Looking Closer at 4,6-Dinitro-2-Aminophenol: More Than Just a Laboratory Curiosity
Historical Development
4,6-Dinitro-2-aminophenol entered the chemistry scene in the early twentieth century, surfacing as researchers delved into explosive compounds and aromatic amines. Chemists searching for nitro-compound applications found themselves producing this molecule as either a deliberate end or a stepping stone to others. Early patents and papers from Germany nodded to this compound when dye chemistry was thriving, especially for the textile industry. Over decades, shifts in industrial focus brought it back into view for different reasons—sometimes pigment, other times pharmaceuticals or explosives. Those open to revisiting older chemistry can appreciate how knowledge moves in cycles, and the story of 4,6-dinitro-2-aminophenol reflects that.
Product Overview
This compound stands out for its distinct structure—two nitro groups and a single amino and hydroxyl set on a benzene ring. Such arrangement builds reactive potential both in synthetic applications and in finished products. A crystalline orange-yellow solid, it earned a spot first in chemical catalogs via the textile dye trade, though it rarely grabs headlines amid more popular compounds. In labs that tinker with aromatic intermediates, you’re likely to spot it on a back shelf or as a pointed suggestion in a synthesis route discussion. Anyone working with advanced pharmaceuticals, sensitive colorants, or analytical reagents has probably come across it, whether they realized its deeper stakes or not.
Physical and Chemical Properties
4,6-Dinitro-2-aminophenol doesn’t shy away from being reactive—its structure tells the story. Dense and colored, it resists easy melting, which creates challenges in processing. Its dual nitro groups pack oxidative punch, while the amino and hydroxyl moieties open doors for further chemical play. Mixes tend to be stable under the right controlled settings, but introduce heat or a strong reducer and its temperament changes fast. Water solubility sits low, though not zero, and solubility in organic solvents varies just enough to be useful for purification or formulation steps. Even seasoned lab folks remind themselves to keep it dry and away from strong bases or acids—the chemistry demands respect.
Technical Specifications and Labeling
Technical data follows familiar patterns: practitioners focus on purity (typically above 95 percent for key work), color, particle size, and the absence of closely-related nitro-amino isomers. In academic settings, emphasis lands on purity for reproducibility. Professional suppliers must adhere to strict GHS compliance, plain hazard statements, and the kind of labeling that leaves nothing to chance. In Europe, REACH registration is a must before quantities grow large. Chemists don’t just want the product—they need to know exactly what’s in each drum or jar: water content, trace solvents, any stabilizers tossed in for safety. Clear labeling can mean the difference between a routine day and disaster, especially when working with materials boasting such energetic components.
Preparation Method
Methods for making 4,6-dinitro-2-aminophenol evolved from trial-and-error nitration of aminophenols with mixed acid, to careful staged syntheses focused on selectivity and safety. Early-on, uncontrolled nitration could lead to a mess—a mix of unwanted isomers and byproducts. Modern chemists lean on methodical addition of acids under cool conditions, tight control of temperatures, and an understanding of how aromatic substitution walks a narrow path between “done” and “ruined.” Some approaches use protection-deprotection strategies on the phenolic or amino group to avoid chaos in the flask. Anyone who has run a batch can tell stories about runaway froth or overreaction, which underscores why process design remains an active area of study. Yield optimization and waste minimization go hand in hand as environmental standards tighten and disposal costs rise.
Chemical Reactions and Modifications
4,6-Dinitro-2-aminophenol isn’t just a static endpoint. Its reactivity under reduction allows chemists to convert it into a variety of diamines, essential steps in dyes and pharmaceutical synthesis. The hydroxyl group enables ether or ester formation, while the nitro groups themselves can shift to amino functions for further progress up synthetic chains. Anyone invested in azo dye production sees its core layout as ripe for coupling reactions. Nitration, reduction, diazotization—each process builds on what’s already a reactive, dense scaffold. Every time a new downstream molecule pops up in published research, odds are that a nitro-aminophenol sits nearby on the synthetic tree.
Synonyms and Product Names
The academic world often prefers “4,6-dinitro-2-aminophenol” for clarity, but trade and catalog listings toss in a variety of synonyms. You’ll see it as DNAP, 2-amino-4,6-dinitrophenol, or in some contexts referred to by dye names if it lands in pigment-related supply streams. Older literature might mention it under slightly misleading titles, so tying CAS numbers to common language remains important for anyone chasing down supply or reading historic patents. As labs pivot toward digital ordering and regulator databases, harmonization of names cuts confusion and ensures the right material shows up on the loading dock—or gets flagged for careful handling at customs.
Safety and Operational Standards
Handling 4,6-dinitro-2-aminophenol brings challenges—no shortcut works here. Nitroaromatic compounds historically raise flags both as potential explosives and for their health hazards. Skin irritation, toxicity on ingestion, dust inhalation—these form the baseline concerns. No professional touches this material without personal protective equipment, full ventilation, and a splash of caution in every gesture. Dust abatement matters just as much as spill protocols. Disposal can’t be casual, and regulatory agencies keep a close watch on waste streams when this molecule features in industrial-scale processes. Safety data literature stresses training, not just box-checking, since the price for a slip-up carries heavy consequences for personal health and environmental impact alike.
Application Area
Anyone with a hand in dye chemistry, pharmaceuticals, analytical reagents, or energetic materials finds use for 4,6-dinitro-2-aminophenol. Its place as a chemical intermediate means that most of its value lies not as a finished good, but as the critical step in crafting something more elegant, more specialized. Textile and pigment markets used this compound as the backbone of vibrant, stable dyes—much prized before environmental rules forced many out of mass production. Research into antimicrobial agents, or as tracers in analytical settings, taps into its dual functional group nature. For the defense sector, its resemblance to nitroaromatic explosives makes it a training compound or a jumping-off point for more complex formulations. Each sector must weigh performance, safety, and cost against regulatory requirements—a balance that grows more complicated as new green chemistry initiatives gain ground.
Research and Development
From my experience tracking chemical innovation, few compounds like 4,6-dinitro-2-aminophenol carry the puzzle-solving potential for researchers looking to do more with less. Lab groups push into greener nitration techniques, searching for catalysts or new solvents that lower hazards and cycle times. Instrumental analysis investigates trace contaminants in batches, keen to reduce operator exposure. Photochemistry groups scrutinize its absorbance and fluorescence pathways, opening doors for imaging and niche analytics. Intellectual property filings hint at modified analogs with tuned reactivity for targeted diagnostics or controlled-release drugs. The R&D community circles back to this molecule whenever new instrumentation or market demand emerges—every rediscovery prompts tweaks to process or application, especially as regulations and expectations shift.
Toxicity Research
Efforts to map out the health profile of 4,6-dinitro-2-aminophenol stretch back decades. Animal testing revealed acute and chronic toxic effects, including potential for methemoglobinemia from nitro group reduction inside the body. Occupational health studies point to skin and airway risk for operators with repeated exposure, especially where dust or vapors accumulate. Regulatory files call out aquatic toxicity, hinting at wider environmental stakes beyond direct human contact. Modern toxicology tools draw finer lines on inhalation limits, metabolic breakdown, and long-term organ impacts. As endocrinology and carcinogenicity assays mature, expectations for robust oversight only climb. Legislators look to the unfolding toxicology data as a guide for action—sometimes shutting down broad applications in favor of tightly-controlled, purpose-driven uses.
Future Prospects
Looking down the line, 4,6-dinitro-2-aminophenol stands at a crossroads between legacy chemistry and forward-thinking innovation. It benefits from a century of expertise, but also carries the baggage of environmental and safety concerns. Green chemistry priorities push researchers to design out hazards and reduce waste, nudging new methods for functionalization and safe handling. Analytical chemists and process engineers discover ways to recycle or destruct unused intermediates safely. Regulation drives creative thinking, not just compliance—the next generation of applications may leverage its unique properties for medical imaging or nano-materials, provided toxicity risks get reined in. There’s reason to believe that with the right oversight, 4,6-dinitro-2-aminophenol can avoid the fate of many historic chemicals discarded for safety or environmental reasons, and instead play a role in more specialized, tightly-regulated sectors.
What is the chemical structure of 4,6-Dinitro-2-Aminophenol?
The Backbone of the Compound
Anyone who’s spent time in a chemistry lab knows that the arrangement of atoms in a molecule can change the way it behaves in more ways than most folks imagine. 4,6-Dinitro-2-aminophenol is a perfect example. This chemical represents a variant of aminophenol, a ring-shaped molecule packed with functional groups that really give it a character of its own.
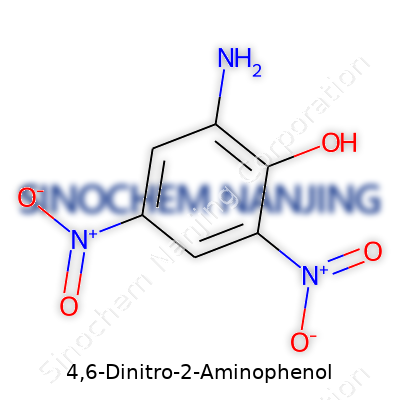
The backbone is a benzene ring, a classic six-membered structure. A lot of chemists can sketch it blindfolded. What sets it apart here are three substituents: two nitro groups and an amino group, plus a hydroxyl group sitting on the ring. In this molecule, the two nitro groups lock into the 4 and 6 positions on the ring, with the amino group at position 2, and the hydroxyl clutching the 1-position. Each of these substituents comes with its own impact on electron density and reactivity, pushing or pulling on the core structure in different directions.
The nitro groups sit like anchors—their sheer electronegativity draws electrons toward themselves, altering the molecule’s overall behavior and reactivity. They have a reputation for slowing down any reactions at nearby spots on the ring, thanks to their powerful electron-withdrawing nature. The amino group at position 2 has a much different flavor: it loves donating electrons, especially toward the ring. The hydroxyl group adds another layer, with its own ability to hand over electrons to the benzene ring.
How the Structure Shapes Use and Risk
This pattern of electron push and pull is not just a mind game for chemists. In practical terms, it can tip the balance between benign and dangerous. Nitro compounds, particularly those with several nitro groups, often appear in the world of explosives and dyes. Chemical labs keep a close eye on compounds like this one. An added amino group nudges it closer to some pharmaceuticals and specialty dyes, yet the two nitro groups remind us not to treat it lightly. Proper safety protocols are a must—those nitro groups can increase toxicity and impact stability.
The arrangement also directs how this molecule gets made and broken down. In the synthesis room, strong acids and bases might act differently around this molecule compared to a plain aminophenol. Since I’ve worked with aromatic compounds before, I’ve noticed how even small rearrangements on the ring shape whether something reacts fast or slow, safe or risky. Engineers and chemists have to assess this structure, planning routes that dodge unnecessary hazards while keeping yields high.
Building Safer Chemical Practices
The unique blend of electron-rich and electron-poor groups on this molecule proves just how much functional groups matter. Companies looking to manufacture compounds with similar structures need to respect both the science and the risks. Using robust personal protective equipment, working with proper fume hoods, and maintaining close monitoring of reaction conditions serve as a good start. Businesses must also watch for waste and byproducts—nitroaromatics, in particular, demand careful disposal to avoid harm to workers and the environment.
On the academic side, experts share data through peer-reviewed journals and regulatory agencies remain vigilant. Open sharing makes it easier for everyone to suggest improvements or spot blind spots. With strong chemistry, practical safety habits, and a little bit of humility about what powerful small molecules can do, safer operations and innovation can walk hand in hand.
What are the common applications of 4,6-Dinitro-2-Aminophenol?
The Role of 4,6-Dinitro-2-Aminophenol in Industry
Factories and laboratories don’t go far before they run into 4,6-Dinitro-2-Aminophenol. This yellowish compound, usually found in crystalline powder form, has carved out a pretty firm niche in certain industries. I remember my early days walking past the dye chemistry labs, and this chemical kept popping up. Turns out, its key applications reflect real, ongoing needs in both traditional and high-tech sectors.
Dye Manufacturing Powers
One of the most active zones for 4,6-Dinitro-2-Aminophenol comes from dyestuffs. Textile plants count on it for specific color formulations that other chemicals can’t produce as reliably. As a chemical intermediate, it transforms during synthesis, taking on the role of a backbone for azo dyes that show up in everything from leather jackets to simple cotton tees. I’ve watched textile dye batches tinted with its derivatives hold their hues even after a dozen washes. Stability and vibrancy in colors make a huge difference for clothing manufacturers since fading dyes mean unhappy customers and returns.
Connection with Pharmaceuticals
Another field relying on this compound is pharmaceuticals. 4,6-Dinitro-2-Aminophenol becomes part of the puzzle in the production of certain specialty drugs. Chemists link it with other molecules, forming more complex structures used in treatments that tackle infections, fevers, or inflammation. While only a part of the multi-step process, its influence can decide the final product’s purity and reliability. Recalling my own chemistry classes, I’ve seen mistakes with the chemical’s quantity or quality throw off reaction yields, leading to long delays in batch approval.
Research and Diagnostic Agents
Beyond more visible industries, research labs rely on 4,6-Dinitro-2-Aminophenol for analytical chemistry. Testing kits and detection reagents incorporate it either to help reveal the presence of metal ions or to serve as a marker during experiments. Accurate results depend on reagents that react clearly and consistently, and this compound pulls its weight. I’ve seen colleagues solve persistent contamination questions in water samples through colorimetric tests, thanks to this chemical’s sensitivity to certain ions.
Concerns and Safety
No discussion about widely used chemicals escapes questions about safety. 4,6-Dinitro-2-Aminophenol can cause health issues if mishandled, as many lab safety posters highlight. Short-term exposure to skin or inhalation of dust can irritate users, and large-scale leaks raise bigger concerns. I always recall my supervisors’ reminders: gloves, goggles, and proper ventilation are essentials, not extras. Factories invest in closed systems and monitoring schemes for just this reason, as workplace health and environmental compliance both demand it.
Moving Forward: Sustainable Solutions
Industry leaders keep an eye on cleaner production methods. Alternatives to nitro-based intermediates have gained attention, yet their price or performance limitations slow the switch. The priority becomes better recycling of process wastes and improved containment — a lesson driven home by regulatory agencies. Research pushes for biotechnological options that mimic these chemical steps, with hopes to ease dependency in the long run. Integrated safety training and routine audits further reduce the fallout from handling potent compounds like 4,6-Dinitro-2-Aminophenol.
Is 4,6-Dinitro-2-Aminophenol hazardous or toxic?
Danger Lurking in a Chemical Formula
Sometimes, chemical names throw up enough red flags that folks barely need a lab coat to react with caution. 4,6-Dinitro-2-Aminophenol sits among those. Working in science-adjacent jobs over the years taught me to pay close attention to any compound loaded with nitro groups. These substances rarely play nice with human health. While they serve useful purposes in explosives, dyes, and specialty manufacturing, tragedy often unfolds during careless handling.
Toxicity Confirmed by Research
Evidence underlines real danger. Studies on dinitrophenols and related compounds show significant toxicity in animals and humans. 4,6-Dinitro-2-Aminophenol hasn’t escaped scrutiny: ingestion or prolonged exposure triggers symptoms like nausea, headaches, and skin rashes. The danger extends beyond discomfort. Nitroaromatic chemicals interfere with the way cells process oxygen. That’s a fancy way of saying even a small dose, breathed or absorbed, can leave people dizzy, disoriented, or worse.
The compound also poses risk to the liver and kidneys. From my time collaborating on safety protocols, I learned that even short-term exposure often sends liver enzymes climbing. Symptoms showing up in the workplace sometimes confused novice workers until testing confirmed the hidden threat lingering on gloves or benches, waiting for a slip-up.
Environmental Impact and Persistence
The risk doesn’t end at the factory door. Runoff from manufacturing or improper disposal exposes wildlife and water supplies to harm. Nitrated organic compounds break down slowly—if at all. Aquatic creatures suffer direct toxicity, and chemicals can work their way up the food chain. Communities living near industrial sites have a right to clean air and water, but without watchful oversight, old habits of careless waste handling continue.
Taking the chemical at face value, it fits into a group the Environmental Protection Agency keeps a close eye on. Runoff can mean local fish functions collapse, with each link in the chain bearing the cost. A quick look back at contaminated waterways worldwide demonstrates the damage isn’t rare or theoretical.
Steps for Reducing Risk
Managing hazards from 4,6-Dinitro-2-Aminophenol starts at the source. Strict workplace controls help, including full protective gear and fume hoods for anyone handling this compound. I’ve seen safety improve in labs that run frequent training and invest in proper equipment. Good habits become routine with reinforcement and strong leadership.
Safe storage matters. Secure containers marked with clear hazard warnings keep surprises to a minimum. Spill kits and regular inspections build another layer of defense. Good practice says never let these chemicals out of sight unless their fate is documented from cradle to grave.
Communities facing industrial neighbors have the right to transparency around discharges. Environmental monitoring, backed by real accountability, pressures organizations to limit pollution. Tough penalties for illegal dumping and public access to safety records form part of the answer.
Final Thoughts
Experience dealing with hazardous substances reminds me that knowledge doesn’t guarantee safety by itself. Clear procedures, vigilant enforcement, and respect for the damage these compounds can inflict protect workers, families, and the environment. No shortcut or gap in oversight can undo harm once it’s happened. That lesson, learned the hard way, deserves repeating wherever dangerous chemicals stand in reach.
What are the storage and handling requirements for 4,6-Dinitro-2-Aminophenol?
Understanding the Risks
4,6-Dinitro-2-aminophenol is a chemical that sparks plenty of discussion in labs and workshops for a reason. It’s sensitive to shock, friction, and heat. People who spend hours handling chemicals like this know the smallest mistake can have outsized consequences—minor carelessness might lead to hazardous situations, even in short timeframes. Experience shows that those taking shortcuts simply do not last long in environments where 4,6-Dinitro-2-aminophenol features in daily tasks.
Proper Storage Conditions Matter
This compound has a known reputation for being unstable at elevated temperatures and when exposed to sparks. A chemical storage room, made for flammable or reactive substances, fits the bill best. Items like this stay safest in tightly sealed glass or high-density polyethylene containers. Corrosion-resistant shelving and dedicated spill trays give extra peace of mind. Temperatures remain around 15 to 25°C—no heaters or direct summer sun nearby. Lights and electrical equipment in the storage space avoid generating sparks. Humidity control becomes essential because moisture can kick off decomposition; those silica packets and dehumidifiers found in many chemical stores play a simple but crucial role here.
Handling Practices That Prevent Trouble
People working with 4,6-Dinitro-2-aminophenol rarely let their guard down. They work on top of static-dissipative mats and wear lab coats made from thick cotton or antistatic materials. Gloves resist both punctures and chemicals—nitrile or neoprene see regular use. Splash goggles remain on, even for short exposure. Supervision becomes routine for new staff, who learn quickly that even a small spill calls for immediate cleanup with proper neutralizers or absorbent materials kept close at hand. Fume hoods regularly get used for any weighing, transferring, or mixing to catch dust and vapors. That keeps the workspace and everyone in it a lot safer. Ventilation systems for these hoods run continuously and see maintenance checks more often than most equipment.
Emergency Preparedness: No Compromises
Universities and industrial sites that deal with 4,6-Dinitro-2-aminophenol drill for emergencies. Fire extinguishers marked for chemical fires, spill kits stocked with neutralizers, and evacuation plans all form part of daily order. Once in a while, a real incident happens—most often because someone skipped a step or acted in a hurry. Having eyewash stations and showers a few steps from any workspace makes a difference. You do not want to run through three doors if something splashes on you or a vapor plume appears.
I’ve watched training pay off in tense moments: one time, the alarm went off after a compound started to smoke. Because everyone knew their roles, the room cleared fast, and the fire crew took quick, safe action. Situations like this bring home how hands-on experience matches up against the unpredictable nature of hazardous chemicals. Prompt response always beats panic.
Building a Culture of Safety
Every chemist, technician, and facilities worker who handles 4,6-Dinitro-2-aminophenol learns two things: there are no shortcuts, and complacency invites disaster. Honest communication about near-misses and regular, evidence-based refresher training helps catch dangerous patterns early. Smart organizations place value on reporting small spills or equipment faults, so fixes come before problems grow.
Solutions Moving Forward
Digitizing inventory logs and access records keeps track of who moves the chemical and when. Investing in self-closing doors and alarms tied to temperature and humidity levels gives another layer of protection. Sharing lessons from minor incidents, instead of hiding them, helps future handling take trends into account. With people learning from experience and clear procedures enforced, risks tied to 4,6-Dinitro-2-aminophenol drop substantially.
Where can I purchase 4,6-Dinitro-2-Aminophenol?
An Overlooked Chemical with Big Questions
Someone asked where to find 4,6-Dinitro-2-Aminophenol. That’s a mouthful, and not something most people pick up at the local hardware store. If you work in research or certain industrial labs, you probably already know sourcing chemicals like this one means jumping through some hoops. You usually can’t just click “buy” online like you do with office supplies. There’s a reason.
Why It's Hard to Buy Certain Chemicals
Many specialty chemicals, especially ones with complex structures or energetic functional groups, raise flags for safety and security reasons. I’ve seen it myself in the lab. Even common reagents require paperwork, approvals, and sometimes a watchful conversation with the chemical supplier on the phone. A lot of this comes from rules designed to keep everyone safe—think about dangerous mishandling, or folks trying to misuse substances for illegal or harmful purposes.
Who Can Actually Purchase This Chemical?
This stuff usually shows up in chemical catalogs like Sigma-Aldrich, Thermo Fisher Scientific, or Alfa Aesar, but it’s not something a person can order for home use. Suppliers almost always check credentials. Companies have to prove they’re registered, labs need detailed research protocols, and buyers go through background checks or account verification. Even universities keep entire departments to handle chemical procurement. These checks matter because substances like dinitro compounds show up on lists that countries use to monitor for controlled precursors or dual-use materials.
Why Oversight Matters
During my own years in academic chemistry, I watched safety officers check inventories with eagle eyes. They care about regulatory standards, but they also want to stop anything dangerous from accidentally winding up in the wrong hands. Some chemicals fit into regulatory categories that trigger even more scrutiny—sometimes, customs officials step in before shipments can even cross a border.
The Right Way to Source Special Reagents
If your project genuinely needs 4,6-Dinitro-2-Aminophenol, contact a legitimate scientific supplier. Get your paperwork lined up. Share details about your research project, compliance standards, and intended use. Sites like Sigma-Aldrich or VWR have clear instructions; emails usually bounce back quickly if something’s off. Never attempt to bypass these channels or pursue unregulated online sources—besides ethical problems, this puts everyone at serious risk.
Support for Science, Caution for Society
Chemical suppliers want to support research and industry, but they live in the real world. Teams work through Know Your Customer (KYC) verifications. Many suppliers have compliance officers monitoring requests, especially if the chemical shows up frequently on law enforcement watchlists. Law-abiding executives, warehouse technicians, and delivery drivers all factor into the oversight ecosystem. It’s a group effort to balance scientific progress and public safety.
Possible Alternatives and Forward Steps
If you’re having trouble with access, ask your organization’s chemical procurement team or safety officer. Sometimes an alternative reagent will work. Approach institutional suppliers, never unregistered sellers. If you ever see a listing on public e-commerce that skips hard questions, that’s a red flag—report it. Every legitimate order, every signature on a shipping manifest, acts as a safeguard against accidents or worse.
Summary
Buying 4,6-Dinitro-2-Aminophenol isn’t like buying printer paper. It takes responsibility, checks, and communication between buyer, supplier, and authorities. These steps keep science moving forward safely, while helping society avoid risks that no lab or community should take lightly.
| Names | |
| Preferred IUPAC name | 4-amino-2,6-dinitrophenol |
| Other names |
2-Amino-4,6-dinitrophenol 4,6-Dinitro-2-hydroxyaniline |
| Pronunciation | /ˈfɔːr.sɪks.daɪˈnaɪ.trə.tuː.əˈmiː.nəˌfiː.nɒl/ |
| Identifiers | |
| CAS Number | 96-96-8 |
| Beilstein Reference | 1740802 |
| ChEBI | CHEBI:51251 |
| ChEMBL | CHEMBL186701 |
| ChemSpider | 8856 |
| DrugBank | DB07907 |
| ECHA InfoCard | 100.013.459 |
| EC Number | 214-657-7 |
| Gmelin Reference | 136300 |
| KEGG | C14320 |
| MeSH | D009624 |
| PubChem CID | 10237 |
| RTECS number | SL8650000 |
| UNII | W1M93W5M3A |
| UN number | UN2020 |
| Properties | |
| Chemical formula | C6H5N3O5 |
| Molar mass | 198.12 g/mol |
| Appearance | Yellow crystalline powder |
| Odor | Odorless |
| Density | 1.71 g/cm3 |
| Solubility in water | slightly soluble |
| log P | 1.14 |
| Vapor pressure | Vapor pressure: 5.20E-7 mmHg at 25°C |
| Acidity (pKa) | 7.07 |
| Basicity (pKb) | 7.75 |
| Magnetic susceptibility (χ) | -54.1·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.718 |
| Dipole moment | 4.98 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 186.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −109.8 kJ mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -1232.6 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, causes serious eye irritation, may cause respiratory irritation, suspected of causing genetic defects. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06, GHS09 |
| Signal word | Danger |
| Hazard statements | H302, H319, H332, H351, H373 |
| Precautionary statements | P261, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 3-2-1-Explosive |
| Flash point | 170°C |
| Lethal dose or concentration | LD50 (oral, rat): 285 mg/kg |
| LD50 (median dose) | LD50 (median dose): 283 mg/kg (oral, rat) |
| NIOSH | NQ6300000 |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | 0.1 mg/m3 |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
2-Aminophenol 4-Nitro-2-aminophenol 2,4-Dinitrophenol 2,6-Dinitroaniline 4,6-Dinitroresorcinol |

