4-Aminophenylarsonic Acid: A Closer Look at Its Past, Present, and Future
Historical Development
Tracing the history of 4-Aminophenylarsonic acid goes hand-in-hand with the rise of organoarsenic compounds in the early 20th century. Around the time when industry started pivoting from artisanal chemistry to industrial-scale synthesis, advances in dye and pharmaceutical chemistry put this molecule on the map. Scientists recognized early on that arsenic’s unique chemistry brought both great promise and significant risk. The work of Paul Ehrlich and the subsequent boom in arsenical drugs led researchers to develop various derivatives, and 4-aminophenylarsonic acid, known for housing both aromatic amine and arsenic groups, rose out of this chemical exploration. It played a role in the agricultural sector as an intermediate in feed additives, though over time concerns about environmental and health impacts took center stage.
Product Overview
What stands out about 4-Aminophenylarsonic acid is its blend of physical robustness and chemical reactivity. Researchers and industrial chemists saw value in a molecule that could be reliably synthesized, stored, and applied without extreme precautions on standard laboratory benches. The crystalline solid form makes it relatively straightforward to weigh and manipulate, but the presence of the arsenic atom always demands a higher level of care compared with less hazardous aromatic amines. The chemical structure grants it a role not just in pure chemistry, but in supporting innovation in crop science and veterinary medicine, opening doors for further research into both beneficial uses and risk management.
Physical & Chemical Properties
This compound generally appears as a solid under standard ambient conditions. It dissolves moderately well in polar solvents, echoing trends seen with similar aromatic acids. The amine and arsenic groups invite targeted functionalization, enabling transformations that create derivatives with different physical behaviors and biological activity. In my work, comparing the color and handling characteristics always brought home the contrast between ostensibly similar compounds — a little tweak in functional group twists everything from melting point to solubility. The molecular formula combines carbon, hydrogen, nitrogen, oxygen, and arsenic, with the full formula confirming both its weight and its diverse range of possible reactions. Importantly, the strong polarity and the acidic hydrogen align with observed reactivity in laboratory settings.
Technical Specifications & Labeling
Working with chemicals like 4-Aminophenylarsonic acid always means paying close attention to labeling. Regulatory markers such as CAS numbers and purity grades help prevent confusion and accidents, especially in larger labs where so many compounds compete for shelf space. If a label fails to communicate the hazards and requirements, the door opens for risky handling. Specs such as assay (the percentage of pure compound), water content, and presence of common impurities matter for research scale-up and industrial applications alike. Knowing exactly what’s in the bottle shapes both experimental consistency and downstream product quality.
Preparation Method
I’ve seen firsthand that the route to 4-Aminophenylarsonic acid often starts with aniline derivatives, either from benzene nitration-reduction sequences or via catalytic amination methods. The challenge comes with getting the arsenic atom into the structure without veering off into side reactions that foul up the product mix or release toxic byproducts. Early methods used inorganic arsenic acid under controlled pH and temperature, leading to stepwise condensation and oxidation. Advances in purification — such as selective crystallization and chromatography — let chemists drive up yields and reproducibility. The key step involves linking the phenylamine with an arsenic acid moiety, and a misstep can quickly escalate into wasted time and dangerous waste.
Chemical Reactions & Modifications
4-Aminophenylarsonic acid opens the door to a wide range of chemical modifications, especially on the amine and arsenic positions. Standard reactions include diazotization and coupling for introduction of additional groups, as well as oxidation or reduction around the arsenic atom for transformations to pentavalent or trivalent arsenicals. Out in the field or a production lab, that flexibility means researchers can tune bioactivity, solubility, or toxicity, depending on the requirements set by the next application. Cross-coupling and acylation reactions lend themselves to crafting new analogs, revealing both the benefit and risk lurking in the molecule’s core. Each step brings new regulatory and safety challenges, especially if scale-up leads to dramatic increases in waste generation.
Synonyms & Product Names
Over the decades, the compound picked up a suitcase of different names. Chemists often refer to it as p-aminophenylarsonic acid or 4-aminobenzenearsonic acid, echoing its structure. Commercially, names like Atoxyl and arsanilic acid have shown up in patents, academic papers, and industrial brochures. This naming mash-up risks confusion for newcomers, especially those less familiar with the quirks of organic nomenclature. From a safety perspective, clarity on names can make all the difference in an emergency or cross-lab collaboration.
Safety & Operational Standards
Day-to-day use of arsenic-based compounds never gets routine in a responsible lab. Even with solid training, it stays front of mind that any exposure to arsenic brings both acute and chronic health risks. Proper personal protective equipment — gloves, lab coats, goggles, and, when dust or aerosols might escape, respirators — belongs on every user. Exhaust hoods, locked storage, and spill kits rank as non-negotiable in both research and industrial contexts. Regulatory bodies at national and global levels impose strict rules for transport, use, and disposal, making compliance a continual part of the workflow. This is not the place for shortcuts: single lapses can cause lifelong harm or expensive remediation.
Application Area
In decades past, agriculture turned to compounds like 4-Aminophenylarsonic acid as animal feed additives, intended to promote growth and fend off certain diseases in livestock. At the time, the thinking focused more on production gains than on the long-term build-up of arsenic in soil and water. The shift in public perception around food safety and environmental health pushed regulators to clamp down on these uses. Outside agriculture, the molecule has drawn interest as a building block for pharmaceutical research and some industrial processes, though ongoing concerns about exposure and waste limit new commercial entrants. That said, the expertise developed in manufacturing and handling this compound carries over into adjacent fields, where lessons learned about containment and hazard reduction steer modern risk management protocols.
Research & Development
University labs and a few industrial innovators keep asking whether there’s a way to harness the beneficial aspects of 4-Aminophenylarsonic acid chemistry without unleashing its worst environmental or health consequences. The molecule still inspires curiosity for its reactivity and potential as a synthetic intermediate, pushing research into greener synthesis techniques or targeted functionalization methods. I’ve seen collaborative studies where chemists, environmental scientists, and toxicologists team up to draw clearer boundaries between practical applications and unacceptable risk. Technology changes and new regulatory frameworks mean that some promising areas, like site-specific drug delivery or advanced material coatings, remain under review until risks can be better quantified or mitigated.
Toxicity Research
No discussion of a compound like this stays complete without facing the realities of toxicity. Arsenic compounds, generally, sit among the most studied and stringently watched substances in the modern world. Studies across multiple species point to acute effects — nausea, vomiting, nervous system disruption — as well as chronic impacts like organ damage and increased cancer risk. The way arsenic embeds in food chains and persists in soils means each new use carries unresolved questions about downstream exposure. Even with modern analytic tools, measuring low-level contamination and predicting biological behavior keeps toxicologists and regulators on their toes. Direct workplace exposure still demands constant monitoring and rigorous adherence to recommended exposure limits, as set by agencies like OSHA and the World Health Organization.
Future Prospects
Looking ahead, the role of 4-Aminophenylarsonic acid in large-scale industry remains clouded by the legacy of its own toxicity. The march of green chemistry pushes researchers toward replacement compounds where possible, and to more sophisticated risk-benefit calculations where no substitute yet exists. Modern advances in catalysis and molecular design might one day unlock ways to neutralize or sequester arsenic byproducts at the point of use, making current risks more manageable. In my experience, interdisciplinary efforts carry the most promise — every innovation in detection, containment, or remediation raises the bar for safe use not just of this compound, but of the broader family of organoarsenicals. Scientists and regulators will keep circling back to the central question: Can we learn from past missteps to find productive, lower-risk roles for these potent, tricky molecules in tomorrow’s technologies?
What is 4-Aminophenylarsonic Acid used for?
A Closer Look at 4-Aminophenylarsonic Acid
4-Aminophenylarsonic acid, known in labs as 4-APA, shows its true colors in agriculture and industry. For decades, folks working close to the land recognized it under brand names in animal feed. It sits in the same chemical family as roxarsone, with both shaped by the goal of boosting animal growth and cutting down on disease in flocks and herds. The reason for this use stands mostly on its antibacterial kick. Small-scale farmers and large producers both leaned on it to keep intestinal parasites at bay, guarding profits at a time when every bushel and crate mattered.
The Double-Edged Sword of Growth Promoters
Adding any arsenic-related compound into food sources started out with more hope than caution. Decades ago, nobody at the feed mill really weighed the risk of these small doses ending up on tables or in rivers. In my own experience growing up around rural communities, talking with older producers always brings up how common medicated feeds used to be. Most believed the government had already cleared the way, so who would doubt the safety? Only much later did independent researchers discover arsenic compounds could linger in meat or find paths into the groundwater.
Journal studies and FDA reviews back this up. Evidence built up by the early 2000s that people eating chicken raised with arsenic feedstock could wind up exposed to low but real traces—never instantly dangerous, but not gone either. The soil beneath long-time chicken barns told similar tales, sometimes showing up as elevated arsenic when tested head-to-head with fields that never saw medicated feed spills. Even when 4-aminophenylarsonic acid and its cousins break down, they can form inorganic arsenic varieties. Those have a well-known reputation for harming health.
Health, Environment, and Moving Forward
Science keeps track of these risks, and the FDA no longer signs off on 4-aminophenylarsonic acid for use in food animals. That shift mattered on both human and environmental health fronts. Childhood exposure to arsenic gets tied to higher cancer rates later, and water systems downstream from livestock barns need constant follow-up checks. This isn’t just an issue for scientists: it lays new burdens on rural communities. Farmers have to pivot, city utilities have to screen for pollutants, and families have to trust the water at the tap.
Switching away from chemicals like 4-APA calls for more than swapping out bags at the feed store. Animal welfare and housing designs take center stage; healthier flocks and herds come from lowering stress, cleaning pens, rotating pastures, and using probiotics in place of antibiotics or arsenicals. It costs time and adds work, but those investments show up in cleaner food and less worry about what’s in the soil.
Accountability and Innovation
4-Aminophenylarsonic acid carved a path for new regulation and greater transparency in what feeds commercial animals. Food producers compete on who can provide clearer sources, and that trust forms the backbone of sustainable farming today. University trials keep searching for ways to stop disease in animals without relying on risky chemicals. The right answer won’t come in a single step, but learning how old tools shaped outcomes keeps the lessons grounded in real effects on health and the earth.
What is the chemical structure of 4-Aminophenylarsonic Acid?
Looking at the Makeup of 4-Aminophenylarsonic Acid
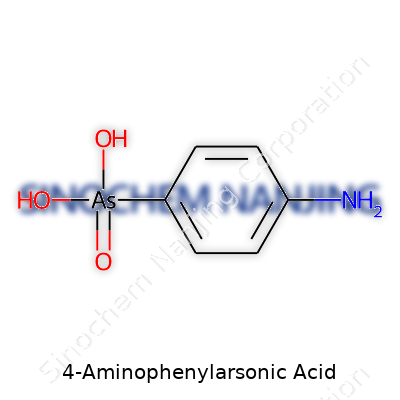
People sometimes see long chemical names and get overwhelmed, but taking apart a name like 4-Aminophenylarsonic acid reveals a lot about the structure. The name gives clues right away. “4-Amino” points to an amino group (-NH2) attached to the fourth carbon of a benzene ring. “Phenylarsonic” says we’re dealing with an arsonic acid group bound to a phenyl ring. Scientists know the formula is C6H8AsNO3.
Simple Breakdown of Components
You start with a benzene ring, a six-carbon aromatic ring found in countless chemistry classrooms—a classic foundation. At position four on this ring, there’s an amino group. The amino group wears a reputation for reactivity and hydrophilicity, making molecules like this keen to interact in water-based environments.
On the same ring, usually at the position where the count begins (number one), you’ll find the arsonic acid group. In structure, this arsonic acid resembles phosphonic or sulfonic acids a bit. The group looks like –AsO(OH)2, giving the molecule both an organoarsenic backbone and an acidic nature because of those two hydroxyls hanging off the arsenic.
The Drawing Beneath the Words
Imagine a ring of six carbons and one hydrogen per carbon, just like old-school benzene. At the “one” spot, swap the hydrogen for an arsonic acid group. Swing your eye three spots over: at the “four,” drop in an amino group. The rest of the hydrogens line the edges. So, you’re left with a flat, rigid shape capped on each end by groups pulling the molecule in different chemical directions.
It’s worth picturing this structure as more than lines and letters. The electron-withdrawing force of the arsonic acid group can dampen the electron-rich benzene, shifting how this molecule reacts compared to others. The amino group brings basicity and hydrogen bonding. Chemists recognize how these features matter when exploring how drugs interact or how compounds break down in the environment.
Where Knowledge Meets Impact
This compound isn’t just a classroom curiosity. As someone who’s worked around both agricultural science and environmental remediation, I’ve seen 4-aminophenylarsonic acid come up in feed additives or discussions about arsenic contamination in water and soil. Its organoarsenic backbone often raises red flags because certain arsenic compounds hold toxicity for humans, plants, and animals. Proper knowledge of its chemical makeup shapes rules for safe use, storage, and disposal.
Academic studies highlight how it breaks down into simpler, sometimes more toxic forms in soils and water. Understanding the arrangement of atoms in 4-aminophenylarsonic acid helps environmental chemists predict how it might react—oxidize, reduce, or bind with minerals. Those predictions guide policies to keep food and water clean, protect farmworkers, and preserve ecosystems. If farmers use these additives, regular screening and switching to less hazardous alternatives make the biggest difference. Regulations usually limit allowable residues, but technology for fast, sensitive detection always deserves more investment.
Looking Ahead: Safer Science and Solutions
Seeing the full structure of 4-aminophenylarsonic acid lets research teams design better analytical methods. Spectroscopy, chromatography, and molecular modeling all lean on those atomic connections for accuracy. Anyone working to reduce the risks tied to organoarsenic compounds starts by understanding the details of this structure. Education, tighter rules, and sharing knowledge between fields all help build a safer agricultural and environmental future. With that atomic snapshot in mind, stakeholders find smarter paths forward—science, farming, and public health all benefit from that clarity.
Is 4-Aminophenylarsonic Acid hazardous or toxic?
Looking at the Science Behind the Substance
4-Aminophenylarsonic acid doesn’t just sound intimidating. This chemical compound has a history mostly tied to agriculture, where it used to feature in feed additives for animals, particularly poultry and swine. Over the years, researchers started asking tough questions about what happens when such substances get into the environment and human food.
Human Health Risks: What Do We Know?
Arsenic raises big, red flags in public health. The problem with 4-aminophenylarsonic acid lies in the breakdown products it creates. Bacteria and other agents in soil and water environments can convert this compound into inorganic arsenic forms, which science has linked to a wide range of health problems. Exposure to inorganic arsenic, even in small amounts over time, can increase the risk of cancers, skin lesions, and cardiovascular disease. The World Health Organization and many national health agencies have made clear: regular exposure to inorganic arsenic in groundwater is a serious hazard, especially in regions with contaminated groundwater.
One point that keeps coming up in studies—farm runoff carries all kinds of things, including these organic arsenic compounds, into nearby water sources. Once there, the transformation into dangerous forms is hard to prevent. Anyone who relies on private wells near such runoff zones faces higher risk, especially children, whose developing bodies are more vulnerable. Studies from the U.S. Environmental Protection Agency show that these risks aren’t a distant concern; they're present in areas with heavy agricultural use.
Environmental Impacts: Not Just a Human Problem
Beyond the personal health concerns, 4-aminophenylarsonic acid and related substances pose serious risks to the ecosystems where they land. Aquatic systems absorb runoff, and as arsenic compounds break down, fish and other animals take them in. Arsenal compounds can then travel up the food chain, disrupting entire ecosystems. As anyone who’s watched a lake struggle with chemical pollution knows, the result hits recreational fishing, tourism, and even property values. Long-term contamination doesn’t wash away quickly.
The impact doesn’t stop in water. Soil can hold onto arsenic for years, affecting crops grown for future generations. The food safety concern grows once you consider that grains, vegetables, and fruit can take up these substances. Farmers and consumers deserve transparency and strong safeguards.
Taking Action: Safer Choices Moving Forward
Many countries took the step to ban or restrict 4-aminophenylarsonic acid in agriculture because of these documented risks. Regulatory decisions stemmed from real findings: increased levels of arsenic detected in meat, eggs, and soil samples. The safest course for consumers is to remain aware of where food comes from and to look for sources that follow strict safety practices. Anyone running a farm or garden can focus on pollution controls, buffer zones, and choosing alternatives to legacy chemicals.
Scientists push for regular groundwater and soil monitoring near old livestock operations. This sort of active observation gives communities the data needed to avoid harm before it becomes a crisis. Policymakers, in turn, need to listen to that data and keep updating what chemicals can be used in food production, keeping public and ecological health at the front of every decision.
How should 4-Aminophenylarsonic Acid be stored?
Down-to-Earth Chemistry
4-Aminophenylarsonic acid is not hanging out on the shelf at your local grocery or pharmacy. In real-world labs, folks work with it for research or as an intermediate, mostly in veterinary or pharmaceutical projects. Scientists learn to treat chemicals like this with some respect, especially since it carries arsenic in its backbone. Nobody wants to discover the hard way that sloppy storage led to cross-contamination, health scares, or regulatory headaches.
Practical Storage Principles
It’s hard to beat a dedicated chemical cabinet for storing 4-aminophenylarsonic acid. My own lab days taught me that an airtight, clearly labeled glass container beats plastic any day for minimizing leaching or reactivity. An arsonic acid compound doesn’t belong near acids or bases, as these can trigger unwanted breakdown or even dangerous gas formation. Dry, cool, and dark conditions slow the odds of the stuff breaking down or reacting with the atmosphere.
The temperature matters for most organic reagents, but with arsenic compounds, there’s a personal and environmental angle. At room temperature, kept away from heat sources, most researchers breathe easier, literally and figuratively. I saw one incident in which storage next to a sunny window ruined both the reagent and parts of someone’s project results. A simple storage slip can cost money and, worse, cause arsenic to end up where it shouldn’t, such as a shared sink or public system.
Human Element: Why Labeling Matters
It’s easy to ignore the little things after a long day. Still, 4-aminophenylarsonic acid deserves clear labeling, complete with hazard information and the date received or opened. Faded tape or scribbled names invite guesswork. I remember a colleague misidentifying an aliquot, which set everyone back two weeks. Good record-keeping matters—not only for compliance, but because it saves someone from cleaning up an arsenic spill months down the line.
Staying organized is part of being a responsible team member. It’s not just about avoiding fines or inconvenient audits; it’s about not putting anyone else at risk. If there’s ever an emergency, the label tells the whole story in a few seconds.
Dealing with Waste and Accidents
Disposing of arsonic acids rarely makes anyone’s list of favorite lab chores, but it’s a job requiring focus. Waste containers designated for heavy metals are a must. Trying to “just rinse it down the drain” isn’t only illegal—holding arsenic above the legal limit can harm water systems and runs straight into local news (and not the kind you want your name in). In my own work, we triple-checked these procedures, and it was worth every extra minute.
If a spill occurs, evacuate, contain, and don protective gear. Teams that drill safety procedures handle these situations much smoother than “by-the-seat-of-your-pants” crews. Name and date all incident documents for traceability, too.
Supporting Safe Science
All of this isn’t just bureaucracy; it’s about maintaining trust. Proper storage prevents dangerous exposure, supports reproducible research, and keeps chemical safety inspectors satisfied. Good stewardship of 4-aminophenylarsonic acid comes down to placing people and the planet ahead of shortcuts.
What are the safety precautions when handling 4-Aminophenylarsonic Acid?
Understanding What’s At Stake
Anyone who’s spent time in a chemistry lab knows the uneasy sense you get when you pull a bottle down and the label says something like 4-Aminophenylarsonic acid. Arsenic in the name sets off alarm bells for good reason. This isn’t an ingredient you want anywhere near bare skin, lunch containers, or your sinuses. In one former workplace, a simple glove rip during an experiment sent a colleague down weeks of medical forms and tests. The lesson: treating compounds like this without respect opens the door to health problems that can linger for years.
Personal Protective Gear Isn’t Just For Show
It’s easy to get lazy about gear, especially if the task looks quick. But exposure to 4-Aminophenylarsonic acid can lead to poisoning through inhalation, skin contact, or accidental ingestion. Nitrile gloves, chemical-resistant goggles, and a lab coat form the first shield. A fume hood is not negotiable either. Our old lab’s cheap, noisy hoods might not have won design awards, but they stopped countless invisible clouds from drifting into lungs.
One thing I always keep at my bench: a face shield for tasks with splash risk. The taste for shortcuts in safety didn’t survive my first close call with a volatile compound.
Clean Habits Prevent Ugly Surprises
Stacking your workspace with unnecessary junk only increases the odds of mixing up contaminated versus clean gear. Separate dirty from clean pipettes, label containers, and bag up disposable gloves right after use. I used to see folks leave powder residue sprinkled near balances—one bump of the elbow, and they’d carry trace amounts home. Invest a few seconds in double-checking everything, especially labels and seals, and you’ll dodge countless problems.
Disposal and Spills: Handle With Respect
Spills aren’t rare; quick action matters. Many labs keep a spill kit nearby, stocked with absorbents and neutralizers. After a spill, we always closed off the area right away, grabbed proper gear, and cleaned it with fresh gloves each time to avoid dragging traces elsewhere. No standard trash can ever takes arsenic compounds. Designated chemical waste bins, with clear hazard labeling, were a must and required regular checks to make sure nothing got missed. Arsenic contamination builds up fast if containers drip or leak.
Knowledge Saves Lives
Training isn’t just paperwork—knowing what to do the moment you catch a whiff of something odd or spot a powder you didn’t notice before can be the difference between a scare and a hospital visit. We kept safety data sheets within arm’s reach, so nobody had to guess what steps to take. Poisoning incidents drop fast when everyone actually reads the sheets and knows how to use the emergency shower, eyewash, and who to call for medical help.
Building a Culture That Cares
Accidents almost always trace back to rushed habits or gaps in knowledge. Management shows it values safety with regular drills, stocked equipment, and crystal-clear signage. Peer pressure helps too; we all took backup checks seriously. The best labs I’ve seen encourage questions and double-checks, turning what could be a mundane rule into a daily habit. The risks aren’t abstract—arsenic exposure has lifelong consequences. Nobody wants to trade their health for a shortcut.
| Names | |
| Preferred IUPAC name | 4-Aminobenzenearsonic acid |
| Other names |
p-Arsanilic acid 4-Arsanilic acid p-Aminophenylarsonic acid Arsanilic acid 4-Aminobenzenearsonic acid |
| Pronunciation | /ˈfɔːr əˌmɪnoʊˌfɛnɪl ɑːrˈsɒnɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | 98-50-0 |
| Beilstein Reference | 1721486 |
| ChEBI | CHEBI:22585 |
| ChEMBL | CHEMBL1433 |
| ChemSpider | 68215 |
| DrugBank | DB03663 |
| ECHA InfoCard | ECHA InfoCard: 100.003.528 |
| EC Number | 215-638-7 |
| Gmelin Reference | 1696350 |
| KEGG | C14257 |
| MeSH | D000635 |
| PubChem CID | 6115 |
| RTECS number | BJ9275000 |
| UNII | TUY4X64I7Y |
| UN number | UN2811 |
| Properties | |
| Chemical formula | C6H8AsNO3 |
| Molar mass | 249.07 g/mol |
| Appearance | White to light yellow crystalline powder |
| Odor | Odorless |
| Density | 1.7 g/cm3 |
| Solubility in water | slightly soluble |
| log P | -1.3 |
| Vapor pressure | 0 mmHg (25°C) |
| Acidity (pKa) | 3.79 |
| Basicity (pKb) | 6.8 |
| Magnetic susceptibility (χ) | -72.0·10⁻⁶ cm³/mol |
| Dipole moment | 4.53 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 250 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −861.4 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -1209.9 kJ/mol |
| Pharmacology | |
| ATC code | QG52AC01 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS05, GHS07, GHS08 |
| Pictograms | SSSR=O)(O)(O)C1=CC=C(C=C1)N |
| Signal word | Warning |
| Hazard statements | Hazard statements: H301, H319, H373 |
| Precautionary statements | Precautionary statements: "P261, P264, P270, P271, P272, P273, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P308+P311, P314, P321, P330, P361+P364, P391, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2-1-0 Health:2 Fire:1 Reactivity:0 |
| Flash point | Flash point: 195°C |
| Lethal dose or concentration | LD₅₀ (oral, rat): 4100 mg/kg |
| LD50 (median dose) | LD50 = 7800 mg/kg (rat, oral) |
| NIOSH | N0797 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of 4-Aminophenylarsonic Acid: "0.5 mg/m3 as As (OSHA) |
| REL (Recommended) | 0.05 mg/m³ |
| Related compounds | |
| Related compounds |
Arsanilic acid Acetarsone Carbarsone Nitarsone Phenylarsonic acid |

