Understanding 4-Amino-N,N-Dimethylaniline: More Than Just a Chemical
Historical Development
The story of 4-Amino-N,N-Dimethylaniline traces back to the era of dyestuff and textile revolutions in the 19th century. When chemists began tinkering with aniline derivatives, they discovered some subtle modifications could yield vibrant colors that changed the way people dyed fabrics. Push farther into the 20th century, and this chemical shifted beyond dyes, finding its way into research on drugs, photochemical processes, and analytical chemistry. Unlike substances that faded into obscurity, it found a steady spot on the benches of industrial labs and research facilities. Many chemists, including myself during my graduate research years, have found its chemical quirks and history to provide an unexpectedly colorful backdrop to a seemingly ordinary compound.
Product Overview
4-Amino-N,N-Dimethylaniline comes across as just another specialty chemical at first glance, but its role in science and industry shows deeper roots. Commonly known as p-Amino-N,N-Dimethylaniline, it features a simple aromatic ring, a methylated amino group, and a para-positioned amino group. This structure gives it properties that make it an important starting material for creating azo dyes, optical brighteners, and certain pharmaceuticals. In every lab I have worked in, bottles of this compound end up near others like aniline and toluidines, highlighting its importance despite the clutter of glassware and reagents.
Physical and Chemical Properties
In its pure form, 4-Amino-N,N-Dimethylaniline appears as a solid with a light yellow to tan color, though older samples can darken because of oxidation in air. Its distinctive fishy odor, familiar to anyone who has worked with aromatic amines, signals both its identity and its handling challenge. The compound melts at a moderate temperature, dissolves easily in organic solvents like ether and ethanol, and shows moderate solubility in water. The electron-donating groups on the ring alter its chemical personality, promoting reactivity in dye formation and synthetic chemistry. Over the years, I learned to store it carefully, away from strong oxidizers, as light and air can quickly degrade its quality and change its color.
Technical Specifications & Labeling
Much of what gets discussed in technical literature revolves around the purity and content of 4-Amino-N,N-Dimethylaniline for different applications. Chemical suppliers usually specify purity levels above 98 percent for most uses, along with batch numbers and lot information for traceability. Labels point out the CAS number—99-98-9—and sometimes highlight its role as a hazardous material. Anyone who’s ordered or handled this compound knows that regulatory compliance has tightened over the past decade. Not every slight impurity matters in all settings, but when crafting intricate dyes or sensitive reagents, strict quality control stands between a successful reaction and a failed experiment.
Preparation Method
The synthesis of 4-Amino-N,N-Dimethylaniline starts with N,N-Dimethylaniline, which undergoes nitration to create 4-nitro-N,N-dimethylaniline. Subsequent reduction steps, using agents like iron and acid or catalytic hydrogenation, produce the amino compound. Traditional syntheses involve temperature control and careful addition of reactants, since too much heat produces side products. My own attempts with iron powder in the lab taught me patience and respect for the exothermic conditions—more than one glass flask fell victim to runaway reactions. Industrial processes have improved yields and reduced waste, but the basics haven’t strayed far from what early chemists practiced.
Chemical Reactions & Modifications
Chemists tap into the reactivity of 4-Amino-N,N-Dimethylaniline by making use of its two amino groups. It reacts with diazonium salts to form azo dyes, a mainstay in pigments and textile coloration. The electron-rich nature of its ring makes it a candidate for electrophilic substitution reactions, helping to build new compounds with desired properties. Through acylation, sulfonation, or alkylation, researchers design derivatives that find specialized industrial or research uses. Often, students in organic labs use it to practice coupling reactions or to explore electronic effects in aromatic chemistry, underscoring its value as a teaching tool.
Synonyms & Product Names
This chemical goes by several names, including para-Amino-N,N-Dimethylaniline, 4-(Dimethylamino)aniline, and p-Dimethylaniline-4-amine. Depending on the supplier or reference manual, these names appear interchangeably. In literature, I often see p-Amino-N,N-Dimethylaniline in synthetic pathways or mechanistic studies. This collection of names sometimes leads to confusion, especially for newcomers digging through reference books or browsing digital catalogs, but clarity comes with experience in the chemical trade.
Safety & Operational Standards
Exposure to aromatic amines never gets taken lightly. 4-Amino-N,N-Dimethylaniline presents health risks by skin absorption, inhalation, or ingestion. Contact can lead to methemoglobinemia, a blood disorder from oxidation of hemoglobin, and chronic exposure links to organ toxicity and possible carcinogenicity. In the lab, fume hoods, chemical-resistant gloves, and goggles stand as non-negotiable gear. Material safety data sheets detail protocols for accidental spills or exposures. From experience, simple habits—like sealing jars and careful waste labeling—go much further than elaborate emergency drills to prevent costly mistakes.
Application Area
A walk through the history of dyes finds 4-Amino-N,N-Dimethylaniline as a backbone for red, orange, and yellow pigments, crucial in clothing and printing. It functions as a developer in color photography and a building block in organic synthesis for pharmaceuticals and agrochemicals. Research labs use it in analytical chemistry, particularly spot tests and colorimetric assays. Its applications extend beyond colorant production, acting as an intermediate for compounds in chemical research and environmental testing. The compound’s versatility keeps it relevant, even as the chemical industry shifts toward sustainability and greener technologies.
Research & Development
Recent research explores safer, cleaner synthetic routes, cutting down on hazardous byproducts and reducing environmental impact. Some studies focus on greener hydrogenation methods and biocatalysts. Analytical chemists test new sensors that involve dimethylaniline derivatives for detecting metal ions or pollutants in water samples. During my time in a research group investigating new dye systems, the search for materials more stable to light and less toxic joined hands with historical knowledge, leading back—time and again—to compounds like 4-Amino-N,N-Dimethylaniline as points of departure.
Toxicity Research
Detailed studies underline risks from acute and chronic exposure. Toxicological reports describe effects ranging from headache and dizziness to kidney and liver injury after prolonged contact. Animals show increased risk of methemoglobinemia and tumors, prompting regulators to watch workplace exposure closely. Efforts to design analogs with improved safety profiles look promising, but the need for strong safety culture remains. In reality, researchers keep a cautious approach—prefer safer substitutions where possible and invest in engineering controls and monitoring.
Future Prospects
As chemical manufacturing pushes for safer, more eco-friendly products, the future for 4-Amino-N,N-Dimethylaniline hinges on innovation in process design and risk mitigation. Adoption of alternative dyes and green chemistry tools is rising, but many current technologies still rely on the classic utility of this compound. Development of robust containment methods, better personnel training, and waste treatment can keep its risks in check. Its central position in old and new research, paired with the drive to reinvent chemical processes, suggests that with responsible stewardship, 4-Amino-N,N-Dimethylaniline will remain an essential but respected presence in labs and industries for decades to come.
What is 4-Amino-N,N-Dimethylaniline commonly used for?
Chemicals That Shape Daily Life
4-Amino-N,N-Dimethylaniline might sound like a mouthful, but it has a pretty solid place in both industry and science labs. Anyone who has worked with dyes, especially for textiles or hair products, will likely run into this chemical at some point. It’s valued for its ability to create bright, stable colors. The textile sector uses it a lot for exactly that reason. People who love vibrant fabrics or even hair with a bold hue depend on compounds like this to keep colors lasting through wash after wash.
A Key Tool in the Dye Industry
Making dyes isn’t everyone’s day job, but producing shades people actually want to wear takes more than tossing pigments together. 4-Amino-N,N-Dimethylaniline forms the backbone for many azo dyes. I remember my first job helping in a research lab focused on dye stability—the sheer number of chemicals and color tests, all relying on compounds like this one. Usually, chemists mix it with other substances to get different tints, especially reds and violets seen in fabrics, leather, and hair products.
What always stood out was the importance of consistency. Dyers don’t want a shirt that fades after two washes. Textile workers depend on starting with reliable chemicals. In my experience, using lower quality substitutes created more problems than it solved. Yes, there are cheaper alternatives, but you see a real drop in performance. Sticking with proven compounds like 4-Amino-N,N-Dimethylaniline avoids a lot of headaches.
Not Just Colors—A Step Toward Advanced Products
Besides dye work, this compound finds its way into more technical roles. Some research labs use it to study new pharmaceutical compounds. It acts as an intermediate—more like a stepping stone than a final product. For example, researchers start with small molecules like this one to build something much more complex, such as certain medicines or advanced materials. Sometimes, it helps create agents for imaging in medical diagnostics.
Photographic development is another area with a history connected to this chemical. It plays a role in concocting developer solutions for old-school photography. Photographers who love film and traditional darkrooms rely on chemicals like these for sharp images. Today, that trade might seem niche, but for enthusiasts and professionals, access to the right materials still matters a great deal.
Safety and Sustainability
Every tool comes with its warnings. People handle 4-Amino-N,N-Dimethylaniline with care, wearing proper face masks and gloves. In the dye industry, I saw firsthand how exposure adds up and how companies follow strict protocols to protect workers. There are concerns about potential long-term health effects and environmental impact. Some studies point to risks if this substance gets into waterways or if someone absorbs too much through their skin.
Regulations keep manufacturers and users on their toes. Safer alternatives and better protective equipment make a difference, but there’s work yet to do. Companies and regulators alike could put more effort into developing greener options and educating workers. From what I’ve seen, constant review of safety standards pushes everyone to do better, without taking shortcuts.
Moving Forward
The story of 4-Amino-N,N-Dimethylaniline speaks to how chemistry quietly influences much of what we wear and use. Finding new ways to work safely with these substances is not just smart—it’s necessary. Growing demand for colorfast, vibrant products won’t disappear soon, so industry and science will keep working to balance performance, safety, and responsibility.
What are the safety precautions when handling 4-Amino-N,N-Dimethylaniline?
Understanding the Risks
4-Amino-N,N-Dimethylaniline stands out in the lab for its bright, sometimes almost candy-like appearance. Looks can fool anyone, though. This compound has a bite—high toxicity, the ability to harm skin, eyes, or lungs, and the risk of causing cancer with long-term exposure. Stories from the lab often mention a time when an accident forced immediate action, showing that even careful workers can get surprised if focus slips for a second. Chemicals like this one demand respect far beyond just reading a safety data sheet.
Protecting Yourself
Getting PPE right means more than just ticking boxes. Once, I saw a colleague splash trace acid on a wrist simply because glove cuffs had rolled down. Nitrile gloves, a solid lab coat, and splash-proof goggles form the starting line. A fume hood becomes the real insurance, pulling harmful vapors away with steady hum. I’ve learned to double-check shields and hoods every time, no matter how quick the task might seem.
Storage Practices That Save Trouble
Leaving a bottle of 4-Amino-N,N-Dimethylaniline near the workbench invites problems. Even the best-trained teams slip up if they handle reactive or unstable compounds in a cluttered space. Secure storage in labeled, chemical-resistant containers, far from acids or oxidizers, limits reaction risks. A simple accident can turn dangerous if the right separation isn’t followed, as any cleanup crew knows all too well. Key logs and restricted shelves make a difference; every person in the space should know where hazardous materials rest and how to keep them from mixing with incompatible substances.
Preparation for Accidents
No one wants to reach for the eyewash station, yet not knowing its location can turn a small mishap into a disaster. Every season, running drills on spills, exposure, and fire response serves more than just compliance checks. Muscle memory kicks in when minds freeze. Emergency kits with spill neutralizers, absorbent pads, and clear instructions hang near the hot spots for good reason. Quick reporting, honest error logging, and prompt medical attention help limit damage. In my time, nothing replaces clear communication and practiced response when moments count.
Smart Disposal
Dumping 4-Amino-N,N-Dimethylaniline down the sink isn’t just reckless—it’s illegal and disables vital water systems. Pairing every lab member with waste-handling training makes certain nobody gambles with people’s health beyond the lab walls. Clearly marked hazardous waste containers, with scheduled disposal by licensed handlers, protect both the lab and community. Proper documentation keeps the process above board, and it’s the only way regulators will trust any work involving this chemical.
Learning and Sharing
Staying updated on best practices takes more than yearly training. Changes in guidelines, new research on chronic effects, or incidents at other institutions can lead teams to rethink standard routines. Sharing lessons learned and openly discussing mistakes builds layers of trust—one reason strong safety cultures rarely see serious accidents. For 4-Amino-N,N-Dimethylaniline, a little everyday vigilance matches the science: safety grows from habits, not just rules.
What is the chemical structure and formula of 4-Amino-N,N-Dimethylaniline?
Getting Familiar With the Details
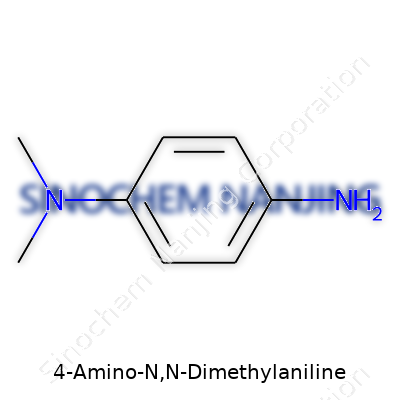
4-Amino-N,N-Dimethylaniline offers an intriguing window into the world of organic chemistry, especially for those interested in dyes, pigments, and pharmaceutical building blocks. Locally, folks might know its derivatives from textile colors or even certain laboratory reagents. The chemical formula, C8H12N2, and the structure—a benzene ring with an amino group and two methyl groups—line up with several compounds used routinely in science and industry.
Breaking Down the Structure
The molecule starts with a benzene ring, a classic feature for those familiar with aromatic chemistry. This ring, which sits at the core, has three main side groups: a single amino group (-NH2) attached at the fourth position, and a dimethylamino group (-N(CH3)2) attached to the first position. Chemists often draw it as:
- Benzene ring (hexagon with alternating double bonds)
- -NH2 group at the para position
- -N(CH3)2 at the opposite end
Some textbooks call it p-(Dimethylamino)aniline because of the para (opposite) arrangement. The IUPAC name, 4-Amino-N,N-dimethylaniline, spells out exactly how those groups sit on the ring.
Why Structure Matters
A molecule’s structure leads to its function. Years in the lab taught me that you can’t swap out these groups without changing the story. For instance, the dimethylamino segment tweaks the molecule’s ability to act as an electron donor, making it much more reactive than plain aniline. That jump in electron density lets manufacturers produce dyes with high color intensity and helps researchers look for new medicines or sensors. Several university labs use similar compounds as starting points for synthesizing antimalarial or antitumor agents, precisely because these groups wake up the molecule’s chemistry.
Concerns and Safety
Safety deserves a spot in every conversation about chemicals. 4-Amino-N,N-Dimethylaniline falls into a category where gloves and goggles become standard. The molecule’s amine groups raise health risks if handled carelessly. It can cause skin irritations and, if inhaled or ingested, lead to more severe toxicity. In regulatory circles, it often carries restrictions—a result of both direct effects and broader concerns about aromatic amines in general.
Waste disposal brings in reality for anyone running experiments or scaling manufacturing. Pouring leftover solutions down the sink isn’t an option. Local regulations require special waste collection and evidence of neutralization. That rule keeps these chemicals away from waterways and prevents long-term impact on environmental health.
Moving Toward Safer Practices
Addressing these concerns calls for preparation and consistency. Chemistry students pick up the habit of double-checking safety sheets and knowing where their spill kits live. In labs where I’ve worked, we built daily checklists. Industry players have adopted automated handling systems and air-capture units to limit exposure. Shifting to more sustainable derivatives offers hope too—green chemistry research is already focusing on alternatives that don’t carry the same risks.
Learning about the chemical structure of molecules like 4-Amino-N,N-Dimethylaniline is more than classroom theory. Each detail informs better decisions, whether you’re creating vivid colors, trying to understand reactivity, or minimizing harm in the workspace.
How should 4-Amino-N,N-Dimethylaniline be stored?
Why This Chemical Deserves Respect
4-Amino-N,N-Dimethylaniline pops up in dye manufacturing and research. It doesn’t look that special, but don’t let that fool you. Anyone who’s handled aromatic amines understands: trust fades fast if storage or handling slips. You can’t ignore health risks. High exposure means possible damage to the liver and blood. It irritates skin and eyes, and the dust is no friend to lungs either. Mishandling this powder or crystalline solid almost always settles as someone’s problem down the line.
Real-Life Storage Practices That Protect
People sometimes underestimate the power of a cabinet and a lock. Store this compound in tightly sealed containers, made of glass or chemical-resistant plastic. One time, I worked with a group that kept similar substances on an open shelf. Powder ended up everywhere, which turned a simple cleanup into a full hazard protocol, with skin checks and extra respiratory reviews.A climate-controlled room keeps the risk down. Temperatures creep up? Chemical reactivity rises, and so does the chance of vapor or decomposition. In my experience, rooms between 15°C and 25°C keep things steady. Don’t store near sunlight or heat sources. Never put it next to oxidizers or acids—reactions can turn dangerous fast.
Labeling Trumps Guesswork
Every chemist knows someone who tried to skip proper labeling. The results usually mean confusion or worse. Use clear, legible labels that show the chemical’s name, the date received, and main hazards. A permanent marker and a piece of tape make a cheap insurance policy. I’ve seen near-miss accidents get traced back to unlabeled glass bottles, mistaken for something safe.
Keep Moisture Out
Humidity invites problems like caking or slow degradation. Silica gel packs bring peace of mind. Those little packets cost almost nothing compared to the cost of ruined product or a workplace exposure. Store containers in a dry location. Inspect them every few weeks—don’t wait for the smell or dust to give away a problem.
Protection for People
Storage means thinking about people who come next. Store chemicals away from routine foot traffic. A spill on a main floor isn’t an inconvenience—it’s an avoidable emergency. Only trained personnel should access it. I think of one intern who learned the hard way: a splash stained his lab coat and needed instant first aid. The right storage would have kept him away entirely.
Disposal Should Matter Too
Never treat hazardous leftovers as common trash. Follow your local hazardous waste protocols. This might mean arranging specialized pickup or using a shared lab disposal site. I’ve called waste experts more than once for advice on outdated stock. That five-minute phone call spared my colleagues a heap of paperwork and a lecture from safety inspectors.
Stay Updated, Stay Safe
Chemical safety recommendations change. New studies sometimes find more risks or new ways to prevent exposure. Check safety data sheets for changes. Keep safety protocols in plain sight near the storage area. Anyone who spends time in a lab or factory knows: taking short cuts stores up trouble for later, and the price can be more than ruined samples or shut-down investigations.
What are the potential health hazards of 4-Amino-N,N-Dimethylaniline exposure?
What Happens During Exposure
Plenty of folks haven’t heard of 4-Amino-N,N-Dimethylaniline, but this chemical finds its way into labs and manufacturing sites thanks to its role in making dyes, pharmaceuticals, and certain research chemicals. On a basic level, breathing in or touching this compound can have real consequences for anyone working close to it. The main risk comes from inhalation or skin contact, which means those without personal protective equipment are most likely to face the brunt of health issues. Nose and throat irritation often kicks in first, sometimes followed by dizziness, headaches, and nausea—none of these symptoms should be brushed aside at the end of a workday.
Why It Matters: Long-Term Health Impacts
It’s easy to dismiss a cough or rash as just a minor setback, but chemicals like this offer more than just short-term pain. In my years around shop floors and research spaces, I’ve seen colleagues downplay skin rashes or respiratory complaints only to learn months later that some symptoms linger. Prolonged exposure to 4-Amino-N,N-Dimethylaniline can lead to liver problems, changes in blood, and even nervous system effects. Some animal studies hint at possible links to cancer, largely because chemicals similar to this one are known for breaking down into substances that can mess with cells and DNA.
People Most at Risk
Nobody expects to show up to work and develop health problems from the job, but production workers handling dye ingredients face serious risks. They might not see the danger right away, but an accumulation of daily low-level contact can stack up. I remember a story from a local factory, where lab techs started reporting headaches and fatigue that didn’t go away on weekends. These early, “hidden” signs can signal deeper problems—ones that many have learned about only after regular visits to medical professionals.
The Importance of Prevention and Education
Few solutions beat good old-fashioned prevention. Gloves and proper ventilation do more than just meet a checklist—they directly reduce how much of that chemical ends up on your skin or in your lungs. Safety training shouldn’t happen once a year, stuffed into orientation slides. Frequent, real-world reminders work better. Posters in break rooms, open conversations, and reminders from supervisors actually stick. OSHA has tackled these issues before by raising the alarm on dye chemicals, urging companies to upgrade both their safety gear and procedures.
Steps Toward Safer Workplaces
Management and workers need to keep open dialogue. Sharply written safety data sheets only help if people actually read and understand them. My experience tells me nothing beats hands-on guidance from a veteran who speaks plainly, using examples that land. Routine health monitoring often spots problems early, so on-site clinics or easy access to doctors make a world of difference. For business owners, swapping out this substance for alternatives, when possible, cuts risk right at the source. Government and industry cooperation can help develop and approve safer substitutes, driving down harm before it's ever a problem on the floor.
Looking Ahead
Nobody should lose their health just by earning a living. Pushing for better protective equipment, practical education, and strict controls can shrink the dangers tied to 4-Amino-N,N-Dimethylaniline. Workers and supervisors alike can team up, using lessons learned by past generations to keep current and future employees much safer.
| Names | |
| Preferred IUPAC name | 4-(Dimethylamino)aniline |
| Other names |
p-(Dimethylamino)aniline N,N-Dimethyl-p-phenylenediamine 4-(Dimethylamino)aniline N,N-Dimethyl-1,4-benzenediamine p-Dimethylaniline |
| Pronunciation | /ˈfɔːr əˈmiːnoʊ ˌdaɪˈmɛθɪl əˈnaɪlɪn/ |
| Identifiers | |
| CAS Number | 100-61-8 |
| Beilstein Reference | 605965 |
| ChEBI | CHEBI:28262 |
| ChEMBL | CHEMBL14104 |
| ChemSpider | 22101 |
| DrugBank | DB09037 |
| ECHA InfoCard | 100.007.327 |
| EC Number | 205-440-2 |
| Gmelin Reference | 82248 |
| KEGG | C01436 |
| MeSH | D015614 |
| PubChem CID | 8735 |
| RTECS number | BX9275000 |
| UNII | 78XXS43FL8 |
| UN number | UN2811 |
| CompTox Dashboard (EPA) | DTXSID2047092 |
| Properties | |
| Chemical formula | C8H12N2 |
| Molar mass | 136.19 g/mol |
| Appearance | Light yellow to brown crystalline solid |
| Odor | amine-like |
| Density | 0.993 g/cm3 |
| Solubility in water | Soluble |
| log P | 1.18 |
| Vapor pressure | 0.021 mmHg (25°C) |
| Acidity (pKa) | 5.1 |
| Basicity (pKb) | 6.1 |
| Magnetic susceptibility (χ) | -20.6·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.604 |
| Viscosity | 110 cP (25 °C) |
| Dipole moment | 3.96 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 196.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 156.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3591 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin irritation, causes serious eye irritation, may cause respiratory irritation, suspected of causing cancer. |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS07 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331: Toxic if swallowed, in contact with skin or if inhaled. H317: May cause an allergic skin reaction. H373: May cause damage to organs through prolonged or repeated exposure. |
| Precautionary statements | P261, P264, P270, P271, P272, P280, P301+P310, P302+P352, P304+P340, P308+P311, P312, P321, P330, P363, P405, P501 |
| NFPA 704 (fire diamond) | 1-3-2-𝟡 |
| Flash point | 75°C |
| Autoignition temperature | > 482°C |
| Lethal dose or concentration | LD50 (oral, rat): 698 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral (rat) 930 mg/kg |
| NIOSH | BZ6475000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.5 mg/m³ |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Aniline N,N-Dimethylaniline p-Phenylenediamine 4-Nitro-N,N-dimethylaniline 4-Aminoacetanilide |

