4-Chlorobenzotrifluoride: A Deep Dive Into Its Role, Risks, and Rewards
Tracing the Story of 4-Chlorobenzotrifluoride
In the world of specialty chemicals, 4-Chlorobenzotrifluoride, known to chemists as PCBTF, shapes modern industry in ways most folks never realize. Decades back, global drive toward stronger, more versatile solvents in coatings, inks, and cleaning agents set the stage for widespread PCBTF production. The story runs parallel to a growing awareness about environmental impact, which forced both manufacturers and users to shift toward chemicals with lower contribution to smog formation and ozone depletion. Stepping back, I remember seeing regulatory conversations swing from nearly ignoring these solvents to scrutinizing every molecular structure, especially as the paint and coatings industry scrambled for alternatives to traditional high-VOC options. As environmental rules tightened up in the United States and Europe, the demand for more "acceptable" solvents pushed PCBTF past the boundaries of a lab oddity into a staple of commercial production lines. Watchdog groups raised ethical flags about each replacement, but industry eyes stayed fixed on PCBTF for its remarkable balance between performance and regulatory compliance.
At Its Core: Physical and Chemical Traits
PCBTF brings together a unique set of traits. It appears as a clear, colorless liquid under standard room conditions and carries a moderately pleasant, sweet odor. Chemically, the molecule features a single chlorine atom attached to a benzene ring, right beside a trifluoromethyl group. This structure grants it a high degree of stability, resistance to acids and bases, and remarkable solvency power. Its boiling point sits higher than many traditional solvents, making evaporative losses less of a concern for workers and businesses. It’s also only very slightly soluble in water, a trait that influences containment and cleanup strategies. The trifluoromethyl group, with its high electronegativity, discourages unwanted reactions with atmospheric ozone, explaining why environmental regulators treat it differently than old-line solvents such as toluene. On the ground, these physical and chemical features translate into concrete advantages—better performance with many resins and pigments, and much lower smog-forming potential.
Preparation and Chemical Reactions: How PCBTF Gets Made and Modified
Chemists looking to make PCBTF tend to rely on straightforward reactions involving chlorination and trifluoromethylation. You take benzotrichloride as a starting material, introduce chlorine through catalytic chlorination, and then swap out a chlorine atom for a trifluoromethyl group using a fluorinating agent under controlled conditions. These steps require steady control of temperature and pressure to yield a product pure enough for industrial applications. The finished molecule stays fairly inert under most use conditions, but it does give synthetic chemists options for downstream modification. For example, in a research lab or specialty plant, the chlorine or fluorinated groups may serve as anchors for building more complex molecules. While this isn’t a chemical most people will tinker with at home, anybody working with dyes, agrochemicals, or pharmaceuticals appreciates the reactivity available at both the chlorine and the benzene nucleus.
Synonyms and Surprising Names
People in industry circles recognize PCBTF by several names. Its main synonyms include 1-Chloro-4-(trifluoromethyl)benzene and para-Chlorobenzotrifluoride. Sometimes you'll see trade names that reflect supplier brands, but the chemical backbone stays the same. Naming confusion still trips up newcomers, especially when regulations or data sheets refer to different synonyms. For anyone trying to understand labeling requirements, knowing these alternate names eliminates a lot of headaches and prevents duplicate inventory stocking.
Real-World Applications: Impact in Everyday Life and Industry
PCBTF primarily finds its home in solvent-based coatings—paints that cover skyscrapers, bridges, and cars alike. Both automotive refinish shops and large-scale manufacturers learned they could rely on PCBTF to dissolve resins and carry pigments, offering strong performance without the regulatory headaches attached to many other aromatic solvents. Its resistance to breaking down lets it act as a carrier fluid in printing inks and some adhesives. In cleaning product formulations, PCBTF strips away grease and residues, especially where stubborn industrial grime challenges more water-based systems. Field service techs count on it as a component in cleaning electronics or precision metalwork, although this carries a different set of risks if ventilation falls short. In consumer-facing products, PCBTF stays further back, mostly appearing in the supply chain rather than household goods—part of what shapes its public profile and debate among advocacy groups.
Technical Specs and What They Mean for Workers
For those handling PCBTF in a manufacturing or laboratory context, a solid grasp of technical data makes all the difference between safe operations and regrettable mistakes. PCBTF boils at around 139°C and sets fire only under strong ignition, pointing to lower flammability risks than lighter solvents. Its moderate vapor pressure means it hangs around in the air long enough to warrant good ventilation, especially in enclosed factory settings. Workers remember that PCBTF doesn't mix well with water, so traditional water-based cleaning won't help in a messy spill. Equipment designers and workplace managers build systems around these facts: proper fume hoods, good gloves, and informed labeling standards all help stem dangerous exposures.
Safety Practices and Industry Standards
Proper handling and exposure control for PCBTF drive safer workplaces and protect nearby communities. Regulatory bodies such as OSHA and their international peers weigh in with exposure limits, usually in the neighborhood of tens of parts per million measured over a standard shift. On plant floors, I’ve watched how teams conduct air quality checks and install ventilation aimed at pulling vapors away from breath zones. Material safety teams stress the importance of storing PCBTF away from incompatible chemicals, like strong oxidizers or open flames, simply because chemical stability shouldn’t be taken for granted when thousands of liters sit in drums. Emergency procedures cover quick containment and cleanup, reinforcing the lessons from countless training sessions. Having met engineers and operators who take real pride in safe workplaces, I see firsthand how following these guidelines prevents long-term health problems not just for workers, but whole communities downstream.
Research, Development, and the Toxicity Question
Ongoing research keeps reshaping our understanding of PCBTF’s risks and benefits. Toxicity data points to moderate acute effects: direct contact can irritate the skin or eyes, and inhaling high concentrations leads to dizziness or drowsiness. Chronic data sets remain less robust, with most studies finding low bioaccumulation potential in animals, but uncertainty lingers over possible links to longer-term health issues with sustained, high-level exposure. As regulations shifted in California, the state flagged PCBTF under its Prop 65 list, sparking debates beyond technical circles. Industry-backed studies suggest no significant carcinogenic risk at levels found in typical workplaces; advocacy groups counter that gaps in data leave room for caution rather than complacency. As a teacher and neighbor, I recognize how toxicity data doesn’t just stay in government filings—it shapes real choices about which products to buy, which jobs to take, and what questions to ask at community meetings.
The Road Ahead: Environmental Impact and Future Prospects
PCBTF stands at a crossroads. Environmental stewardship calls for a sharper look at how chemicals linger in air, water, and soil. Scientists continue to dissect PCBTF’s breakdown pathways, aiming to pin down its fate and effects if it slips past containment at a factory or job site. Early findings indicate a degree of persistence, with degradation more likely in air than groundwater. Policy shifts in the US or Europe will probably depend not just on new toxicity data, but on broader pushes toward circular economies and nontoxic workplaces. Meanwhile, research groups hunt for even lower-emission solvents, pushing PCBTF closer to obsolescence in some markets and into new specialty niches in others. My time spent talking with both older chemists and the next generation of researchers shows an industry unwilling to settle for partial solutions. Collectively, we shape the story of PCBTF by asking tougher questions, demanding more transparent safety data, and never dropping the guard against chemical complacency. What happens next matters not just to companies and workers, but to future generations inheriting the chemical choices we make today.
What is 4-Chlorobenzotrifluoride used for?
What It Is
4-Chlorobenzotrifluoride is a word you don’t hear every day unless you work in a chemistry lab or on a factory floor. This compound, with its mouthful of a name, ends up playing a bigger role in daily life than most people imagine. Made up of a benzene ring, a chlorine atom, and three fluorine atoms attached to a carbon, it shows up as a volatile, colorless liquid. If you’ve ever been near its vapors, the sharp, almost medicinal smell lingers in your nose.
How Companies Use 4-Chlorobenzotrifluoride
Most people never see 4-Chlorobenzotrifluoride in pure form on a store shelf. Industry, though, uses this compound as a building block. Manufacturers need it when making agrochemicals, pharmaceuticals, and dyes. A cornfield sprouting impossibly green after a spray or a vivid fabric that doesn’t fade in the sun? There’s a fair chance 4-Chlorobenzotrifluoride played a hidden part. It’s a starting point for other chemicals, especially where chemists want to tack on a strong fluorine group. You’ll also find it in some specialty paints, varnishes, and coatings. Here, the chemical helps resins stick to surfaces and protects against weathering. In the past, I’ve watched paint technologists tinker for days, searching for that perfect balance of shine and scratch protection. They ask suppliers for chemicals that can stretch a product’s performance, and 4-Chlorobenzotrifluoride often comes up in their list.
The Real-World Value
Many of us care about sturdy, long-lasting goods—no one wants cars or farm tools covered in rust after a year outside. That’s where this chemical quietly offers value. By blending it into polymers and coatings, factories make their goods less likely to crack, peel, or change color. Fluorinated compounds such as this one repel oils and water, adding another layer of protection. The medicine cabinet also benefits. Certain drug molecules need a chlorine or fluorine piece in just the right spot, and 4-Chlorobenzotrifluoride helps researchers get there. Next time you pick up a pill, it helps to remember that some of the chemistry behind safe and stable medicines starts with simple, versatile compounds such as this.
Weighing Safety and Environmental Cost
No story about industrial chemicals feels honest without addressing risk. 4-Chlorobenzotrifluoride is flammable and can irritate the skin and lungs. Workers need gloves and proper ventilation. Regulatory agencies track how much gets handled and released, knowing that persistent chemicals can build up in water and soil. In the nineties, I recall reading reports about how halogenated solvents drifted into streams. Since then, rules tightened, forcing companies to recycle and contain chemical wastes better. Studies show 4-Chlorobenzotrifluoride breaks down slowly in nature, so safety measures remain a priority.
Looking Forward
Cleaner and safer chemistry sits high on the agenda. Today, manufacturers explore catalysts and reaction conditions that trim waste or keep dangerous byproducts out of the air. Alternatives to chlorinated solvents get more research funding than ever. At the same time, material scientists continue to rely on chemicals like 4-Chlorobenzotrifluoride for breakthroughs in durability and design. The push for greener options walks a tricky path—balancing progress and protection.
Solutions in Practice
Companies can invest in closed-loop systems, using less fresh material and cutting emissions. Routine monitoring and strict labeling keep workers safer. Chemists also test biodegradable substitutes, though few match the stability and functional punch of trifluoromethyl compounds. Sharing research and pushing for international standards helps everyone move forward together. We all rely—sometimes unknowingly—on good chemistry, so it pays to keep asking where our industrial staples come from and where they end up.
What is the chemical formula and structure of 4-Chlorobenzotrifluoride?
Chemical Identity that Matters
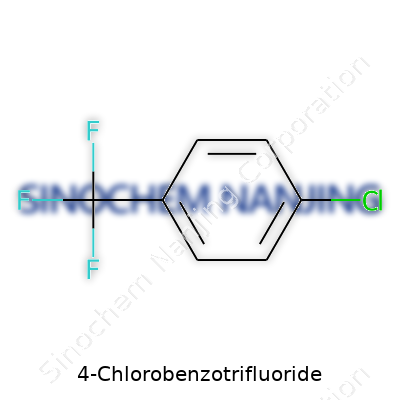
4-Chlorobenzotrifluoride has a chemical formula of C7H4ClF3. In basic terms, it’s a benzene ring—a classic shape in organic chemistry—carrying both a chlorine atom and a trifluoromethyl group. In the shorthand of chemists, the structure arranges the chlorine atom at the fourth position relative to the trifluoromethyl group. Picture the benzene ring, count off positions around the hexagon, and you’ll see the symmetry of putting the chlorine opposite the trifluoromethyl group.
Getting Clear about Structure
The best way to think about the structure: the benzene ring forms the core. Attach a trifluoromethyl group (–CF3) at position one. Directly across from it lands the chlorine atom (–Cl), at position four. The rest of the ring holds hydrogen atoms. It’s simple and predictable, which helps researchers spot its identity with certainty. This arrangement gives the molecule both stability and a set of physical properties that industry finds useful.
Practical Uses Rooted in Chemistry
Paints, coatings, and specialty chemicals manufacturers lean on 4-Chlorobenzotrifluoride for its solvent qualities and its ability to stand up to harsh conditions. The compound’s chemical resilience comes from the fluorine atoms—bonds with carbon in the trifluoromethyl group resist breaking apart. Chlorine’s presence further changes the behavior of the molecule, boosting its usefulness in creating intermediates for other chemicals such as pharmaceuticals and agrochemicals.
Handling Concerns and Health Stances
Anyone handling organic compounds needs to pay attention to both health and environmental effects. As someone who’s spent time in research labs working with halogenated solvents, good ventilation, protective gloves, and correct disposal practices always form part of the routine. Inhalation or prolonged skin exposure carries risks, so it pays to follow safety data sheet advice. Information from the U.S. Occupational Safety and Health Administration and the European Chemicals Agency highlights the need for caution, especially since trifluoromethylated aromatics can linger in the environment.
Environmental Impact and Solutions
Fluorinated aromatics are known for their persistence. They don’t break down easily. Responsible usage means limiting the amount released during manufacturing and choosing methods that capture waste. Investing in improved scrubbing systems, recycling solvents, and switching to less hazardous alternatives for minor uses can cut down on environmental exposure. Researchers keep searching for ways to design greener synthetic routes and to degrade leftover organofluorines safely. Transparent documentation and meticulous tracking build trust between manufacturers, regulators, and neighbors living near production sites.
Weighing Value and Responsibility
4-Chlorobenzotrifluoride brings real value to chemistry and materials science, offering properties that make some processes more efficient and durable. That same stability also creates responsibility. It’s easy to forget how small spills and escapes add up over time. My own work showed that small changes in lab practice—better labels, double-checking inventory, swapping out gloves sooner—can make a big difference. Regulators and businesses owe it to themselves and the public to keep safety and transparency front and center as they use and manage this compound. Staying ahead of both science and standards is the way forward.
Is 4-Chlorobenzotrifluoride hazardous or toxic?
Understanding 4-Chlorobenzotrifluoride
4-Chlorobenzotrifluoride sometimes pops up in paint thinners, cleaning fluids, and the chemistry behind making other compounds. On the surface, the name alone might set off alarm bells. People hear “chlorine” or “benzene” in a chemical’s name and quickly start wondering if they should steer clear or worry about the safety of their environment.
Looking at Health and Safety Evidence
Safety sheets hand out warnings for this compound. Direct skin contact can trigger irritation, and eyes exposed to it might sting or even turn red. If a person inhales too much vapor, headaches, dizziness, or nausea can follow pretty fast. Respiratory complaints tend to hit more often indoors, especially around poor ventilation or spills. Swallowing isn’t common, but if it does happen, expect abdominal pain or even vomiting. Long-term, repeat contact with strong solvents dries skin out and may crack it. A few folks I’ve worked with in the lab saw mild dermatitis after cleaning up spills too quickly without gloves.
Workers in industrial settings usually run into this chemical more than most consumers. The U.S. Environmental Protection Agency classifies it as a volatile organic compound and watches workplace exposure carefully. While there’s no strong connection yet to cancer or genetic problems, that doesn’t mean ignoring safety guidelines. The weight of evidence from safety studies still describes it as an irritant, not a carcinogen.
Toxicity and the Bigger Picture
On the toxicity scale, 4-Chlorobenzotrifluoride carries more risk than vinegar, less than pure benzene. Rats and mice exposed to high doses in testing labs showed some central nervous system symptoms, but nothing uniquely alarming compared with many other industrial solvents. High doses killed lab rats, but only at amounts far beyond what workers breathe on a plant floor or painters encounter on a job site. At the same time, we shouldn’t shrug off or ignore it—today’s accidental splash or spill could set off tomorrow’s allergic skin reaction for someone who isn’t careful.
Some small spills evaporate quickly, yet the chemical can stick around in air in low concentrations, especially if the weather’s humid or the workspace stays stuffy. In community settings, air monitoring near factories helps ensure workers and neighbors avoid long-term health problems. Having worked in places where air quality monitoring ran non-stop, folks felt a lot safer with detailed reports and quick responses to any jump in levels.
Reducing Risks in Workplaces and Homes
Prevention always beats treatment. Good protective gloves, eye covers, and running exhaust fans or open doors block most problems before they start. Training workers—making sure they know how to store, pour, and dispose of the stuff—cuts down on accidents. Reading the safety instructions before using these chemicals sounds obvious, but too many skips right past them. At home, sticking with pre-mixed products rather than raw chemical ingredients goes a long way toward safety.
Cleanups deserve just as much care. Treat every small spill as though it could burn or trigger a cough. Used solvents never belong in a sink. Taking the time to drop them off at hazardous waste pickup points protects both the environment and water supply. Taking that extra moment prevents future headaches—literally and figuratively—and keeps everyone healthier in the long run.
What are the storage and handling requirements for 4-Chlorobenzotrifluoride?
Why Safe Storage Matters
Working with 4-Chlorobenzotrifluoride means more than grabbing a drum and sticking it in a corner. I've walked through enough sites to see that chemical safety starts and ends with respect for the little details. This has always meant a secure, cool, and well-marked storage area. Even if it doesn’t look threatening, this solvent can turn into a real headache if left in a sun-baked lot or near sources of ignition.
Keeping drums in a dry spot, away from open flames and incompatible chemicals, closes the risk window. A solid concrete floor makes spills easier to handle, while proper ventilation keeps fumes out of your breathing zone. It never hurts to keep containers tightly sealed — solvents like this don't just evaporate, they take safety out the window along with them. I always make sure that the labeling is clear. No one should have to play detective when they need to know what's inside a drum.
Personal Protection Isn’t Overkill
Many people tend to think of gloves and goggles as overkill, but if you’ve lost count of how many splashes you’ve dodged, you’ll never skip the gear. Chemical-resistant gloves and goggles save a trip to the eyewash and worse. I keep a spill kit nearby and encourage everyone else to know where it lives. In my experience, the time to look for emergency eyewash or showers is well before someone needs them.
Containment, Spills, and Cleanup
You never plan a spill, but you should prepare for one. I’ve seen 4-Chlorobenzotrifluoride trickle from a leaky valve and soak into packing because someone thought “hand tight” meant “tight enough.” Sorbent pads, chemical waste drums, and working procedures keep that leak from becoming an environmental nightmare. You put wet rags, gloves, and debris in designated waste containers so the job doesn’t turn into a hazardous waste puzzle later on.
Good Practices Pay Off
Chemical stability gets a lot of attention in the textbooks, but practicality wins the day. Regular checks for corrosion or signs of wear on containers help spot trouble early. I've built a habit of logging drum locations and expiry dates. It sounds tedious, but records can stop a scramble during audits or when a question comes up about safe shelf life.
The law sets a high bar for safe chemical storage. The United States Occupational Safety and Health Administration (OSHA) lays out clear rules for flammables and hazardous chemicals. Local fire codes can sometimes push requirements even higher. Following the rules saves more than fines; it prevents fires and health problems. According to the National Fire Protection Association, chemical fires often start with routine neglect or complacency around ignitable solvents.
Room for Improvement
No storage practice stays perfect forever. Suppliers roll out improved container materials. Sensor technology makes remote leak detection cheaper. I believe in reviewing chemical handling policies each year, especially after new incidents or near-misses. Team feedback matters. Most improvements in our procedures began with someone pointing out a small flaw or asking the right question. You can’t afford to wait for a major accident to push meaningful change.
What is the CAS number of 4-Chlorobenzotrifluoride?
Spotlighting the CAS Number: 98-56-6
4-Chlorobenzotrifluoride, known to chemists and folks in industry circles under the code 98-56-6, pops up in labs, paint shops, and manufacturing spaces more often than most realize. The CAS number isn’t just a jumble, it's a key identifier that gives a chemical its fingerprint. Type "98-56-6" into a chemical registry and all the information tied to this compound lays itself out—everything from its safety profile to physical form and uses around the globe.
The Chemical Behind the Number
The compound itself carries a sharp set of properties. It’s got a strong trifluoromethyl group and a chlorine atom tucked on a benzene ring, bringing both reactivity and stability. In daily experience, you find it turned into solvents for paints and coatings, and it helps the dye and pharmaceutical trades churn out new products. In my own time messing with various solvents for paint thinning and stain removal, this one stood out—a whiff tells you enough to handle it with caution.
Why the CAS Number Matters
Accuracy in chemical identification keeps lab benches and big industrial tanks safe. Picture two containers—labels worn down, smell nearly identical. Without the right CAS number, someone can mix up a batch that could ruin a run of paint or, worse, trigger a safety incident. In 2019, the European Chemicals Agency ran a review of several aromatic solvents and flagged wrong labeling as a hazard in about 7% of facility checks. A misstep in identification doesn’t just cost money, it hikes up the health risks and the chance of environmental trouble.
Safety and Health: More Than Just a Number
This chemical packs enough punch that safety data sheets give clear warnings. Exposure to its vapor can irritate the eyes and nose. Working with it in a closed space, you feel the difference right away. Regulations like OSHA call for air monitoring, solid ventilation, and defined storage protocols. In smaller shops, a simple fume hood and gloves go a long way, but those steps only help if workers actually follow them. Reading the CAS number off the drum doubles as a mental nudge to check each handling step.
Fixing What Goes Wrong
Mislabeled drums and confused shipments pop up, even in companies with strict warehouse routines. Training can slip and lead to a whole supply chain mistake. Printing the CAS number on every secondary label and running regular cross-reference checks between purchase, transport, and storage gets ahead of the problem. A digital inventory system that flags errors based on CAS numbers saves headaches for everyone from the loading dock to the back office.
Responsible Practices Moving Forward
Using 4-chlorobenzotrifluoride responsibly takes both up-to-date knowledge and a hard look at how procedures play out on the floor. Groups like the American Chemical Society keep recommending practical steps, like pairing digital records with barcodes tied to each chemical’s CAS code. Coming from experience, taking the minute to confirm a CAS number like 98-56-6 on both a bottle and a delivery log often heads off bigger mistakes. In the end, the number isn’t just a technical detail—it's the foundation that keeps chemicals sorted, workers safe, and products legitimate.
| Names | |
| Preferred IUPAC name | 1-chloro-4-(trifluoromethyl)benzene |
| Other names |
PCBTF p-Chlorobenzotrifluoride Parachlorobenzotrifluoride 1-Chloro-4-(trifluoromethyl)benzene 4-Chloro-α,α,α-trifluorotoluene |
| Pronunciation | /ˈklɔːr.oʊ.bɛn.zoʊ.trɪˈfluː.raɪd/ |
| Identifiers | |
| CAS Number | 98-56-6 |
| Beilstein Reference | 1209246 |
| ChEBI | CHEBI:82127 |
| ChEMBL | CHEMBL31867 |
| ChemSpider | 9429 |
| DrugBank | DB14084 |
| ECHA InfoCard | ECHA InfoCard: 100.011.677 |
| EC Number | 208-890-7 |
| Gmelin Reference | 101874 |
| KEGG | C14316 |
| MeSH | D002681 |
| PubChem CID | 72885 |
| RTECS number | CB9275000 |
| UNII | 97Q6RV5F2Y |
| UN number | UN2234 |
| Properties | |
| Chemical formula | C7H4ClF3 |
| Molar mass | 181.56 g/mol |
| Appearance | Clear, colorless liquid |
| Odor | Aromatic odor |
| Density | 1.34 g/mL at 25 °C (lit.) |
| Solubility in water | Insoluble |
| log P | 3.7 |
| Vapor pressure | 3.2 mmHg (25°C) |
| Acidity (pKa) | ~43 |
| Basicity (pKb) | Product not basic |
| Magnetic susceptibility (χ) | -54×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.496 |
| Viscosity | 1.30 mPa·s at 25 °C |
| Dipole moment | 3.44 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | '340.3 J·mol⁻¹·K⁻¹' |
| Std enthalpy of formation (ΔfH⦵298) | -584.4 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2028.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07,GHS09 |
| Signal word | Warning |
| Hazard statements | H315, H319, H335 |
| Precautionary statements | P210, P261, P264, P271, P280, P301+P312, P303+P361+P353, P305+P351+P338, P337+P313, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 2-2-0 |
| Flash point | 54°C |
| Autoignition temperature | 705°C |
| Lethal dose or concentration | LD50 Oral Rat 13,000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 > 6800 mg/kg |
| NIOSH | CN9840000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | REL (Recommended Exposure Limit) of 4-Chlorobenzotrifluoride is "2.5 ppm (15 mg/m³) as a 10-hour TWA". |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Benzotrifluoride 3-Chlorobenzotrifluoride 2-Chlorobenzotrifluoride 4-Bromobenzotrifluoride 4-Fluorobenzotrifluoride 4-Nitrobenzotrifluoride |

