Commentary: Demystifying 4-Amino-N,N-Dimethylaniline Oxalate—From Lab Bench to Modern Applications
Historical Development
The past shapes every tool, chemical, and material on the laboratory shelf, and 4-Amino-N,N-Dimethylaniline Oxalate traces its lineage to the decades when organic synthesis became central to industrial chemistry. Back in the late nineteenth century, researchers started tinkering with anilines, trying to amplify dyes and streamline their production for textiles. Later variations followed on the heels of the first synthetic purple dye and brought new dimensions to molecular design, especially through the hands of German chemists exploring aniline derivatives. By the 1950s, laboratories worldwide counted on 4-Amino-N,N-Dimethylaniline not just for color work but also as a stepping stone for more complex molecules—who could have imagined its oxalate salt would find a place in both academia and industry in years to follow?
Product Overview
4-Amino-N,N-Dimethylaniline Oxalate is not a household name, but ask a chemist and most will recall it as a useful reagent in both organic synthesis and as a building block in specialty pigment production. It looks unassuming—usually a pale crystalline powder or sometimes slightly off-white. Its true value stretches far beyond the bottle; it shines through in the way it enables the production of dyes, influences research in pharmaceuticals, and supports analytical chemistry. Every time this compound enters a reaction, its role feels essential for moving science—or even entire industries—a step forward.
Physical & Chemical Properties
You notice certain details with your own eyes: a faint odor, compact crystals, and a sample that dissolves readily in water and several organic solvents. On the working bench, its stability under standard conditions makes it fairly easy to handle, though moisture can sometimes affect the sample's shelf life. Temperature changes bring subtle shifts, and labs that use this salt know to avoid letting the environment swing too much. Its melting point sits comfortably below many industrial reagents, suggesting a reactivity profile that fits a myriad of synthetic needs. Operators appreciate its solubility and purity, which often challenge other aromatic amines; when in solution, it offers up opportunities for controlled reactions and easy monitoring—a detail you only learn through hands-on hours at the bench, not just from data sheets.
Technical Specifications & Labeling
In high-stakes environments, mislabeling means wasted time or, worse, unwanted reactions. Bottle labels typically display the full IUPAC name, hazard symbols mandated by the Globally Harmonized System, and concentration if it’s a solution. Chemists keep an eye out for batch purity, which impacts downstream applications, and trace contamination from the oxalate salt, which sometimes matters in analytical projects. While some debate the value of chasing exceptionally high purities versus “good enough” for most applications, anyone deep in color chemistry or pharmaceuticals knows that small differences in impurity profiles can matter. Clear, precise labels play a hidden but critical role in making sure mistakes don’t happen, especially for early-career researchers new to the rigor of chemical handling.
Preparation Method
Producing 4-Amino-N,N-Dimethylaniline Oxalate draws from the chemistry playbook of aromatic amine synthesis, with a twist for those attempting scale-up. The parent amine, 4-Amino-N,N-Dimethylaniline, forms through methylation of the corresponding nitro compound, often using formaldehyde and formic acid or reductive amination routes. Acidification with oxalic acid produces the oxalate salt, giving a product that’s easier to purify and handle than the free base. Recrystallization steps follow—there’s no substitute for that careful, hands-on purification that comes from years of lab work. Mistakes in stoichiometry crop up often in student labs, proving that no recipe works perfectly every time without some adjustment.
Chemical Reactions & Modifications
Work with this compound and its reactions unfold like a story. The nucleophilic -NH2 group and the dialkylated nitrogen offer windows for further functionalization. Often, reduction, diazotization, or electrophilic aromatic substitution reactions take center stage, allowing the molecule to serve as a core for azo dye synthesis or for creating tailored intermediates in medicinal chemistry. Each pathway teaches a chemist hard lessons—sometimes side products predominate, at other times yield fades because tricky reagents react inadequately. These practical challenges shape every decision and reinforce the need for constant adaptation, not rote execution of protocols.
Synonyms & Product Names
In the world of specialty chemicals, synonyms cause more confusion than most outsiders imagine. 4-Amino-N,N-Dimethylaniline Oxalate shows up in catalogs and journals as p-(Dimethylamino)aniline oxalate, N,N-Dimethyl-p-phenylenediamine oxalate, or simply DMAPA oxalate. Each vendor and journal seems to prefer their own variant, and double-checking CAS numbers saves headaches when cross-referencing old literature. Students get tripped up by these multiple identities, but with experience comes quick recognition of labeling habits in every lab or institution.
Safety & Operational Standards
Chemical safety has never been more important—especially for aromatic amines, which sometimes exhibit toxicity and mutagenicity. Mishandling could spell disaster not just for the immediate user, but also for those sharing research space. Standard practice requires gloves, eye protection, and careful attention to storage away from strong oxidizers. Fume hoods aren’t just recommendations—they save lungs and skin. Those with experience know that lapses, even on slow days, can lead to accidents or chronic issues. Regular training and simple checklist routines build habits that stick, outlasting even the most detailed written protocols.
Application Area
Many see 4-Amino-N,N-Dimethylaniline Oxalate as just a reagent, but its impact reaches far into dye manufacture, colorimetric analysis, and medicinal compound design. The dye industry celebrates its ability to generate vivid colors; analytical labs leverage its sensitivity in detecting chlorine or nitrite, especially when rapid color changes are needed for quantification. In pharmaceutical research, its presence marks a pathway toward more advanced nitrogen-containing drugs. Each of these uses comes wrapped in challenge—finding just the right reaction condition, scaling from milligram to kilogram, or chasing regulatory requirements for trace contaminants—all push research further and faster than planned at the outset.
Research & Development
The pace of change in chemical research never slows. Recent work looks to push the properties of 4-Amino-N,N-Dimethylaniline Oxalate into new frontiers, often searching for more selective synthetic routes or tuning the stability for broader use. Teams attempt to cut down on by-products, refine yields, or improve purification—less waste counts for more than cost savings, as sustainability scrutiny intensifies every year. New techniques, like flow chemistry and green solvents, turn up in R&D meetings, offering glimpses of less hazardous, more efficient processes. Crucially, academic-industrial partnerships bring fresh ideas that rarely fit textbooks but often fit the real needs of users.
Toxicity Research
Questions about safety can make or break a compound’s future. 4-Amino-N,N-Dimethylaniline and its derivatives have generated health studies for decades, with researchers diving into enzyme interactions and metabolic fate in living systems. Evidence points to possible liver toxicity and raises eyebrows about its risk to those using large quantities over a career. Researchers advocate for alternatives in sensitive applications, and the pressure sharpens safety protocols everywhere the molecule persists. Calls for more comprehensive long-term studies and clear labeling won’t end while laboratory work and industrial use persists—no chemical, no matter how old or established, escapes this scrutiny these days.
Future Prospects
Sustainable chemistry now shapes the dialogue about every specialty chemical, and 4-Amino-N,N-Dimethylaniline Oxalate faces the same pressures as the rest. Companies and universities both feel the need to minimize waste, boost atom economy, and seek safer alternatives. Engineers explore new synthetic pathways to cut environmental risk; academic groups look at structure modifications that could preserve function without as many health warnings. Ultimately, this compound’s future may hinge on whether the benefits in color synthesis, detection work, and pharmaceutical research keep outpacing the pressures from toxicity and regulatory oversight. Innovation—both in process and in policy—carries the most weight, and there’s no shortcut to the steady, hands-on work driving both.
What is 4-Amino-N,N-Dimethylaniline Oxalate used for?
Why Chemists Rely on 4-Amino-N,N-Dimethylaniline Oxalate
In research circles, 4-Amino-N,N-Dimethylaniline Oxalate shows up on the pages of chemical catalogs. This compound, made for the lab bench more than the household, takes a real role in dye chemistry. It adds value to color laboratories, serving as an intermediate for synthesizing certain dyes, especially those tied to azo and methine colors. This stuff helps chemists shape colorants that end up in products we see every day: printer inks, fabric stains, plastics, and sometimes scientific test kits.
Back in the lab, the compound delivers a chemical structure that links well during synthesis. This lets researchers create new color molecules, check reaction pathways, and even design molecular tags used in some diagnostic tests. Spending part of my early career assisting in synthetic chemistry, I watched how small tweaks in compounds like this could change a molecule’s color, light absorption, or stability. One small change in the amino group and, suddenly, a whole new palette emerges. Maybe a chemist’s hands carry more stains than a painter’s, but the goal remains the same: create the hues society wants, from bright reds to deep blues.
Role in Analytical Science
Quality control labs sometimes turn to this oxalate salt for chemical tests. Analytical chemists know that specific agents react with target substances, producing colors that speak volumes about purity or concentration. For example, some redox titrations and trace metal detections use derivatives of this molecule. Its sensitivity to light and reducing agents helps build assays where a visible color tells you—almost instantly—that something is present or missing.
I bumped into this during a stint with an environmental testing group. Trace metal tests for water quality in some older methods called for color-forming agents built on aromatic amines. Handling these tests, you quickly learned to treat every powder with respect, and always double-check the right safety data sheets.
Health and Environmental Impact
Stories about aromatic amines and health risks go back decades. Evidence points to the possibility that certain compounds in this class could affect health if used improperly. Industrial hygiene codes require special storage and serious attention to personal protective equipment. Long gloves, closed containers, strong ventilation—these aren’t just suggestions; they’re musts. I remember the safety drills and the conversations with health officers about keeping exposures close to zero.
Out in the world, regulators keep tabs on manufacturing and handling. Proper disposal means incineration at facilities set up for hazardous waste, not tossing in everyday trash. One slip at the lab bench or in the waste stream, and the risks grow: contaminated ground water, chemical exposures, or even lingering traces in recyclable plastics. Lab managers and environmental professionals keep pushing for greener alternatives—safer colorants, less toxic reagents, and recycling programs that work.
Room for Improvement
Many chemists work today on new dyes or analytical agents that deliver bright colors and accurate tests, but with less toxicity and easier breakdown in nature. Grants and regulations encourage this shift. Training grows ever more thorough, because everyone—from students to career scientists—shares the goal of safer workplaces and cleaner rivers.
The journey doesn't stay in the lab. Every new discovery in this space offers the promise of more sustainable colors and safer chemistry in the products the world relies on, from diagnostics to fashion to industrial design.
What is the chemical formula and molecular weight of 4-Amino-N,N-Dimethylaniline Oxalate?
The Formula: Cutting Through the Chemistry Jargon
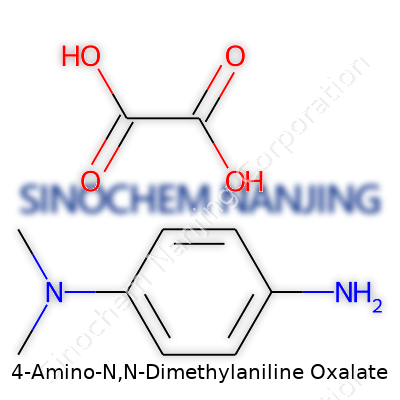
In the world of organic chemistry, small changes in molecules can make a big difference. 4-Amino-N,N-Dimethylaniline Oxalate brings together an aromatic amine and an oxalic acid salt. If you map it out, the free base structure is C8H12N2, but when it teams up with oxalic acid, the salt becomes C8H12N2·C2H2O4. In simpler terms, the oxalate part acts like an anchor, keeping the amine from misbehaving.
Put together, each formula unit holds eight carbons from the aniline portion, two more from oxalate, a dozen hydrogens, two nitrogens, and four oxygens. That tells you there's both an organic backbone and enough oxygen to make it a true salt.
Molecular Weight: It’s Not Just a Number
Add up the atoms. Carbon gives 12.01 g/mol and there are 10 of them. Hydrogen chips in 1.01 g/mol times 14. You get 2 nitrogens at 14.01 g/mol and 4 oxygens at 16.00 g/mol. Total it out. The molecular weight sits at about 236.26 g/mol. That's bigger than the free base by almost exactly the amount contributed by the oxalic acid.
This number might seem like trivia, but molecular weight often marks where theory meets reality. Chemists use it every day. Whether it's packing the right mass into a reaction vessel or calculating dosing for research, the wrong value wrecks results. I’ve watched skilled bench scientists toss out data after weighing out too much or too little, all because of a misprint in a supplier sheet.
Why Accuracy Means Everything in the Lab
Trust in a reported chemical formula saves hours for people in the lab. I’ve helped graduate students trace a mysterious reaction yield back to a typo in molecular weight. A simple swap from free base to oxalate salt made the experiment look off by more than 30%. Dollars, time, and nerves were lost. The right formula, the right weight, and results look a lot less foggy.
Beyond the bench, chemical data gets plugged into industry databases, patent filings, and drug development pipelines. One missed atom means expensive miscommunication. When companies get raw materials, purity and weight become legal matters. Any mistake balloons costs or risks compliance headaches with regulatory watchdogs. If a process chemist runs a batch based on a faulty formula, the cascade can run up six figures in damages or recalls.
Building Trust With Reliable Chemical Data
Groups like Sigma-Aldrich, Merck, and TCI publish everything online because trust only builds with transparency. The scientific community demands more than just numbers—they expect robust sourcing, method notes, and even MSDS backup to confirm molecular weights and structures. I always double-check new stocks against authoritative chemical registries or journal references before using them. Reputable databases like PubChem or ChemSpider help avoid repeating old mistakes.
Solutions for Fewer Headaches
The answer isn’t just better note-taking. Digital tools now auto-calculate molecular weights and cross-check known structures. Training early-career researchers to inspect formulas and registry data before purchase can slash errors. Instructors should walk students through manual molecular weight calculations using chemical handbooks or online calculators before trusting supplier catalog numbers. Many errors fade when fresh eyes scrutinize protocols and data sheets.
In the end, clear and accurate reporting of chemical formula and molecular weight powers reliable science. It also prevents the domino effect that a single slip of pen or database entry could invite. The details matter, and nobody should be content until those details match up across every resource relied on in the lab and beyond.
How should 4-Amino-N,N-Dimethylaniline Oxalate be stored?
A Chemist’s View from the Bench
Spending time in the lab, I’ve handled a long list of chemicals, sometimes arcane, occasionally infamous. 4-Amino-N,N-Dimethylaniline Oxalate doesn’t turn heads the way mercury or hydrofluoric acid do, but take it lightly and you risk unexpected trouble. Respect in storage makes all the difference, and years working with complex organics underlined this: small details keep people safe as much as any glove or fume hood.
Sensitivity and Main Risks
Compounds in this family can show sensitivity to light and air. I’ve seen bottles change color or clump when someone shrugs off a tightly closed stopper, or leaves stock near a sunny window. These changes often signal degradation, which invites waste and surprises—sometimes hazards, sometimes unreliable results.
The material doesn’t tolerate moisture well. Oxalates draw in dampness, turning powders into sticky messes. A humid storeroom usually announces itself—clumsy lids, swollen seals, crystals caked at the rim. Once moisture creeps in, break-down accelerates, and weighing out a clean sample becomes a headache.
Temperature and Light Matters
Refrigerated storage has always proven to be the best option. Colleagues stashing oxalate salts at room temp sometimes circle back to find them unfit for purpose. Low temperatures slow chemical reactions, keeping the material stable. None of my mentors recommended storing sensitive amines above 8°C, and for good reason—treating them like table salt only guarantees short shelf life.
Opaque or amber bottles serve as a safeguard. Exposure to regular lab lighting, not even direct sunlight, can spark decomposition, sometimes releasing fumes that make the storeroom reek. Stored the right way, the material lasts long enough for whole research projects; carelessness sends stock to waste disposal after just a few months.
Container Choices—A Dose of Reality
Glass wins over plastic in my experience. Polyethylene jars often allow slow seepage of air—and over the years, this adds up. Screw caps with tight-fitting seals, accompanied by a layer of parafilm, keep the oxalate powder dry and untouched by ambient oxygen. Labels, always clear and date-stamped, prevent guessing games during busy stretches.
Practical Handling and Spill Management
Most accidents don’t happen during experiments; they happen during storage or waste handling. Keeping the oxalate in small, clearly labeled containers helps. Opening only what you need ensures the bulk quantity stays untouched and uncontaminated. Once, a well-meaning tech tried to scoop powder with a damp spatula. Moisture ruined the whole batch. Clean, dry tools remain non-negotiable.
If you spill any, treat it like you would with any finely ground salt that might be toxic: damp towels for cleanup, gloves on, plus immediate disposal in proper chemical waste containers. Ventilation helps, too. Many storerooms lack airflow; small spills linger in the air and trigger headaches or worse.
The Broader Why—People and Results
Good storage habits reflect a deep respect for both science and personal safety. Laboratories lose research hours every year from unstable chemicals and minor exposures. I’ve seen promising projects halted by something as basic as a poorly capped bottle. Organizations like OSHA echo these points, backing up common sense with clear policies. A tightly run storeroom isn’t just a feature of big labs; it’s a baseline set by good practice.
Factories and research labs around the world echo the same priorities: shield stock from air, light, and moisture, keep things cool, handle with awareness. Whether someone works in pharmaceutical synthesis, dyes, or analytical chemistry, the lessons translate across the board. Safety and reliability stem from choices made before any experiment starts.
What is the purity level of the 4-Amino-N,N-Dimethylaniline Oxalate product?
Why Purity Shapes Research Outcomes
The moment you see a chemical reagent like 4-Amino-N,N-Dimethylaniline Oxalate on a supplier’s website, questions about quality jump to mind. Chemists, pharmacists, and lab techs all want to know—how pure is this stuff, really? In my years working inside both teaching labs and applied research outfits, nothing causes more headaches than an impure starting material. Dead ends, garbled data, or ruined syntheses often trace back to lackluster reagents. Purity isn’t a side issue—it directs the whole experiment.
Standard Purity Levels: What You See and What You Get
Reagents often land on the bench labeled as “analytical grade,” “reagent grade,” or simply with a percentage indicating their purity. Most credible suppliers list 4-Amino-N,N-Dimethylaniline Oxalate as at least 98% pure. This percentage means by mass, nearly every gram in the bottle is what it says on the label. The rest comes from manufacturing leftovers—salts, traces of solvents, or incomplete reaction products. A published assay or certificate of analysis usually backs up the claim.
Folks involved in sensitive sectors—like pharmaceuticals or forensics—rarely settle for less than 98%. The remaining small percentage might seem minor. Yet, even trace impurities sneak into reactions, skew instrument readings, or trigger false positives. In teaching labs, maybe such high standards aren’t always practical, but in my own work with sensitive photochemicals, pushing toward that upper limit brought peace of mind and reproducibility.
How Purity Gets Verified
Gone are the days of taking suppliers purely at their word. Any reputable company runs batches through techniques like HPLC, NMR, or mass spec. These tools slice up molecular profiles and spotlight minute impurities far better than older colorimetric tests or melting point checks. Experienced chemists know how to read these batch reports. If a supplier can’t produce this data or wants to dodge pointed questions, consider that a red flag. Quality environments demand transparency, not guesswork.
Back in my own graduate research, a series of contamination-driven failures taught me to always double-check each reagent. I tested even “fresh, sealed” bottles before big syntheses. A fifteen-minute check with thin-layer chromatography stopped some costly mistakes and kept projects moving.
The Bigger Picture: Practical Routes to High Purity
Some users might find themselves wrestling with a subpar lot. This isn’t the end of the road. Recrystallization—dissolving and re-forming the product—often pushes purity higher. This old-school practice, still around for good reason, can make all the difference, especially for a compound like 4-Amino-N,N-Dimethylaniline Oxalate which shows solid, stable properties useful for such purification.
Choosing known suppliers and steadily asking for certificates bolsters reliability. Labs that frequently rely on custom syntheses develop relationships with trusted vendors and implement in-house quality checks. Looking at the broader supply chain, country of manufacture, batch documentation, and logistics all play into the trust calculus, especially after years of disruptions shown by the pandemic.
High-purity input paves the way for real discovery. Without it, not even the sharpest analysis or most innovative method can correct systemic bias. For scientists and industrial users alike, asking sharp questions about every product lot marks the only reliable starting point.
Are there any safety or handling precautions for 4-Amino-N,N-Dimethylaniline Oxalate?
Understanding the Substance
4-Amino-N,N-Dimethylaniline Oxalate rarely shows up in your average office, but it plays a role in research and some specialized industrial processes. Its structure signals possible health concerns. A compound like this isn’t something to take lightly, and old stories from chemistry labs make that obvious.
Why Precautions Matter
Seeing colleagues get careless and ending up with irritated skin, headaches, or worse, teaches a person to take safety seriously. This compound falls into a group of organic amines known to cause trouble if handled the wrong way. The oxalate part doesn’t help—oxalates can irritate tissue and even cause kidney stones if people face chronic exposure. A spill or mistake, even a small one, leads to consequences the user may regret down the line.
The Real Dangers
4-Amino-N,N-Dimethylaniline Oxalate can absorb through the skin. It doesn’t need much time to irritate or bring on allergic reactions. Accidentally inhaling dust or vapor delivers another set of problems: sore throat, headache, and potential damage to deeper tissues in severe cases. Swallowing? That marks a real emergency. Even indirect exposure matters; this substance loves to cling to surfaces and contaminate equipment.
Safe Storage Practices
Lessons from years in shared labs always point to storage as a first line of defense. Keep these materials in tightly sealed containers, away from acids, strong oxidizers, and anything reactive. Shelves must stay dry and cool. Too often, sloppiness with temperature or nearby chemicals turns a safe stockroom into a ticking time bomb. Label everything in large, clear print. One wrong grab could send an inexperienced lab mate to the emergency room.
Personal Protective Equipment
Anyone working with this substance shouldn’t settle for vinyl or latex gloves—nitrile provides better resistance. Eye protection isn’t optional; goggles with side shields block any chance of a splash hitting your face. Wearing a lab coat, and leaving it at work, stops the compound from hitchhiking home on your clothes. Good ventilation helps; open windows and fume hoods reduce chances of breathing in any fumes or fine particles. Don’t skip these steps, even if the work seems routine.
Cleanup and Waste Disposal
Small spills demand action right away. Paper towels don’t cut it. Use absorbent materials labeled for chemical cleanup and seal them in a bag for hazardous waste disposal. Never pour leftovers down the drain. Local rules often require sealed, labeled containers headed to specialized waste sites. People ignoring these regulations risk fines and harm to public water sources. Remind colleagues and new staff at every turn—the rules keep everyone safer.
Training and Culture
Maybe the best precaution is a healthy respect for what’s on the workbench. Encourage honest conversations about accidents and near-misses. Lab leaders set the tone. One slipup or shortcut sets an example, good or bad, for the team. The best-run labs I’ve seen invest in regular training—even refreshers for seasoned workers. People who recognize the dangers become better at protecting not just themselves, but also everyone else in the space.
Building a Safer Routine
Treat every moment with hazardous materials like a drill for a bigger emergency. Double-check gloves. Inspect goggles for cracks. Test your knowledge of cleanup protocols before disaster strikes. Spread these habits through the organization, and incidents drop—sometimes to zero. Smart, cautious handling of 4-Amino-N,N-Dimethylaniline Oxalate isn’t just about rule-following—it’s about respect for your own health and for those working alongside you.
| Names | |
| Preferred IUPAC name | 4-amino-N,N-dimethylaniline ethanedioate |
| Other names |
4-Amino-N,N-Dimethylaniline oxalate p-(Dimethylamino)aniline oxalate N,N-Dimethyl-p-phenylenediamine oxalate N,N-Dimethyl-4-aminobenzene oxalate p-Amino-N,N-dimethylaniline oxalate |
| Pronunciation | /fɔːr-əˈmiːnoʊ ˌɛn ˌɛn daɪˈmɛθɪlˈænɪˌliːn ɒkˈseɪleɪt/ |
| Identifiers | |
| CAS Number | 16380-47-1 |
| Beilstein Reference | 136489a |
| ChEBI | CHEBI:87211 |
| ChEMBL | CHEMBL4135872 |
| ChemSpider | 160856 |
| DrugBank | DB08722 |
| ECHA InfoCard | 07eaa996-bb4b-484a-aba3-48a2f5e07d27 |
| EC Number | 6108-16-3 |
| Gmelin Reference | 92952 |
| KEGG | C14647 |
| MeSH | D08.811.277.040.330.200.150.180 |
| PubChem CID | 168843 |
| RTECS number | BW6650000 |
| UNII | 66064TVE5E |
| UN number | 2811 |
| CompTox Dashboard (EPA) | DTXSID2058288 |
| Properties | |
| Chemical formula | C8H12N2 · C2H2O4 |
| Molar mass | 278.29 g/mol |
| Appearance | Light yellow to light brown crystalline powder |
| Odor | odorless |
| Density | 1.18 g/cm³ |
| Solubility in water | Soluble in water |
| log P | -0.07 |
| Acidity (pKa) | 6.1 |
| Basicity (pKb) | 7.74 |
| Magnetic susceptibility (χ) | -45.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.663 |
| Dipole moment | 3.54 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 274.5 J·mol⁻¹·K⁻¹ |
| Pharmacology | |
| ATC code | D06BA08 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause allergic skin reaction. |
| GHS labelling | GHS05, GHS07 |
| Pictograms | GHS07 |
| Signal word | Danger |
| Hazard statements | H302 + H312 + H332: Harmful if swallowed, in contact with skin or if inhaled. |
| Precautionary statements | Precautionary statements: P261, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 2-2-0 |
| Lethal dose or concentration | LD₅₀ (oral, rat): 600 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral (rat) 1650 mg/kg |
| NIOSH | DJ9350000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 4-Amino-N,N-Dimethylaniline Oxalate: Not established. |
| REL (Recommended) | 0.05 mg/m³ |
| Related compounds | |
| Related compounds |
4-Amino-N,N-Dimethylaniline N,N-Dimethylaniline p-Phenylenediamine Aniline 4-Aminophenol |

