4-Bromoanisole: From Early Chemistry Labs to Modern Innovation
Historical Development
4-Bromoanisole began showing up in academic literature over a century ago during an era when organic chemistry was still carving out its niche. Early researchers recognized its value as a building block in complex molecule synthesis, especially in pharmaceutical discovery. Insights from the post-war chemical boom led to rapid adoption across industry, as aromatic bromides like 4-Bromoanisole offered unique reactivity that wasn't easily matched by other compounds. Chemical manufacturers soon began refining its production, and by the late 20th century, nearly every university research lab and fine chemicals company had bottles of this compound. Small innovations in purification and reaction techniques, like improved distillation and crystallization, helped bring 4-Bromoanisole into the toolkit of bench chemists, making it a reliable participant in cross-coupling reactions and library synthesis projects.
Product Overview
Today, 4-Bromoanisole shows up as a colorless to pale yellow liquid, usually with a faint, sweet aromatic odor. Labs rely on its stable, predictable properties as a substrate in transition metal-catalyzed coupling reactions, as well as in other organic transformations. Its reputation rests on consistency: bottles of this compound arrive with detailed labeling, sometimes showing batch-specific spectra, because downstream customers often use it to make high-value drug intermediates or agricultural molecules. Quality suppliers test every shipment for both chemical purity and isomer content. Clients include pharmaceutical researchers, materials scientists, and fine chemical processors who prioritize bench-ready inputs.
Physical & Chemical Properties
With a molecular formula of C7H7BrO and a molecular weight close to 187, 4-Bromoanisole holds a boiling point around 230°C and a melting point near -1°C. Its density hovers around 1.5 g/cm³, so it feels significantly heavier than water. The compound dissolves easily in most organic solvents—diethyl ether, dichloromethane, acetone—but barely budges in water, reinforcing its organic preference. Chemists note its low vapor pressure and moderate toxicity, making it manageable but not something to treat lightly. The structure places the methoxy group on one end and the bromine directly opposite, giving it both electron-donating and electron-withdrawing functionality—details that drive much of its value in synthetic chemistry.
Technical Specifications & Labeling
Lab suppliers usually guarantee a minimum purity of 97%, with leading brands pushing purity above 99%. Impurities often show up as unreacted anisole or isomeric bromination products, both picked up in routine HPLC or GC analysis. Batch documentation now includes full spectra—1H NMR, mass spec, sometimes even IR—especially for regulated industries. Labels highlight proper storage guidelines: keep away from heat or sunlight, prevent moisture exposure, and seal bottles tightly. Regulatory data covers hazard pictograms and GHS-compliant handling warnings. Customers expect real clarity here because the slightest contamination can derail sensitive reactions.
Preparation Method
Production most often employs electrophilic aromatic substitution, starting from anisole. Classic approaches use bromine or other brominating agents under carefully controlled conditions to direct the reaction to the para position, yielding the desired 4-bromo product with high selectivity. Advances came with milder brominating agents—NBS (N-bromosuccinimide) came into play thanks to its improved safety and easier waste management. The isolation process calls for distillation or extraction steps followed by purification either through column chromatography or recrystallization. Each manufacturing facility tweaks protocols based on scale, local environmental laws, and available raw materials, but the core strategy hasn't shifted much in decades.
Chemical Reactions & Modifications
The aryl bromide group sits ready for a range of cross-coupling reactions. Suzuki-Miyaura and Buchwald-Hartwig processes rely heavily on compounds like 4-Bromoanisole to introduce complexity into molecules, forging carbon–carbon or carbon–nitrogen bonds under palladium catalysis. Many researchers appreciate how the methoxy group moderates reactivity, making it possible to manipulate selectivity and yields. Reduction, nucleophilic substitution, and oxidative transformations unlock further diversity—each method expands the utility of the starting material, letting chemists fine-tune products for pharmaceutical screening or materials testing. In hands-on research, students often use 4-Bromoanisole as a starting point to master techniques in aromatic substitution, seeing firsthand the practical advantages of a well-behaved substrate.
Synonyms & Product Names
You'll spot this compound in lab catalogs under several names, including 4-methoxybromobenzene, p-bromoanisole, and para-bromoanisole. CAS number 104-92-7 helps cut through ambiguity, especially in international trade. Despite a handful of subtle spelling differences across catalogs or regions, chemicals with this structure and registry code all point to the same product. Researchers sometimes prefer to call it by its more systematic title, 1-bromo-4-methoxybenzene, to avoid confusion with similar aromatic compounds.
Safety & Operational Standards
Safety data calls for gloves, goggles, and a well-ventilated hood, as skin contact and inhalation both carry toxic potential. Labels highlight the risks: irritation, moderate systemic toxicity, and environmental concerns if spills happen. In university and industrial settings, chemical hygiene plans require training on spill containment, storage, and waste management before anyone handles 4-Bromoanisole. Modern suppliers ship it in secure, leak-proof containers designed for chemical compatibility. Disposal procedures follow strict regulatory tracks, as improper incineration or sewer disposal can push brominated organics into the environment, where they resist breakdown.
Application Area
Research labs across pharmaceuticals, agrochemicals, and material science departments turn to 4-Bromoanisole for its solid record as a synthetic intermediate. Medicinal chemists build on its core to create new molecules for screening, often looking to tweak the aromatic ring for improved activity or metabolic stability. Crop science teams experiment with altered versions to pursue next-generation pesticides or plant hormones. Polymer scientists leverage its cross-coupling potential for specialty resins and liquid-crystalline materials. Fine chemical producers often scale up tried-and-true syntheses that start from this compound, cementing its place in the modern organic chemistry toolkit.
Research & Development
Contemporary research often circles around new catalytic methods for installing or replacing the bromine atom with alternative functional groups. Scientists investigate metal-catalyzed borylations, aminations, and carbon–carbon couplings to expand the reach of 4-Bromoanisole, seeking ways to make complex molecules in fewer steps. Efforts also target greener bromination methods to reduce byproducts and environmental impact. Computational chemists model different reaction pathways using density functional theory, picking apart electronic effects from the methoxy and bromine groups to better predict how new transformations might unfold. Intellectual property filings track inventive chemistries, with patents staking out ground around application-specific process tweaks that save cost, improve yield, or enhance safety.
Toxicity Research
Animal and cell culture studies show moderate acute toxicity, with metabolic processing leading mostly to harmless metabolites, although rare cases of sensitization or liver impact have cropped up in rodent models at high doses. Environmental scientists monitor for hazards in aquatic systems, since brominated aromatics resist rapid breakdown and have the potential to bioaccumulate. Data collected across several decades provides a clear map for safe handling—threshold limit values, permissible exposure levels, and cleanup standards make up the backbone of regulatory cautions. Strict site controls help prevent accidental release or chronic exposure in chemical plants and research facilities.
Future Prospects
Demand for 4-Bromoanisole isn’t fading; new pharmaceutical and advanced materials development keep pushing for clever, efficient access to functionalized aromatics. Startups and established firms both chase catalytic breakthroughs that use this compound as a launchpad to more complex scaffolds. Regulatory scrutiny around hazardous brominated compounds nudges the market toward greener syntheses and improved waste management practices. Automated synthesis platforms include 4-Bromoanisole among their favorite starting points, as its reactivity and reliability factor into both proof-of-concept and scale-up campaigns. Chemistry students will keep reaching for this bottle, as its straightforward structure and textbook reactivity offer practical lessons that outlast trends.
What is 4-Bromoanisole used for?
Chemistry’s Hidden Backbone
If you’ve walked through a research lab, odds are you’ve never heard anyone wishing for a bottle of 4-Bromoanisole at the top of their lungs. Still, behind the scenes, this pale yellow liquid shows up all over synthetic chemistry. I’ve seen fellow chemists reach for it mostly in the hunt for efficient routes to complex molecules. Its structure puts a bromine atom next to an anisole group—this combination gives it a kind of versatility that rarely gets the spotlight.
Chemical Building Block
I always remember my old professor calling compounds like this the “Lego bricks” of organic chemistry. In my own experience, 4-Bromoanisole gets used as a starting material, especially in making larger molecules through something called cross-coupling reactions. Scientists love those reactions—Suzuki, Stille, or Heck—since they let you glue together fragments in ways nature can’t manage alone.
Pharmaceutical companies often rely on these building blocks because drug discovery churns through so many possible molecular combinations. Without compounds like 4-Bromoanisole, the hit rate when searching for new medicines would drop. Researchers tested its pathways in the past decades when antihistamines, antifungals, and even possible cancer treatments got their first syntheses.
Big in Flavor and Fragrance
Sometimes it’s easy to forget how much basic chemistry ends up in the products we use every day. Perfume makers and flavor chemists don’t talk much about raw inputs, but I’ve toured a few facilities that quietly order tons of chemicals like this one. It gets used to make other aromatic chemicals—think vanilla and anisole derivatives—that wind up in vanilla ice cream, cola, or luxury perfumes.
The reason comes down to selectivity. If a process uses 4-Bromoanisole, chemists can attach different groups exactly where they want, making sure the smell or taste is just right. Mistakes with these starting ingredients mean the end product feels off, either too synthetic or just unpleasant.
Challenges in Handling
Working with 4-Bromoanisole isn’t a walk in the park. It’s not particularly toxic compared to some lab chemicals, but its volatility and persistence can turn tidy benches into sticky messes, and breathing it over hours isn’t great for your health. That’s why labs with regular use install strong local ventilation and carefully track storage. Mistakes feel small in the moment, but over months or years, you see the impact—smells lingering, health complaints rising, and occasionally violations of regulations.
Companies have pushed for safer handling protocols. Years ago, I spoke with a safety manager who insisted on sealed transfer systems when moving aromatic bromides around. The investment made a difference—lower insurance claims, better working conditions, fewer headaches for everyone.
Solutions and Future Outlook
The demand for these types of intermediates won’t slow, since modern drugs and consumer goods depend on rapid chemical synthesis. Looking ahead, the industry keeps pressing for “greener” routes, finding ways to recycle or reuse bromo-based reagents, and using automation to keep exposure to a minimum. My hope is more academic labs adopt these methods early, so the transition gets easier and safer for everyone down the road.
What is the molecular formula of 4-Bromoanisole?
Zeroing In on the Details
People often discuss molecules like blueprints, but behind each one sits a purpose. For 4-Bromoanisole, its structure gives chemists clues about what it can do and how it fits into larger projects, especially in pharmaceuticals and materials science. I've come across this compound in a teaching lab, where understanding its whole formula gave us an edge on how to safely use it and what it could make in future syntheses.
What’s Inside Matters
Every atom in 4-Bromoanisole tells a story. Its molecular formula is C7H7BrO. That means it carries seven carbons, seven hydrogens, one bromine, and one oxygen. This combination lends itself to a family of aromatic compounds. The “anisole” part springs from its methoxy group (–OCH3), and the bromine lands at the para spot—the fourth carbon away from the methoxy group—resulting in interesting reactivity.
Real Practicality, Not Just Theory
Working with 4-Bromoanisole isn’t abstract. It shows up as a starting material for building complex molecules. In process chemistry, for example, the para-substituted ring system lets researchers direct reactions to create new pharmaceuticals or chemical intermediates that wouldn’t be possible with a blank benzene ring. Having bromine hanging off the ring means you get opportunities for palladium-catalyzed cross-coupling reactions—Suzuki or Heck reactions build bigger molecules with precision. That’s not just textbook talk: these reactions power the development of everything from new medicines to advanced plastics and electronic materials.
Safe and Responsible Use
Handling chemicals like 4-Bromoanisole calls for respect and attention. The number and position of atoms in its formula also influence safety. The bromine atom gives it more heft and changes how it interacts with both people and the environment. Researchers and manufacturers have to keep this in mind, especially when disposing of waste. Brominated compounds bring extra regulations, as they may persist in nature. Lab safety guidelines, personal protective gear, and careful disposal limit exposure and environmental impact. Tools like Chemwatch or SDS databases support smart, responsible practice, helping keep people safe while handling and disposing of materials properly.
Teaching and Learning Value
Teachers use the structure and formula of 4-Bromoanisole to walk new students through the basics of organic chemistry nomenclature, reactivity, and functional groups. Learning from real molecules like this bridges the gap between abstract concepts and hands-on understanding. A student figuring out why the formula reads C7H7BrO will nail down important ideas about hydrocarbon rings, functional groups, and halogenation. This foundation opens doors to more advanced synthetic projects later on.
Looking Forward
Working with molecules at the atomic level isn’t just about memorizing letters and numbers. It’s about seeing where that knowledge takes us—safer labs, cleaner processes, smarter products. Industry and academia keep putting effort into greener alternatives, recycling brominated waste, and developing safer analogues. These steps aim to keep 4-Bromoanisole useful, but also manageable, in labs and beyond.
Is 4-Bromoanisole hazardous or toxic?
What Is 4-Bromoanisole?
4-Bromoanisole looks like another chemical in a long list cooked up in labs for industry and research. Chemists use it as an intermediate, especially in pharmaceuticals and fine chemicals. But safety never takes a back seat, either in a factory or a college workspace, so it makes sense folks ask questions about its hazard profile.
Common Encounters and What Science Says
I’ve mixed batches of 4-Bromoanisole in grad school, so memories of those heavy vapor smells stick with me. The Safety Data Sheet gets clear on this compound: its fumes cause irritation and overexposure risks headache or nausea. Swallowing isn’t smart—expect a sore stomach at least, and potentially more serious problems for children or pets. Skin doesn’t love it, either. The stuff doesn’t dissolve well in water, so spills can linger unless handled quickly.
According to the European Chemicals Agency, tests show low acute toxicity, so small splashes likely won’t put someone in grave danger. But “low acute toxicity” doesn’t equal harmless. Over time, repeated exposure builds up risk. Inhalation can aggravate asthma or underlying lung issues, and chronic handling without gloves or goggles leaves skin dry and red.
Recognizing the Limits of Research
Peer-reviewed studies point out data gaps. Long-term animal testing hasn’t been done on the same scale as for bigger-name chemicals like benzene. No strong proof ties 4-Bromoanisole to cancer, birth defects, or big reproductive harm, but the absence of evidence rarely puts it on the “safe” list. Many imported chemicals arrive with SDS documents not always vetted by Western agencies, so it pays to check labeling and lean on trusted sources.
Practical Safety Tips From Experience
Decades in chemistry labs left me pretty strict: I always use gloves, goggles, and a serious fan or hood. If a bottle drops, sand or kitty litter absorbs it better than a rag. Don’t pour leftovers down the drain—municipal water plants won’t catch it and small amounts slip out to rivers and streams. Some countries regulate industrial use tighter but a lot of hobby chemists skip formal training and face greater risk because they cut corners.
I’ve seen friends lose feeling in their hands from repeated chemical handling. That’s why undergrad labs echo with warnings, posters, and tales of what happens when people ignore proper clean-up or trade PPE for short-term ease. Cancer risk or not, chemical burns, dizzy spells, and lifelong skin trouble are real enough.
What Can Be Done Differently?
Better ventilation in small labs keeps air fresher. Hood filters catch volatile chemicals like 4-Bromoanisole before a whiff becomes a headache. Substituting less volatile compounds helps where possible. Manufacturers must update SDS sheets, and supervisors should walk through storage rules monthly instead of relying on annual reviews. Schools can put samples or demo kits in locked drawers, tracking sign-outs to discourage casual misuse.
On the policy level, more research is needed. Testing health effects, gathering user reports, and enforcing disposal standards make a difference. Until regulatory bodies fill the research blanks, it comes down to vigilance on the ground.
Looking Ahead
Working with chemicals like 4-Bromoanisole brings responsibility. Good habits and respect for safety processes lower risk—no matter how small the danger appears on paper. Until the science catches up, treating every unfamiliar bottle with caution isn’t just wise—it’s non-negotiable.
What are the storage conditions for 4-Bromoanisole?
Why Storage Conditions Impact Chemical Quality
4-Bromoanisole stands out in many organic chemistry projects. It brings flexibility to labs, often finding its way into pharmaceutical and agrochemical work. But quality doesn’t stay put on its own. After years of sifting through supply chain issues, I’ve found sloppy storage invites trouble nobody wants—degraded product, lost time, unnecessary risk.
What Experience Shows About Reliable Storage
Every chemical has its quirks, 4-Bromoanisole included. No fancy tricks, just basic respect for stability. Start with a tightly closed container. I once saw a batch left in a poorly sealed jar, and the subtle ether smell soaked the whole room, raising safety eyebrows right away. Avoiding that comes down to proper seals and good habits.
Temperature shifts throw these molecules for a loop. Leaving 4-Bromoanisole in a spot where the sun blasts through a window never ends well. Persistent warmth invites slow breakdown. A cool, steady spot—think a standard laboratory chemical storage cabinet away from heat sources—works best. Consistency does more than fancy technology in this environment.
Shielding from Moisture and Air
A careless mistake in my early years taught me how moisture creeps in, even in seemingly clean spaces. 4-Bromoanisole doesn’t like water. Humidity, even in small amounts, kicks chemical changes into motion. For this reason, keep it away from open windows and steamy lab corners. Desiccators or silica gel packets in the storage cabinet buy time and save headaches.
Oxygen in the air nudges the compound to degrade slowly. Every open-lid moment counts. Strive to limit how often the container is opened. This attention to detail adds up, especially when working with sensitive intermediates or planning multiple steps ahead in synthesis.
Safe Handling and the Human Side
No story about chemical storage skips over safety. Gloves, goggles, lab coats—they’re not just for show. Spills happen quickly, even with experienced hands. Ready access to a fume hood makes cleanup and transfer less stressful. I learned from watching a careless moment escalate that small spills still expose coworkers to unexpected risks.
Labeling counts as much as the actual storage itself. Clear, non-fading labels prevent nasty surprises during busy moments. I’ve grabbed the wrong bottle before—luckily, I caught it before using it for a reaction. Every lab should stick to a system for checking dates, rotating stock, and logging what comes in and out.
What Industry Best Practice Looks Like
Regulators and trusted chemical suppliers agree: store 4-Bromoanisole in a well-ventilated, dry, cool spot, away from incompatible substances. No magic, just discipline. It pays to avoid storing it near strong oxidizers. At one job, sloppy neighbors shelved chemicals too close together, and the resulting cross-contamination nearly ruined a week of work. Segregation matters.
Insurance companies and auditors track incidents caused by poor storage. They report fewer accidents in facilities that take routine checks seriously. That matters for business reputation as much as personal health.
Real Solutions from Real Practice
Routine trumps complexity. Build daily habits—double-check seals, log movements, inspect for leaks. Keep moisture at bay, avoid temperature swings. Good storage isn’t about spending more money on fancy gear; it’s about using what’s there with focus and care. In my experience, well-run labs look and feel boringly tidy. That’s where projects succeed and nobody gets hurt.
What is the purity and grade of 4-Bromoanisole offered?
Why Purity Matters in Everyday Chemistry
Purity stays on everyone’s mind in the lab. Not because people are picky, but because it shapes results and safety. 4-Bromoanisole, with its clean, almost sweet-smelling aura, ends up in a surprising number of products—in dyes, perfumes, and pharmaceuticals. Its role isn’t limited to labs that cook up molecules for fun. Poor purity turns simple reactions into messes. Anyone who has spent their afternoon troubleshooting a failed experiment knows this headache well. An impurity that seems innocent on paper will sometimes throw a whole process off track. Small numbers can mean big problems.
Understanding Grades Offered on the Market
Not all 4-Bromoanisole out there arrives in the same shape. Chemical suppliers set out different grades. You’ll see phrases like ‘analytical grade’, ‘reagent grade’, and ‘technical grade’. Analytical grade gives peace of mind—it's tested to meet the strictest purity standards, usually upwards of 99%. For research that pushes boundaries or demands certainty, this grade is what chemists grab from the shelf. Reagent grade lands in the same neighborhood, though slight traces of extra stuff may slip by. Technical grade sometimes dips below 98%, aiming to serve large-scale industry, where traces of unrelated compounds pose less risk for the final output.
Personal Experience with Supplier Choices
On busy lab days, I’ve reached for both analytical and reagent jars. Cheap options tempt, but sometimes you notice a faint color, a stubborn residue, or the structure not matching expectations in NMR or GC-MS. These reminders show why purity isn’t just a line in a data sheet, but a safeguard for endpoints. Reactivity, shelf life, and even basic safety depend on what else sneaks into a “pure” bottle. One friend of mine ran a cost-saving synthesis using a lower-grade 4-Bromoanisole, only to spend twice as long cleaning up an unruly byproduct. It left a clear impression: cutting corners brings trouble down the road.
Purity Assurance: Direct from the Source
Suppliers know that transparency about purity isn’t negotiable. Good companies lay out chromatograms, NMR traces, and Certificates of Analysis, not just glossy marketing claims. These documents spell out precise contents—sometimes down to a few parts per million—so everyone knows what sits in the flask. I've learned to ask for documentation up front, and to view any reluctance as a warning sign.
Quality Control in the Real World
While the process from factory to warehouse involves many hands, careful control at each step makes a real difference. Community reviews help steer choices—some suppliers get a reputation for consistency, others for letting subpar batches slip through. From experience, even small lapses in drying, sealing, or shipping can pull purity down, even when the batch started fine.
Improving the Standard
Doing better isn’t just about stricter checks by the supplier; users can play a role by reporting quality problems and sharing independent testing. Collaboration between user and supplier keeps everyone honest and drives best practices. Whenever doubts surface about a chemical’s grade, a quick check with a TLC plate or a scan on a spectrometer clears them up and avoids lost time.
Finding the Balance
Choosing the right purity means weighing budget against a project’s need for reliability. Most researchers walk this tightrope by talking with colleagues, reviewing shared data, and sometimes running their own checks. At the end of the day, purity isn’t only about numbers on a label. It’s the difference between progress and wasted effort.
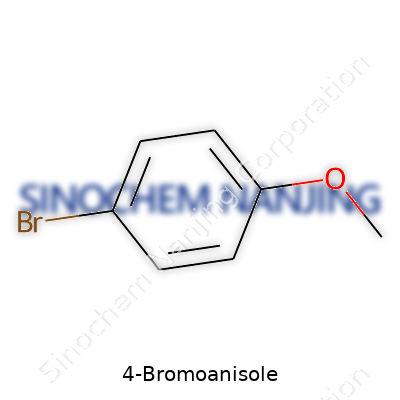
| Names | |
| Preferred IUPAC name | 1-Bromo-4-methoxybenzene |
| Other names |
p-Bromoanisole 1-Bromo-4-methoxybenzene 4-Methoxybromobenzene |
| Pronunciation | /ˌfɔːrˌbroʊˈmæ.nəˌsoʊl/ |
| Identifiers | |
| CAS Number | 104-92-7 |
| 3D model (JSmol) | ``` 4-Bromoanisole JSmol 3D Model String: C1=CC(=C(C=C1Br)OC) ``` |
| Beilstein Reference | 1208734 |
| ChEBI | CHEBI:74243 |
| ChEMBL | CHEMBL141111 |
| ChemSpider | 16702 |
| DrugBank | DB04261 |
| ECHA InfoCard | 03b3c209-570a-4d21-88f8-72fd4c5c4e48 |
| EC Number | 211-213-6 |
| Gmelin Reference | 795957 |
| KEGG | C05601 |
| MeSH | D000402 |
| PubChem CID | 12053 |
| RTECS number | BR4025000 |
| UNII | 32N-1PPGH |
| UN number | UN2667 |
| CompTox Dashboard (EPA) | 4-Bromoanisole (EPA CompTox Dashboard): **DTXSID1021487** |
| Properties | |
| Chemical formula | C7H7BrO |
| Molar mass | 187.04 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Density | 1.495 g/mL at 25 °C (lit.) |
| Solubility in water | Insoluble |
| log P | 2.9 |
| Vapor pressure | 0.3 mmHg (20 °C) |
| Acidity (pKa) | 18.3 |
| Magnetic susceptibility (χ) | -69.0 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.553 |
| Viscosity | 0.828 cP (20°C) |
| Dipole moment | 3.90 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 354.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -56.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4257.7 kJ/mol |
| Pharmacology | |
| ATC code | Not assigned |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P210, P261, P280, P305+P351+P338, P370+P378 |
| Flash point | Flash point: 113°C |
| Autoignition temperature | 90 °C |
| Lethal dose or concentration | LD50 (oral, rat): >5000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 >2000 mg/kg |
| PEL (Permissible) | Not established |
| REL (Recommended) | 36 months |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Anisole Bromobenzene 4-Iodoanisole 4-Chloroanisole 4-Fluoroanisole Phenol Para-bromoanisole |

