, smoother runs, and fewer headaches down the road.
Closing on the Details that Matter
Boiling points sit at the intersection of safety, efficiency, and progress. 4-Ethylpyridine, with its 162°C boiling threshold, challenges teams to stay sharp and respect the data. It reminds us that chemical properties deserve careful attention, not just for productivity, but for health and security every day.
| Names | |
| Preferred IUPAC name | 4-Ethylpyridine |
| Other names |
4-Ethylpyridine 4-Ethyl-pyridine para-Ethylpyridine p-Ethylpyridine |
| Pronunciation | /ˌfɔːrˌiːθɪl.pɪˈrɪd.iːn/ |
| Identifiers | |
| CAS Number | 100-47-0 |
| Beilstein Reference | 87378 |
| ChEBI | CHEBI:38103 |
| ChEMBL | CHEMBL234378 |
| ChemSpider | 68233 |
| DrugBank | DB04220 |
| ECHA InfoCard | 100.057.882 |
| EC Number | 207-975-3 |
| Gmelin Reference | 8727 |
| KEGG | C06217 |
| MeSH | D000075634 |
| PubChem CID | 9577 |
| RTECS number | UY9625000 |
| UNII | YXGCYTYYOY |
| UN number | UN2438 |
| Properties | |
| Chemical formula | C7H9N |
| Molar mass | 107.16 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | unpleasant, pyridine-like |
| Density | 0.948 g/mL at 25 °C(lit.) |
| Solubility in water | Soluble |
| log P | 1.51 |
| Vapor pressure | 1.4 mmHg (at 25 °C) |
| Acidity (pKa) | 5.98 |
| Basicity (pKb) | 5.86 |
| Magnetic susceptibility (χ) | -38.2·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.508 |
| Viscosity | 1.017 cP (20°C) |
| Dipole moment | 1.63 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 297.4 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 93.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3075.6 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Danger |
| Hazard statements | H226, H301, H311, H315, H319, H331, H335 |
| Precautionary statements | P210, P261, P280, P301+P312, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 3-2-2 |
| Flash point | 52 °C (126 °F; 325 K) |
| Autoignition temperature | 430 °C |
| Explosive limits | Explosive limits: 1.1–8.9% |
| Lethal dose or concentration | LD50 rat oral 400 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 630 mg/kg |
| NIOSH | SR2710000 |
| PEL (Permissible) | PEL: 2 ppm (8 mg/m³) |
| IDLH (Immediate danger) | IDLH: 100 ppm |
| Related compounds | |
| Related compounds |
Pyridine 2-Ethylpyridine 3-Ethylpyridine 4-Methylpyridine 4-Propylpyridine |

