The Real Story of 4-Chloronitrobenzene: Past, Present, and the Path Ahead
Looking Back: Roots of 4-Chloronitrobenzene
Peering into the chemical archives, discoveries like 4-chloronitrobenzene pop up as reminders of how much industry has changed. This compound once found its way into factories during the rapid growth of modern synthetic chemistry in the nineteenth and twentieth centuries. As scientists started manipulating the benzene ring through nitration and halogenation, 4-chloronitrobenzene emerged as both a challenge and an opportunity. The use of nitroaromatics across Europe and later in North America tracks directly with shifts in technology: more reliable production of fine chemicals brought substances like this from academic curiosity into the realm of commercial staples.
Unpacking the Product: What Is It, and Why Use It?
Ask folks in dye or agricultural chemical labs what makes 4-chloronitrobenzene matter, and the answer usually boils down to versatility and reactivity. Its molecular design—a nitro group and a chlorine atom seated oppositely on a benzene ring—delivers a platform for building more complex molecules. Companies seeking stable, reactive intermediates often keep a barrel or two on hand. It doesn’t dazzle on its own but plays a central role in bridging gaps between raw materials and finished products.
The Physical and Chemical Heart of the Matter
Holding a sample of 4-chloronitrobenzene, you’ll spot a pale yellow crystalline solid. There’s not much of a smell, though a faint chemical sharpness hints at its potent nature. With a melting point around 83 degrees Celsius and moderate solubility in many organic solvents, the compound handles heat fairly well in controlled environments. Its nitro group offers ready sites for reduction, amidation, or replacement, and the chlorine can slip off or swap with relative ease. This duality—reactive yet stable enough for storage and transport—explains its persistent appeal for research and manufacturing.
Technical Details and Honest Labeling
Labels on containers spell out strict guidelines. Purity often exceeds 98 percent, since impurities can ruin later reactions. Batch numbers, handling precautions, and regulatory warnings crowd every side. Legal standards for labeling in most regions demand real clarity due to the dangers involved, from toxicity to environmental persistence. Regulatory agencies—think REACH or the EPA—pay especially close attention to labeling accuracy, because sloppiness leads to incidents nobody wants. In my own work, the headache often comes from reconciling local and global standards; companies need extensive training just to keep up.
Crafting 4-Chloronitrobenzene: The Lab to Plant Route
Industrial chemists synthesize 4-chloronitrobenzene by first nitrating chlorobenzene with a blend of concentrated nitric and sulfuric acids. This reaction needs close control—too little heat, and the mixture stalls; too much, and heat runs away, with obvious dangers. Over time, improvements in reactor design and process automation helped push yields higher while cutting waste. Before modern environmental standards, leftover acids and side products sometimes ended up in rivers or landfill. Experience—and regulation—shifted habits toward closed systems and on-site neutralization, reducing the compound’s environmental footprint.
How It Reacts: Routes to New Products
Chemists value 4-chloronitrobenzene as a starting block. The chlorine atom gets swapped out in nucleophilic aromatic substitution reactions to create new amines and phenols. The nitro group converts cleanly into an amine after hydrogenation, letting scientists build anilines or important pharmaceutical intermediates. Sometimes, companies modify the compound with sulfur- or oxygen-containing groups. In the dye sector, 4-chloronitrobenzene unlocks vivid azo and disperse dyes through simple coupling reactions. In herbicide manufacturing, further substitution and reductions quickly lead to active crop protection agents. The flexibility echoes a core lesson from organic lab classes: one well-placed group on a benzene ring can shape entire industries.
Other Names: Tracking a Compound Across Borders
Names multiply depending on the setting. On bills of lading or inventory sheets, you might spot synonyms like p-chloronitrobenzene or 1-chloro-4-nitrobenzene. Chemists sometimes write "para-chloronitrobenzene" out of habit, using shorthand from undergraduate lectures. International trade demands clarity—one mislabel can send a shipment to regulatory limbo. That makes accurate naming more than bureaucracy; it's the last stop before someone handles the wrong chemical with tragic consequences.
Staying Safe: Handling Hazards With Respect
Experience dealing with hazardous intermediates like 4-chloronitrobenzene teaches respect. Exposure risks cover skin, eyes, and the respiratory tract. Long-term mishandling causes chronic health issues, so gloves, goggles, and proper ventilation are a must. Safety standards require regular monitoring—air quality, skin checks, and spill training. Modern automation helps reduce direct contact, yet the human factor still matters most: one tired operator who skips a glove can send the whole plant scrambling. International safety guidelines keep evolving as more toxicity data emerges and as workplace health grows in public consciousness.
Where 4-Chloronitrobenzene Finds Work
Venture into a dye manufacturing site or an agrochemical plant, and you’re likely to cross paths with this compound. In dye chemistry, it anchors everything from basic colorants to specialty dispersive dyes. Farmers benefit from its role in herbicide precursors, while pharmaceutical industries sometimes reach for it to build complex intermediates. Its utility extends to rubber chemicals, antioxidants, and even some specialty plastics. I’ve seen research labs order small batches for exploratory projects, pushing the compound in entirely new directions, from advanced polymers to nanomaterials.
Research and Development: The Push for Progress
Research rarely stands still. Academic labs keep searching for greener synthesis routes—alternate solvents, milder nitration mixtures, and better catalysts all gain attention. Some startups aim to overhaul manufacturing, sketching out continuous flow reactors that trim waste and boost yield. Computational chemists dive deep into reaction pathways, hunting for the next big shortcut. In my experience, even small process improvements can save vast resources once scaled up, proving that curiosity-driven work has real value when it leaves the lab bench.
Toxicity: Understanding the Real Risks
Toxicologists and industrial hygienists devote serious energy to mapping how 4-chloronitrobenzene affects organisms. Animal studies show liver and kidney stress with repeated exposures. Occupational studies reinforce the message: chronic exposure needs prevention rather than cure. Protective measures—closed systems, fume hoods, personal monitoring badges—have become normal on any production line. Water and soil monitoring near manufacturing sites grew much stricter after environmental mishaps in the twentieth century. Transparent toxicity research keeps workers and communities safer than ever, though the fight for stricter oversight continues.
Looking Forward: Potential and Responsibility
As industries chase new efficiency and sustainability targets, compounds like 4-chloronitrobenzene face more scrutiny. Some companies explore biobased feedstocks or electrochemical alternatives, aiming to leave fossil resources behind. Others study ways to recover and recycle chemical byproducts, nudging the process toward a real circular economy. Researchers share data more openly now, pushing for international best practices. The future sits in a crossroads: scientists, regulators, and the public must balance demand for advanced materials with a tighter grip on health and safety. 4-chloronitrobenzene will probably stick around as a core chemical building block, but with cleaner processes, smarter safety protocols, and more transparent risk communication guiding its path forward.
What are the main uses of 4-Chloronitrobenzene?
Breaking Down the Basics
4-Chloronitrobenzene pops up in conversation mostly in chemistry circles, but it reaches much farther than textbooks and laboratories. Its story starts as a pale-yellow solid that comes from nitrobenzene using chlorination. Chemists figured out how to make it in the late 1800s, but it is in the twentieth and twenty-first centuries that this compound became a quiet workhorse for manufacturing, medicine, agriculture, and colorants. The number may not mean much to a person outside a lab, but looking deeper paints a picture of why it matters.
Making Pharmaceuticals Possible
It struck me just how often a small change in a molecule steers a whole industry. With 4-Chloronitrobenzene, you see this in how it builds up the backbone of several everyday medicines. Factories convert it to things like 4-chloroaniline, and then push farther into drug ingredients. Some antihistamines and painkillers begin with this chemical, as do antiseptics and certain blood pressure pills.
Many people rely on these lifesaving or life-improving drugs without knowing how they start out. What seems distant or harsh from the outside—handling a chemical that requires gloves and goggles—leads to capsules or creams in your bathroom. High-quality standards and strict environment controls stand between the hazards of the source chemical and the final product on pharmacy shelves. Still, the chain begins with molecules like this, and better traceability in the supply chain means higher safety for users.
Chemistry Behind Colors
I remember learning where the bright colors of shirts or backpacks came from. 4-Chloronitrobenzene’s role in dyes hits especially close to home for anyone working with textiles. Companies turn it into intermediates like 4-chloroaniline, then use those to create vivid colors used on wool, cotton, or synthetic fibers. The compounds born from it hold onto fabric well, resist washing, and survive sunlight. Techniques keep evolving, and chemists seek safer, less polluting routes, but this building block remains essential.
Protecting Crops and Controlling Pests
Seasons change, and crops stand at risk from insects and disease. Farmers face pests that can shave off entire yields within a few weeks. Agri-chemicals help out, and 4-Chloronitrobenzene sits in the background of several of these solutions. It forms part of the process in making herbicides and insecticides, keeping gardens, parks, and fields productive. Regulations run strict due to environmental and health concerns—manufacturers need to report and manage waste products and residues throughout the cycle.
Challenges and New Directions
Using an industrial chemical on this scale brings up real concerns about waste, health, and environmental effects. Accidents and spills can happen, and these highlight the need for monitoring and enforcement. Real progress comes from updated filtration, recycling solvents, and even better chemical paths that cut down on hazardous byproducts. Recent years have shown movement toward less toxic dyes and greener pharmaceuticals, but it takes ongoing effort, funding, and public awareness.
So, each time someone takes allergy medicine or notices the colorfastness of a favorite T-shirt, there’s a good chance 4-Chloronitrobenzene played a hidden role. Its presence may not make headlines, but the drive to make its use safer and cleaner continues to shape everyday life around the world.
What is the chemical structure and formula of 4-Chloronitrobenzene?
Getting to Know the Basics
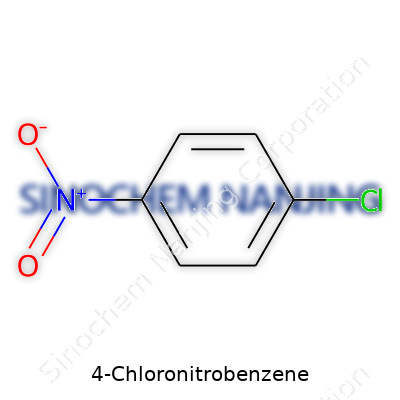
In the world of aromatic chemistry, 4-Chloronitrobenzene stands out among simple compounds. Its structure delivers a unique combination that sparks interest in many science classrooms and labs. This yellow crystalline chemical owes its form to the benzene ring at its core, with a nitro group and a chlorine atom attached in a para relationship. The chemical formula is straightforward: C6H4ClNO2.
The Structure – Clear and Simple
Anyone who has explored organic molecules will recognize this form. Take a benzene ring—a hexagon of six carbons—and attach a nitro group (NO2) at one carbon. Directly across the ring, hook up a chlorine atom (Cl). The molecule isn’t crowded; each group sits in the para position, which keeps the chemical stable and easy to work with in a synthesis setting.
To picture it: count around the ring, place NO2 at the first spot, then Cl at the fourth. The positions matter in organic chemistry. If you swap the groups or place them next to each other, you’ll get a totally different chemical with new traits and reactivity. This precise arrangement earns it the name 4-Chloronitrobenzene (sometimes called para-chloronitrobenzene).
Why Structure Shapes Usefulness
As I remember from my own experience in undergraduate labs, handling these chemicals teaches a few things fast about safety and precision. The nitro group brings strong electron-withdrawing power, affecting the reactivity of the ring. The chlorine atom, a halogen, offers versatility for later chemical reactions. Together, these two groups make the compound valuable for further synthesis, whether that’s dyes, pharmaceuticals, or rubber processing chemicals.
Looking up historical usage data, large amounts of 4-Chloronitrobenzene have supported industry work for well over half a century. The chemical doesn’t break down easily under simple conditions, and its unique structure forms a building block for more advanced molecules. Production usually follows careful chlorination and nitration of benzene, each step requiring strict control to prevent dangerous byproducts.
Safety, Handling, and Environmental Responsibility
Treating 4-Chloronitrobenzene with caution isn’t just good lab practice; it’s a must. Its yellow color gives a quick visual cue, but safety data sheets show it can irritate skin, eyes, and respiratory tracts. Long-term exposure risks point to the need for well-ventilated environments and protective gear. As chemical industries move toward greener solutions, finding ways to recycle spent chemicals or break them down into harmless products should be a top priority.
Efforts to substitute safer alternatives in high-volume uses have surfaced since the late 1990s. Some chemical plants pilot bio-based or catalytic processes to reduce environmental footprint. Sharing research findings can drive positive change, but it takes commitment across manufacturing, regulation, and education.
Building Better Practices Through Chemistry
Learning about the formula and shape of 4-Chloronitrobenzene leads to a deeper understanding of organic synthesis, safety, and the push for sustainable industry. The molecule reminds me that, in chemistry, how atoms fit together decides so much more than maybe expected: safety, performance, even the cost. By focusing on both the structure and its impact on people and the environment, the chemical industry and educators can make smarter choices every step of the way.
Is 4-Chloronitrobenzene hazardous or toxic?
Unpacking the Risks of 4-Chloronitrobenzene
Factories have relied on raw chemicals for decades, pushing boundaries in dyes, pharmaceuticals, and pesticides. 4-Chloronitrobenzene stands out here. While it’s become a staple in large-scale chemical reactions, there’s a darker side: risk. It’s easy to focus on the benefits of strong intermediates, but overlooking their danger invites harm to workers, communities, and the environment.
What Makes This Chemical Harmful?
Most people never come across 4-chloronitrobenzene at home, but in a production setting, exposure can happen during manufacturing, packaging, or spills. Skin contact, inhalation, or even accidental ingestion can all trigger serious health problems.
This chemical doesn’t just irritate. If it touches skin, severe burns often develop after even a short time. Workers might notice their skin itching or blistering. Inhalation can affect breathing, causing coughing, dizziness, or tightness in the chest. Some research shows that long-term or repeated exposure ties into blood disorders, particularly methemoglobinemia—a dangerous condition where oxygen delivery in the body drops off.
It’s not only people who face harm. 4-chloronitrobenzene can stick around in soil and water. Fish and small animals suffer when levels rise, disrupting local food chains and polluting resources needed by entire towns.
Concrete Evidence of Hazard
Regulators in the US and Europe have flagged this compound as hazardous. The Occupational Safety and Health Administration (OSHA) and the European Chemicals Agency both list it as toxic. Swallowing small amounts puts vital organs at risk. Lab studies back this up: rodents exposed to vapors frequently develop organ damage or tumors.
Cleanup teams and workplace safety managers must take its risks seriously. Protective gear—special gloves, goggles, and respirators—go a long way to protect skin and lungs. I’ve seen coworkers develop nasty rashes because they skipped gloves for just a few minutes, thinking they’d “be fine.” Those were harsh lessons.
Reducing Harm, Protecting the Future
You don’t need a science degree to understand that prevention makes sense. Keeping spills off factory floors, installing tight-sealing pipes, and double-checking valves can stop leaks before they start. Routine air monitoring gives early warning signs, so smaller issues get attention before large groups fall ill.
Community leaders and plant managers build public trust by sharing what steps they take to prevent chemical exposure. Local populations have a right to know evacuation routes and have some say about plants within their neighborhoods.
Better Alternatives and Smart Choices
For companies willing to invest upfront, seeking out less hazardous alternatives can save money and goodwill long-term. Employees tend to stay with companies that show concern for well-being—not just productivity—and communities are more likely to welcome future development.
Environmental advocacy groups suggest researching greener synthesis methods. Some labs have explored swapping in slightly different chemical ingredients that are easier to break down, cutting down persistent, toxic leftovers.
Hazardous doesn’t mean inevitable disaster—if people weigh benefits against risks and put action behind promises, workplaces and neighborhoods can both thrive. Trust comes from transparency, solid training, and refusing to cut corners.
What safety precautions should be taken when handling 4-Chloronitrobenzene?
Why 4-Chloronitrobenzene Deserves Respect
4-Chloronitrobenzene counts as one of those chemicals you don’t want to take lightly. With its pale-yellow crystals, it looks harmless at first glance, but it packs quite a punch in terms of health risks. As someone who’s worked hands-on with a range of hazardous substances, I’ve learned the hard way that shortcuts cost more than time—they can cost health. The main thing with 4-Chloronitrobenzene is its toxicity: it irritates the eyes and skin, harms the liver and kidneys, and vapor exposure can cause headaches, dizziness, and even blue-tinted skin from methemoglobinemia. Each spill or puff of dust isn’t just a nuisance—it’s a risk that can add up over the years.
Personal Protective Equipment Isn’t an Afterthought
Before going near this compound, gloves and safety goggles need to be on, not just somewhere in the room. I’ve seen chemical-resistant gloves turn a serious scare into nothing more than a cleanup task. Nitrile gloves work well, and polycarbonate goggles protect against unexpected splashes. A well-fitted lab coat with long sleeves gives a useful added layer, and for larger amounts or open handling, a face shield never goes amiss.
Respiratory protection calls for more than a dust mask. If there’s a risk of breathing in dust or fumes, a half-face or full-face respirator with organic vapor and particulate cartridges should come out of the cabinet. Many underestimate airborne exposure, but you can’t see or smell everything dangerous in the air.
Handling and Ventilation Count for a Lot
The best lab setups keep 4-Chloronitrobenzene inside a fume hood. Hoods and proper local exhaust draw harmful vapors away from you and other workers. In smaller shops, at the very least, keep the space well-ventilated. We used to check airflow before every shift—simple habits like that have kept accidents low.
Spills sometimes happen, and quick cleanup makes a difference. Don’t sweep up with a dry broom—dust just gets into the air and lungs. We use a HEPA vacuum or damp cloths for smaller spills. Work surfaces stay clean, clear, and clutter-free, reducing the chance that an open bottle gets knocked over or overlooked.
Safe Storage and Responsible Usage
4-Chloronitrobenzene stores best in tight, clearly labeled containers, kept out of direct sunlight and away from acids or bases. Strong acids and bases can trigger unwanted reactions. Never trust a container unless the label matches the contents, and don’t use makeshift lids or cracked jars. Storage cabinets made for toxic chemicals keep the workplace safe overnight—and proper signage helps during emergencies.
Disposal takes just as much care. Tossing this compound down the drain or in regular garbage threatens both people and the environment—industrial waste contractors handle it safely. Always keep waste separate and record what goes in each container.
Training and Vigilance Make the Difference
No safety protocol replaces good training. In every facility I’ve helped inspect, teams who run regular chemical handling drills see fewer incidents. Real hands-on demonstrations beat long-winded manuals every time. That culture of safety spreads, and new employees learn by example.
If there’s only one takeaway: respect 4-Chloronitrobenzene as a real hazard, not just another jar on the shelf. Take a couple extra minutes to grab the right gear or double-check the workspace. Over time, that mindset saves bodies—and livelihoods.
How should 4-Chloronitrobenzene be stored and transported?
A Chemical with Risks That Deserve Respect
4-Chloronitrobenzene isn’t just another industrial chemical. Its solid crystalline form, faint yellow color, and sharp odor warn you straight away that it means business. This chemical finds its way into dyes, pharmaceuticals, and pesticides. The flipside? Strong evidence links it to skin irritation, respiratory issues, and long-term harm if mishandled. Years of work in labs and warehouses have shown just how quickly small mistakes around this compound can snowball. So, safe storage and transport call for precise steps, not shortcuts.
Keep It Dry, Cool, and Far from Hazards
One lesson stands out: never cut corners with the environment where you store this stuff. Humidity and heat make it unstable, and sealed containers keep trouble away. Steel drums or tightly capped plastic barrels work well, but corrosion resistance is a must—4-Chloronitrobenzene and moisture never mix nicely. Workers—whether new to the job or seasoned—have seen containers sweat through even mild temperature spikes, creating fumes that linger in storage rooms. So, always look for a space with solid ventilation, and push it away from sunlight, acids, bases, or anything combustible. Flames and static sparks spell disaster here.
Labeling and Separation Save Lives
Busy warehouses get sloppy, but that’s exactly what 4-Chloronitrobenzene counts on to make a mess. Bold, unmistakable labels warn everyone what sits inside each drum or barrel, and you separate it from food, animal feed, and basic supplies. Even after years working with chemicals, I’ve seen near-misses come from drums moved by people who missed a faint warning tag. Use big, chemical-resistant markings, and update them if you switch containers. Avoid stacking too high or too close together. Spills love cramped quarters, and if a leak happens, you want to spot it early.
Safe Handling and Protective Gear
Skin contact brings rashes, and inhalation brings coughing fits, so personal protective equipment becomes your friend. Gloves, goggles, and fitted respirators keep risk at bay. Plenty of companies spend on chemical-resistant suits for a reason—labor laws expect no less now. Spills call for sawdust or inert absorbents, not water, because this chemical reacts unpredictably with moisture. Training matters just as much as any piece of gear. I’ve watched training drills turn into real emergencies, and the well-prepared always stay a step ahead of chaos.
Transport: Keep It Tight and Above Board
Regulations treat 4-Chloronitrobenzene as hazardous cargo. That label isn’t just legalese; it reflects lived reality on the road and rails. Drivers who haul these shipments must know their routes, have emergency numbers handy, and check containers for cracks before setting out. Regulations forbid mixing this load with food or household goods. Vehicles stay locked tight, with loads braced to prevent shifting and ruptures. If a transit company ignores these protocols, it puts towns, rivers, and workers in the line of fire. History is rich with examples where ignored guidelines triggered evacuations and lawsuits. Proper paperwork and ongoing training are the best shields against these risks.
Every Step Matters—From Start to Finish
End-to-end, handling 4-Chloronitrobenzene is never just a box-ticking exercise. It depends on real respect for its power. Storage space, containers, protective equipment, handling routines, labels, and paperwork—every detail counts. Lives, livelihoods, and local environments hang in the balance each time this chemical moves. Pragmatic care, learned from both regulations and hard-won experience, forms a line of defense worth protecting.
| Names | |
| Preferred IUPAC name | 1-Chloro-4-nitrobenzene |
| Other names |
p-Chloronitrobenzene p-Nitrochlorobenzene 1-Chloro-4-nitrobenzene 4-Nitrochlorobenzene PNCB |
| Pronunciation | /ˌklɔːr.oʊˌnaɪ.troʊ.bɛnˈziːn/ |
| Identifiers | |
| CAS Number | 100-00-5 |
| Beilstein Reference | 1209222 |
| ChEBI | CHEBI:34645 |
| ChEMBL | CHEMBL14344 |
| ChemSpider | 1099 |
| DrugBank | DB16303 |
| ECHA InfoCard | ECHA InfoCard: 100.003.022 |
| EC Number | 208-892-5 |
| Gmelin Reference | Gmelin Reference: 151360 |
| KEGG | C14110 |
| MeSH | D002756 |
| PubChem CID | 7901 |
| RTECS number | CZ1400000 |
| UNII | 90E9605O8O |
| UN number | UN1578 |
| CompTox Dashboard (EPA) | DJ13JL86VJ |
| Properties | |
| Chemical formula | C6H4ClNO2 |
| Molar mass | 157.55 g/mol |
| Appearance | Pale yellow crystals |
| Odor | Odorless |
| Density | 1.36 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 2.61 |
| Vapor pressure | 0.03 mmHg (25°C) |
| Acidity (pKa) | 13.0 |
| Basicity (pKb) | 10.0 |
| Magnetic susceptibility (χ) | -57.0 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.613 |
| Viscosity | 1.46 mPa·s (at 25 °C) |
| Dipole moment | 3.51 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 324.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 48.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1402 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | D06AX02 |
| Hazards | |
| GHS labelling | GHS02, GHS06, GHS08, GHS09 |
| Pictograms | GHS06,GHS09 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H331, H335, H410 |
| Precautionary statements | Precautionary statements for 4-Chloronitrobenzene: "P260, P264, P270, P271, P273, P280, P301+P312, P304+P340, P305+P351+P338, P310, P312, P330, P363, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-0-☠️ |
| Flash point | Flash point: 131°C |
| Autoignition temperature | 160 °C |
| Explosive limits | Explosive limits: 1.3–8.0% (in air) |
| Lethal dose or concentration | LD50 oral rat 640 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral (rat) 640 mg/kg |
| NIOSH | 1019 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of 4-Chloronitrobenzene: **1 mg/m³** |
| REL (Recommended) | REL: 0.1 mg/m³ |
| IDLH (Immediate danger) | 50 ppm |
| Related compounds | |
| Related compounds |
Nitrobenzene 4-Chloroaniline 4-Bromo-nitrobenzene 4-Iodonitrobenzene 1,3-Dichloro-4-nitrobenzene |

