The Science and Significance of 4-Dimethylamino-6-(2-Dimethylaminoethoxy)Toluene-2-Diazonium Zinc Chloride Salt
Looking Back: The Route to Modern Diazotization
Scientists didn’t always have ready access to the complex azo compounds that shape today’s colorants, photosensitive papers, and laboratory indicators. Research on diazonium compounds traces back to the mid-1800s, beginning almost by accident. The discovery of diazonium salts arose from tireless, sometimes risky trial and error with aromatic amines and nitrous acid—a period when the risk of unexpected explosions plagued every experimentalist. It’s easy to forget how recent these advances are. The leaps made in dyestuff chemistry, particularly in Germany and the UK, turned color from a natural rarity to an industrial staple. The evolution toward salts like 4-dimethylamino-6-(2-dimethylaminoethoxy)toluene-2-diazonium zinc chloride reflects a focus on linking stability, solubility, and performance, not only for bright shades but also for more reliable reactions in the laboratory.
What Defines This Compound?
The drawn-out name hides real complexity. This molecule offers more than basic labeling. The diazonium group is exceptionally reactive and, combined with extra electron-donating substituents like dimethylamino and ethoxy, it turns this salt into a powerhouse for both classic and novel transformations. The addition of zinc chloride stabilizes the otherwise sensitive diazonium, giving chemists enough control to push this material into various applications, whether making lake pigments or designing new chemical sensors. In my view, every time I’ve handled such salts, I’ve appreciated the convenience compared to the more temperamental, explosively unstable diazonium compounds that shaped my undergraduate lab experience.
What It Looks and Feels Like in the Lab
Chemically, the salt lands between crystalline and powdery, often yellowish or orangey, with a faint odor that sometimes calls up childhood memories of musty storerooms and faded textbooks. It dissolves fairly well in water, especially when the zinc chloride content is optimized. Reaction behaviors remind me of the unpredictability that always lurks with diazonium chemistry—a splash, sudden warmth, or stray contaminant can make things lively, sometimes alarmingly so. It doesn’t just sit idly in storage. Humidity or organic matter might start a reaction, so safe handling really means treating every step as a potential hazard. Over the years, manufacturers have standardized technical grades and labeling, laying out precise details for minimum purity, storage conditions, and shelf-life. That reassures research teams but doesn’t remove the need for common sense and personal protection.
On the Shoulder of Technical Progress
I remember the shift as standardized protocols started rolling in, especially in Europe and North America. Barrels arrived stamped not only with purity and lot numbers but also elaborate instructions and warning symbols. These labels keep evolving, but at their root, they reflect the constant struggle between industrial convenience and laboratory safety. Today’s packages bring QR codes with direct access to data sheets, but what matters is that content never gets buried beneath administrative bloat. As research demands grow, smaller sample packaging and tailored blends have gained traction in academic circles and startup labs, where overhead room for error shrinks fast.
Piecing Together the Chemistry
Crafting this salt takes fine-tuned timing and precision. Synthesis builds on diazotization: a reaction of aromatic amines (specifically the dimethylamino-toluene derivative) with sodium nitrite and hydrochloric acid under tightly-controlled cold conditions. The resulting diazonium chloride pairs with zinc chloride to stabilize the product—an essential step, since stray temperature spikes or poor mixing can turn a promising batch into a failed, sometimes dangerous mess. The method forces anyone attempting it to be patient and attentive; those who rush, or skip best practices, sooner or later pay for their shortcuts by losing product, or worse. These are lessons I learned with burnt fingers and wasted afternoons in university research groups.
What Always Happens Next: Chemical Reactions and Experimentation
This chemical doesn’t hang around for long in its “pure” state. In practice, labs use it in coupling reactions, bringing out vivid azo dyes on the way to pigments that paint our everyday objects. The extra electron-donating groups offer resonance effects, supporting rapid and reliable reactions—most famously as partners in the synthesis of advanced dyes and photoactive films. Whether attached to aromatic compounds like resorcinol, phenol, or naphthols, every tweak to the structure translates into vivid shifts in color or sensitivity. This potential for modification—swapping, elongating, or altering its side chains—drives innovation in sectors as different as forensic science and solar film design. I’ve watched research groups invent clever modifications, aiming for brighter shades, ultraviolet resistance, or molecular sensing capability, sometimes getting as close to a patent as red tape and funding allow.
The Many Names of One Compound
Jargon shifts rapidly in chemistry, and this salt goes by various handles: some call it “dimethylaminoethoxy diazonium chloride with zinc,” while older research sticks to legacy catalog numbers or abbreviations. Some suppliers mark their batch as a “Zinc-stabilized Fast Salt,” especially for pigment work. Industry naming often chases marketing trends, cycling through specialized synonyms as new applications pop up. At root, the science speaks loudest—every new name or trade alias begins with the same parent structure and core hazards.
Staying Safe: Standards Aren’t Optional
Anyone who’s spent time with diazonium salts knows to work slow and stay cautious. Even with zinc stabilization, these compounds can decompose violently, especially if left warm, dry, or mixed with contaminants. Strict adherence to standardized handling practices isn’t a chore; it’s a necessity. Gloves, goggles, and fume hoods represent minimum requirements. Modern safety data speaks to air reactivity, light sensitivity, and, in some cases, unexpected allergic responses. In my view, stories of avoidable accidents always trace back to skipped steps or overconfidence. Better technology and industry watchdogs have brought down incident rates, yet accidents and exposures still crop up for those treating such salts as “ordinary.” Many in my circle remember classmates burned or scared off chemistry for good by underestimating these quirky, high-strung molecules.
In the Field: How This Compound Gets Used
Large-scale industrial progress draws much of its magic from precisely these sorts of specialized molecules. The salt steps up most in the pigment and dye industries, especially for textiles, synthetic leathers, and specialty inks where fastness and color intensity set the product apart. Its fine-tuned molecular structure gives deeper tones and longer-lasting brilliance than many alternatives, adding value that manufacturers can’t easily substitute out. Beyond dyes, the compound gets pressed into service as a diagnostic tool in analytical labs, where reactive diazonium groups help pinpoint the presence of certain organic substrates. I have seen patent disclosures making use of its photoreactivity in inks for circuit board etching or even in exotic biomedical imaging probes. No one in specialty chemistry stays untouched by the edge this class of molecules confers.
The Never-Ending Curiosity: Research and Development Directions
Researchers keep exploring new corners of diazonium chemistry, chasing better stability, greener synthesis, and enhanced single-molecule sensitivity in sensor applications. Most recently, eco-friendly stabilizing agents compete with zinc chloride for environmental approval. Green chemistry mandates have researchers tinkering with solvent systems, aiming to minimize energy input and toxic byproducts. Many research teams now focus on nanoencapsulation and solid-state reactions, creating safer routes to handle and deploy otherwise dangerous reagents. In conferences and workshops, newcomers swap notes with veterans, learning which routes shave hours off syntheses, or which byproducts signal success or failure. From cancer diagnostics to OLED design, each tweak to this molecule’s backbone potentially sets a new benchmark for performance or safety.
Hazards Under the Microscope: Toxicity Research
Every advance brings a responsibility to understand risk. Ongoing toxicity studies suggest diazonium salts, no matter how stabilized, can threaten health through accidental inhalation or contact. Chronic exposure in industrial settings links historically to urinary tract cancers, though modern controls and substitution efforts have slowed the rate of such cases. Animal studies point to mutagenic risk with improper protective measures. For years, academic lore featured cautionary tales—some true, some exaggerated—of careless students ending up in the emergency room. Real data supports many of these anecdotes, and industrial hygienists now keep close tabs on air quality, uptake, and handling procedures. The push for greener chemistry partly grew out of the long, hard look experts took at the silent legacy of legacy dyes and pigments.
What Comes Next?
Progress won’t simply mean more of the same. The next big leap may come from hybrid molecules—mixing diazonium groups with new stabilizers to open up safer, wider-ranging uses. Pushes to eliminate toxic metals from everyday chemistry may dethrone zinc chloride, yet adoption moves slowly because end-users want proven results. Research funding trends hint at priority for sustainable chemical production and closed-loop manufacturing, both of which may soon trickle down to the azo dyes sector. For anyone in specialty chemicals, the story of 4-dimethylamino-6-(2-dimethylaminoethoxy)toluene-2-diazonium zinc chloride salt isn’t over. As regulation, demand, and technical discovery keep changing, we’ll keep adapting, striving always to balance creative innovation with responsibility—not just in the lab, but out in the world it colors.
What is 4-Dimethylamino-6-(2-Dimethylaminoethoxy)Toluene-2-Diazonium Zinc Chloride Salt used for?
Color Formulation: The Real Value in Dye Chemistry
This mouthful of a compound—4-Dimethylamino-6-(2-Dimethylaminoethoxy)Toluene-2-Diazonium Zinc Chloride Salt—plays its part in the world of dyes. In my years watching the textile supply chain, I’ve seen factories rely on specialized diazonium salts to turn bland cloth into fabric loaded with character. Here’s the gist: this chemical shows up as a key component in producing azo dyes, a family responsible for some of the most vivid reds, oranges, and yellows in standard synthetic textiles.
A number of synthetic coloring processes depend on diazonium salts’ knack for coupling with other aromatic compounds. The efficient reaction produces stable bonds and strong, reliable colors. Fabric manufacturers don’t just chase a pretty result. They want color that clings to cotton or wool through wash after wash, even when exposed to sunlight. Without chemicals like this, those colors would fade fast—or wash out entirely.
Industry Practices: Why Precision Matters in Coloring
A company working with large textile orders needs consistency. When working with wool, silk, or synthetic fibers, dye houses weigh the exact mixture for each batch. I’ve seen operators tear their hair out from uneven shades or early wash-out. The chemistry behind their work solves those problems, often relying on a stable intermediate such as this diazonium salt. It dissolves well, handles predictably, and lets technicians match hues from small samples all the way to tons of dyed cloth.
Besides textile dyeing, the field of printing inks benefits from this compound. Printers look for color that stands up against smudging and light exposure. In the graphic arts, no one wants a brilliant magazine cover that dulls in a week. By tackling the coupling reactions with stable diazonium intermediates, ink makers can offer bright, reliable inks for high-end publications and packaging.
The Environmental Angle: Trade-Offs and Future Solutions
Production and use of functional dyes come with a load of environmental baggage. Synthetic dyes carry a reputation for tough wastewater, often loaded with persistent organic molecules. Back when I first visited dye houses, chemical runoff wasn’t a big talking point. Times changed. Now, people press for greener processes, whether by pre-treating effluent or using better-controlled reaction conditions.
Research into biodegradable dyes and advanced catalytic treatments can cut the environmental impact. Some groups look for enzyme-based systems that mimic biological dye pathways, reducing dependence on metals and harsh oxidants. Others fine-tune diazonium chemistry to lower temperatures and reuse solvents, trimming energy needs and waste.
Handling and Worker Safety: No Room for Shortcuts
I remember safety procedures as stricter every year, and with good reason. Diazonium salts carry explosive risks in dry conditions. Factories enforce careful moisture controls and ventilated storage. Zinc chloride adds a separate set of health protocols to keep workers protected from irritation and toxicity. Without these layers of caution, the price for bright color could come out of workers’ health or local ecosystems.
Embracing Change in Chemical Manufacturing
People count on durable chemicals like this diazonium salt to keep their textiles and prints looking sharp. At the same time, there’s a growing appetite for transparency and environmental responsibility. Smarter research, tighter controls, and full adherence to safety guidelines will decide how well this industry balances market demand with real environmental care.
What are the safety precautions when handling this compound?
Respect the Science by Protecting Yourself
Many folks working in labs or industrial settings deal with substances that demand respect because of real risks. Once I nearly learned this lesson the hard way with a fuming acid, ignoring a splash shield for a split second—my lab coat bore the brunt but woke me up fast to the basics. Good gear saves skin and vision, and it’s not up for debate. Gloves suited to the job, tight goggles, a proper lab coat, and sturdy shoes all form your first line of defense. The confidence to handle a tricky bottle begins with a barrier between you and the worst a chemical could offer.
Why Ventilation Isn’t Just a Box to Check
Chemical vapors creep into lungs quietly. In a stuffy room, fumes build fast—at work, one simple whiff can turn a regular day upside down. Investing in a fume hood or local exhaust isn’t overkill. It’s routine. I always test the airflow before popping a lid or mixing anything volatile. If your eyes sting or you pick up a strange smell, that means trouble is already in the air. More than once, I've seen someone skip the hood “just for five minutes,” only to set off an evacuation. Keeping air moving could be the one thing between routine work and a medical emergency.
Training Isn’t Optional
Labs change, protocols shift, and new people join fast. I’ve watched new hires guess their way through a procedure, only to realize nobody showed them the right spill cleanup methods. Continuous training isn’t a luxury; it’s survival. Getting clear instructions on what to do if a bottle cracks or a beaker boils over makes the difference between cleaning up safely and turning a small accident into a disaster. Veterans on the floor—like I became after a few years—owe it to everyone to pass along these habits. It’s better to ask three times than get careless once.
Safe Storage Makes for Safe Handling
Out of sight doesn’t mean safe. Neatly shelved chemicals, separated by shelf and clearly labeled, reduce the urge to grab whatever is closest in a rush. As a student, I saw acids stored much too near bases—one clumsy mix up brought the fire department in. Flammable liquids belong away from spark sources and inside proper cabinets. Strong oxidizers and solvents can’t sit together. It takes a bit more work to set up the right way, but it spares everyone a red-faced moment or worse.
Preparation Turns Panic Into Action
Spills and splashes happen, despite the best intentions. Having a shower, eye wash, and spill kit close can turn a critical moment into a fixable one. At one point, a colleague splashed a caustic compound—she knew to use the eyewash fast and called our safety lead, so she escaped without lasting harm. Keeping emergency contacts and procedures right by the door makes them easy to find, not just for regulars but for visitors as well.
Make Culture Matter
Working safely with hazardous compounds starts with a culture of care and accountability. Speak up if you spot a shortcut or sloppy storage. My experience taught me that safety grows out of everyday decisions, not just manuals. If everyone chips in, a lab or shop becomes less tense and more productive. When we treat every bottle and beaker with a little respect, the workday ends with everyone leaving whole.
How should 4-Dimethylamino-6-(2-Dimethylaminoethoxy)Toluene-2-Diazonium Zinc Chloride Salt be stored?
Understanding the Substance
Handling chemicals like 4-Dimethylamino-6-(2-Dimethylaminoethoxy)Toluene-2-Diazonium Zinc Chloride Salt isn't something you just figure out as you go along. This one falls into the category of diazonium salts, which means it can break down fast, releasing nitrogen gas. This sort of reactivity causes a real headache if you don’t store it right. Not only could you lose valuable material, the release of gas can make for dangerous situations. Folks who work around these chemicals, from chem majors to lab techs, have seen cabinets buckle or lids pop due to missed details.
The Role of Temperature and Moisture
Everyone who’s handled a reactive chemical knows high temperatures shorten shelf life. This compound breaks down even faster in a warm space. Room temperature isn't good enough. My own experience in shared academic labs taught me that someone always “forgets” to double-check the fridge settings, which could spell disaster for a batch of diazonium salt. Chemists with more gray hairs than me recommend a storage fridge set below 5°C, with strict logs to keep an eye on who last opened the door. Humidity throws in an extra curveball. Any dampness lets water creep in and start decomposing these diazonium salts. It doesn’t take much for a clumpy powder or fizzing vial to ruin your morning. Keep everything sealed, with silica gel or another desiccant in the container. That’s a detail I learned the hard way—lost a few samples before I made the habit stick.
The Container Makes a Difference
Plastic rarely stands up to chemicals for long, especially when you’re storing compounds that might react with traces of the container. Glass bottles, with chemically-resistant lids, have stood the test of time. Each one gets clearly labeled, with dating and hazard information. Sometimes good labeling falls through the cracks. In a real lab, one poorly marked bottle can lead to all sorts of mix-ups. Proper labeling, often double-checked at every transfer, really isn’t just bureaucracy—it’s what saves you from the “unknown white powder” mystery that no one wants to solve.
Protecting Against Light and Contamination
UV light gives another fast lane to decomposition for this salt. Store the bottles in a solid, opaque cabinet, not a handy shelf under bright lights. After one sunbeam incident in an undergrad lab, even the teaching assistants started taping over glass doors. Doing your own quick light-test in each storage space can reveal overlooked spots. Work with clean gloves, and avoid opening the main supply each time you dose a reaction—divide out small samples so you don’t expose the whole batch to air or moisture every single day.
Safe Access and Responsibility
Access control isn’t just about regulation. Fewer hands in the storage fridge means less chance of mishandling. Trained staff know the importance of secondary containers in case something leaks or breaks. Some places log entries by hand, others use keycards, but the logic remains: limit the risk, check the inventory regularly, and make sure anyone on your crew or in your lab understands the hazards. Training sessions help drive home the point that even a tiny quantity of certain chemicals can put the whole space at risk if you get sloppy. A regular quiz or walkthrough isn’t anyone’s favorite, but they keep safety habits sharp.
Building a Good Chemical Culture
Most labs talk about safety, but not every lab truly lives it. A good culture isn’t about fancy posters or overwhelming policies. Daily routines—like logging fridge temps, swapping out desiccants, making sure every bottle has a clear date—build habits that keep everyone safe and work flowing. Every new chemist brings new eyes, and the most experienced hands know things can turn ugly if shortcuts sneak in. Recognize when something feels off, talk to coworkers, and never treat safe storage as a side task. There’s no prestige in fixing messes that never needed to happen in the first place.
What is the chemical formula and molecular weight of this product?
Why These Numbers Matter
Every product used in labs, factories, and even homes starts with a formula. That formula—those tiny clusters of letters and numbers—tells a big story. For chemists, engineers, or quality managers, the chemical formula lays out exactly what’s inside the bottle or the barrel. No assumptions, just straight facts. Take sodium chloride: NaCl. Two elements, sodium and chlorine, bond together in a fixed ratio. This fingerprint of sorts doesn’t change from one factory to another. If you know the formula, you know what reactions it can cause and how it might behave next to other compounds.
The molecular weight sits right next to the formula in importance. Think of it as the sum of each atom’s mass in the formula, measured in grams per mole. If you have NaCl again, sodium weighs about 22.99, chlorine about 35.45, so together, 58.44 grams per mole. With that number, scaling up becomes possible. Say you want to make a solution for an experiment. With the molecular weight, you weigh out the exact amount needed for a precise result. No guesswork; the numbers do not lie.
Making Informed Choices
Safety and compliance ride on these details. I’ve seen cases where a missing molecular weight led to dosing errors, and the result was wasted product and sometimes dangerous spills. Rules from regulatory bodies like the FDA or EPA do not exist to burden anyone. They push for this information because lives and resources depend on it. For shipping hazardous chemicals, for example, transporters need to know exactly what’s inside. The label must match the true contents, or people could get seriously hurt. The formula and weight tell responders what precautions to take in case of leaks or accidents.
In my years working with quality systems, the products backed by strong documentation almost always reached the customer without problems. Plants using clear chemical formulas and molecular weights tracked fewer recalls and customer complaints. Hospitals only work with suppliers who publish this information. That peace of mind keeps patients safe. Teachers, too, won’t let students run experiments without the right math. One wrong assumption changes the outcome for everyone in the room.
How to Improve the Situation
Companies can set up digital systems for recording and sharing these details with buyers. You don’t need the fanciest tools. A PDF with certificate of analysis will do the trick, as long as the right information is on there: chemical formula, molecular weight, purity, and supplier contact. QR codes on packaging can link to verified lab results. Small companies might partner with third-party labs to audit and validate their data. This investment pays back with customer trust and fewer legal headaches down the road.
On the user’s side, it helps to double-check certificates and only source from trusted makers or distributors. If a supplier won’t tell you the formula and weight, there’s a reason to walk away. Labs should train new staff not just in handling materials, but also in checking paperwork and watching out for inconsistencies. It’s not just bureaucracy. It can mean the difference between success and disaster.
Complete, accurate, and accessible chemical information gives every part of the supply chain a foundation to build on. Anyone who handles, processes, or sells chemicals gains from knowing exactly what they’re dealing with, both for safety and smarter business.
Is this compound available in different purities or grades?
Real Effects from Real Differences
Every time someone asks if a chemical compound is available in different purities or grades, a serious conversation about quality and purpose opens up. As someone who has worked in a lab and in industry, I can tell you: small differences in purity lead to huge differences in outcomes. It’s not only about scientific accuracy—it's about safety, money, and sometimes even public health.
Why Purity Isn’t Just a Number on a Label
A lot of people imagine purity as some kind of arbitrary scale, but in reality, the stakes run pretty high. Pharmaceuticals, for instance, require a level of consistency and cleanliness that’s far stricter than standard industrial applications. Trace contaminants in pharmaceutical-grade compounds can lead to unpredictable side effects. Food-grade purity, too, gets governed by stringent regulations. Meanwhile, technical or industrial grades get used in processes where small amounts of impurity won’t skew results or endanger users.
Years ago, I saw a situation where using an industrial-grade solvent in a food processing plant ruined an entire batch of product. The minuscule difference in purity included one contaminant that reacted badly with food ingredients. Regulatory bodies, including the FDA and EPA, don’t recommend grade distinctions as mere guidelines—they’re legal requirements in many scenarios.
Where the Real Hazards Lie
Purity links directly to risk. Laboratory researchers learn this lesson early: choosing the wrong grade brings headaches, ruined experiments, or even dangerous reactions. Impurities serve as wild cards—sometimes causing explosions or illnesses that catch even skilled hands by surprise. The Thalidomide disaster of the twentieth century, though more about chirality than gross purity, underlines how the tiniest differences in makeup can claim lives.
Cost, Waste, and Environmental Impact
Labs and factories buying higher purity than necessary face runaway costs. But going cheap with the wrong grade creates more troubles—faulty products, regulatory violations, and environmental contamination. The electronics industry, for example, invests heavily in "semiconductor grade" chemicals. Even a slight contaminant can turn a promising new circuit into scrap metal.
Waste management grows complex as impurities climb. Treatment plants burn energy removing by-products that originated from lower-purity input materials. The more pure the starting material, the simpler and greener the disposal at the back end.
The Solution: Match Grade to Need—and Never Guess
It’s tempting to save a few bucks on raw materials, but risk rarely pays off. Every industry, from winemaking to vaccine production, needs a careful matching of chemical grade to application. Standards set by groups like ASTM, USP, or ISO exist for a reason. Labs and factories must always check technical data sheets, supplier certifications, and safety documentation. Relying on guesswork leads to batch recalls, sick workers, or angry regulators.
Transparency matters, too. Suppliers must let customers know exactly what they’re getting—no one should have to chase down a certificate of analysis or struggle to get straight answers. In my own work, demanding this clarity up front saved projects and, sometimes, saved lives. The next time purity comes up, don’t treat it like a boring footnote. A careful choice up front brings peace of mind and better results down the line.
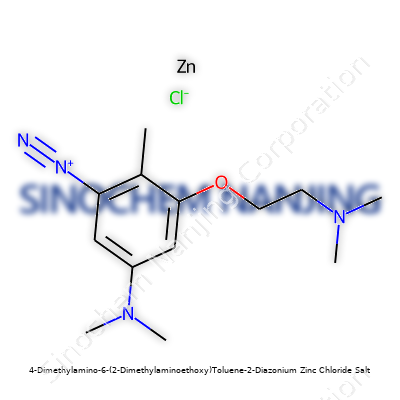
| Names | |
| Preferred IUPAC name | [4-(Dimethylamino)-6-[2-(dimethylamino)ethoxy]-2-(diazanyliumyl)toluene]zinc dichloride |
| Other names |
Fast Garnet GBC C.I. 37210 C.I. Azoic Diazo Component 48 Garnet GBC Salt C.I. Developer 35 |
| Pronunciation | /daɪˌmɛθɪl.əˈmiːnoʊ sɪks tuː daɪˌmɛθɪl.əˈmiːnoʊ ɪˈθɒksi ˈtɒljuːiːn tuː daɪˈæzəniəm zɪŋk ˈklɔːraɪd sɒlt/ |
| Identifiers | |
| CAS Number | 21436-97-5 |
| 3D model (JSmol) | `3Dmol:'Cn1c(ccc(c1N=NC2=CC=CN(C)C2)C)OCCN(C)C.Cl[Zn]Cl'` |
| Beilstein Reference | 1239760 |
| ChEBI | CHEBI:52234 |
| ChEMBL | CHEMBL2105938 |
| ChemSpider | 23070002 |
| DrugBank | DB13335 |
| ECHA InfoCard | ECHA InfoCard: 100_210_799 |
| Gmelin Reference | 89697 |
| KEGG | C17055 |
| MeSH | D008089 |
| PubChem CID | 139642043 |
| RTECS number | GF8750000 |
| UNII | J7A8B84VFS |
| UN number | UN3432 |
| CompTox Dashboard (EPA) | C463471 |
| Properties | |
| Chemical formula | C13H22N4OZnCl3 |
| Molar mass | 637.65 g/mol |
| Appearance | light yellow to yellow solid |
| Odor | Odorless |
| Density | 1.2 g/cm3 |
| Solubility in water | Soluble in water |
| log P | -2.7 |
| Acidity (pKa) | 10.1 |
| Basicity (pKb) | pKb: 5.1 |
| Magnetic susceptibility (χ) | -35.0 x 10^-6 cm³/mol |
| Refractive index (nD) | 1.575 |
| Dipole moment | 8.05 Debye |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 589.82 J·mol⁻¹·K⁻¹ |
| Hazards | |
| Main hazards | Harmful if swallowed; causes skin irritation; causes serious eye irritation. |
| GHS labelling | GHS07, GHS09 |
| Pictograms | GHS07,GHS09 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H334, H335, H400 |
| Precautionary statements | P261-P264-P271-P272-P280-P302+P352-P305+P351+P338-P333+P313-P337+P313-P362+P364 |
| NFPA 704 (fire diamond) | 1-2-0-酸 |
| Lethal dose or concentration | LD50 (oral, rat) >2000 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1600 mg/kg (rat, oral) |
| NIOSH | Not Listed |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | 0.3 mg/m³ |
| Related compounds | |
| Related compounds |
Benzenediazonium chloride Tosyl chloride 4-N,N-Dimethylaniline 2-Dimethylaminoethanol Diazotized aromatic amines |

