4-Amino-N,N-Dimethylaniline Sulfate: Exploring Its Life and Utility
Historical Development
Chemistry books once told stories about the industrial revolution’s impact on dye production. In those days, the need for new colorants sent chemists scouring through aniline derivatives. 4-Amino-N,N-Dimethylaniline, later paired with sulfate to improve stability, emerged as a result of this push. The pursuit for brighter, more durable dyes in textiles and inks drove this compound’s early synthesis. Its role expanded beyond color. Lab benches and industrial plants found new uses as researchers uncovered how its unique amine structure could serve as a building block for other substances. Every breakthrough depended on practical handling and keen observation, rather than just theoretical dreaming. So, 4-Amino-N,N-Dimethylaniline sulfate earned its spot in both the history of chemistry and the toolbox of research.
Product Overview
Unlike flashy tech gadgets, chemicals like 4-Amino-N,N-Dimethylaniline sulfate sit quietly behind the scenes, making things work in pharmaceuticals, dyes, and analytical testing. This salt appears as a fine, colorless to slightly yellow powder, but don’t be fooled by its plain appearance. It steps up whenever a methylated aniline with extra reactivity is needed. Factories rarely value appearance, but they do remember reliability, and this compound delivers on that front.
Physical & Chemical Properties
You won’t spot 4-Amino-N,N-Dimethylaniline sulfate glimmering under a microscope, but its attributes pack a punch in the lab. This compound dissolves smoothly in water, a result of the ionic sulfate partner pulling its weight. Heat it up enough, and it slowly breaks down, releasing that signature amine aroma many chemists recall from long afternoons in university labs. As for reactivity, that para-amino group and dimethylation put it somewhere between an intermediate and a starting material in organic syntheses. The white to slightly yellow tint tells you about its purity and handling—exposure to air or impurities darkens it over time. The density and melting point, boring as they sound, matter for process engineers weighing batches and setting up production lines.
Technical Specifications & Labeling
Regulatory agencies impose strict labeling standards, and for good reason. Precise names, hazard warnings, and batch numbers prevent dangerous mix-ups. On a shelf, this sulfate salt bears the chemical name, molecular formula, CAS number, and hazard classification. This kind of rigorous detail reflects the hard lessons from years of chemical accidents and near-misses. Chemists and quality assurance teams rely on honest, accurate labeling to keep people safe and production running smoothly.
Preparation Method
Industrial manufacturers usually synthesize 4-Amino-N,N-Dimethylaniline by methylating p-phenylenediamine, followed by Sulfate addition through simple acid-base reactions. It’s a workhorse process: react, extract, purify, crystallize. Careful temperature control prevents side reactions that would spoil the product or yield hazardous byproducts. Filtration separates pure salt from the reaction mess. Every step in that sequence was born out of trial, error, and experience gained in the field. This isn’t million-dollar innovation, it’s boots-on-the-ground chemistry.
Chemical Reactions & Modifications
Once you have 4-Amino-N,N-Dimethylaniline sulfate, you gain a versatile handle in organic transformations. Its amine group takes on diazotization, forming intermediates for azo dyes, which color everything from plastic toys to medical stains. Throw the molecule into a coupling reaction, and it’ll latch on to aromatic systems, building chains that take on new color and function. Its methyl groups also give it extra resistance to some oxidative degradation, stretching its value in demanding chemical environments. Dotted around hundreds of patent filings, you’ll see this compound stepping in as a key intermediate. Its adaptability keeps it in these recipes long after trendier reagents flop.
Synonyms & Product Names
Say 4-Amino-N,N-Dimethylaniline sulfate in a lab meeting and you might get blank stares unless you add the synonyms: Dimethyl-p-phenylenediamine sulfate, or DMAPA sulfate. In the dye trade, variations on these names fill order books and invoice sheets. The pile of synonyms can slow down regulatory compliance, so familiarity with each name prevents errors and keeps inventories straight. Some older texts refer to it by less formal nicknames, a relic of looser practices that have since tightened up for safety reasons.
Safety & Operational Standards
Handling this compound calls for respect. Skin or lung exposure leads to irritation or worse, so gloves, goggles, and fume hoods stay non-negotiable. Facilities commit to safety data sheets detailing risks, symptoms of overexposure, and steps for potential spills. Emergency protocols—flushing, proper disposal, and regular audits—reflect decades of workplace hard-learned experience. Environmental laws restrict the discharge of amines and sulfates into wastewater, guarding public health and ecosystems. From laminar flow benches in analytical labs to the drum unloaders at chemical plants, operational standards form the backbone of safe, responsible use.
Application Area
Industry relies on 4-Amino-N,N-Dimethylaniline sulfate most heavily for dye stuff production. The molecule acts as a stepping stone for synthesizing colorants that wind up in textiles, paper, and diagnostic reagents. It serves as a part of oxidative hair dyes, laboratory assays, and niche pharmaceutical formulations. Analytical chemistry procedures—especially those detecting trace metals or testing for peroxides—take advantage of this compound’s electron-rich amine. Pharmaceutical research sometimes roams into this territory, exploring derivatives for drug molecules and bioactive compounds. Behind every application, practical problems like cost, purity, and traceability matter as much as theoretical possibilities.
Research & Development
The R&D teams I’ve seen in action give 4-Amino-N,N-Dimethylaniline sulfate hard scrutiny, searching for efficient syntheses, better handling characteristics, and reduced toxicity. Among the biggest recent pushes, green chemistry stands out—teams hunt for solvent systems and catalysts with less waste and lower energy inputs. Academic labs look at how subtle tweaks in the structure affect dye shades and stability. Pharmaceutical scouts keep an eye out for new therapeutic angles, tracking how methylated amines change biological activity profiles. Engineers test high-throughput methods, scaling up lab reactions into commercial processes. Real improvements here ripple out through supply chains, cutting costs, and improving safety.
Toxicity Research
Discussions around aniline derivatives always circle back to toxicity. Animal studies and cell assays show that N,N-dimethylated anilines can impact blood and liver function. Chronic exposure can lead to methemoglobinemia and other problems, especially for workers handling these products without proper protections. Regulatory bodies, including the EPA and ECHA, monitor workplace exposure and set strict permissible levels. In the lab, toxicology testing pushes forward, looking for ways to modify molecular structure and cut risk. Every safety improvement follows a close reading of past mistakes, from uncontrolled releases to misdiagnosed health impacts. These stories fuel honest conversations about risk and prevention.
Future Prospects
Looking ahead, applications may broaden as industries demand specialty dyes, sensors, and analytical reagents with sharper performance traits. Sustainability goals nudge the field toward new preparation methods that minimize hazardous byproducts and promote safe reuse or disposal. Digital tools and automated reactors could shorten development cycles, making it easier to test tweaks in molecular structure. Pressure from governments and consumers for safer, greener chemicals won’t let up, so researchers will continue hunting for less toxic alternatives without sacrificing performance. The journey won’t be quick. It’ll involve persistent, detail-oriented work, combining lessons from the past with creative thinking for the future of specialty chemicals.
What is 4-Amino-N,N-Dimethylaniline Sulfate used for?
Unpacking the Real-World Value
4-Amino-N,N-Dimethylaniline sulfate holds a key spot in the toolbox of labs, industries, and research teams. The compound, best known for its noticeable purple tint in certain reactions, plays a reliable part in a number of analytical and synthetic tasks. With background in chemistry, I’ve worked around the practical side of chemicals like this. It’s more than just a long name on a bottle — it gets things done in the lab.
Key Role in Dye and Pigment Chemistry
You’ll often see 4-Amino-N,N-Dimethylaniline sulfate pop up in the production of colorants. Textile makers rely on anilines for dyes, and this compound provides a starting block for several vivid shades. It acts as an intermediate, letting chemists tweak its structure to design color molecules. The affinity for fibers and resistance to washing make the resulting dyes valuable in daily life — shirts, towels, and upholstery draw on this sort of chemical groundwork.
Sensor and Detection Solutions
Lab analysts prize this amine for its use in spectroscopy. The compound interacts with oxidizing agents in test kits, creating color changes that point to certain biochemical or environmental substances. Hospitals and water quality labs both benefit, using it in test strips or solutions. Changes in color can clue technicians in to toxins, pollutants, or even glucose levels, a fact that keeps the compound in steady demand.
Pharmaceutical Building Blocks
Drug companies and academic groups sometimes use 4-Amino-N,N-Dimethylaniline sulfate as a scaffold. Its structure lends itself well to modification. Medicinal chemists take advantage of this versatility, exploring derivatives in trial runs against bacteria, fungi, or even tumor cells. While not a blockbuster drug itself, the molecule serves as a foundation for inventing new medicines or diagnostic reagents.
Printing and Photographic Applications
Printing inks and some classic photographic developers draw on aniline-based compounds for stability and deep color. In darkrooms, developers formulated with aniline derivatives provide consistent results. Even as digital takes over, specialty applications in art photography still pick these chemicals for the unique properties they bring.
Safety and Environmental Impact
Regulation surrounds the use of aromatic amines for a reason. Prolonged contact with 4-Amino-N,N-Dimethylaniline sulfate can pose health risks, with skin and respiratory exposure leading to issues. Working with this chemical in my own time, I stuck to fume hoods and gloves — overstating safety would be impossible in this case. Waste water from dye factories, if left untreated, spells trouble for local streams and wildlife. Many industrial producers invest in closed-loop systems and scrubbing technologies to reduce runoff. Given the strict eye from regulatory agencies such as OSHA and the EPA, safe handling and proper disposal remain non-negotiable.
Responsible Innovation and Moving Forward
Looking to balance utility with public well-being, researchers and manufacturers hunt for greener pathways. Alternatives inspired by natural dyes or more biodegradable amine analogs hold promise. Organic chemists work on newer catalysts and reaction methods to minimize unwanted byproducts. These choices can lead to a cleaner industry while still delivering the colors, tests, and medicines people rely on.
What are the safety precautions when handling 4-Amino-N,N-Dimethylaniline Sulfate?
Working With an Eye on Safety
In any chemistry lab, certain substances carry extra risk. 4-Amino-N,N-Dimethylaniline Sulfate is one of those chemicals that reminds you accidents can happen fast. This compound shows up in dye manufacturing and research settings, with toxic and potentially carcinogenic properties. Whenever I step into a lab that uses it, the mood turns focused and deliberate.
Recognizing Real Hazards
The risk goes beyond stains on your lab coat. Direct skin contact or inhalation can bring on irritation or more serious problems. The SDS for this compound highlights harm to the respiratory system, skin, and eyes. Ingestion brings about headaches, nausea, and more severe toxicity. Cancer concerns aren’t just warnings: the International Agency for Research on Cancer has flagged related aromatic amines.
Basic chemistry work teaches everyone to treat strong chemicals with respect. Over the years, I’ve seen incidents from carelessness—one wrong move, a forgotten glove, or a hasty pipetting job. Once my own colleague ended up with burns by relying on old nitrile gloves. It’s a harsh lesson to learn, and it happens quickly.
Personal Protective Equipment Matters
The best defense starts with good PPE. In my experience, always wear gloves rated for chemical resistance, not the thin medical kind from drugstores. Splash goggles keep eyes safe, and a face shield handles larger risks. A proper lab coat with sleeves over your gloves keeps splashes away from skin. I choose nitrile or laminated gloves and check the manufacturer’s data, not just the shop label. Doubling up on gloves in busy labs cuts down on careless accidents.
Fume hoods aren’t just recommendations; they’re mandatory with this substance. Even quick measurements send fine dust into the air. Breathing enough of it over time can do a number on your lungs, so ventilation stays at the top of my checklist every session.
Handling, Storage, and Cleanup
Measure chemicals with tools that stay dedicated to this job, then wash them thoroughly. I store 4-Amino-N,N-Dimethylaniline Sulfate tightly sealed in a cool, dry space that keeps acids, oxidizers, and food-grade materials far away. Every label shines clear so nobody grabs the wrong bottle by mistake.
Any time I spill a pinch, out come absorbent pads and disposable wipes. I grab a lab partner so neither of us works alone. The area gets cleaned with care, not speed, and all waste goes in a labeled, hazardous waste bin for certified disposal. Nobody pours this down the drain.
Training and Emergency Prep
Labs with strong safety cultures run regular drills, keep eyewash stations loaded, and train on emergency showers. More than once, practice kept panic from ruining a response to a real splash. Staff refresh training so that new faces on the team know exactly what to do, even in a hurry. An effective safety approach doesn't rely on guesswork—facts from the SDS, experience from colleagues, and open communication make up the smartest prevention.
Practical Steps Make the Difference
Chemistry brings big discoveries and challenges, but only with the right handling can people stay out of the ER. An honest look at past mistakes and a readiness to speak up about risks change outcomes for the better. Safety isn’t something you check off—it stays active with every experiment and every bottle uncapped.
How should 4-Amino-N,N-Dimethylaniline Sulfate be stored?
Understanding the Risk
In labs, 4-Amino-N,N-Dimethylaniline Sulfate shows up as a white to grayish powder, mostly used in dye production and lab synthesis. It can be pretty irritating to skin and eyes, and it forms dust that isn’t great for lungs. For anyone dealing with chemicals every day, this stuff reinforces the lesson that respect for proper storage isn’t about following a rulebook – it’s about coming home safe.
Direct, No-Nonsense Storage
Chemicals don’t forgive shortcuts. Don't leave this compound on a shared shelf or anywhere it could cross paths with strong oxidizers, acids, or a sunny window. My chemistry training hammered home one point: keep it cool, dry, and out of light. Cabinets with tight-sealing doors and clear hazard labeling cut the risk of accidental mix-ups. Humidity is a huge enemy here. I have seen jars bloom with lumps overnight in damp rooms, which means a ruined batch or unseen risk building up.
Containers Matter
Polyethylene or glass containers kept upright do the job. Screw tops that seal tightly keep out air and moisture. I always avoid reaching for cracked lids or old bottles with labeling worn off – it’s not worth trusting fate to guesswork. Chemical-resistant gloves and goggles always come out. A single splash or spilled pinch lying on a bench never ends with a shrug; accidents happen in the split second you break focus.
Clear, Reliable Labeling
Handwritten notes on masking tape got old quick. Permanent, legible labels that list the compound’s name, the date received, and hazard symbols allow everyone a second look before touching anything. I once had a coworker with a habit of sloppy labeling, and it nearly turned into a serious exposure after a switch-up in similar-looking jars. It’s all about keeping each other in the safe zone.
Your Storage Space and Routine
Walk into a well-managed storage room and you’ll notice order – flammable cabinets separate from oxidizers, spill trays matched to the chemicals’ properties, inventory logs checked off after every shift. I like to take ten minutes at the end of each week to tidy up, check for leaks, and update inventory sheets. No one ever gets in trouble for double-checking, but skipping a small check can lead to major messes.
Disposal and Spill Plan
Even the best storage plans hit snags. If a container starts to leak, there’s no wisdom in waiting around. Use designated chemical spill kits and notify your safety officer, then ventilate the area well. Never try to sweep powder spills into a trash bin. Lab safety protocols usually call for an absorbent material, followed by specialized waste containers. This keeps the cleanup safe for everyone who walks through that lab door in the future.
Improving Everyday Safety Culture
Proper chemical storage amounts to more than just locking away a bottle. It’s about making habits, building trust with coworkers, and treating every container like it matters. Safety instructions written on the wall aren’t enough; drills, walk-throughs, and open talks about mistakes used to make the difference in every busy lab I worked in. The next time you store 4-Amino-N,N-Dimethylaniline Sulfate, remember that real safety grows out of routines and shared experience.
Practical Solutions for Reliable Storage
Invest in sturdy cabinets with secure locks, maintain updated inventory records, and train new lab members on specific protocols for this compound. Speak up if you spot a risk and encourage colleagues to do the same. Secure storage never feels like a burden once it turns into everyone’s habit – and that’s how you keep risks from turning real.
What is the chemical structure and formula of 4-Amino-N,N-Dimethylaniline Sulfate?
Getting Past the Name: What's in This Compound?
4-Amino-N,N-dimethylaniline sulfate might not be a name that rolls off the tongue, but this compound tells a real story in molecular chemistry. Plenty of folks have crossed paths with similar molecules while working with dyes, pigments, or organic synthesis. Before diving in, breaking down the name pulls back the curtain on its structure.
The backbone is an aniline ring — that classic benzene ring with a single amino group tacked on. Here, the twist comes with two extra methyl groups on the nitrogen. It means, at its core, it looks like this: the benzene ring anchors everything, the amino group attaches at the fourth carbon (para position), and the nitrogen grabs two methyl tags, making it a tertiary amine. That’s where the “N,N-dimethyl” part steps in. The structure is further stabilized as a sulfate salt, which matters for both its stability and water solubility.
Chemical Structure and Formula
To those tackling the raw details, 4-Amino-N,N-dimethylaniline itself has the formula C8H12N2. The sulfate part rolls in as a hydrated salt. As the sulfate salt, the complete formula is usually written as (C8H12N2)2·H2SO4. Two molecules of the amine pair with one molecule of sulfuric acid. In line structure form, it looks like a benzene ring with an amino group (-NH2) at position four and two methyl groups attached to the nitrogen atom. More simply: the sulfuric acid acts like glue, linking the amine molecules together as a solid, crystalline salt.
Why Knowing Structure Matters in Everyday Chemistry
Plenty of uses pop up for aromatic amines. I remember working in a small industrial lab where we handled compounds like this for dye production. The structure controls everything: solubility in water, how fast it reacts, and even how it interacts with enzymes or metals. The sulfate salt, in particular, gives the compound more punch in terms of handling — it’s less volatile, safer to transport, and dissolves well in water. That’s huge when labs try to avoid unnecessary risk. It also limits exposure to the free base form, which can be more hazardous due to volatility or toxicity.
The structure also hints at the reactivity. Aromatic amines can form vivid colors when reacting with other chemicals. This property forms the backbone of their role in dyes and indicators. Chemists trust these predictable behaviors, knowing the location of each group on the ring and how they influence electron flow.
Debating Safety and Environmental Impact
There’s a flip side to this convenience. Aromatic amines sometimes find themselves linked to challenges in health and environmental circles. Studies have shown some related compounds have carcinogenic risks. Regulatory agencies in the US and EU regularly update lists of safe allowable levels for these chemicals. Lab safety training stresses gloves and extraction fans, not just for personal safety, but for anyone who shares downstream drinking water or food sources.
Looking Toward Better Handling and Use
Addressing potential issues means using less hazardous alternatives when possible or enclosing production systems to catch stray particles and vapors. Newer dye technologies sometimes rely on different chemistries, aiming to cut out the risk without sacrificing performance. Education plays a role, too. Equipping those in classrooms and factories with strong labeling, clear protocols, and up-to-date research keeps everyone safer — and that means fewer surprises.
Are there any specific disposal guidelines for 4-Amino-N,N-Dimethylaniline Sulfate?
Why Proper Disposal Isn’t Optional
4-Amino-N,N-Dimethylaniline Sulfate shows up in labs for dye making, organic synthesis, and sometimes for research on chemical sensors. It’s not some kitchen scrap—this compound brings health and environmental hazards. The stuff reacts strongly with oxidizing agents, and can irritate the skin, eyes, or even mess up your breathing if inhaled. Some studies list its potential to harm aquatic life, which nudges us to take disposal seriously. I’ve seen careless disposal decisions—poured down drains, tossed in general trash—that sparked workplace safety scares. This isn’t just about rules. It’s about responsibility to the people next door and the planet.
Straightforward Guidelines and Tried-and-True Steps
If you crack open the Material Safety Data Sheet (MSDS) for this compound, clear instructions jump out: treat as hazardous waste. I always double-check whether local, state, or national bodies—like the EPA in the U.S.—have extra rules. Local regulations might ask for bringing chemical waste to a certified hazardous waste center or getting in touch with a contractor.
Diluting the compound in water or tossing it down the drain feels tempting, especially in small labs. That shortcut risks harming water treatment plants and downstream wildlife. A lot of folks underestimate how tiny traces from hundreds of labs can add up to real ecological damage. In my work with lab groups, we’ve collected every bit in sealed, labeled containers built to handle chemicals. No gaps, no shortcuts. The process includes marking the container with the compound name, concentration, hazard class, and the date. Many waste collection services won’t pick up poorly labeled containers, and handling delays cost everyone extra money and hassle.
Supporting Workers and the Environment
Lab workers get the most direct exposure. Gloves, goggles, and a chemical fume hood act as the first line of defense. Still, accidents happen. If a spill shows up, I’ve found it’s smart to have spill kits handy and actual training—not just a stapled safety pamphlet. The more people practice the steps, the less panic during a real accident.
Big institutions—universities, hospitals, contract labs—run regular hazardous waste pickups. If money stands in the way, some smaller outfits pool waste for bulk pickup to cut costs. Some regions even run “hazardous household collection days” for small quantities from teaching labs or DIY science groups. Where I grew up, a nearby town treatment plant had open weekends for safe chemical waste drop-off. The old timers working there taught newcomers what goes in which drum, and why it makes a difference.
Doing It Right: Core Solutions
Safety starts with strong habits: label, separate, never mix unknowns, and always check the latest local laws. Some people argue these steps eat up time, but in the long run, they prevent lost productivity and health costs. I’d argue that updating disposal training and clear signage in every lab or storage area cuts down on mistakes. Institutions and local authorities who design straightforward pickup options get better participation and cleaner results. Public labs sharing tips and offering community help line up everyone on the right side—science, safety, and the environment.
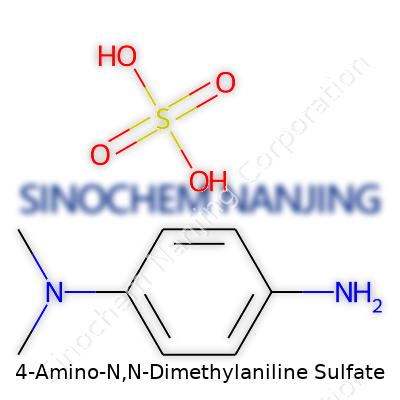
| Names | |
| Preferred IUPAC name | 4-amino-N,N-dimethylanilinium sulfate |
| Other names |
N,N-Dimethyl-p-phenylenediamine sulfate Dimethylparaphenylendiamine sulfate N,N-Dimethyl-1,4-benzenediamine sulfate 4-(Dimethylamino)aniline sulfate 4-Aminodimethylaniline sulfate |
| Pronunciation | /ˈfɔːr əˈmiːnoʊ ˌdaɪˈmɛθɪlˈænɪliːn ˈsʌl.feɪt/ |
| Identifiers | |
| CAS Number | 618-68-6 |
| 3D model (JSmol) | `3D model (JSmol)` string for **4-Amino-N,N-Dimethylaniline Sulfate**: ``` CS(=O)(=O)[O-].CN(C)c1ccc(N)cc1 ``` |
| Beilstein Reference | 604939 |
| ChEBI | CHEBI:38942 |
| ChEMBL | CHEMBL1230129 |
| ChemSpider | 20740772 |
| DrugBank | DB14147 |
| ECHA InfoCard | 03e5cf46-46a9-4fe1-b745-c3c832b2a7b2 |
| EC Number | 205-488-0 |
| Gmelin Reference | 1630687 |
| KEGG | C16586 |
| MeSH | D05.750.078.730 |
| PubChem CID | 85914 |
| RTECS number | BZ9625000 |
| UNII | 9Z6R2T5A4B |
| UN number | UN2811 |
| CompTox Dashboard (EPA) | DTXSID80896644 |
| Properties | |
| Chemical formula | C8H12N2·H2SO4 |
| Molar mass | 246.32 g/mol |
| Appearance | Gray to brown solid |
| Odor | Odorless |
| Density | 1.223 g/cm3 |
| Solubility in water | Very soluble in water |
| log P | -0.24 |
| Acidity (pKa) | 5.1 |
| Basicity (pKb) | pKb ≈ 10.77 |
| Magnetic susceptibility (χ) | Negligible |
| Viscosity | Viscous liquid |
| Dipole moment | 4.04 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 324.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -3610 kJ/mol |
| Pharmacology | |
| ATC code | D06BA10 |
| Hazards | |
| Main hazards | Harmful if swallowed, inhaled or absorbed through skin. Causes skin and eye irritation. May cause allergic skin reaction. Suspected of causing genetic defects. |
| GHS labelling | GHS07, GHS09 |
| Pictograms | GHS07,GHS09 |
| Signal word | Warning |
| Hazard statements | H302 + H312 + H332: Harmful if swallowed, in contact with skin or if inhaled. |
| Precautionary statements | P261, P264, P280, P301+P312, P302+P352, P305+P351+P338, P322, P330, P337+P313, P501 |
| NFPA 704 (fire diamond) | 2-3-0 |
| Flash point | > 260 °C |
| Autoignition temperature | 390 °C |
| Lethal dose or concentration | LD50 Oral Rat 698 mg/kg |
| LD50 (median dose) | 860 mg/kg (rat, oral) |
| NIOSH | JN8225000 |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | REL: 0.5 mg/m³ |
| IDLH (Immediate danger) | No OSHA IDLH |
| Related compounds | |
| Related compounds |
p-Phenylenediamine N,N-Dimethylaniline Aniline 4-Aminophenol 4-Nitro-N,N-dimethylaniline |

