4-Cyanobenzoic Acid: More Than a Building Block
Historical Development
Learning about the story behind 4-cyanobenzoic acid gives context to its value in modern chemistry labs. Researchers in the early twentieth century were on the hunt for organic compounds that could push the boundaries of dye and pharmaceutical research. Benzonitriles, with their strong -CN group, piqued attention since they opened up new reaction pathways. In this environment, scientists managed to synthesize 4-cyanobenzoic acid, amassing both academic curiosity and practical demand. The drive to produce affordable intermediates for polymers, pigments, and specialty chemicals set a fire under industrial chemists, and this particular molecule found its place as a sturdy link in the supply chain for multiple sectors. Year after year, chemists continued to adapt and improve its synthesis, shaving off steps, cutting waste, and squeezing more yield out of each reaction.
Product Overview
Just looking at 4-cyanobenzoic acid, with its benzene ring dressed with a carboxylic acid group and a cyano group—both placed opposite each other—one can tell this is not another simple organic acid. The molecule blends aromatic chemistry with functional group versatility. In practice, manufacturers turn to this chemical as a core intermediate for making specialty polymers and active pharmaceutical ingredients. Its robust structure stands up well in synthetic schemes that need both stability and a handle for further chemical reactions. In my years working with organic synthesis, I’ve seen the compound show up classically in projects that need strong molecular scaffolds. Formulators also value its ability to work seamlessly in multi-step synthetic routes. All these factors have helped make it a regular item on chemists’ shopping lists in labs and production plants.
Physical & Chemical Properties
4-cyanobenzoic acid presents itself as a white to pale crystalline powder, reflecting a purity that’s essential for high-quality research and production. It melts at a temperature high enough to make it manageable but not so stubborn as to require excessive heating, typically in the range above 200°C. The compound carries a faint, characteristic odor and dissolves modestly in water, performing better in organic solvents. Chemically, the combination of carboxyl and cyano groups means the molecule can undergo both nucleophilic substitution and electrophilic activation. The aromatic ring helps maintain a certain rigidity, allowing for tight packing in crystalline form and slow, steady dissolution rates. It’s the stability, both chemically and physically, that ensures consistent behavior whether the compound is being stored, transported, or incorporated into further synthesis.
Technical Specifications & Labeling
In practice, industries demand precise specifications for 4-cyanobenzoic acid. This usually means rigorous testing for purity, which can exceed 99 percent in analytical and pharmaceutical-grade material. Real laboratories need this level of clarity to ensure reproducibility. Labeling on containers typically highlights batch number, purity level, potential impurities—such as residual benzoic acid or meta-cyano analogs—and proper storage temperatures to avoid degradation. I’ve seen quality control teams press suppliers hard for certificates of analysis that detail every detectable contaminant. As a buyer or user, looking for well-labeled containers and transparent specifications not only prevents nasty surprises but also supports downstream compliance, especially where product recall risks carry heavy consequences.
Preparation Method
Sourcing 4-cyanobenzoic acid relies on methods rooted in proven organic chemistry. One common route involves the benzonitrile core, which undergoes carboxylation at the para position. Another practical approach involves controlled oxidation or hydrolysis of 4-cyanotoluene using specific catalysts. The development of catalytic systems, especially those minimizing environmental hazards, has been key. Factories look to processes that reduce solvent use, cut out particularly toxic chemicals, and boost yield with minimal energy input. These considerations matter hugely for both cost and environmental footprint. In my observation, technicians now lean toward greener procedures in keeping with regulatory pressure and the pop-up of environmentally conscious procurement policies.
Chemical Reactions & Modifications
The unique combination of the nitrile and carboxylic acid functionalities on the benzene ring allows for a broad swath of possible chemical manipulations. The cyano group converts into amines, amidines, or carboxyls under the right conditions, giving a chemist plenty of flexibility for downstream synthesis. The carboxyl group reacts readily in condensing reactions, such as forming esters or amides. Drawing from my own work at the bench, I’ve found 4-cyanobenzoic acid especially effective as a precursor for heterocyclic compounds. It opens up paths to medicinal chemistry research or new polymer developments just by switching up the reaction partners or conditions. Its role as a parent structure for ligands, especially in coordination chemistry, also deserves mention, since the molecule offers dual points for metal binding or structure extension.
Synonyms & Product Names
Searching literature and supplier catalogs often means wading through a host of names for the same chemical. In academic and commercial circles alike, this molecule surfaces as p-cyanobenzoic acid, para-cyanobenzoic acid, 4-cyanobenzoate (for its salt), or even as 1-cyano-4-carboxybenzene. Sometimes packaging references CAS numbers to clear up confusion. From my desk, making sense of synonyms avoids the mix-ups that can derail an experiment or procurement process. Multi-lingual catalogs and global trade often complicate things, so verifying the full IUPAC or CAS identifier helps keep large projects on track.
Safety & Operational Standards
Lab and plant safety remains pivotal when dealing with aromatic nitriles, particularly ones with reactive groups. 4-cyanobenzoic acid, though not acutely toxic under normal lab use, demands respect. Teams handling this powder wear gloves and eye shields, not just by policy, but from years of experience with skin and mucous membrane irritation. Dust control, good ventilation, and fume extraction become standard. After several years handling bench chemicals, I can say that training everyone—young students and seasoned pros alike—on the risks of nitrile ingestion or inhalation greatly reduces health incidents. Documentation and access to Material Safety Data Sheets go hand-in-hand with safe storage plans, especially for facilities managing high volumes.
Application Area
4-cyanobenzoic acid fills vital gaps in many supply chains—its impact shows up in industries as varied as pharmaceuticals, advanced polymers, electronics, and liquid crystals. One clear example comes from the production of liquid crystalline polyesters, where the rigid aromatic backbone contributes to the unique mechanical and optical properties. Research into new pharmaceuticals often drafts this compound as a starting point for anti-inflammatory and anticancer agents. Through my interactions with product engineers in specialty plastics, I learned that resin manufacturers chase molecules like this for their thermochemical durability. Even pigment and dye industries count on the stability and reactivity of this compound to create hues that resist fading. Its breadth of use rests on its molecular toughness and readiness to adapt to chemical tweaks.
Research & Development
Curiosity about the properties and modifications of 4-cyanobenzoic acid keeps driving research. Universities and industrial R&D centers still look for new functional materials rooted in this aromatic acid. Researchers test its derivatives in polymers that exhibit tailored flexibility or improved conductivity. The field of medicinal chemistry stays busy, checking new substitutions or coupling partners for potential pharmaceutical breakthroughs. For several years, patents continue rolling out on ester or amide derivatives intended for next-generation products. I’ve found that a robust chemical like this, with proven synthetic accessibility, often takes its place as a “go-to scaffold” for teams racing against competitors in the specialty materials arms race.
Toxicity Research
Every new or long-used chemical prompts safety and toxicity evaluation. For 4-cyanobenzoic acid, animal studies and in vitro screens focus on effects such as irritation, allergic reaction, or organ impact after chronic exposure. There’s greater awareness these days about the need for comprehensive risk assessment that goes beyond just acute exposure. Experience in the chemical industry shows that regulatory agencies expect companies to keep up-to-date toxicity studies to back up their safety files. Studies on metabolites, especially those breaking down in water or soil, force companies to think about end-of-life and eco-toxicological risks. Discussions I’ve had with environmental health officers remind me that transparency and early reporting on new hazard findings build trust with both regulators and the public.
Future Prospects
Looking ahead, I see plenty of promise for 4-cyanobenzoic acid in supporting new technology rollouts—from advanced display materials to sustainable plastics. The trend toward recyclable, high-performance polymers keeps creating space for molecules with sturdy aromatic backbones. Demand in pharmaceuticals, especially for intermediates with ready modification potential, will likely keep this compound in focus. As industry pivots towards green chemistry, future processes for making and modifying 4-cyanobenzoic acid will need to be cleaner and more resource-efficient. In my circle, researchers already weigh the environmental costs of synthesis alongside the material properties, searching for renewable feedstocks and non-toxic reagents to push things forward.
What are the main applications of 4-Cyanobenzoic Acid?
Finding Value in Chemical Building Blocks
Some chemicals quietly support a wide range of industries without ever making headlines. 4-Cyanobenzoic acid is a prime example. Its story hits close to home for anyone who has spent time in a research lab or worked in manufacturing settings where details matter. This compound, with its cyanide group attached to a simple aromatic ring, does more than just sit on a shelf.
The Role in Polyester and Liquid Crystal Polymers
The first place I noticed the real-world use of 4-cyanobenzoic acid came during my early work with polymer synthesis. It shows up in the production of specialty plastics known as liquid crystal polymers. These aren’t the plastics you find wrapped around groceries—they live in the world of electronics, where strength and stability under heat are gold. Circuit manufacturers count on these polymers for making tiny but powerful electronic parts.
The reason this acid matters lies in its contribution to producing rigid rod segments within the polymer chains. The presence of the cyano group helps the polymer align and form ordered structures. These unique properties drive improved mechanical strength and thermal performance. Think connectors inside your smartphone or automotive electrical systems that keep running after years exposed to heat and vibration—the backbone in those connectors comes in part from compounds like this.
Pharmaceutical Intermediates and Fine Chemicals
Not every chemistry breakthrough changes the world overnight, but building blocks like 4-cyanobenzoic acid show their worth in drug discovery and production. Pharmaceutical manufacturers use it as an intermediate—a key step in synthesizing complex molecules for therapies targeting inflammation, cancer, and metabolic disorders. In these cases, reliability, purity, and consistent supply take center stage.
Sitting at a bench surrounded by glassware, I saw firsthand how one small misstep in an intermediate synthesis could throw off an entire process. That’s why researchers often return to trusted chemicals like this one. They streamline the conversion into benzoic acid derivatives with different substitutions, paving the way to new and promising drug candidates.
Dyes, Pigments, and Agrochemicals
Color design in textiles or everyday products often traces back to dye and pigment synthesis powered by unusual building blocks. 4-cyanobenzoic acid plays a role here too. Its cyano group acts as a foundation for linking with other chemical groups, creating brighter, longer-lasting dyes and pigments. Color quality, resistance to fading, and environmental stability all benefit from these innovations. In the textile factories I have visited, products that pass wear-and-wash testing owe a debt to thoughtful chemistry like this.
Markets also turn toward this compound in the search for new herbicides and pesticides. Its structure provides a versatile starting point for attaching different functional groups, opening doors to more selective and safer agricultural chemicals. In conversations with agronomists, I’ve heard repeated questions about how to boost productivity with fewer environmental risks. Molecular tweaking starting from 4-cyanobenzoic acid aims to move that needle in the right direction.
Challenges and How We Move Forward
No single chemical solves every problem. Cost controls, handling safety, and environmental impact all push the industry to refine processes. Green chemistry initiatives look for cleaner synthesis routes to 4-cyanobenzoic acid, slashing toxic byproducts and increasing yields. Researchers and manufacturers can partner more closely, sharing data to identify safer catalysts and solvents. As the field leans into more sustainable practices, every small step adds up.
In day-to-day work, tracking purity, trace impurities, and performance during end-use matters just as much as any new invention on paper. Experienced staff, clear protocols, and open feedback loops help shift a compound like 4-cyanobenzoic acid from a line item in a catalog to a key enabler in manufacturing, health, and technology.
What is the chemical formula and molecular weight of 4-Cyanobenzoic Acid?
Unpacking Its Structure: Chemical Formula
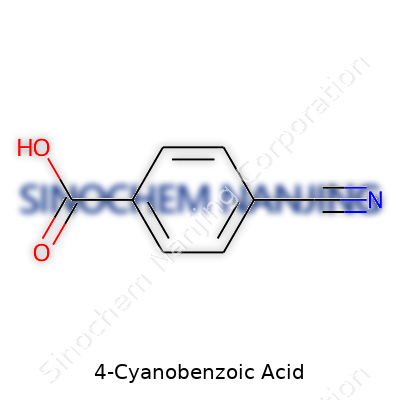
4-Cyanobenzoic acid sounds like a mouthful, but break it down, and you find the formula C8H5NO2. This means it has eight carbon atoms, five hydrogen atoms, one nitrogen atom, and two oxygen atoms. The “cyano” in its name hints at a nitrile group attached to the fourth carbon on the benzene ring, sitting right across from the carboxylic acid group. Look at a picture of its structure—the sharp, pointed nitrile sticks out, marking the difference between this compound and regular benzoic acid.
Molecular Weight: Why It Matters
The molecular weight of 4-cyanobenzoic acid stands at 147.13 g/mol. Chemists use this number all the time, making calculations for reactions, scaling up processes, or measuring out the right dose in pharmaceutical labs. Say you’re mixing chemicals for a new drug, or just running an experiment in organic chemistry class—knowing the formula and weight means you’re not fumbling around blind. It means you measure things confidently and get repeatable results.
Real-World Uses & Importance
Anyone working in material science or pharmaceuticals may come across this molecule. I’ve seen it mentioned in research about liquid crystal compositions—think electronic displays with super sharp images. Folks in industrial work use it as a building block for dyes and specialty polymers too. The cyano group on that benzene ring makes it very reactive, opening the door to create complex organic compounds with tailored properties.
Safety comes up, especially handling raw chemicals. 4-Cyanobenzoic acid isn’t classified as highly toxic, but like a lot of organic acids, it can be an irritant. A simple slip—forgetting gloves or a mask—leads to irritation or more serious exposure. I always say respect for these materials starts with knowing exactly what’s in your flask or beaker, down to its molecular makeup and weight.
Challenges and How to Tackle Them
One challenge with chemicals like this is safe storage and correct disposal. It’s easy for labs to let old containers sit forgotten on a shelf. Negligence leads to hazardous spills or accidents. Regular inventory checks and well-labeled storage solve most of this problem. I’ve seen labs that set digital reminders and keep a clear logbook stay much safer than those that don’t.
Education is another big factor. Teaching young scientists not just the theory, but the practical side—handling, labeling, disposal, and what those numbers on the bottle really mean—makes a big difference long-term. Many accidents don’t come from malice, but from not knowing. Simple facts, such as the formula and the molecular weight, become the foundation for safe and smart chemistry.
Connecting the Details: Why Accuracy Counts
Details matter in chemistry. Just one missing hydrogen atom can change the whole story of a compound. That’s why knowing 4-cyanobenzoic acid as C8H5NO2, 147.13 g/mol, and understanding what that means goes beyond memorization. It’s about a respect for the craft, whether making something new in a research lab or just learning the ropes in a classroom.
How should 4-Cyanobenzoic Acid be stored?
Why Safe Storage Really Matters
Anyone who’s handled fine chemicals will tell you the story doesn’t end after the delivery arrives. Keeping a compound stable takes more than tossing it on a shelf and walking away. With 4-cyanobenzoic acid, the way the bottle sits in a storeroom can matter a lot in the long run. If a lab tech opens a jar and finds clumps, discoloration, or a strange smell, it’s not just a waste of cash—it's a headache for safety and experiment reliability. Those risks only grow if the storage routine gets sloppy.
The Practical Guidelines That Make a Difference
Storing 4-cyanobenzoic acid calls for some simple habits, but they add up to real peace of mind. I’ve seen too many workplaces skip the warning labels or let the bottles drift onto the wrong shelf. That makes a mess for anyone who walks in later and assumes things are in order. Let’s cut down on confusion from the start.
The acid always belongs in a cool, dry spot. Any excessive warmth can nudge this solid toward breaking down over time. Moisture in the air brings its own set of problems—powders clump, unwanted reactions creep in, and the purity slips out the door. So a lidded jar and a dedicated cabinet really earn their keep, especially in labs with poor air circulation or wild temperature swings.
Using containers made from glass or high-quality plastic ups your chance of holding on to that white, granular look. Metal containers should stay on the sidelines: they can react, or sometimes corrode from trace contaminants. Screw-caps with a proper gasket shut out humidity, but only if users rebuild the habit of sealing the jar tight every single time. In my first lab job, unsealed jars were a recurring pain point, and every mistake chipped away at product shelf life.
Light can fade a compound’s quality more than most realize. Storing 4-cyanobenzoic acid away from direct sunlight—inside an opaque or amber plastic bin—brings extra protection, especially in smaller facilities that double as classroom spaces. A window shelf easily ruins weeks of care in just a few afternoons.
Labeling and Separation: Keeping Order Sensible
Unlabeled bottles cause more trouble than almost any other kind of error. Hand-written tape wears thin and falls off, and I’ve seen people reach for the wrong compound just because the label was vague or too faded to read. Every jar deserves a clear label, noting the chemical name, concentration (if it’s a mixture), date of receipt, and the supplier. These bits of information outlast memory, and can keep regulators or safety officers content on inspection day.
Separating acids from bases cuts down on reaction risk from splashes and fumes. Even though 4-cyanobenzoic acid doesn’t grab headlines for volatility, accidental mixing with the likes of sodium hydroxide can turn a quiet storage room into a spill scene. It isn’t just about chemistry knowledge—simple shelving arrangements and color-coded bins eliminate guesswork, especially at the end of a long week.
Smart Precautions for Long-Term Stability
Opening a fresh jar always beats finding a neglected one that’s gone off, but budgets don’t always allow for one-use bottles. Rotating inventory helps, and anyone managing a chemical storeroom can benefit from sticking newer stock at the back and moving the older jars to the front. Routine checks, once a month or so, pick up early warning signs like new odors or crystallization at the rim.
Investing a few minutes in sound storage keeps risk at bay. The process is simple, but the results ripple through experiments, shelf life, and lab safety every day.
Is 4-Cyanobenzoic Acid hazardous or does it require special handling?
Looking Closely at Chemical Safety in Everyday Labs
My years working alongside chemists, students, and safety officers taught me that the world of organic compounds holds plenty of hidden challenges. Take a closer look at 4-cyanobenzoic acid. You won’t find it on the list of household names, but its presence in certain manufacturing or research spaces brings up questions worth addressing. Getting comfortable around chemicals like this leads to trouble, so it makes sense to check their fine print before bringing them near the lab bench.
Understanding the Risks with 4-Cyanobenzoic Acid
This solid, with a feathery white appearance, evaporates zero alarm in the way it looks. Still, what counts isn’t how something sits in a jar, but what happens when it interacts with the body and environment. Chemists know that benzoic acid derivatives aren’t as harmless as they appear. Ingesting or inhaling powders like this usually irritates mucous membranes and airways. Eye and skin exposure creates irritation and sometimes even more serious reactions with prolonged contact.
Many safety datasheets classify 4-cyanobenzoic acid as an irritant. It does not fall into the same hazard zones as acids like hydrofluoric or industrial cleaning-grade hydrochloric, but it doesn’t belong in your average high school science classroom either. According to regulatory documents, swallowing or inhaling its dust may cause discomfort or damage to tissue. The compound doesn’t appear to be acutely toxic in the way pesticides or cyanide salts are, but irritation can still sideline work in a busy lab, especially for those with pre-existing allergies, asthma, or sensitive skin.
Why Special Handling Matters
Working for years in R&D teams, I see accidents happen mostly on ordinary days—most from small oversights, not spectacular blunders. Glove changes, forgotten masks, or half-empty chemical labels contribute more to hospital visits than caustic acids ever do. Even with something that doesn’t fizz or burn on contact, underestimating PPE leads to preventable injuries. Proper gloves, goggles, and dust masks lower risk. Good ventilation lets lab colleagues breathe easier, literally.
Disposal also challenges even seasoned chemists. Flushing 4-cyanobenzoic acid down the drain, or tossing it into regular waste, invites regulatory headaches and raises red flags for local water treatment plants. Regulations from the EPA and OSHA steer clear about specifics for every niche organic, but any benzoic derivative with a cyanide group deserves a closer look. Even with low acute toxicity, accidental release or large-scale spills draw attention fast in tight-knit lab communities.
Clear Thinking Helps Guide Best Practices
The answer to safe work with this acid—like many middle-of-the-road lab reagents—comes from habit, not heroics. Get eyes on current safety datasheets. Store acids and organic compounds in closed containers, away from basic solutions or oxidizers. Sweep up spills gently, never dry-brushing or stirring dust into the air. Put in ventilation upgrades if dust seems to hover longer than it should.
Training makes the difference, especially for new hires or rotating students. Rather than assuming “not immediately hazardous” means “harmless,” encourage a culture of real caution. Annual retraining or walk-throughs with a safety officer highlight blind spots. If something feels off—the dust smells odd or a reaction takes a strange turn—speaking up quickly catches trouble before it spreads.
Room for Improvement and Ongoing Vigilance
Labs using chemicals like 4-cyanobenzoic acid can always raise their safety bars higher. That means clearer labels, accessible spill kits, and clear reporting paths for accidents, no matter how minor. Labs can collaborate with health and safety offices to minimize storage and improve disposal tracking. Every student or researcher benefits from guidelines built on real-life experience, not just rulebooks.
I’ve watched careful planning pay off with safer spaces and fewer incidents—even with mundane substances. Whether for new researchers or seasoned chemists, keeping respect for the chemicals we use daily keeps everyone working at their best.
What is the purity grade available for 4-Cyanobenzoic Acid?
What Purity Levels Mean to Chemists
In the lab, purity is everything. If someone has ever tried to run a reaction with a chemical that isn’t clean, frustration sets in fast. 4-Cyanobenzoic acid crops up in syntheses for pharmaceuticals, dyes, and sometimes liquid crystal materials. Most suppliers advertise a purity above 98%, labeling the grade “analytical” or “high purity.” You'll see 99% and, in some cases, even 99.5%. That tiny percent makes a real difference, especially where impurities can ruin downstream processes or skew analytical data.
Understanding What’s Hiding in the Gaps
Working as a research chemist, it’s clear those extra decimals in purity reports aren’t just sales fluff. That last 0.5% might sound minor, but extra stuff—like unreacted starting material, solvents, or trace metal contaminants—brings trouble. These interferences show up in melting points, NMR spectra, and yields. Once, purifying a sample meant to be “99%” pure, I found more than a trace of phthalic acid mixed in. That single mistake wasted days.
Suppliers such as Sigma-Aldrich, Alfa Aesar, and TCI typically list 4-cyanobenzoic acid, offering routine analytical data: melting point between 234–238 °C, HPLC or titration data for purity. Their certificates spell out water content, heavy metals, and sometimes specific organic impurities. If a project calls for higher standards, like trace analysis or electronic-grade work, researchers have to dig for specialty lots or even re-crystallize on their own. There’s no shortcut—chromatograms and certificate of analysis reports aren’t optional reading.
Why Grades Above 99% Matter in Practice
Being on a bench team, I have watched a small impurity in a key intermediate toss a whole week’s results. Pharmaceutical work demands the tightest controls. Less-than-99% content forces extra purification that not only loses time—it can cause headaches with environmental waste and tight budgets. In a competitive industrial setting, even a supposed 98.5% product often goes through more checks before entering the process.
Using the right purity depends on the lab’s tolerance for impurities. If the acid serves as a reference standard or as a reactant in a sensitive sequence, anything below 99% weakens outcome reliability. Not every project pushes for that precision—routine undergraduate labs sometimes use reagent-grade stock around 97-98%. But advanced development skips the risks and pays the premium for the cleanest lot.
Evaluating What’s Available and What to Ask For
Anyone in procurement should press for the current lot’s COA. Trusted vendors supply lot-level certificates showing at least titration or HPLC data. Some sellers simply report “minimum 98%” with no detailed analysis—those questionable lots usually hang around at lower price points. I’ve seen groups regret not choosing a higher-purity supplier after dealing with unexplained NMR peaks or failed couplings.
Handling issues of purity means asking about the testing methods—HPLC, NMR, and elemental analysis go further than “meets specification.” For work that can’t tolerate cross-contamination, it makes sense to ask about the cleaning procedures at the supplier’s facility. When quality falls short, re-purification methods like recrystallization from ethanol or water offer a backup, though they eat up lab time.
Solutions to Common Purity Concerns
For teams running high-stakes chemistry, it pays to check for batch-to-batch consistency. Keeping a direct line to your supplier’s technical support builds trust and sometimes unlocks higher-purity material stored for niche clients. Tight record keeping, methodical screening, and not settling for “adequate” quality protect results. In a world where reproducibility drives discovery, tight purity control on every order makes a difference.
| Names | |
| Preferred IUPAC name | 4-Cyanobenzoic acid |
| Other names |
4-Carboxybenzonitrile p-Carboxybenzonitrile p-Cyanobenzoic acid 4-Cyanobenzoesaeure 4-Cyanobenzoate |
| Pronunciation | /ˌfɔːr.saɪˌænoʊ.bɛnˈzoʊ.ɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | [619-80-7] |
| Beilstein Reference | 91192 |
| ChEBI | CHEBI:20441 |
| ChEMBL | CHEMBL16130 |
| ChemSpider | 2027 |
| DrugBank | DB08540 |
| ECHA InfoCard | 100.016.498 |
| EC Number | 207-325-2 |
| Gmelin Reference | 5294 |
| KEGG | C06585 |
| MeSH | D017110 |
| PubChem CID | 9980 |
| RTECS number | CU1400000 |
| UNII | RG9L1T2A91 |
| UN number | UN3077 |
| Properties | |
| Chemical formula | C8H5NO2 |
| Molar mass | 147.13 g/mol |
| Appearance | White to off-white powder |
| Odor | Odorless |
| Density | 1.32 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 1.44 |
| Vapor pressure | 0.0000057 mmHg (25°C) |
| Acidity (pKa) | 3.54 |
| Basicity (pKb) | 1.86 |
| Magnetic susceptibility (χ) | -29.6·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.626 |
| Viscosity | 1.34 g/cm³ (25 °C) |
| Dipole moment | 3.99 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 221.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -16.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3451 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, causes serious eye irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H315, H319, H335 |
| Precautionary statements | Precautionary statements: P261, P264, P271, P272, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P362+P364, P501 |
| NFPA 704 (fire diamond) | 2-1-0 |
| Flash point | 180 °C (356 °F; 453 K) |
| Autoignition temperature | 580°C |
| Lethal dose or concentration | LD50 (oral, rat): 3120 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 > 2000 mg/kg |
| PEL (Permissible) | Not established |
| REL (Recommended) | Recrystallization (ethanol, water) |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Benzoic acid Terephthalic acid Isophthalic acid 4-Aminobenzoic acid 4-Nitrobenzoic acid 4-Methylbenzoic acid 4-Formylbenzoic acid |

