An Editorial Commentary on 4-Chlorobenzoyl Chloride
Historical Development
4-Chlorobenzoyl Chloride has roots that stretch back to the days when chemists began uncovering the broader utility of aromatic carboxylic acid chlorides. Over the decades, researchers saw the benzoyl chloride core as a blank canvas, perfect for building useful intermediates. Through routine chemical tinkering, someone swapped in a chlorine atom at the para position, giving rise to this specific compound. Laboratories in Europe, North America, and Asia recognized quickly that this modification wasn’t just a novelty. The result created a bulk chemical with attractive traits, and synthetic organic chemistry gained a new workhorse. Whether in pharmaceuticals or the dye industry, this substance quietly shaped the toolkit of chemists everywhere. That journey from niche curiosity to industrial mainstay speaks to its adaptability and lasting demand.
Product Overview
With a sharp odor and a reputation for subtle aggression towards both tissue and lab glassware, 4-Chlorobenzoyl Chloride hardly wins any popularity contests outside of its synthetic chemistry admirers. Still, look beneath the surface, and its value stands out. As a reagent, it slips into acylation reactions, building bridges towards more complex molecules. There aren’t many extra bells and whistles, just a well-defined benzoyl backbone and a single chlorine atom doing more than meets the eye. The bottle isn’t flashy, and warnings on the label fill more space than marketing slogans. But the professionals who use it rarely care about appearances; they chase results, and this reagent rarely lets them down.
Physical & Chemical Properties
At room temperature, 4-Chlorobenzoyl Chloride doesn’t hide its acidity. It appears as a pale to colorless oily liquid. Vapors sting the nose, and just a whiff reminds chemists to reach for a fume hood. It boasts a melting point around 23–24°C and boils well above water’s point at nearly 230°C. The molecule comes equipped with an acyl chloride group, making it hypersensitive to moisture. Drop some water nearby and expect a fizzing reaction, coughing out hydrogen chloride gas and forming 4-chlorobenzoic acid. Reactivity doesn’t stop there, as the benzene ring brings stability, while that para-chlorine substituent quietly tips the compound toward a blend of reactivity and robustness.
Technical Specifications & Labeling
Serious manufacturers describe 4-Chlorobenzoyl Chloride with specs focusing on purity, which typically hits upwards of 98% in reputable chemical supply chains. Labels scream CAUTION for good reason—users must navigate corrosivity and irritant nature with respect. On any lab shelf, this liquid comes sealed in amber bottles or corrosion-resistant drums, never loose and open. Batch numbers, manufacturing dates, and tightly regulated storage guidelines fill the fine print of the safety data sheet, more so than for many common reagents. Regulations covering hazardous substances often include it by name, and labs storing it long-term train their teams in spill cleanups and emergency responses.
Preparation Method
4-Chlorobenzoyl Chloride’s synthesis usually starts with 4-chlorobenzoic acid. Splashes of thionyl chloride or phosphorus pentachloride convert the acid to the acyl chloride, giving off volatile side products like sulfur dioxide or phosphorus oxychloride. In a modern chemical plant, this reaction happens under carefully controlled conditions—low moisture, steady stirring, and precise temperature controls. Once the reaction winds down, the crude product gets distilled, and impurities are stripped out, usually leaving a fairly pure liquid. Sometimes technicians add inhibitors or store the compound over desiccants, keeping it dry in storage for the longest possible shelf life.
Chemical Reactions & Modifications
The biggest draw of 4-Chlorobenzoyl Chloride isn’t its appearance or smell, but its near-infinite potential as a synthetic intermediate. That reactive acyl chloride group makes it hungry for nucleophiles. It bonds with alcohols to build esters, and with amines to form amides, while nucleophilic substitution on the aromatic ring remains limited due to the electron-withdrawing chlorine and acyl groups. Chemists use these traits as a shortcut in multistep synthesis. If a pharmaceutical chemist needs to add a 4-chlorobenzoyl group onto a bioactive scaffold, this compound paves the way. In research, tweaks to the core structure have produced analogues that lead to novel drugs, dyes, and agricultural chemicals. Despite its straightforward formula, the compound adapts almost endlessly depending on what a chemist needs to build.
Synonyms & Product Names
Old-timers in organic labs often refer to 4-Chlorobenzoyl Chloride by a mouthful of other names—p-Chlorobenzoyl Chloride, para-Chlorobenzoyl Chloride, or 4-CBC for short. The systematic name, 4-chlorobenzenecarbonyl chloride, sums up what the structure holds, even if most professionals use the fastest form in conversation. These names appear on bottles, papers, and regulatory lists, so paying attention to synonyms keeps mistakes at bay. While there’s no magic to the nomenclature, everybody who works in organic synthesis recognizes the pattern, and the substance rarely gets confused with safer or less reactive chlorinated intermediates.
Safety & Operational Standards
No matter how skilled the chemist, there’s never a good time to cut corners with 4-Chlorobenzoyl Chloride. Contact with skin or eyes spells trouble—burns, redness, pain. Inhalation can kick off coughing fits or worse, especially in tight spaces. Fume hoods, gloves, goggles, and sturdy lab coats become the bare minimum. Acid neutralizers and stretchers for chemical burns remain nearby, and spill training kicks in before the first bottle gets opened. Regulations in well-run labs treat this substance as a priority hazard, not just another bottle on a shelf. Proper labeling, ventilation, compatible storage, and routine checks for leaks or degraded seals make sure nobody gets an unpleasant surprise. Disposal calls for care too, since pouring leftovers down a drain or regular trash would create an even bigger problem for the team and the environment.
Application Area
4-Chlorobenzoyl Chloride works best when it’s used with skill and respect. Drug developers turn to it when synthesizing key intermediates for active compounds. It is favored in the creation of agrochemical building blocks, particularly where halogenated benzoic structures need precise placement. Dye and pigment producers also reach for it during acetylation or amidation steps. While it doesn’t often appear in consumer-facing products due to its corrosiveness and reactivity, its fingerprints show up in the pharmaceutical, agricultural, polymer, and fine chemical markets. Each of these fields values its reliability and functional versatility, and chemists see it as a bridge to more complex, high-value molecules.
Research & Development
Many current research trends explore ways to tame 4-Chlorobenzoyl Chloride’s aggressive side, searching for gentler reaction conditions that yield high-purity products without excess waste. There’s a steady stream of innovations around catalytic approaches, lower temperature synthesis, and green chemistry adaptations that reduce hazardous byproducts. In medicinal chemistry, scientists keep mining the molecule’s potential. Modified derivatives pave pathways to new antibiotics, anti-inflammatory drugs, and enzyme inhibitors. Each successful lab experiment recasts 4-Chlorobenzoyl Chloride as more than just a stepping stone—it’s a gateway to coveted patent claims and untapped pharmaceutical therapies.
Toxicity Research
Toxicologists approach 4-Chlorobenzoyl Chloride with the determination to map out risks, not fear. Exposure studies track skin and respiratory impacts. Severe irritation forms an immediate line of concern, and accidental exposure in industrial settings draws prompt intervention. Long-term health impacts from chronic low exposure receive scrutiny, but routine studies keep most of the danger at the acute level. Ecotoxicity comes up in water and soil environments, so regulators continue to pressure for tight containment practices during manufacture, use, and disposal. Toxicity data steers regulatory agencies to call for robust training programs and detailed documentation for everyone who handles the material, not just to tick a box but to make a real difference in lab and factory safety.
Future Prospects
Looking forward, 4-Chlorobenzoyl Chloride stands at a crossroads shaped by both chemistry and policy. Cleaner synthesis processes will drive adoption in responsible facilities, rewarding research that replaces harsh reagents and slashes emissions. Advances in automation promise less human exposure, while digital workflows flag storage and handling issues before accidents develop. Fields like pharmaceuticals lean hard into structural analogs built from this compound, and as researchers push the boundaries of molecular design, the toolkit only grows. The demand isn’t likely to disappear, but new regulations, eco-friendly directives, and competitive pressures mean every batch tells a story—not just about what got made, but how it got there. For a molecule first drawn up generations ago, the next chapters are likely to be the most transformative yet.
What is the chemical formula of 4-Chlorobenzoyl Chloride?
Understanding 4-Chlorobenzoyl Chloride
4-Chlorobenzoyl chloride often pops up in labs and chemical plants for good reason. Its chemical formula—C7H4Cl2O—is not just a set of numbers and symbols; it shapes the way this compound reacts and behaves. The molecule comes from benzoyl chloride, with a chlorine atom attached to the fourth spot on the benzene ring. Straightforward as it seems, this tiny tweak gives the compound its unique identity.
Why the Formula Matters
I remember my early days in university chemistry—one wrong atom on paper, and the lab results would send us back to square one. In industry, mistakes like that translate to lost hours and added costs. For 4-Chlorobenzoyl chloride, each part of the formula plays a role. The chloride groups hold energetic potential for making new bonds. This is why chemical manufacturers, especially those producing pharmaceuticals or specialty polymers, keep an eye on precise ingredient lists. Getting one atom wrong doesn't just mess up a formula sheet—it risks product safety, purity, and compliance with regulations.
Applications and Safety
Take drug synthesis: the presence of two chloride atoms—one on the aromatic ring, one sticking out from the carbonyl group—lets scientists build up or modify complex molecules. A batch of anti-inflammatory medications may rely on the accuracy of this reagent. In polymer chemistry, that same structure lends itself to forming specialty polymers with properties tailored for strength or resistance.
Handling this compound is never a casual affair. I’ve seen chemical burns firsthand from mishandling reactive chlorinated compounds. The formula C7H4Cl2O signals strong reactivity—especially with water, leading to releases of irritating or toxic gases such as hydrogen chloride. Proper training and equipment become non-negotiable. Lab teams rely on clear labeling and up-to-date material safety data sheets. Each symbol in the formula ties directly to the safety routines set in workplaces across the globe.
Quality, Trust, and Research
Researchers count on the reliability that comes from understanding the chemical structure. Peer-reviewed journals demand verification down to the molecular level, and regulatory bodies expect documentation that confirms products perform as claimed. Building trust starts with the basics, and a correct chemical formula forms the foundation for any experiment or production run. In my time reviewing research, quality control slips have led to retractions and lost credibility.
Improving Clarity and Outcomes
Better education around reading and writing chemical formulas pays dividends. Students and professionals benefit from checking, double-checking, and referencing trusted resources such as PubChem or peer-reviewed handbooks. Digital resources simplify formula validation, but continuous investment in training keeps everyone sharp.
Companies and educators who promote a culture of accuracy see fewer safety incidents and higher customer satisfaction. I’ve come across facilities where routine peer review and open communication around chemical identities save time and money in the long run. In my view, investing in foundational understanding creates a ripple effect—improving research reliability, product safety, and professional integrity.
References
1. PubChem Database, 4-Chlorobenzoyl chloride, NCBI.2. United States National Library of Medicine, Hazardous Substances Data Bank.3. Ullmann’s Encyclopedia of Industrial Chemistry.
What are the main uses or applications of 4-Chlorobenzoyl Chloride?
The Backbone Behind Many Chemical Creations
If you’ve ever been curious about what makes modern chemistry tick, 4-chlorobenzoyl chloride sits pretty close to the top. Its punch comes from the combination of a benzene ring, chlorine, and a reactive acyl chloride. You don’t find this chemical grabbing headlines, but its fingerprint turns up everywhere—especially once you start tracing the origins of everyday products and essential medicines.
Building Blocks in Pharmaceuticals
I’ve watched scientists rely on 4-chlorobenzoyl chloride as a go-to intermediate in drug synthesis. This isn’t lab trivia—it really matters to how quickly new medicines reach people. Chemists value this compound because it lets them introduce a “chlorobenzoyl” group, allowing new structures through acylation. Several pain relievers and antibacterial agents take shape around this transformation. Each time researchers tweak the molecule, they open the door to better drugs, improved bioavailability, and sometimes, fewer side effects. It’s not glamorous, but it’s critical to seeing progress in the pharmacy aisle.
Agrochemicals: Feeding a Growing World
The race to feed more people safely and efficiently touches almost everyone I know in agriculture. 4-chlorobenzoyl chloride supports that work because it helps chemists build herbicides and insecticides. Crops face constant pressure from weeds and pests. Adding the right chemical groups using this compound makes it possible to target threats more precisely. It clears the path for modern products like selective weed killers—tools that help farmers grow more food with less waste and lower environmental impact. Society enjoys safer produce in part because of careful chemical design at this step.
Specialty Chemicals: From Ink to Polymers
Anyone who’s worked with dyes or specialty coatings has probably crossed paths with 4-chlorobenzoyl chloride’s legacy. It acts as a starter for advanced dyes and pigments. These give vibrant color to textiles and stable finishes to plastics. Its use stands out in the world of polymer chemistry, especially for high-performance plastics and resins. I’ve seen industries cut waste and boost longevity in their materials, all while shielding electronics or automotive parts. The compound’s reactivity makes it easier to get durable, heat-resistant products that last.
Laboratory Workhorse
Even in smaller lab settings, this chemical takes on a big role as a reagent. Researchers exploring new compounds or vetting organic reactions will often reach for 4-chlorobenzoyl chloride. It unlocks routes to new molecules—steps that keep lab benches busy and open up paths for both academic and industrial discoveries.
Addressing Health and Safety
One thing I always stress is that strong chemicals call for respect. 4-chlorobenzoyl chloride can burn skin and eyes, and its vapors don’t mess around. Experts make a real difference—proper ventilation, gloves, and training help keep people safe. Some companies now explore greener synthesis pathways to cut down on hazardous byproducts. These advances can limit exposure and cut costs from handling hazardous waste. Broad adoption of safer practices in manufacturing keeps everyone—from chemists to consumers—out of harm’s way.
What Comes Next?
Innovation rarely means tossing out proven tools. Instead, I see efforts focused on using 4-chlorobenzoyl chloride more thoughtfully. Developing catalysts to lower reaction temperatures, swapping toxic solvents for water-based ones, or finding ways to recycle byproducts—these solutions build a more responsible chemical industry. End-users, including pharmacy buyers and farmers, rely on vendors who back up their products with real transparency. At the end of the day, strong safety standards and better chemistry open up opportunities for everyone. A world built on smart chemistry works for all of us, not just for those with a lab coat and a pipette.
What are the storage and handling requirements for 4-Chlorobenzoyl Chloride?
Why Its Storage Takes Serious Attention
Nobody enjoys unnecessary hazards, especially where strong chemicals like 4-Chlorobenzoyl Chloride enter the picture. This compound, common in industrial organic synthesis, won’t hesitate to put workers at risk if basic safety gets ignored. I’ve watched colleagues underestimate how fast a volatile or corrosive chemical can stir up trouble—they learn quick by example or sore eyes.
Direct contact with moist air or water sparks a nasty reaction. The fumes alone can irritate eyes, skin, and lungs, so any carelessness leads to more than just a cleanup headache. I’ve seen storage rooms overheat, causing bottles to hiss ominously. No one forgets the smell after that. Keeping this stuff secure isn’t overkill. It’s self-preservation.
Location Matters: Always Cool and Dry
No one stores perishable food next to a heater; the same goes for reactive chemicals. A dedicated chemical storage area works best. Solid, well-ventilated shelving in a cool corner stops temperature swings and moisture build-up. Humidity goes up—trouble follows. Strong odor catchers or monitoring tools help spot leaks long before a crisis.
From my own hands-on days, glass containers with Teflon-lined caps keep out oxygen and humidity and laugh off chemical corrosion. Metal reacts; plastics can melt. Most accidents I’ve heard about began with picking a cheap container or neglecting the cap. Cost-cutting doesn’t balance out the spill risk.
Direct Handling: Suits, Gloves, and Fume Hoods
Thin disposable gloves barely register as protection around chemicals like this. I always go for nitrile or long cuffed rubber gloves—skin contact is no joke. Face shields and goggles protect eyes from even the tiniest splash or mist. A proper lab coat covers up arms that stray within the danger zone.
Pouring or transferring, I always use a certified fume hood. The fumes sting and linger fast. Respirators with well-matched cartridges help when ventilation won’t cut it, especially during larger transfers. Rushed work, loose lids, and skipping a fume hood make toxic exposure a guarantee. Safe workflow becomes instinct fast after seeing white clouds rise from a beaker and set off the lab alarm.
Disposal and Cleanup: Don’t Wing It
Waste disposal calls for special containers labeled as hazardous. Pouring leftovers down the drain invites legal headaches and environmental harm. I collect spills using absorbent material rated for acid chlorides, not whatever mop sits closest. Using neutralizing powders shrinks the danger, but a phone call to hazardous waste professionals makes the real difference.
Training and Labels Change Everything
Everyone working near reactive chemicals benefits from clear, frequent training. I once watched a new hire try to wipe up a spill with paper towels—a lesson in better onboarding followed. Labels with dates, contents, and warning symbols give everyone fair notice.
Chemical safety boils down to systems—never shortcuts. Most stories of chemical mishaps have roots in a skipped label, missing glove, or a shortcut during a transfer. Skills honed by repetition, quality storage, and strong habits keep production running and people healthy.
What are the safety hazards and precautions for using 4-Chlorobenzoyl Chloride?
Understanding the Real Risks
4-Chlorobenzoyl chloride doesn’t sound threatening until it’s in a flask in front of you, ready to splash or fume. This compound, common in labs and chemical plants, shows up in pharmaceutical synthesis, dye manufacturing, and agrochemical production. The risks become clear as soon as that strong, acidic odor hits. Skin contact causes burns. A single whiff of the vapor sends your throat itching and your nose running. Spilled on a bench—or worse, your hand—it reacts with water, producing hydrochloric acid fumes powerful enough to turn a careless moment into an emergency.
What Makes It So Hazardous?
Workers handling 4-Chlorobenzoyl chloride face more than just simple irritation. It eats through gloves if given the chance. On the skin or in the eyes, it doesn’t just sting; it causes deep, painful burns. Breathing it in can trigger coughing fits and damage airways. A less-discussed hazard involves its reaction with water, producing toxic, corrosive gases that spread quickly in a closed space. Even the dust can irritate eyes and lungs, adding another layer of risk during any transfer or packaging.
Everyday Precautions That Matter
Hard-earned experience in the lab taught me to respect chemicals with a reputation. 4-Chlorobenzoyl chloride is at the top of that list. Rushing is never worth it. The right habit starts with the right gear. Always reach for goggles that seal tightly, not just open safety specs. A lab coat seems basic, but when the splash comes it’s the barrier between you and a nasty burn. Chemical-resistant nitrile or neoprene gloves work better than the standard latex kind, which dissolve fast and leave hands exposed.
Lab ventilation saves more than your comfort. A good fume hood pulls vapors away before they hit your lungs. If you smell its pungency, that means the system isn’t strong enough. Spills need quick attention, but never with water. Dumping a bucket of water on the mess only turns the liquid into stinging gas. Use absorbent pads rated for corrosive chemicals, and keep containers sealed and stored below eye level on spill trays.
Better Training, Fewer Accidents
No instruction manual covers everything. Good training means seeing the risks for yourself and hearing about what’s happened before—someone elsewhere, years ago, who let their guard slip. Lessons stick better that way. Newcomers benefit from walking through every step under a watchful eye, not just reading safety sheets. On-the-job stories make the dangers real and memorable.
Responsible Handling Doesn't End in the Lab
Disposal often gets overlooked, even though real danger remains once the bottle’s nearly empty. Leftovers and contaminated gear need proper containers, clearly labeled, waiting for safe pickup—not tossed into the regular trash. Waste treatment technicians rely on that early effort, and so do the folks working down the line in sanitation and environmental cleanup.
Stronger Systems Mean Safer Work
Every time a worker gets careless or a company skimps on training or protective equipment, the risks don't just affect one person. Mishaps result in hospital trips, missed work, and sometimes lasting injuries. Proper investment in safety gear and regular hands-on education turns risky compounds like 4-Chlorobenzoyl chloride into manageable tools instead of workplace hazards. Companies that back up their talk with real systems see fewer accidents and a reputation for safety. That matters much more than cutting corners or working a little faster.
What is the physical appearance and purity specification of 4-Chlorobenzoyl Chloride?
What Stands Out About 4-Chlorobenzoyl Chloride?
Walking into a chemical storeroom, the first thing you notice with 4-chlorobenzoyl chloride is how plain it looks at first glance. It usually shows up as a clear to pale yellow liquid. That pale yellow shade isn’t just for show; it hints at its stability and how it's been stored. Over time, any color deepening would signal contact with moisture or light — both enemies of this compound. Sometimes even a little bit of it splashed on a bench leaves behind a lingering, pungent smell. That’s the chloride group in action, right there, announcing its presence.
The Issue With Purity
Researchers and glassware veterans care about purity for a simple reason: reactions don’t lie. Impurities sneak into every experiment’s data set, sometimes ruining dozens of hours of work. High-purity 4-chlorobenzoyl chloride tends to run above 99%, often verified by gas chromatography or titration. Roll out a product below 98%, and you might as well be spinning your wheels. You start seeing unpredictability in yields because some side reactions crop up, thanks to those extra contaminants.
Industrious labs and big pharmaceutical facilities have learned that taking shortcuts with purity standards traps them in endless troubleshooting. Consistent quality checks keep downtime to a minimum. My own time in an organic synthesis lab taught me that a smaller batch from a less reputable supplier often tests out with water impurities or leftover solvents. Water loves to get cozy with acid chlorides and throw off weird, corrosive by-products like hydrochloric acid. That smell of bitterness in the air? A sign somebody went cheap on purity.
Safety and Handling: More Than Just Gloves
Pick up a bottle of 4-chlorobenzoyl chloride and the first thing you’ll notice after the smell is the warning on the label. Skin contact burns. Vapors hit the nose and lungs hard. The yellow tint gets noticed, not just for technical reasons, but because it signals degradation, which makes fumes even nastier. Big glove boxes, good exhaust hoods, and careful refrigeration keep things safer. The real headache comes when moisture finds its way in. Decomposition kicks off, producing hydrochloric acid and putting glassware, metal, and even skin at risk.
Chasing Higher Purity: What Actually Works?
Instead of relying just on supplier specs, labs regularly run their own test analyses. Titration helps to check the acid content, giving a quick answer about water contamination. Gas chromatography pinpoints organic impurities. Anyone serious about research takes this double-check approach, especially for scale-up or critical synthesis work. Last month, during a round of QC for a multi-step synthesis project, our team caught a batch that barely hit 96.5%. That’s enough to wreck an entire sequence, sending days of progress straight into waste disposal.
Trusting a reputable manufacturer for consistent sourcing and storing the product in moisture-tight containers goes a long way. If you keep the bottle tightly sealed, away from light, and avoid plastic stoppers (which sometimes get eaten by the corrosive fumes), you'll see less color change and virtually no loss in purity over short periods.
| Names | |
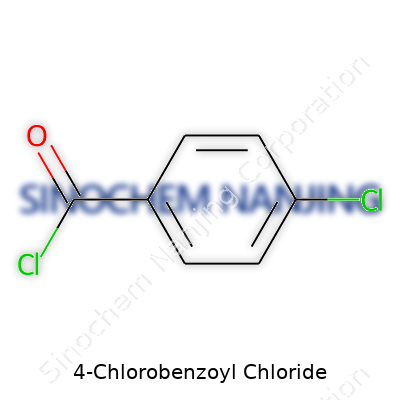
| Preferred IUPAC name | 4-chlorobenzoyl chloride |
| Other names |
p-Chlorobenzoyl chloride 4-Chlorobenzenecarbonyl chloride p-Chlorobenzenecarbonyl chloride 4-Chlorobenzoic acid chloride 4-Chlorobenzoylchloride 4-Chlorobenzencarbonyl chloride |
| Pronunciation | /ˈklɔːr.oʊ.bɛnˌzoɪl ˈklɔːr.aɪd/ |
| Identifiers | |
| CAS Number | 122-01-0 |
| 3D model (JSmol) | `3D model (JSmol)` string for **4-Chlorobenzoyl Chloride** (C7H4Cl2O): ``` Clc1ccc(cc1)C(=O)Cl ``` |
| Beilstein Reference | 1208551 |
| ChEBI | CHEBI:85021 |
| ChEMBL | CHEMBL12208 |
| ChemSpider | 11095 |
| DrugBank | DB14230 |
| ECHA InfoCard | 100.006.171 |
| EC Number | 211-958-1 |
| Gmelin Reference | Gmelin Reference: 142165 |
| KEGG | C01508 |
| MeSH | D002678 |
| PubChem CID | 7488 |
| RTECS number | CD0700000 |
| UNII | 4X6B2H971N |
| UN number | UN1752 |
| Properties | |
| Chemical formula | C7H4Cl2O |
| Molar mass | 190.00 g/mol |
| Appearance | White to light yellow crystalline powder |
| Odor | Pungent odor |
| Density | 1.38 g/mL at 25 °C |
| Solubility in water | Reacts with water |
| log P | 2.9 |
| Vapor pressure | 0.4 mmHg (20 °C) |
| Acidity (pKa) | 13.3 |
| Magnetic susceptibility (χ) | -67.5 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | nD 1.568 |
| Viscosity | 1.474 cP (20°C) |
| Dipole moment | 2.96 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 309.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -205.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -622.6 kJ/mol |
| Pharmacology | |
| ATC code | Not assigned |
| Hazards | |
| Main hazards | Corrosive, causes severe skin burns and eye damage, harmful if inhaled, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H302, H314, H332 |
| Precautionary statements | P261, P264, P271, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P321, P363, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-0-W |
| Flash point | 78 °C |
| Autoignition temperature | 250 °C |
| Lethal dose or concentration | LD50 oral rat 2300 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 1410 mg/kg |
| NIOSH | DN6300000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.05 mg/m³ |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Benzoyl chloride 4-Chlorobenzoic acid 4-Chlorobenzaldehyde 4-Chloroaniline Benzoyl bromide |

