4,4'-Methylenedianiline: A Deep Dive into Its Role and Risks
Historical Development
Looking back, the story of 4,4'-Methylenedianiline runs parallel with breakthroughs in polymer science. Researchers in the mid-1900s started looking for sturdy materials for aerospace and electronics, which led them to aromatic diamines. Chemists figured out that this molecule, with its two amino groups linked by a methylene bridge between benzene rings, could anchor more complex structures. Once it entered the scene, it became a key player, especially in the rise of polyurethanes and advanced epoxy resins. Over the decades, labs and factories both shifted toward its use thanks to the toughness and resilience it brought to end-products.
Product Overview
4,4'-Methylenedianiline, sometimes shortened to MDA, stands as a workhorse for making polyimides, polyurethane foams, and epoxy hardeners. The molecular structure gives it outstanding promise for creating materials that resist heat and stress. In industrial settings, MDA’s solid form usually appears as a pale yellow crystalline powder or sometimes flakes, depending on how it’s processed. Packagers tend to ship it in drums lined with polyethylene, ensuring moisture and oxygen keep their distance until someone opens it up on the plant floor.
Physical & Chemical Properties
Anyone who’s handled MDA quickly notices its distinct appearance and odor. It melts at roughly 88°C and boils at about 398°C, showing decent thermal stability. The compound dissolves in polar solvents but barely budges in water, thanks to its aromatic rings. Reactivity comes primarily from the amine groups, which react strongly with acid chlorides and anhydrides, giving it a reputation for reliability in polymerization. While it doesn’t ignite easily, MDA decomposes at high temperatures, releasing fumes you don’t want near unprotected lungs.
Technical Specifications & Labeling
Every shipment of this chemical wears a label that warns of its potential hazard. You’ll find the UN number 2651 stamped on containers, along with hazard pictograms showing health and environmental risks. Specifications usually require purity above 99% for most technical uses—a figure reached through care in recrystallization and purification. Regulations in the US, EU, and Asia require Safety Data Sheets and clear markings for personal protective equipment (PPE) during storage and handling. There’s no room for slapdash labeling because regulatory compliance isn’t optional.
Preparation Method
The usual route for producing MDA starts with aniline and formaldehyde. Acid catalysis turns these into a bis-aniline intermediate, finally yielding the target diamine after water washes and vacuum filtration. Labs stick close to this method because of efficient yields and cost control. Anyone scaling production up to the metric ton scale has to juggle reaction heat, exotherms, and the final isolation of pure crystals. In my experience, even small shifts in pH or temperature during synthesis can cut quality or cause problems when it’s time for downstream usage.
Chemical Reactions & Modifications
Once in hand, MDA’s amino groups let chemists branch out to all kinds of derivatives. Epoxy resin manufacturers rely on its reaction with epoxides to create thermoset polymers that tolerate severe heat and pressure. Polyimide synthesis harnesses MDA’s dual reactivity to deliver lightweight insulation in electronics and aerospace. Selective hydrogenation transforms it into aromatic polyamines for more niche applications. These reactions aren’t just academic—the properties of the finished plastics or adhesives depend on precisely how these planar benzene rings and amino groups fit together.
Synonyms & Product Names
This compound travels under several aliases. Chemically, it’s called 4,4'-Diaminodiphenylmethane—a mouthful, but precise. Some product labels list simply “MDA” or “Methylenedianiline.” Look at older manufacturing guides and you’ll sometimes find “Bis(4-aminophenyl)methane.” Global chemical suppliers may attach their own trade names, but the structure remains the same, and buyers learn to focus on batch certificates instead of branding.
Safety & Operational Standards
Coming into contact with 4,4'-Methylenedianiline is no joke. The chemical carries a strong warning for potential liver toxicity and carcinogenic effects. Factory workers suit up with gloves and goggles, work under fume extractors, and follow rigid protocols to avoid breathing in dust or vapor. Good ventilation remains critical, and spills demand immediate cleanup with protective gear. Regulatory authorities don’t pull punches: production and handling require detailed risk assessments, consistent PPE, regular health checks on staff, and strict record-keeping. In my own lab days, even a tiny amount spilled meant stopping work for a full decontamination and record update. For worldwide shippers, rules extend to transport labeling and notification.
Application Area
Industrially, MDA earns its keep in several core areas. Polyurethane foam makers count on it as a precursor for rigid insulation that lines buildings and refrigerators. Epoxy resin producers lean on MDA as a curing agent for everything from circuit boards to protective coatings. The aerospace industry prizes its ability to create high-temperature polyimides—materials that remain stable inside jet engines and under the skin of space vehicles. Electrical insulation, adhesives for structural materials, and even advanced composites for sport and defense sectors all trace back to 4,4'-Methylenedianiline chemistry. Hard-wearing, heat-resistant plastics like these often make the difference between something that works for a season and something that remains reliable for decades.
Research & Development
Research around MDA doesn’t rest. Universities and corporate labs both dig into safer process methods and greener alternatives. Some recent academic work explores catalysts that might lower the environmental footprint during synthesis. Other studies focus on structural tweaks—modifying the aromatic rings to create new polymers with adjustable flexibility or flame resistance. In personal experience collaborating with a materials lab, new initiators and alternative diamines steadily pop up, but none quite match the performance profile of tried-and-true MDA for specific electrical and engineering applications. Big advances may only come as researchers balance trade-offs in toxicity, long-term stability, and cost.
Toxicity Research
Years of toxicological studies ring a consistent bell: repeated exposure to MDA poses real health threats. Animal tests show clear links to liver damage and, at high enough doses, increased tumor rates. Workers in industries that use it demand strong monitoring, with biological markers tracked in blood and urine. Even trace amounts escaping into wastewater prompt calls for remediation and better containment. There’s an ongoing push for alternatives, especially in Europe, as regulatory science sharpens its focus on endocrine disruption and cumulative exposure in sensitive populations.
Future Prospects
The future for MDA looks complicated. On one side, next-generation composite materials and electronics ask for exactly the chemical backbone MDA offers, pushing research teams to develop enhanced safety, recycling, and containment protocols. Industry teams chase formulations with lower human and environmental hazards, exploring methyl-free or bio-based alternatives for the same role. Public regulators push harder for cleanroom manufacturing, traceability, and worker health plans. I’ve seen increasing crossover between academic chemistry and commercial scaling as demands for sustainability ramp up. Scientists and plant managers both know: new solutions will need to match the performance MDA delivers, or it’ll remain a tough act to follow, keeping risk management front and center for years.
What is 4,4'-Methylenedianiline used for?
What It Does
Walking through any manufacturing hub, you’re bound to cross paths with 4,4’-Methylenedianiline, also known as MDA, without even realizing it. This chemical helps shape everyday items, sturdy machinery, and even high-tech gear, thanks to its essential role in making polyurethane foams and epoxy resins. MDA reacts with other chemicals to form hard plastics and foams that end up in insulation panels, auto parts, and protective coatings.
Pushing the Boundaries of Materials
Engineers use MDA mainly to make polyurethane, a versatile plastic found in many products. In my shop class days, we relied on polyurethane for its toughness and flexibility, perfect for workbenches, shoe soles, and the wheel casters that survived years of abuse on our carts. MDA gives polyurethane its backbone, making materials sturdy enough to handle pressure and wear. Factory floors depend on this type of plastic not just for longevity, but also for handling chemicals and heat.
Beyond polyurethane, MDA steps into the world of epoxy resins. Picture the demanding needs of circuit boards, paints that seal ships from the ocean’s destruction, or adhesives that hold airplane wings together. These applications require resins that don’t crack or melt easily. This is where MDA comes in, delivering resistance to both heat and chemicals, making sure products last longer and work better in tough environments.
The Risks No One Should Ignore
As durable and useful as MDA is, its dangers don’t just stay behind factory doors. It raises real concerns in the workplace. Handling MDA can damage the liver, impact the immune system, and cause allergic reactions. The International Agency for Research on Cancer has listed it as a possible human carcinogen. In 1979, eight workers in Epping, England, suffered liver damage after MDA contaminated their bread at a bakery. This didn’t just spark outrage—it changed rules and the way industrial chemicals get managed.
Today, companies must keep workers safe by using protective gear, improving ventilation, and making sure only those trained for the job work with compounds like this. At my old plant, we spent extra time in training, learned to double-check procedures, and anyone handling dangerous chemicals had a backup watching over their shoulder. Policy matters—regulatory agencies like OSHA and the European Chemicals Agency enforce strict controls so that accidents become rare, not routine.
Finding Better Ways Forward
Factories rely on MDA, but that reliance brings responsibilities. Researchers keep looking for safer substitutes that still perform well. Some new aromatic amines and greener epoxies have already started replacing MDA in places like electronics or medical devices. Switching over isn’t always easy or cheap, but protecting health always costs less than dealing with accidents or lawsuits later.
Looking at the big picture, innovation pushes industry forward, but vigilance and care must follow every step. The story of MDA reminds us how much progress depends on balancing invention with common sense and caution, using hard-won lessons to keep workers and customers safe while building the future.
What are the health hazards of 4,4'-Methylenedianiline?
What Is 4,4'-Methylenedianiline?
4,4'-Methylenedianiline, or MDA for short, crops up in a world full of coatings, adhesives, and resins. Known for its role in hardening industrial plastics, it lurks behind the scenes in places like epoxy production or polyurethane manufacturing. Most people never see it. Workers do. Those who handle tanks, pumps, or parts coated in epoxy run smack into MDA almost daily. Its chemical popularity keeps it in the background of modern life, but its reputation in medical journals paints a rougher picture.
How Does 4,4'-Methylenedianiline Impact Health?
Direct exposure turns up a mess of health problems. The eyes, skin, lungs, and liver take most of the hits. Odd rashes, yellowing skin, and deep itching don’t stay hidden for long if uncovered skin touches this compound. Sometimes, brown patches and blisters follow, making it look more like a chemical burn than an allergic reaction. MDA slides through the skin, not just on top of it. Once inside, it doesn't leave quietly. The liver takes the beating. Lab tests in Europe tie MDA exposure to hepatitis-like illness, marked with jaundice and lasting fatigue.
Breathing in the dust or vapor can irritate airways and cause headaches, coughing, and trouble concentrating. You don’t need a heavy cloud for trouble—the substance leaves a mark even in small doses over time. The liver keeps showing up as the organ on the front line. Occupational health reports detail workers sent to the hospital, bloodwork showing signs of liver stress after being on the job for only a few shifts. Chronic exposure over months or years stacks up, and long-term effects include decreased liver function, nausea, and sometimes even more serious results.
Cancer Risk and Long-Term Concerns
Repeated exposure ranks as a possible human cancer risk, according to the International Agency for Research on Cancer (IARC). The agency placed 4,4'-Methylenedianiline in Group 2B, which means there’s enough animal research to justify strong caution for people. Scientists studying rats and mice saw liver tumors after feeding or injecting them with it over many months. While human cases haven’t been as conclusive, the signal in animal studies pushes workplace regulators to urge more care.
In practice, this means workers get blood tests, facility managers order spill kits, and government agencies keep tabs on job sites using MDA. Wear gloves, use ventilation fans, watch cleanup closely—these steps aren’t just for “best practice” checklists. They cut down on real cases of chemical-induced sickness.
Tackling the Problem: Simple Steps for Safer Workplaces
Safe handling makes the biggest difference. Switching to less dangerous substitutes helps where possible, but MDA hasn’t vanished from critical manufacturing. Real benefit comes from clear education, tight handling protocols, and independent audits of how companies manage spill risks or air quality. Regular health checks for workers, quick reporting of skin or liver symptoms, and access to safer personal protective equipment all stack the odds in favor of safety.
Paying attention to the stories of injured workers, and the data clinics gather, cuts through the technical jargon. These accounts remind everyone that the white powder sitting next to a tool bench carries consequences. It isn’t just a workplace compliance issue—it's a matter of human health and long-term wellbeing.
How should 4,4'-Methylenedianiline be stored and handled safely?
The Uncomplicated Truth Behind 4,4'-Methylenedianiline
Working around chemical hazards forces folks to ask a basic question: what’s the safest way to handle them? 4,4'-Methylenedianiline, a substance found in jobs from plastics to epoxy hardeners, can threaten health if short-cuts creep into storage and hands-on work. Plenty of people, myself included, remember the first time a safety officer held up a dense yellow jug and made us read the label front to back. That eye-opener sticks with you because this stuff rewards the careful and punishes the careless.
Respecting the Hazards
Science says 4,4'-Methylenedianiline can irritate the skin, affect the liver, and linger in the body. Inhaling dust or vapors brings trouble fast. Ignoring this risks long-term harm that no short-term productivity can justify. When rules seem strict—think gloves, goggles, lab coats, proper ventilation—they’re born from hard lessons. Workers at chemical plants and busy workshops learn to build these steps into their routine until it feels automatic.
Building Strong Storage Habits
Store this chemical in a cool, dry spot out of sunlight—away from food and regular foot traffic. The best containers lock tight and don’t corrode, so glass, stainless steel, or certain plastics work better than cheap solutions. Clear labels aren’t up for debate. They tell everyone, from seasoned pros to late-shift cleaners, what’s inside and what not to mix or mishandle. Locks or access controls keep wandering hands and treasure hunters from sneaking a look, which helps limit both accidents and long-term health impacts.
Hands-On Handling Practices
Putting on nitrile gloves forms the start line. Splash-proof goggles back up your vision in case things go sideways. Some shops set up dedicated stations under fume hoods or with solid exhaust fans so no one wonders about exposure. Personnel training gets forgotten sometimes, but seasoned workers know that short refreshers cut down on confusion and keep everyone safe. My old shop sent us yearly to listen to someone walk us through the dos and don’ts—never just for insurance but because folks genuinely cared about our health. This creates a culture where questions get answered, and mistakes are rare.
Seeking Smart Change
No chemical safety rulebook works if no one reads it. Supervisors who reframe these as a lifeline instead of a block to the bottom line influence the entire workplace. Digital systems now track storage temperature and monitor leaks better than eyes alone ever could. Easy-to-find eye wash stations, spill kits, and emergency procedures posted at every door make all the difference in a pinch. Looking back, each real spill or near miss followed the same script: someone cut a corner, and someone else paid for it.
Strict local regulations already guide many shops, but strong common sense must match the rules on paper. Workers who understand why 4,4'-Methylenedianiline demands respect don’t just recite protocols—they make them part of the job’s rhythm. As in so many parts of life, the small steps—closing a cap, cleaning a glove, running a vent—write the biggest difference in health and safety.
Is 4,4'-Methylenedianiline regulated or restricted by law?
Understanding the Risks and Why Rules Exist
4,4'-Methylenedianiline, or MDA, doesn’t roll off the tongue, but it turns up in industries shaping the world around us. It pops up in polyurethane manufacturing, helps create epoxy resins, and even supports cement production. Chemicals like this often look like tough, reliable partners in building stronger products. The issue with MDA is that it has a history of harming people who handle it without proper precautions. Years ago, research tied MDA to liver toxicity and changes in genetic material. Evidence grew that it could cause cancer in humans. For regulators, that raised enough red flags to force change.
Governments don't usually move swiftly on industrial chemicals unless people start getting sick. That's what happened in Norway during the 1970s. Workers exposed on the job developed liver disease. The link was clear enough to make authorities step in. The European Union followed, classifying MDA as a carcinogen. Companies in the EU face strict limits on how much MDA can be present in workplaces, and workers get checked for exposure on a regular basis. The Reach Regulation puts it on the "Substances of Very High Concern" list, which means companies have to track it closely and publicly declare its use above tiny concentrations. This isn't just paperwork—it's a tool to push safer alternatives.
Rules on the Books: What Changes for Business?
In the United States, MDA lands on the Environmental Protection Agency’s Toxic Substances Control Act inventory. The Occupational Safety and Health Administration doesn’t mess around here: airborne exposure gets capped, and skin exposure is tightly controlled. Workers receive respirators and protective gloves when even a whiff could mean trouble. The Department of Transportation slaps hazardous material labels on shipments. As a result, companies can’t just toss around MDA like salt at dinner; violation brings fines and legal headaches.
On the other hand, in places with weaker enforcement, people pay the price. I worked in a factory years ago where safety meant wearing the same gloves until holes appeared. Companies like ours didn’t want to slow down or pay for better equipment. It’s easy to talk about personal responsibility, but many workers never get the full story about what these chemicals can do to their bodies until it’s too late. Real regulation makes companies invest in better training and equipment, which might cost more upfront but saves on health costs and lawsuits down the line.
What Can Be Done?
Few people outside of specialized manufacturing circles know about MDA or why it’s so closely watched. Still, history shows that failing to pay attention lets companies cut corners. Workers deserve the right to know what they’re being exposed to. Legislation should stay public and easy to find, so no one ends up in the emergency room just for doing their job. Alternatives to MDA exist, but swapping them into production lines doesn’t always happen overnight. Incentive programs and grants could push more companies to make the switch.
At the end of the day, MDA’s story isn’t about obscure chemicals or distant factories. It’s about people and the choice to put worker safety over cutting costs. Protecting employees should never take a back seat to easy profit, and laws on MDA work best when everyone knows why they’re there in the first place.
What are the environmental impacts of 4,4'-Methylenedianiline?
Digging Into the Problem
Ask anyone who’s spent time near an industrial plant about strange smells in the air, and talk often turns to chemicals like 4,4'-Methylenedianiline, or MDA. This chemical, known for its role in the production of polyurethanes and epoxy resins, finds its way into the ground and water much easier than many realize. It lingers, sometimes for weeks, before anyone in charge starts asking questions about where the fumes or neon-colored puddles are coming from.
What Science Shows About MDA’s Environmental Footprint
MDA doesn't just break down and disappear. Scientists have reported that it tends to persist in water and soil. Certain bacteria break it down, but the process takes a long time, especially outdoors where weather keeps shifting. In rivers and groundwater, MDA spreads out and has the potential to travel far from the source. For wildlife, this spells trouble. Studies linked exposure to lower growth rates in fish and problems with their livers. Frogs exposed in their formative stages don't fare any better.
Direct Human and Ecosystem Impact
People who work with MDA, or even live near sites where it spills, sometimes speak about skin rashes or breathing issues. The impact runs deeper for those relying on well water drawn near contaminated spots. The effects aren’t just about dry facts and figures; for farmers and fishermen, contaminated water can threaten both livelihood and food security. Crops pull up water that may carry traces of MDA, impacting the food chain in subtle ways that go ignored until a careful study uncovers the link.
Why This Problem Demands Real Solutions
In my neck of the woods, once chemicals enter the landscape, worry sets in fast. No one wants to think about the invisible damage. Local water authorities struggle to remove complex industrial chemicals like MDA, and regular treatment plants rarely catch everything. Research from the EPA points out that MDA stays stubbornly persistent, especially without specialized filtration.
The challenge gets compounded by legacy pollution. Old storage tanks, forgotten barrels, and leaky plumbing built decades ago turn into slow-moving threats. Regulators and corporations trade blame, and the cleanup crawls along with too little urgency.
Chasing Realistic Fixes
Communities and industry leaders do have tools for making things better. The first step comes through transparency. Sharing what chemicals flow through local plants—and the risks they pose—forms the bedrock of trust. Water utilities could upgrade their filtering technology, drawing from methods used in places that handle tougher contaminants.
Regular soil and water testing around current and former MDA hotspots helps keep everyone informed. Public funding for cleanup might not sound glamorous, but these investments prevent far bigger health bills down the line. Industry can double down on containment—barriers, improved storage practices, and spill-response drills.
Personal experience shows that people, given the facts and a say in what happens next, bring energy and urgency to environmental issues. Real progress begins with stopping new leaks and refusing to sweep decades-old pollution under the rug. The story of MDA’s environmental footprint isn’t just about chemicals in the air or water—it’s about people, jobs, and the places that shape a community.
| Names | |
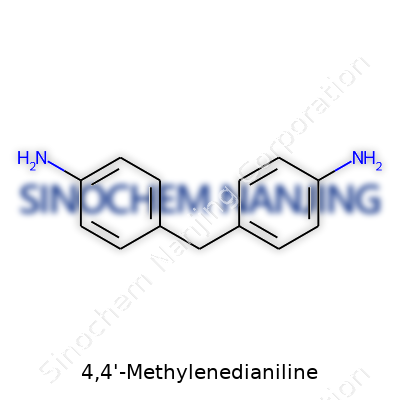
| Preferred IUPAC name | 4,4'-Methylenedianiline |
| Other names |
MDA Diaminodiphenylmethane Bis(4-aminophenyl)methane |
| Pronunciation | /ˌmɛθ.ɪˌliːn.di.əˈnɪl.iːn/ |
| Identifiers | |
| CAS Number | 101-77-9 |
| Beilstein Reference | 1733773 |
| ChEBI | CHEBI:41264 |
| ChEMBL | CHEMBL140463 |
| ChemSpider | 8219 |
| DrugBank | DB01544 |
| ECHA InfoCard | ECHA InfoCard: 100.003.221 |
| EC Number | 202-974-4 |
| Gmelin Reference | 68439 |
| KEGG | C06581 |
| MeSH | D008774 |
| PubChem CID | 7419 |
| RTECS number | NL0175000 |
| UNII | NDP84L285V |
| UN number | 2811 |
| CompTox Dashboard (EPA) | DTXSID2021351 |
| Properties | |
| Chemical formula | C13H14N2 |
| Molar mass | 198.27 g/mol |
| Appearance | Light brown flakes or crystals |
| Odor | amine-like |
| Density | 1.31 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 0.76 |
| Vapor pressure | 0.01 mmHg (25°C) |
| Acidity (pKa) | 3.72 |
| Basicity (pKb) | 10.26 |
| Magnetic susceptibility (χ) | \-74.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.663 |
| Viscosity | 2.6 cP (20 °C) |
| Dipole moment | 3.74 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 255.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 77.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3210 kJ·mol⁻¹ |
| Hazards | |
| Main hazards | Toxic if swallowed, in contact with skin or if inhaled; causes damage to organs; suspected of causing cancer. |
| GHS labelling | GHS02, GHS05, GHS06, GHS08 |
| Pictograms | GHS05,GHS08,GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H317, H318, H334, H350, H360Fd, H373, H400 |
| Precautionary statements | P201, P202, P260, P264, P270, P273, P280, P301+P310, P302+P352, P308+P313, P321, P330, P405, P501 |
| NFPA 704 (fire diamond) | 3-1-0 |
| Flash point | 302 °C |
| Autoignition temperature | 520°C |
| Lethal dose or concentration | LD50 oral rat 630 mg/kg |
| LD50 (median dose) | LD50 (median dose): 930 mg/kg (oral, rat) |
| NIOSH | BW4100000 |
| PEL (Permissible) | 0.01 ppm (OSHA PEL, TWA) |
| REL (Recommended) | 0.82 mg/m3 |
| IDLH (Immediate danger) | IDLH: 10 mg/m³ |
| Related compounds | |
| Related compounds |
Aniline Diphenylmethane MDI (Methylene diphenyl diisocyanate) Benzidine |

