4-Chloro-m-Cresol: A Close Look at a Ubiquitous Chemical
Historical Development
Tracing the journey of 4-Chloro-m-Cresol requires respect for the chemical industry’s progress over the last century. Early researchers found cresols in coal tar distillates around the late 19th century. With advances in organic chemistry, halogenated derivatives drew attention for their antimicrobial activities. Laboratories around the globe, from Germany to the UK, began manipulating cresol molecules, adding chlorine atoms to enhance performance. Through trial and error, chemists at the bench discovered 4-Chloro-m-Cresol packed a reliable punch against bacteria and fungi. Today’s manufacturing reflects both that curiosity and years of trial, spanning everything from antiseptics to industrial formulations.
Product Overview
Walk through any industrial supply catalogue and 4-Chloro-m-Cresol will not hide. This white to off-white crystalline substance, also called para-chloro-meta-cresol, keeps popping up—medical disinfectants, wood preservatives, and even cosmetic products keep finding value in its broad-spectrum abilities. Its roots run deep in healthcare, especially as a talking point in formulating safe but potent germicides. If you ever swabbed an abrasion and caught a peculiar phenolic scent, you might have been using a soap or ointment built around it.
Physical & Chemical Properties
The compound’s chemical formula, C7H7ClO, reveals a building block with just enough tweak to make a difference. Crystalline granules flow easily, dissolving well enough in alcohol or ether, not so willingly in water. Scientists favor these properties: the melting point hovers around 63°C, the boiling point passes 225°C, and the compound stays stable under most storage conditions. With its modest solubility and aromatic ring, 4-Chloro-m-Cresol escapes easy detection in the field but retains plenty of power in a controlled application.
Technical Specifications & Labeling
If you have handled a drum or bottle in a research or industrial setting, you might recall cautionary symbols on the label—warnings linked to skin and eye contact. Labels reflect legal and scientific standards worldwide, especially noting purity levels above 98% and identifying codes for customs and laboratory systems. The pleasant notion of a neat list of certifications and quality grades dissolves fast if someone ignores safe handling guidelines. This chemical needs respect from every operator, from lab techs up to warehouse managers, to avoid unnecessary accidents and ensure consistent output for any batch sent out.
Preparation Method
Making 4-Chloro-m-Cresol calls for precise chemistry. Usually, the story starts with meta-cresol, sourced from coal tar or petrochemical routes. Chemists then introduce chlorine, often using controlled chlorination reactions. Industrial reactors allow tight temperature and agitation control, helping minimize byproducts like 6-chloro variants or unwanted isomers. The reaction mixture goes through a series of washes and crystallizations to yield a pure product. These steps require vigilance, not only to hit purity targets but to minimize environmental impact—chlorinated waste streams, if mishandled, can spur regulatory headaches and long-term pollution problems.
Chemical Reactions & Modifications
Anyone who’s tinkered in a synthesis lab knows how a slight tweak opens doors. Modifying 4-Chloro-m-Cresol’s structure creates analogs tailored for niche use. The phenolic ring welcomes alkyl and halogen substitutes—chemists use these tweaks to shift antimicrobial strength or solubility. In labs, reactions like etherification or esterification frequently pop up in process notes, as manufacturers chase improved compatibility with emulsifiers, binders, or active pharmaceutical ingredients. Reductive or oxidative treatments alter its reactivity or decompose pollutants in wastewater. These modification techniques demonstrate chemistry’s knack for evolutionary, not just revolutionary, change.
Synonyms & Product Names
4-Chloro-m-Cresol slips behind plenty of masks in trade and research. Synonyms run from PCMC—a common abbreviation in pharmacopeia literature—to 4-chloro-3-methylphenol, reflecting its ring structure. Other names show up in regulatory filings or safety sheets, so nobody should make assumptions by glancing at just one label. Awareness of these aliases becomes critical, especially in cross-border supply chains, regulatory submissions, or global R&D projects.
Safety & Operational Standards
Chemistry can serve or harm depending on respect for protocols. In the workplace, I’ve watched colleagues rigorously suit up before handling 4-Chloro-m-Cresol: gloves, goggles, and proper ventilation are non-negotiable. The compound’s potential to irritate skin, eyes, or respiratory tracts draws a line—ignore it, and the consequences come quick. Guidance from global standards—like OSHA or REACH—implements specific exposure limits and disposal routes. Stories circulate about mishaps from lax storage, teaching everyone that routine diligence outlasts luck. The environmental angle demands equal seriousness; effluent treatment, regular spill drills, and staff refreshers anchor best practices.
Application Area
Look down the list of products in most hospitals, veterinary clinics, or food processing plants, and 4-Chloro-m-Cresol claims a spot. Its key appeal remains broad-spectrum antimicrobial punch, so you’ll see it in disinfectant sprays, ointments, handwashes, and antifungal creams. Agriculture leans on it, too—wood treatments and preservatives draw on its ability to push back rot and insect damage. Even the paint and polymer sector calls on it, preventing microbial spoilage before the end user ever opens a can. Every sector weighs cost, regulations, and mounting consumer concern for greener substances, so the chemical’s inclusion reflects both its power and persistent need for oversight.
Research & Development
Over the past decade, I’ve watched research teams pivot toward safer and more effective substitutes, but 4-Chloro-m-Cresol’s niche persists. Journals report on tweaks that boost its selectivity, reduce allergenic risk, or bolster performance against emerging pathogens. Some teams have mapped out detailed interaction studies at the cellular level, hoping to pull apart the fine line between stopping microbes and irritating human tissue. Analytical chemists craft better test methods for residue monitoring in food and water, showing how modern labs blend classic organic chemistry with high-tech detection to track even trace quantities.
Toxicity Research
The discussion around safety never stops at basic labeling. Chronic exposure brings questions beyond the immediate burns or sniffles. Animal studies point to liver and kidney effects in high doses, and researchers debate possible links to allergic reactions or long-term organ stress. In the field, mishaps sometimes lead to spills or workplace accidental exposures—a sobering reminder of the difference between theory and practice. Ongoing toxicology studies play a real-world role, guiding workplace limits and regulatory updates. The dialogue between toxicologists and industry gets loud anytime new data emerges, which keeps the safety conversation anchored firmly in today’s reality rather than resting on older data.
Future Prospects
Anyone paying attention knows environmental health and human safety now shape product futures. Demand for antimicrobial performance surges during disease outbreaks, and yet regulatory scrutiny only tightens. Green chemistry initiatives gather steam, seeking less toxic alternatives without giving up microbial control. Start-ups pitch bio-based phenolic alternatives, and some big players invest in new synthesis methods to reduce waste or energy consumption. Even so, 4-Chloro-m-Cresol’s proven record keeps it on the roster, especially in legacy products. Expect ongoing debate between innovation, risk reduction, and commercial inertia as science and policy together chart its next chapters.
What are the main uses of 4-Chloro-M-Cresol?
The Backbone of Disinfectants
Ask anyone who’s ever scrubbed their hands raw after a long shift in a hospital or clinic, and you’ll hear the same thing—protection starts with a good disinfectant. 4-Chloro-M-Cresol plays a big part here. Most non-alcohol hand washes, wound cleansers, and surface sprays in clinics rely on its strong antimicrobial character. This is evident in its inclusion in critical wound care products and soaps around the world. With drug-resistant germs on the rise, proven agents like this are relied on by people on the front lines—not because regulations say so, but because lives are at stake. The World Health Organization keeps 4-Chloro-M-Cresol on its list of recommended biocidal substances in healthcare, recognizing its value against a wide spectrum of bacteria and fungal cells.
Guarding Everyday Products
Take a walk down any drugstore aisle—many deodorants, lotions, and even tattoo aftercare formulas owe their shelf-stability to this preservative. Mold, yeast, and other spoilage risks pop up fast in water-based creams. With a low dose of 4-Chloro-M-Cresol, formulas avoid contamination and stay safe for skin. Anyone who has opened a jar of cream that smelled a little “off” knows how important this is. Safe, effective preservation avoids the need for harsh alternatives or endless refrigeration.
Use in the Paint and Leather Trades
Fungus and mildew thrive in more places than we think—paints, adhesives, leather goods. Builders rely on treated paints, and shoemakers trust their tanned hides. Left unchecked, organic growth can ruin products and create health hazards indoors. Paints for damp cellars, leathers for heavy-duty boots, and construction adhesives commonly carry this preservative for that exact reason. Practicality drives the decision here: it’s not about brand image, but about preventing ruined materials and unhappy buyers.
Pharmaceutical and Veterinary Solutions
A trip to any rural clinic or farm will show 4-Chloro-M-Cresol in bottles labeled for animal wound rinse and topical antiseptics. Its low toxicity makes it a reliable choice for treating livestock and pets where careful dosage is crucial. Injections and creams often contain it to prevent spoilage and cross-contamination. For pharmacists and veterinarians who know how quickly bacteria spread in warm, moist conditions, this small molecule means peace of mind—and healthier animals.
Addressing Public Concerns and Moving Forward
Widespread use always comes with questions. Folks raise concerns about overuse, allergies, and possible impacts on the environment. Research from the European Chemicals Agency suggests safe levels for skincare and cleaning, and studies keep pushing for greener alternatives or safer blends. Product safety teams and consumers both win when information gets shared clearly and new rules reflect the latest science. Some cosmetics now feature plant-based preservatives, but 4-Chloro-M-Cresol remains a reliable mainstay for many applications. Companies can invest in improved packaging and formulation science to reduce waste and keep formulations safe, while regulators and scientists stay on top of long-term impact research.
Keeping an Eye on Safety
In my own work with hospital supply chains, I saw firsthand the careful tracking required for ingredients like this—nobody wants accidental overdosing or accidental mixing with incompatible chemicals. Proper training, labeling, and tracking keep staff safe and products effective. End users play a role too—following directions and choosing trusted brands.
Is 4-Chloro-M-Cresol hazardous or toxic?
Understanding What We’re Using
4-Chloro-M-Cresol isn’t something most people talk about over coffee. Still, folks who work in labs, hospitals, or factories might know it as a preservative or disinfectant. Its job often involves killing microbes in products or on surfaces. Big deal, right? It helps keep things from growing mold or bacteria. Yet not everything toxic smells bad or feels strange on your hands. Plenty of chemicals, including this one, can fly under the radar until trouble pops up.
Health Hazards Don’t Always Knock on the Door
In my years around medical supplies and disinfectants, labels like “caution” or “irritant” rarely tell the whole story. The thing is, 4-Chloro-M-Cresol calls for respect. Breathing in its dust or vapors can leave lungs irritated and eyes watery. Skin doesn’t always get off easy, either—redness and rashes can follow close behind. Safety data isn’t buried in fine print to get ignored, it’s there because accidental splashes can lead to chemical burns. Mix the stuff in a small, poorly ventilated space and you’ll quickly learn how strong it can get.
The Long-Term Picture Wins Every Time
Repeated exposure doesn’t act friendly on the body. Some workers have told me about headaches, loss of focus, and chest tightness after regular contact. A big concern: evidence shows possible links to organ damage, especially in the liver and kidneys, with high or frequent exposure. Animal studies point out risks that shouldn’t get swept aside—those trials guide workplace safety for a reason. In a world where corners sometimes get cut, people need to know the stakes.
Environment Pays the Price Too
Wash 4-Chloro-M-Cresol down the drain and it doesn’t disappear. Wastewater treatment plants can only handle so much. Most of these molecules stick around and build up in rivers, hurting fish and aquatic insects. Tests have shown 4-Chloro-M-Cresol disrupts life cycles, even at lower concentrations. This chemical acts like a double-edged sword—great for knocking out bacteria, tough on everything else that shares the water. Whenever a cleanup crew finds high levels downstream from factories, nobody acts surprised. There’s a lesson here about keeping both people and ecosystems in mind with every product we use.
No Silver Bullet—Stay Smart and Safe
Experience tells me that the right gear and training always beat improvising. Gloves, goggles, and working near a proper ventilator matter. Ventilation shouldn’t be treated like an afterthought. Regular air checks in work areas can catch leaks or spills before they cause harm. Plenty forget about skin contact until a rash appears—over-the-counter remedies won't fix the root issue. Responsible employers do more than hand out pamphlets. They invest in good labeling, real training, and proper disposal programs to keep contamination in check.
Striking a Balance
My time around chemicals shaped the way I think about everyday risks. Many things get their value from proper use and respect for limits. 4-Chloro-M-Cresol belongs in this category. It plays a useful role, but it isn’t worth risking health or the environment just to save a few bucks or minutes. Selecting safer alternatives or asking tough questions before buying or using powerful chemicals can make a real difference. Protecting workers, water, and wildlife depends on moving past good intentions to practical habits.
What is the recommended storage condition for 4-Chloro-M-Cresol?
Chemicals Like 4-Chloro-M-Cresol Matter More Than Labels Suggest
Picture a small amber glass bottle with a faded label in a high school science lab. It’s easy to walk by a chemical like 4-Chloro-M-Cresol and think all bottles can stay anywhere as long as the lid is tight. In reality, every detail about storage helps keep people, products, and the environment safe.
Why the Right Storage Changes Outcomes
4-Chloro-M-Cresol often shows up in disinfectants and as a preservative. It isn’t the most dramatic chemical, but the risks are real if corners get cut. Vapors can irritate airways. Skin contact brings rashes or burns. Tossing a container onto any old shelf puts janitors, lab techs, or anyone handling it in a tough spot. Even worse, improper storage sometimes causes unplanned reactions or dangerous fumes.
A well-ventilated, cool, and dry space always beats just shoving things into a random cabinet. Moisture and heat don’t play well with this chemical. A humid storeroom or strong sunlight can slowly degrade the material or make it sweat out hazardous vapors. If you’ve ever opened an old storeroom on a summer day, you know how stuffiness can slap you in the face—the same stuffy air can create problems for chemicals like this.
Secure the Space: Access and Labels Matter
There’s a temptation to leave lesser-known chemicals on an open shelf. Good practice means locking them away, keeping curious hands off. A locked cabinet with clear labels helps the team spot exactly what they’re holding so that no one grabs the wrong item in a hurry. Labels should share both the chemical’s name and quick hazard warnings, not just fine print.
Prevention Trumps Cleanup: Responding Before Trouble Starts
Once I visited an old university storeroom where nearly everything coexisted on sagging shelves. Corrosives sat next to organics. Spills happened so often that you could see the stains and rings from years past. Each time the janitor cleaned, there was a risk of chemical mixing. Segregated storage by hazard class makes sure no one has to gamble that nothing will react.
4-Chloro-M-Cresol, for example, should not sit near strong oxidizers or acids. A cabinet clearly labeled for “organics” with separation from “corrosives” or “oxidizers” makes it easier to spot mistakes before they happen, even when new staff or students join.
Personal Protection—Not Just a Rule, but a Habit
Whenever anyone pulls a bottle from the shelf, gloves and eye protection come first. Even after years working with chemicals, I still double-check my gloves for holes. Accidents come out of nowhere, usually because someone rushed or assumed nothing bad would happen “just this once.” Habits built around caution pay off every single day.
Solutions Make the Difference
Relying on written policies isn’t enough unless people understand why those rules exist. Routine walkthroughs, inventory checks, and training help all staff remember how much every step matters. Engineers, custodians, and teachers benefit from practice drills with spill kits and eye-wash stations. Regular training gives everyone confidence to handle unexpected situations without panic.
It pays off to think of chemical storage as more than compliance—a good storage setup protects health, saves costs, and shows respect for the people managing chemicals daily.
What is the chemical formula or structure of 4-Chloro-M-Cresol?
Understanding 4-Chloro-M-Cresol at a Glance
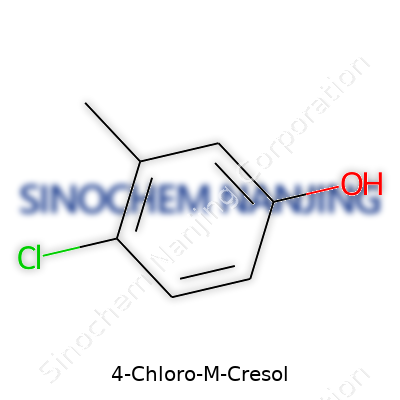
4-Chloro-M-Cresol shows up in places many people might not expect, especially in cleaning and disinfectant products. Its chemical formula is C7H7ClO. Anyone with a basic science background might remember that this kind of notation tells us a lot: the molecule has a benzene ring, a methyl group (–CH3), a chlorine atom, and a hydroxyl group (–OH). For this chemical, the “4-chloro” bit points to where the chlorine attaches on the ring, while “m-cresol” points to the position of the methyl and hydroxyl groups.
Drawing it out, you get a benzene ring. The methyl group and hydroxyl group sit next to each other at what chemists call the "meta" position; the chlorine atom sticks to the spot across from the methyl, on the fourth carbon from the starting point. So, the chemical structure isn’t just trivia for science professionals — it tells us how the molecule acts in real life.
The Real-World Impact of Structure
Shape affects function. 4-Chloro-M-Cresol gets used for disinfection because that exact pattern of atoms disrupts cell walls and proteins in bacteria and fungi. Specific bonds and placements lend this molecule the “bite” that takes down germs.
That structure also influences its water solubility, which means it can mix into cleaning solutions and work well on dozens of surfaces. Sometimes I’ve seen this ingredient in hand soaps in hospitals, where strong and dependable disinfection goes a long way. Scientists chose this molecule over similar ones like o-cresol or p-cresol because the chlorine at the fourth position boosts antimicrobial action without causing too much irritation for human skin. Chemists have studied comparative results and found this arrangement hits that sweet spot between power and safety.
Balancing Benefits and Health Risks
No chemical comes without trade-offs. With 4-Chloro-M-Cresol, people do need to watch for skin sensitization and possible allergic reactions. I’ve talked with health professionals who say patch-testing cuts down on problems, especially for folks working daily with concentrated solutions. Survey work from peer-reviewed journals backs this up: cases of skin reaction pop up mostly with high exposures, not everyday handwashing.
Regulators in Europe and the United States have set strict limits for concentrations in cosmetics and cleaners. The chemical provides fast-acting, broad-spectrum coverage against bacteria, but its effect on the environment and long-term exposure make manufacturers rethink dosages. Analytical chemists run studies in real-world settings to track breakdown in wastewater, keeping pollution in check.
What Does the Future Hold?
Fresh questions arise as new strains of bacteria evolve resistance to disinfectants. Industry partners team up with university research labs to study these trends, often searching for combinations of chemicals that lower the need for intense concentrations of any single ingredient.
Education does a lot here, especially for workers who handle cleaning agents every day. Clear labeling and easy-access safety training lower risks on the job. From chatting with folks in cleaning services to reading published safety reviews, it’s clear more awareness leads to fewer problems across the board.
So, knowing about the chemical formula and the story behind each atom in 4-Chloro-M-Cresol pays off. It connects experts, regulators, and everyone who puts trust in disinfectants.
Are there any specific handling precautions for 4-Chloro-M-Cresol?
Understanding the Risks
4-Chloro-M-Cresol shows up in a lot of industrial and laboratory work. It serves a clear purpose: effective as a disinfectant and preservative. Take a quick look at any safety sheet, and the warnings around this chemical pop right out. Once, during college, I ignored the burning sensation from a single drop brushing my skin. Lesson learned. The compound stings. Skin contact can trigger itching, redness, or much worse if you’re careless over time. Inhaling dust or vapors can irritate the nose and throat, sometimes sparking trouble in the lungs. Swallowing by accident—rare in a lab, sure, but not impossible—could bring serious consequences.
Why Gloves Make a Difference
Nitrile gloves go a long way. For those of us getting hands-on, skipping gloves feels tempting if the job seems small. Don’t do it. Friends working in chemical plants have all had stories about coworkers who let their guard down, often thinking rinsing their hands afterwards will keep them safe. Chemicals soak in fast. Regular gloves stand up better than latex or vinyl. One colleague kept a stash by the bench and insisted everybody pair up and check each other during messy projects. Taken together, habitual glove use shrinks risk and builds habits you rely on.
Eye and Face Safety
A splash in the eye can cause instant regret. Lab goggles—real ones, not those flimsy glasses from hardware stores—stop that disaster. In one public health survey, chemical-related eye injuries often came from inconsistent eye protection. Use goggles that fit snugly and cover the sides. For larger-scale work or open containers, add a face shield. Keeping the face barrier in place means you never scramble to the eye wash, praying there’s no lasting damage.
Ventilation Beats Tough Fumes
Sometimes, the sharp odor of a substance signals trouble. Working with 4-Chloro-M-Cresol in a confined space, you notice the smell quickly builds up. Poorly ventilated areas raise your risk. Fume hoods draw vapors away from your breathing space. An experienced lab tech once saved us from a rough day by spotting a blocked hood vent—small checks like this matter. Fact: steady airflow can decide whether you spend the evening headache-free or not.
Storage Habits That Prevent Accidents
People just starting out often tuck bottles wherever there’s space. That just increases danger. Store containers tightly sealed, well labeled, and out of direct sunlight. Mixing with incompatible substances sparks nasty reactions. Keeping everything organized prevents a rushed mix-up. I once saw a colleague use painter’s tape to label hazardous bottles even if the writing was faded—those habits slow mistakes and keep everyone safer.
Cleaning Up and Responding to Spills
No matter how careful everyone feels, spills still happen. Small ones can be wiped up fast, but only with gloves and disposable materials. For larger spills, use absorbent pads made for chemicals and toss them as hazardous waste. Know the location of eyewash stations and emergency showers. Run regular drills so nobody hesitates if something goes wrong.
Training Makes It Routine
All the safety gear and signs help, but real safety sticks after steady training. New team members need reminders and walkthroughs. Senior staff can model good habits, showing that none of these precautions ought to slide. Building a routine around safety with 4-Chloro-M-Cresol—just like strapping in a seatbelt—pays off every day.
| Names | |
| Preferred IUPAC name | 4-chloro-3-methylphenol |
| Other names |
4-Chloro-3-methylphenol PCMC para-Chloro-meta-cresol 4-Chloro-3-cresol p-Chloro-m-cresol |
| Pronunciation | /ˈklɔːrəʊ ɛm ˈkriːsɒl/ |
| Identifiers | |
| CAS Number | 59-50-7 |
| Beilstein Reference | 1207744 |
| ChEBI | CHEBI:34041 |
| ChEMBL | CHEMBL153907 |
| ChemSpider | 5937 |
| DrugBank | DB14039 |
| ECHA InfoCard | 03f085de-df87-4a43-958e-911d0049a51b |
| EC Number | 204-385-8 |
| Gmelin Reference | 83969 |
| KEGG | C06535 |
| MeSH | D002756 |
| PubChem CID | 6970 |
| RTECS number | GO8575000 |
| UNII | R7GV7H988D |
| UN number | UN3077 |
| CompTox Dashboard (EPA) | DTXSID3023143 |
| Properties | |
| Chemical formula | C7H7ClO |
| Molar mass | 142.57 g/mol |
| Appearance | White to yellowish crystals or powder |
| Odor | Phenolic |
| Density | 1.18 g/cm³ |
| Solubility in water | Slightly soluble in water |
| log P | 2.78 |
| Vapor pressure | 0.004 hPa (25 °C) |
| Acidity (pKa) | 9.43 |
| Basicity (pKb) | 7.05 |
| Magnetic susceptibility (χ) | -79.7×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.558 |
| Viscosity | Viscous liquid |
| Dipole moment | 2.71 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 118.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -159.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4458.7 kJ/mol |
| Pharmacology | |
| ATC code | D08AE10 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H315, H318, H335 |
| Precautionary statements | P261, P264, P271, P272, P273, P280, P302+P352, P305+P351+P338, P310, P321, P332+P313, P333+P313, P337+P313, P362+P364, P391, P501 |
| NFPA 704 (fire diamond) | 3-2-0 |
| Flash point | 113 °C |
| Autoignition temperature | 120°C |
| Lethal dose or concentration | LD50 oral rat 1830 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1830 mg/kg (oral, rat) |
| NIOSH | B017 |
| PEL (Permissible) | PEL: 5 mg/m3 |
| REL (Recommended) | 1 mg/m³ |
| Related compounds | |
| Related compounds |
3-Chlorophenol 4-Chlorophenol 4-Chloro-2-methylphenol m-Cresol Phenol |

