4-Chloro-2-Nitrotoluene: Examining Its Path and Place in Modern Chemistry
Historical Development
4-Chloro-2-nitrotoluene, an aromatic compound with both chlorine and nitro substituents on a toluene backbone, found its footing in organic chemistry a long time ago. This substance crept into textbooks as chemists explored routes for more functional dyes and pharmaceuticals. Research in the late 19th and early 20th centuries started mapping out nitrated aromatic compounds and their chlorinated cousins, mostly in pursuit of industrialization. The boom in synthetic dyes and agrochemicals put compounds like this one in the spotlight. Chemical factories grew on the backbone of such substituted toluenes, promising cheaper production of useful intermediates and the launchpad for even more complex molecules. Chemists in industrial labs, well before the digital revolution, spent countless hours developing purification and preparation protocols to tame both the usefulness and notorious reactivity of aromatic nitro compounds.
Product Overview
When scanning the world of nitrotoluenes, this compound—4-chloro-2-nitrotoluene—always stands out because of its particular substitution pattern. A methyl group on the benzene ring, chlorine clinging to the fourth carbon, a nitro group in the ortho position. What this mix produces is an electrophile with two strong functional handles: the nitro group, bulking up the electron sink, and the reactive chlorine. These features attract synthetic chemists, who often chase complexity from such straightforward starting blocks. Specialty chemical makers continue to supply it as an intermediate for the manufacture of pigments, agrochemicals, and pharmaceuticals, but its appeal doesn’t stop there. It helps researchers push boundaries in what can be achieved with controlled reactivity.
Physical & Chemical Properties
This isn’t a glamorous molecule, but it’s reliable. In the bottle, it appears as a yellowish solid or crystalline powder, stubbornly hydrophobic and only slightly soluble in water. That solubility profile suits several organic solvents such as methanol or dichloromethane. A boiling point high enough to allow careful heating, a melting point that reveals how tightly molecules pack together—the physical data all points toward a robust, easily handled solid at room temperature. The chemical nature, weighted with both nitro and chloro groups, makes it a reliable electrophile for substitution reactions but stable enough that it can survive typical handling, storage, and transport. It’s not prone to explosion under normal conditions, making it safer than some of its close relatives. Still, the nitro group marks it as one to treat with respect. In my experience, nitro-aromatics rarely forgive those who get too casual.
Technical Specifications & Labeling
Every batch moving through commerce carries clear labeling, usually in line with national and international systems. From the moment the drums arrive in the warehouse, the hazard pictograms and chemical identifiers remind everyone involved of what they’re dealing with: toxicity risks, strong odors, bright color—these all communicate something important. The specifications for purity are strict once this makes its way into the supply chains of dye factories or pharmaceutical plants. It doesn’t just affect product yield, but also the downstream safety. Analytical testing, usually through GC or HPLC, gives buyers and users the confidence that every shipment meets expectations. I’ve seen operations grind to a halt because a drum missed a technical specification or labeling step. In a workplace that runs on tight schedules, those labels become as vital as the chemical itself.
Preparation Method
In a laboratory or a plant, chemists rely on clear, time-proven methods to synthesize 4-chloro-2-nitrotoluene. The most common route involves chlorination of 2-nitrotoluene or the nitration of 4-chlorotoluene with nitric and sulfuric acid. Temperature control demands careful attention, especially to avoid over-chlorination or runaway nitration, both of which would trash the product and, much worse, create dangerous side products. I remember the stories from older chemists who described clouds of acrid gas and vigorous reactions when procedures went sideways. These days, continuous monitoring and automated dosing lines prevent most incidents, but there’s no room for complacency. Washing, crystallization, and purification by recrystallization or distillation refine the product till it meets lab or industrial standards.
Chemical Reactions & Modifications
Chemists value 4-chloro-2-nitrotoluene largely because it’s a launch point for more tailored molecules. Nucleophilic substitutions are the first stop—removing or replacing that chlorine with an amine, methoxy, or a host of other groups. The nitro group often turns into an aniline derivative via reduction, opening the door to azo dyes, pharmaceuticals, or fungicides. Even the methyl group acts as a synthetically useful position for oxidation into benzaldehyde or benzoic acid derivatives. Each group can be manipulated selectively, letting synthetic chemists proceed stepwise to more advanced molecular targets. I’ve seen academic groups design multi-step syntheses starting from compounds just like this one, putting to the test not just the limits of reactivity but also the patience and thoroughness of their research teams.
Synonyms & Product Names
This chemical goes by several aliases, which can trip up even seasoned researchers. Names like 1-Methyl-4-chloro-2-nitrobenzene, 4-chloro-o-nitrotoluene, and sometimes alternative numbers like “para-chloro-ortho-nitrotoluene” show up in literature or on chemical suppliers’ lists. CAS numbers offer some relief, acting as a universal translator. In the market, packaging may feature any of these, especially if imported or sourced from different suppliers, so cross-checking product names avoids mishaps. In a field where minor changes in substitution matter a lot, getting the right synonym linked to the correct structure is far more than a clerical issue.
Safety & Operational Standards
Nobody who works around nitroaromatics takes lab safety for granted, and 4-chloro-2-nitrotoluene proves why. The nitro group brings toxicity concerns, particularly for long-term exposure or mishandling. Goggle-wearing and glove use are standard—more for the health of chemists than to follow some abstract rule. Fume hoods remain a staple, as this compound can volatilize or produce off-gassing at higher temperatures. Any spill or residue needs immediate cleanup since nitrotoluenes don’t mix well with an open cut or extended skin contact. Safety data sheets form the backbone of handling, but personal vigilance rounds out the best protection. Chemical waste disposal, always a headache, draws extra scrutiny to keep contaminated material far from regular landfill pathways. This chemical, like many intermediates from older branches of industry, acts as a reminder that the past didn’t always dwell on environmental aftershocks. Current regulations put the emphasis where it belongs: protecting workers and surroundings alike.
Application Area
Dye and pigment manufacturing still circle back to 4-chloro-2-nitrotoluene, especially in producing azo dyes and colorants for textiles, plastics, and inks. Agrochemical producers use it to start off synthetic routes toward fungicides, herbicides, and even growth regulators. The pharmaceutical industry sometimes taps it for intermediates in the synthesis of drugs that need a chlorinated aromatic ring or a precursor to nitroanilines. I’ve talked to process chemists who appreciated how it unlocked quick assembly of core scaffolds for medicinal chemistry programs, moving from small-scale synthesis to production scale-up with relative ease. Research institutions lean on it as a reliable vector for new reaction methodology studies, and sometimes even in environmental chemistry to investigate the breakdown of persistent aromatic residues.
Research & Development
Over the decades, synthetic chemists and process engineers dove deep into developing new catalysts and milder reaction conditions for transforming compounds like 4-chloro-2-nitrotoluene. The focus rests on efficiency, safety, and minimizing environmental burden. Industry labs devote plenty of time to optimizing reactions, aiming for less hazardous reagents and lower energy use. I’ve seen innovative attempts to use green solvents and biocatalysis for both the preparation and modification of this compound. Computational chemistry, for its part, assists in predicting reaction outcomes and minimizing the need for large-scale trial and error. R&D teams examine both the upstream (how to make it more safely) and downstream (how to convert it into valuable products with less waste). Industry conferences showcase advances from process engineers who relentlessly shave costs, reduce waste, and push selectivity to new heights.
Toxicity Research
Experience with 4-chloro-2-nitrotoluene—and most nitrotoluenes for that matter—teaches caution. Repeated studies show toxic impacts, particularly on blood and liver function in mammalian hosts. Workforce health studies from older chemical plants revealed cases of chronic exposure causing anemia and related health problems. Researchers dig into both acute and chronic effects, not just to comply with regulations but to assess real health risks tied to exposure. Animal models show dose-dependent toxicity, with special concern for extended low-dose exposure typical in industrial workplaces. Agencies list clear precautions, but ongoing work still looks for gaps, such as the cumulative impact of long-term, low-level exposure and breakdown products in the environment. Safety innovation in this area moves beyond compliance: new air monitoring systems, wearable detectors, and better training drills build safer working environments.
Future Prospects
Environmental regulation keeps driving change, challenging chemists and manufacturers to design better, less hazardous production pathways and cleanup strategies. The legacy of aromatic nitro compounds reminds everyone how the past shaped today’s safety culture and environmental standards. The next decade will bring a mix of green chemistry, smarter waste management, and new uses discovered as chemists map out more selective reactions and safer derivatives. Automation and digital monitoring will tighten controls and squeeze out process inefficiencies. As research groups and companies develop alternatives to traditional routes, 4-chloro-2-nitrotoluene may see new uses or be shaped into even more valuable downstream products. Every step forward requires balancing the practical needs of manufacturing with the imperatives of worker safety and ecosystem health.
What are the main uses of 4-Chloro-2-Nitrotoluene?
Real Role in Everyday Goods
4-Chloro-2-nitrotoluene sounds like something found in a locked cabinet at the back of a school chemistry lab, but this compound stands behind plenty of objects folks use every day. In the chemical world, it’s not the flashiest name, yet its influence stretches across industries. At its core, it gets produced to offer a reliable intermediate for creating other molecules—often the kind that drive the world of color, protection, and crop care.
Dyes That Stick, Fabrics That Last
Textile workers and dyers depend on color that doesn’t fade after a wash or two. Several types of azo dyes owe their resilience to compounds built using 4-chloro-2-nitrotoluene. In practice, the chemistry allows the creation of colors that don’t bleed or run, keeping jeans blue and shirt prints crisp. Without this kind of stability, fast fashion would be even more fleeting, and textile waste would grow. Studies from industry groups—like those cited in the Journal of Textile Science—point out that the backbone furnished by intermediates like this toluene derivative helps keep color fastness and resistance in wide production.
Protecting Crops, Feeding the World
Growers look for pesticides and herbicides that protect crops without flavoring food or harming the soil. Several well-known agrochemicals start their journey from the building block offered by 4-chloro-2-nitrotoluene. Chemists use this compound for synthesizing products that deter insects, fungi, or weeds. Farm productivity has gone up sharply over the decades, partly because of these targeted crop protectants. Of course, safe handling matters. Companies and suppliers need to rely on proper warnings and oversight so misuse doesn’t spoil ecosystems. Evidence from food safety agencies around the world supports tighter oversight of ingredients and intermediates like this one to make sure they don’t wind up where they don’t belong.
Medicines and the Search for Cures
The pharmaceutical sector often uses 4-chloro-2-nitrotoluene as a starting point to create bigger, more complex molecules for medicines. Antibiotics, painkillers, and specialty drugs sometimes owe their backbone to this base compound. Chemists like having stable, tested intermediates around because they provide a known quality in each batch—critical for drugs where impurities spell danger for real people. Over the years, regulatory checks and third-party labs—like those endorsed by the FDA or EMA—establish strict standards on everything from initial chemicals to the end pill on the shelf.
Unlocking Better Chemistry, Responsibly
It’s tempting to look at a single chemical and just see hazard, but tools like 4-chloro-2-nitrotoluene unlock creative solutions to wide-ranging problems: feeding growing populations, curing sickness, and brightening textiles with stable color. This only works when production, storage, and transport line up with responsible practices. For everyone from the lab technician to the logistics manager, following global safety guidelines and investing in regular audits cuts risk. There’s always room for safer substitutes too—green chemistry researchers search for less toxic alternatives that still deliver on the job.
Promoting Awareness and Safety
Education about chemicals like 4-chloro-2-nitrotoluene can help balance their positive contributions with safety needs. For workers around these compounds, training and protective equipment matter. For communities nearby, clear risk communication clears up questions before they become problems. This kind of openness builds trust and keeps the focus where it belongs: using chemistry’s benefits for real improvements in daily life, without sidestepping the responsibility that always rides along.
What are the safety precautions for handling 4-Chloro-2-Nitrotoluene?
Why Care Matters in the Lab
Many chemicals get handled every day, but 4-Chloro-2-Nitrotoluene stands out because exposure can cause serious trouble for skin, eyes, lungs, and even long-term health. The yellow crystal dust looks pretty harmless to someone new in the lab, yet breathing it in or getting it on your skin can do real damage. People want to get through a shift safely and avoid health issues down the line, so good safety habits become second nature.
Personal Protective Equipment Saves Skin and Lungs
No one should ever handle this substance without quality gloves. Nitrile or neoprene gloves block most of the contact. Chemical splash goggles don’t leave much to chance, protecting eyes from burns or irritation. For the face and upper chest, lab coats and splash-proof aprons offer solid coverage. Respirators are not just for smoky fires—they play a big role if fumes or dust clouds appear, since breathing in particles carries health risks that linger for years. A decent lab ensures fume hoods work well, catching particles at the source so air stays clean.
Good Habits Make All the Difference
Before starting, check the safety data sheet. It’s the manual you never skip. People forget to wash their hands enough, especially after pulling off gloves, but chemical dust clings to skin and moves fast. Eating or drinking anywhere near work areas brings chemical residue right to your mouth. I’ve seen a few chemists get sick simply from snacks near their bench, even though they thought they were careful. Simple habits, like taping up cuts and never rubbing your eyes in the lab, go a long way to staying healthy.
Proper Storage Limits the Danger
Storing 4-Chloro-2-Nitrotoluene requires attention. Keep it in sealed, labeled glass bottles. Clear, durable labels warn anyone nearby what’s inside, in case of spills or emergencies. Lock storage away from heat, direct sunlight, and strong acids or bases. If a container cracks or leaks, the chemical can get loose, react dangerously, and contaminate other supplies. Emergency showers and eye wash stations stand ready to handle spills or accidental splashes—no one expects accidents, yet being prepared turns a scary moment into a safe recovery.
Cleanup and Waste: The Often-Ignored Step
Cleaning up after each experiment closes the safety loop. Use special pads—not paper towels—to pick up any spilled powder or droplets. Dispose of rags and pads in chemical waste bins, never the regular trash. Waste handling rules keep the next person safe and stop pollution before it starts. Even small mistakes, like missing a sprinkle of dust near the balance or dripping liquid down a bench leg, can create problems later. Working in a team that values these steps adds an extra shield.
Protect People, Not Just Protocols
Staying safe in chemical work doesn’t run on checklists alone. It takes real care, teamwork, and learning from those who’ve handled risky materials for years. Respect for 4-Chloro-2-Nitrotoluene—and for each other—keeps serious accidents in stories rather than reality. Simple routines, smart gear, and shared responsibility make every experiment a safer experience.
What is the chemical formula and structure of 4-Chloro-2-Nitrotoluene?
Digging Into the Formula
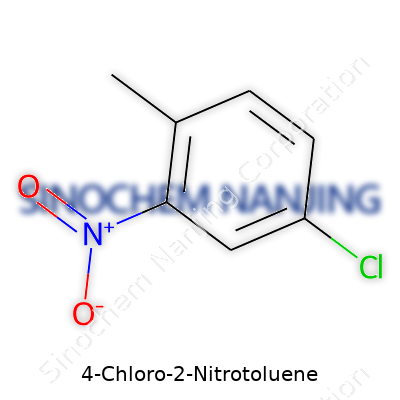
Chemistry doesn’t give a free pass to anyone trying to cut corners. Every atom in a molecule matters. With 4-chloro-2-nitrotoluene, every piece fits into a specific spot, and the formula C7H6ClNO2 reflects that arrangement. At its core, this compound traces back to the benzene ring, but it’s not just plain old benzene. An extra jolt comes from three specific groups— a methyl group, a nitro group, and a chlorine atom — each attached to different points on the ring.
Breaking Down the Structure
I picture molecules as neighborhoods, where each group moves in and changes the way the street feels. In 4-chloro-2-nitrotoluene, start counting at the methyl (toluene) group as house number 1. Nitrogen’s nitro group lands at position 2, right next door. Chlorine takes the fourth spot, which pulls on the neighborhood in its own electrifying way. This leaves a very distinct structure: benzene’s backbone with a –CH3 on carbon 1, –NO2 on carbon 2, and –Cl on carbon 4.
Drawing this out explains much of its reactivity in real chemical work. Steric hindrance, electronic effects, and reactivity all grow out of how those groups get along (or argue) with each other across the carbon ring. Experienced chemists spot how these positions steer reactions, sometimes blocking one side, sometimes making a corner very much open for business. A nitro’s electron-withdrawing power will usually slow down attacks from electrophiles, while the methyl group’s electron-donating ability gives a little boost right around its side.
Why Placement Matters in Chemistry
It’s a lesson I learned in the lab the hard way: swapping positions even slightly on a ring like this changes more than textbook numbers. The arrangement of chlorine and nitro decides what reactions this molecule can take. For instance, trying to do a nucleophilic substitution? The presence of nitro right next to the methyl group throws a wrench into many plans. Explaining away mistakes in synthesis often comes down to what group is where.
In research labs that handle aromatic nitro compounds, safety jumps to the front of everyone’s mind. 4-chloro-2-nitrotoluene sits at the intersection of many industrial needs, especially in dye and pharmaceutical manufacturing. Labs keep strict protocols because nitro compounds can turn dangerous—fire hazards, toxic byproducts, and stubborn residues are part of everyday life for chemists. I’ve seen teams review every step, sometimes two or three times, before scaling up production.
Finding Answers in the Details
Chemists benefit from hard facts. 4-chloro-2-nitrotoluene dissolves in most organic solvents but avoids water. Its melting point floats around 54–56 °C, giving a clue for handling and storage. Structure determines each of these properties. The same holds for larger questions: how it reacts, how it should be stored, the steps that keep workers safe.
For those working with chemicals on a regular basis, structure isn’t just an academic point. Misunderstanding the arrangement or formula leads to wasted money, lost time, and sometimes health risks. If you don’t see every atom’s job, you miss important clues in how to use these compounds most effectively. It’s not about memorizing — it’s about applying that structure to real decisions, every day, under real-world pressures.
Building Safer, Smarter Labs
Every day, chemists and health experts work together to refine protocols. Improving ventilation, personal protective gear, and reaction monitoring makes dealing with compounds like 4-chloro-2-nitrotoluene safer. Updated reference materials help new team members grasp why placement of functional groups guides lab choices. Education never ends in this field, since even small changes can set off major reactions.
Staying informed and working together with colleagues — that’s the most reliable formula for progress in chemistry and manufacturing. The old saying holds true: know your chemicals inside and out. The shape, the structure, and the practical risks all add up to smarter, safer work for everyone involved.
How should 4-Chloro-2-Nitrotoluene be stored?
Why Safe Storage Really Matters
4-Chloro-2-Nitrotoluene packs some punch as a chemical. Plenty of people in manufacturing and research know it as a compound with some sharp teeth, causing trouble for skin and lungs, and occasionally sparking fires if it finds the wrong neighbor. Years ago, I walked into a storage area that smelled a little sweet, a little harsh, and found a loosely capped drum leaking yellow powder. Nobody wants to find themselves in that situation. Once anyone handles chemicals like this, memory serves as a loud reminder—smart storage can mean the difference between a regular workday and a major problem.
Keep Cool, Keep Dry, Stay Isolated
This compound prefers the quiet life: cool, dry spaces keep it stable. Heat invites breakdown and spills kick up fumes no one wants near their eyes or lungs. The best setup heads underground, in ventilated rooms that avoid high temperatures. Old warehouses and hot attics give this stuff too much rope. Picture a labeled drum, sealed tight, nowhere near a steam pipe or sunlit window.
Humidity also brings headaches. Moisture reacts with plenty of chemicals, and strong condensation drips can cause small pools. I once saw cardboard drums go soft from damp air, risking leaks. Swap out anything cardboard for thick plastic or rust-proof metal. The peace of mind comes from double-checking those lids after every use and writing logging entries for every access. Messy records and loose seals tempt fate.
The Enemies: Fire, Acid, and Open Containers
Besides warmth and water, strong acids and oxidizers have to stay away. Storing 4-Chloro-2-Nitrotoluene near bleach or peroxide cheers up the risk for a chemical reaction. Workers tell stories about fires starting in labs because someone mixed up shelves. Always post storage rules right on the door—one glance for anyone who enters. Shelves should stay neat, with wide gaps between different chemicals, no exceptions for "just for a day."
Fire safety grabs top priority. Choose spaces with sprinkler systems, no open flames, and plenty of room between drums. If regulations allow, tuck a Class D fire extinguisher nearby, and teach folks how to use it. Community fire departments appreciate lists of stored chemicals, so prepare copies for first responders. Neighbors down the block also sleep easier when they know hazardous compounds won’t spill into nearby drains.
Solutions Rooted in Training and Equipment
Good personal protective equipment beats bravery. Gloves, goggles, and a strong-smelling sense of caution work together. Every lab worker and warehouse manager should run through emergency drills every season. I've seen folks laugh off drills until the one day alarms go off for real—the calm practice pays off.
Modern inventory systems track each container, catching slow leaks or shipments that sit too long. Chemical suppliers offer special storage cabinets designed for specific hazards; use them. Having spill kits, absorbents, and dedicated disposal bins right on-site turns minor mishaps into quick fixes, not evacuations. Even a simple habit, like washing hands after each use, helps hold back the risk.
Looking Out for the Next Shift
Storing 4-Chloro-2-Nitrotoluene means caring about the folks on the next shift, the cleaning crew, and the community that lives nearby. Right storage habits earn trust and keep accidents rare. I’ve found that once management commits to safety and backs it up with equipment and training, workers pick up good habits and problems shrink. Chemical hazards don’t always make the news, but smart, intentional storage keeps them in the corner, right where they belong.
What are the potential health hazards of 4-Chloro-2-Nitrotoluene exposure?
The Trouble with Chemical Exposures
Stories about toxic chemicals often feel distant, but for workers in chemical plants, the risks have real consequences. 4-Chloro-2-nitrotoluene, used to make dyes, pigments, and drugs, comes with very real health hazards. Many people don’t think about what goes into the dyes that color our clothes or the chemicals that find their way into finished products. These chemicals rarely make news unless there’s a spill or an accident, but the hazards remain, day after day.
Health Risks That Come with the Job
Exposure to 4-chloro-2-nitrotoluene can cause irritation to skin, eyes, and lungs. Breathing in the dust or vapor may bring on headaches and dizziness. In the lab, I remember handling similar nitro compounds—everyone wore gloves and lab coats, the smell sharp and unmistakable. Short-term discomfort is a warning sign. Reports link repeated exposure to damage to the liver and kidneys, organs that work around the clock to clear toxins. Chemicals like this target the blood, lowering its oxygen-carrying power and bringing on symptoms such as weakness, blue skin, and fatigue. Anyone working near vats or in poorly ventilated spaces faces higher risks. Chronic exposure tends to work quietly, building up damage over months or years.
Why It Matters Beyond the Factory
Safety isn’t only a concern for workers. The communities around chemical plants carry their own risks. Contaminated water or soil, drifting dusts during transport—these hazards reach beyond the plant’s fences. Incidents remind us that accidents don’t respect property lines. News stories from industrial towns tell of families with similar health complaints, from nosebleeds to unexplained fatigue. In places where environmental monitoring slips, companies sometimes ignore rules meant to keep people safe. The legacy left in soil and water sticks around, as these compounds break down slowly in the environment.
Gaps in Regulation and Knowledge
Even though regulatory bodies set limits and issue safety data, chemical exposure often slips under the radar in smaller or less regulated industries. Not every facility has strict ventilation or hands-on safety training. Some workers don’t get regular medical checks. I’ve met people who never received details about what they handled daily, making it tough to push for better safety practices. Risk increases where people don’t know what’s in the air or on their gloves.
What Can Be Done?
Preventing exposure to 4-chloro-2-nitrotoluene calls for personal and shared responsibility. Proper protective gear, good ventilation, and safe storage form the front lines of protection. Training needs grit—real stories combined with facts hammer home the importance of compliance. In my experience, teams work better when everyone understands the "why" behind safety rules. Government inspections play a needed role. Speed bumps come from budget cuts and loopholes, but public pressure can move things forward. Community groups and health advocates help hold companies accountable when shortcuts tempt decision-makers.
Tracking exposure and reporting symptoms make a difference for early detection. Simple blood tests catch red flags before the worst damage sets in. Regular reviews ensure procedures grow with new science. At its core, health protection depends on respect—for the people, for the truth in good data, and for every lesson learned from injury or close call.
| Names | |
| Preferred IUPAC name | 1-Chloro-4-methyl-2-nitrobenzene |
| Other names |
2-Methyl-4-chloronitrobenzene 4-Chloro-o-nitrotoluene 2-Nitro-4-chlorotoluene 4-Chloro-2-nitromethylbenzene NSC 401882 |
| Pronunciation | /ˈfɔːr-klɔːr-oʊ-tuː-naiˈtrɒl-tɒl.juːˌiːn/ |
| Identifiers | |
| CAS Number | 89-59-8 |
| Beilstein Reference | 1209452 |
| ChEBI | CHEBI:84287 |
| ChEMBL | CHEMBL154451 |
| ChemSpider | 14928 |
| DrugBank | DB16080 |
| ECHA InfoCard | 03d3d3b7-0ad8-4f42-89ba-4de906e2f9d8 |
| EC Number | 210-819-4 |
| Gmelin Reference | 7780 |
| KEGG | C14346 |
| MeSH | D002828 |
| PubChem CID | 79247 |
| RTECS number | XZ3150000 |
| UNII | Q25P44V6S1 |
| UN number | UN3442 |
| Properties | |
| Chemical formula | C7H6ClNO2 |
| Molar mass | 157.56 g/mol |
| Appearance | Light yellow crystalline solid |
| Odor | Aromatic odor |
| Density | 1.38 g/cm³ |
| Solubility in water | Insoluble |
| log P | 2.9 |
| Vapor pressure | 0.016 mmHg (25°C) |
| Acidity (pKa) | pKa = 12.9 |
| Basicity (pKb) | pKb = 13.7 |
| Magnetic susceptibility (χ) | -50.3·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.5950 |
| Viscosity | 6.2 cP (20°C) |
| Dipole moment | 3.65 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 218.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 129.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3811 kJ/mol |
| Pharmacology | |
| ATC code | |
| Hazards | |
| GHS labelling | GHS02, GHS07, GHS09 |
| Pictograms | GHS02, GHS07, GHS09 |
| Signal word | Warning |
| Hazard statements | H302 + H315 + H319 + H332 + H335 |
| Precautionary statements | P261, P264, P273, P280, P301+P312, P305+P351+P338, P337+P313, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-1 |
| Flash point | Flash point: 108°C |
| Autoignition temperature | 531°C |
| Explosive limits | Lower: 1%, Upper: 7% |
| Lethal dose or concentration | Lethal Dose (LD50) oral/rat: 1230 mg/kg |
| LD50 (median dose) | LD50 (median dose): 2100 mg/kg (oral, rat) |
| NIOSH | CN5775000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 4-Chloro-2-Nitrotoluene: 1 mg/m³ |
| REL (Recommended) | REL (Recommended): 2 ppm (10 mg/m3) |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
2-Nitrotoluene 4-Chloro-2-nitroaniline 4-Chlorotoluene 2-Nitrobenzyl chloride 2-Chloronitrobenzene |

