4,4-Dimethyl-1,3-Dioxane: A Practical Perspective
Historical Development
Stories about chemicals often start with a single-minded pursuit of utility. 4,4-Dimethyl-1,3-Dioxane, once a small curiosity in the vast family of cyclic ethers, has grown out of classic organic synthesis roots. Early days, organic chemists searched for stable dioxanes that could be manufactured without fuss and that wouldn’t fall apart in the flask. Back in those years, substituting the dioxane ring seemed a clever way to avoid some notorious instability—temperature sensitivity, unwanted polymerization, dangerous side reactions. Adding two methyl groups at the 4-position created a molecule with bulkier dimensions and gave it new possibilities. Its emergence didn’t happen overnight, but as research labs explored new cyclic solvents and intermediates, 4,4-Dimethyl-1,3-Dioxane took on a place as more than a chemical oddity.
Product Overview
Most people rarely encounter 4,4-Dimethyl-1,3-Dioxane by name, but its influence slips quietly into manufacturing streams—from specialty resins to select flavors and fragrances. This dioxane typically appears as a stable, colorless liquid, suitable for bench chemistry or scaled-up processing. As a cyclic acetal, its reactivity and physical properties lend themselves to both practical industrial work and academic tinkering. The addition of two methyl groups at the 4-position shifts its character from simple dioxane, granting a bit more resilience against breakdown and a subtle change in its boiling point. If you've worked in a lab setting, you recognize these small structural changes matter. They allow certain syntheses or applications to operate with less risk, and that can reduce accidents or lost batches over time.
Physical and Chemical Properties
Nothing replaces handling a substance to get a real feel for it, but reliable data gives confidence before opening the bottle. 4,4-Dimethyl-1,3-Dioxane offers a moderate boiling point and a density close to that of water, which means it doesn't present some of the handling headaches that more volatile organics do. Its solubility profile answers for itself: a modest mix with water, but broad compatibility with organic solvents. The high flash point brings a noticeable safety bonus, especially for crowded labs with other flammable materials around. You can see, both from literature and bench experience, its chemical stability holds up under a span of conditions, although strong acids or oxidants should be kept away. Its mild, almost sweet odor points to its acetal makeup. Work in process chemistry, and you’ll notice how acetal stability can enable or undermine a project depending on reaction conditions.
Technical Specifications and Labeling
Accurate chemical identification always matters, but regulatory compliance adds another layer these days. Labels, both digital and physical, carry CAS numbers and precise naming—so 4,4-Dimethyl-1,3-Dioxane usually travels with its CAS RN 1540-85-8. Some suppliers offer purity grades defined by trace water or residual solvents, reflecting how well the material fits electronics, pharma, or fine chemicals work. The safety info on these labels sets expectations: proper ventilation, goggles, gloves. From personal experience, having precise, readable labeling on dioxane derivatives in a workshop spares time and brainpower under deadline pressure, so high-contrast hazard icons or robust handling guidance are a must.
Preparation Method
The dioxane ring doesn't come together by magic. Chemists usually walk the tried-and-true routes: acid-catalyzed cyclization of diols or controlled condensation reactions. For 4,4-Dimethyl-1,3-Dioxane, methyl-substituted precursors—most often neopentyl glycol—undergo acid-promoted acetalization with an aldehyde, commonly formaldehyde or one of its polymers. Timing and water removal matter a lot, since incomplete reactions lead to all kinds of pesky alcohol by-products. As someone who has run both bench and plant-scale condensations, the main challenge lies in getting the right ratio of reactants and the right acid strength to drive the transformation forward without bubbling up unwanted side reactions. Classic approaches use Dean-Stark apparatus for water removal, though more modern processes sometimes use azeotropic distillation and greener catalysts. Make no mistake, acetal synthesis rewards careful monitoring.
Chemical Reactions and Modifications
The real value of this dioxane isn't just as a static molecule. It acts as both building block and protector. The ring resists most bases and mild acids, but stronger acids break it back down, releasing methyl-substituted alcohols or aldehydes. Smart synthetic chemists harness this: protect a reactive diol as a 4,4-dimethyl-1,3-dioxanyl acetal through harsh steps, then quietly deprotect at the end. Modifications target the dioxane's ring: either tinker with substitutions on the methyl groups or swap out one of the carbons for selective labeling. In organometallic routes, dioxanes occasionally act as ligands or solvents, stabilizing sensitive reagents during dry reactions. All these little transformations add up, and each one calls for a fresh look at both reactivity and downstream complications.
Synonyms and Product Names
Naming questions haunt even experienced chemists. 4,4-Dimethyl-1,3-Dioxane answers to several variations. Some catalogs use "dimethyldioxane," others prefer systematic names like "2,2,6,6-Tetramethyl-1,3-dioxane" or simply “Tetramethyldioxane.” Trade names exist, but the most consistent label outside lab notebooks sticks with the IUPAC form. Wrong synonyms slow down online sourcing or cross-checking safety literature. My own work has stumbled over suppliers listing the substance under old or regional names—forcing more late-night literature sessions to confirm the correct structure before signing off on an order.
Safety and Operational Standards
Every labmate knows: safety shouldn't go out the window for the sake of convenience. 4,4-Dimethyl-1,3-dioxane offers a safer profile than some smaller cyclic ethers like tetrahydrofuran, mostly because of its lower volatility and less aggressive flammability. Even so, gloves and fume hoods stay mandatory, since inhalation still carries risks for those working long hours. Chronic exposure—to vapors or accidental skin contact—remains under-studied. Fire codes require proper storage in tightly capped bottles far from open flames or oxidizers. Lab protocol always calls for spill kits and first aid nearby. Those who’ve seen the aftermath of careless solvent handling rarely forget the importance of well-maintained eye wash stations and solvent-resistant aprons. Following these standards means everyone, from first-year student to tenured professor, stays clear of unnecessary health scares or cleanup marathons.
Application Area
Chemicals like this one don’t survive decades of product shelf life unless they bring something worthwhile to the table. In synthetic chemistry, 4,4-Dimethyl-1,3-Dioxane shows up as a reagent, protecting group, or specialty solvent. It anchors protective groups in the synthesis of pharmaceuticals, helps create spirocyclic intermediates for agrochemicals, and sometimes stabilizes flavors or fragrances in the food industry. The acetal backbone shields functional groups during multi-step syntheses; without such protection, more sensitive alcohols and ketones might never make it to the last step untouched. Some paint and coating formulations lean on these dioxanes for their solvency and long-term stability. Anyone who spends days developing new molecules knows such specialty chemicals offer a shortcut through difficult sequences and let researchers focus on innovation rather than fighting side reactions.
Research and Development
R&D teams never stop pushing for more sustainable, robust, or cost-effective processes. In academic labs, 4,4-Dimethyl-1,3-dioxane draws attention as a model compound to test new catalytic cycles or as a testbed for developing greener protecting group chemistry. Industrial R&D wants to optimize its synthesis or reclaim its by-products for downstream value. Lately, people focus on improving atom economy and ditching corrosive acid catalysts for zeolites or solid acids that promise cleaner waste streams. Collaborations between academia and industry uncover new application areas in biotech, nanotech, and advanced materials. My own time in industry research taught me the value of such incremental breakthroughs: shave a few cents per kilogram off production, cut hazardous waste in half, or build a reuse loop—these victories add up.
Toxicity Research
Public information about the toxicity profile of 4,4-Dimethyl-1,3-Dioxane tends to be limited compared to higher-profile solvents or additives. Whatever data is available, it suggests manageable toxicity in standard lab usage, though as with many cyclic ethers, longer-term chronic effects haven't been ruled out. Animal studies hint at minimal acute effects at moderate doses, but large-scale inhalation or ingestion leads to expected narcotic symptoms and potential organ disruption. Responsible researchers document these effects and push for more rigorous animal-free methods for assessment. There’s a real drive for better understanding of breakdown products in both landfill and aquatic environments, since partly hydrolyzed dioxanes can persist or lead to unpleasant bioaccumulation. Personal experience reminds me that regulatory agencies grow harsher each year with trace-level solvent contamination in food or consumer goods, so periodic review of toxicity reports matters.
Future Prospects
Tomorrow’s chemical toolbox demands versatility and a lighter footprint on health and environment. Research in cyclic acetals heads toward more sustainable ways of making and using dioxane derivatives. For 4,4-Dimethyl-1,3-dioxane, the outlook remains positive in specialized syntheses and new material science projects. Green chemistry efforts keep searching for solvent systems that match performance with reduced volatility and toxicity. Advances in catalytic recycling might turn acetal-based reagents into genuinely sustainable choices. Meanwhile, bio-based versions or derivatives open up markets where both regulatory and consumer expectations keep climbing. The lessons from the past—care with process conditions, transparency in labeling, commitment to safety—still guide every smart decision about how to handle and deploy these useful chemicals in the decades to come.
What is 4,4-Dimethyl-1,3-Dioxane used for?
What Drives Its Use?
Few people give much thought to odd-sounding chemical names unless they show up in headlines or cause trouble. 4,4-Dimethyl-1,3-dioxane rarely gets attention, yet this molecule quietly finds its way into everyday life. Its main draw comes from its stable molecular shape, which allows companies to use it in specialized processes where reliability matters.
Common Applications in Industry
Paint manufacturers look for ingredients that hold up under varied conditions. Additives like 4,4-Dimethyl-1,3-dioxane give paints and coatings better resistance to moisture and help them last under heat or chemical exposure. By improving durability, this chemical lengthens the lifespan of painted surfaces in high-use environments.
In the cleaning and degreasing world, 4,4-Dimethyl-1,3-dioxane pops up as a solvent. Its structure lets it dissolve oils, greases, and some polymers that water alone ignores. Factories that depend on machine maintenance and heavy-duty cleaning tools often lean on effective solvents to get the job done quickly and safely. Keeping machines free of debris not only protects the equipment but also preserves worker safety.
Plastics and resins sometimes need a boost to perform in tough settings or to gain particular properties. Specialty chemicals like this dioxane variant help tweak those plastics, adding flexibility or altering how they handle exposure to sunlight or chemicals.
Looking at Health and Environmental Considerations
Experience has taught me and anyone who has spent time in laboratories that even stable solvents deserve respect. Chemical exposure can surprise you with unexpected health impacts if handled carelessly. 4,4-Dimethyl-1,3-dioxane can irritate the eyes or skin and demands proper storage to avoid accidental release. Protective gloves and eye gear should never get skipped, and work spaces need good ventilation.
Chemicals moving from industry to the environment create headaches down the line. Companies must track solvents like this from purchase to disposal. Loose handling can mean contamination of soil and water. Environmental investigations over the years have shown how quickly minor leaks or dumping escalate. Laws and rules exist, but effective oversight and practical training for workers help prevent accidents before they have a chance to snowball.
Improving Safety and Reducing Impact
Shifting toward greener chemistry stays on the minds of both regulators and honest business owners. Newer alternatives sometimes replace traditional solvents in response to environmental and health concerns. In labs, swapping in a safer chemical, switching to closed-loop systems that catch vapors, or recycling spent solvents cuts down risk. Proper disposal—never dumping down the drain or tossing in regular trash—reduces pollution worries.
Companies upgrading to better training, investing in closed transfer systems, and watching their waste output more closely lead by example. For years, meeting room conversations about safety protocols felt routine, but after witnessing a few close calls, the message stuck: people keep each other safe by treating every step with care. Keeping track of every chemical, no matter how obscure, protects both workers and the public.
Weighing the Future
Every chemical has a story, shaped by how people use it, safeguard against its risks, and search for better options. 4,4-Dimethyl-1,3-dioxane proves useful in focused applications, but safe handling, tighter controls, and a willingness to seek alternatives remain crucial for industries that count on specialty chemicals. People who learn the names—and the reality—of these compounds play a real part in protecting health and the planet.
What are the safety precautions when handling 4,4-Dimethyl-1,3-Dioxane?
Respect the Substance: It’s Not Just Another Chemical
Anyone working with 4,4-Dimethyl-1,3-Dioxane needs to know the risks, not just on paper or in a safety data sheet, but right there in the lab or on the workbench. This isn’t a harmless cleaner. Breathing vapors or skin contact can cause irritation or, in higher doses, more serious problems. In my experience, skipping the routine protections—like goggles or gloves—often comes from “just this one quick task” thinking. Then someone gets a rash, wild coughing spell, or worse. So I always grab nitrile gloves, chemical splash goggles, and a proper coat before opening the bottle. Even veterans slip up, but repeated reminders and seeing real-life incidents drive the point home.
Keep It Contained: Fume Hood Isn’t Optional
Vapors from 4,4-Dimethyl-1,3-Dioxane don’t just linger—they require proper ventilation. I’ve seen people try to work on an open bench “just for a minute," trusting a cracked window. A fume hood fixes the issue. It felt like a hassle the first time I locked everything away under the sash, but knowing how quickly these vapors can affect air quality changes your attitude. Airborne toxins build up fast in a small lab or shop, and personal stories about dizziness or headaches remind me why the fume hood cannot be an afterthought. The right ventilation keeps fumes out of your lungs and away from coworkers.
It Only Takes a Splash: Don’t Underestimate Jacket and Face Shield
Handling this chemical gets messy during transfers or spills. Once, a manager rushed through pouring and landed drops on their forearm. It turned red on contact. Now I always double up—a long-sleeved lab coat and sometimes a face shield if the volume is higher. Even if the documentation calls it "low risk," skin and eyes aren’t worth rolling the dice. Quick access to an eyewash station or emergency shower feels unnecessary until you need it. Every second counts, so I check these before starting work.
Label and Store Like It Matters—Because It Does
Cluttered shelves, faded labels, and poorly sealed bottles turn a controllable risk into panic mode during an emergency. Chemicals like 4,4-Dimethyl-1,3-Dioxane hide in old jars or look like water. In my old lab, a mislabeling incident could have mixed a reactive acid with dioxane. Now, all containers carry legible, current labels with hazard symbols. I store them away from oxidizers and acids, always in a cool, ventilated cabinet. These steps prevent little slips from becoming disasters.
Proper Waste Disposal Isn’t Optional
Pouring leftover dioxane down the drain or tossing it in the trash might save time, but it poisons water and soil. I watched a team get fined after ignoring guidelines—no one avoided the consequences, even managers. Authorized hazardous waste bins, handled by trained staff, are the only way out. Check spill kits are ready and absorbents on hand, so everyone responds quickly if something tips over.
Training and Emergency Response: The Real Line of Defense
No substitute exists for knowing the emergency plan and practicing it as a team. Lab drills always felt tedious until one day, a spill caught us off guard. The process—evacuate, contain, call for help—ran smoothly because we had rehearsed. Basics like knowing where the fire extinguisher or first aid kit sits make more difference than any manual. It’s this culture of safety—not just rules and gear—that actually keeps people healthy. Honest sharing of lessons learned, clear instructions, and watching out for each other strengthen every safeguard the chemical industry builds on.
What is the chemical formula of 4,4-Dimethyl-1,3-Dioxane?
Understanding the Structure
The name might sound complicated, but 4,4-Dimethyl-1,3-dioxane falls into a group of chemicals that show up across many industries and laboratories. Its formula, C6H12O2, sums up the balance of elements in this molecule: six carbon atoms, twelve hydrogens, and two oxygens. People who’ve ever worked with chemicals for research or manufacturing often know that tiny changes in structure—where those atoms fall—can turn something simple into a crucial tool or an irritating impurity.
Real-World Uses and Relevance
This compound isn’t just a neat summary on a lab report. I’ve seen 4,4-Dimethyl-1,3-dioxane used as a solvent and intermediate, especially where a touch of water resistance or stability makes a difference. Compared to other dioxanes, this one’s methyl groups help control volatility and reactivity. In industrial labs, those details can mean safer handling and better product consistency.
Researchers use this compound to study reactions that mimic how more complex materials behave. For anyone making specialty polymers or tweaking pharmaceutical candidates, having a predictable, stable compound to serve as a reference makes life a lot simpler. In past projects, when we needed to try out new antioxidants or to boost the shelf life of sensitive blends, this type of molecule’s balance of properties played a big part.
Health and Environmental Considerations
Chemicals like 4,4-Dimethyl-1,3-dioxane rarely sit untouched—they often end up as byproducts, lingering in waste streams if systems don’t catch them. Its cousin, 1,4-dioxane, earned a reputation as a water contaminant that resists easy cleanup and causes concern at trace levels. Unlike some compounds, the two extra methyls on 4,4-Dimethyl-1,3-dioxane change how quickly it degrades and how likely it is to show up in groundwater. Yet, caution never hurts. Each time I’ve seen colleagues deal with trace impurities, I’m reminded how quickly regulators can change standards as health data evolves.
People living near chemical plants or research centers shouldn’t have to worry about long technical names in their water or air. Rigorous waste handling and targeted filtration methods prove more effective than hoping for the best. In my own work, using up-to-date equipment for monitoring emissions and strict inventory controls limited unnecessary releases—real results over theoretical promises.
Solutions That Make a Difference
Keeping up with regulations helps, but making improvements at the source brings real gains. For manufacturing and research facilities, routine audits, better waste management, and investment in modern treatment systems turn chemical stewardship from an afterthought into standard practice.
Switching to less persistent chemicals sometimes requires adjusting processes or partnering with outside experts, but these changes offer long-term benefits. In training new chemists, I’ve found that building a mindset around responsibility eclipses memorizing regulatory codes; practical steps like regular leak checks and transparent documentation do more to protect both workers and the wider community.
4,4-Dimethyl-1,3-dioxane might sound obscure, but smart practices keep these types of chemicals from becoming tomorrow’s headlines. Leaning on fact-based approaches and a watchful eye keeps chemistry both productive and safe.
What are the storage conditions for 4,4-Dimethyl-1,3-Dioxane?
Why Storage Matters More Than You Think
Most people don't give much thought to chemical storage until something goes wrong. With a compound like 4,4-Dimethyl-1,3-Dioxane, careful storage avoids wasted money, work stoppages, and injuries. This chemical shows up in specialty syntheses and research labs, and sometimes in production settings. Its stability depends on some basic but important conditions that shouldn’t get overlooked.
What Science Says About Protecting This Chemical
Let’s keep things real—4,4-Dimethyl-1,3-Dioxane is a liquid, and while it doesn't break down in air the way some peroxides do, light and warmth nudge it toward trouble. The liquid can give off fumes if the temperature creeps up, so pitting it against heat sources turns routine storage into an accident waiting to happen.
A lot of folks, especially those new to chemistry, treat “room temperature” as a guarantee. The truth is, lab rooms run warm or cool depending on season, geography, and whether someone left equipment running nearby. A safe bet is a space kept under 25°C, with lower temperatures favored in crowded labs or buildings where the heating swings hard between winter and summer.
Moisture and Air: Hidden Enemies
4,4-Dimethyl-1,3-Dioxane doesn’t love water. Moist air creeping into a bottle leads to rust on metal caps, diluted contents, or property changes nobody tracks until quality control fails. Using strong, well-sealed glass bottles or high-quality plastic containers blocks most of that trouble right at the start. Compressed lids and clear date labels help, because leaks and old stock sneak up on people who don’t check regularly.
Shelving Choices Can’t Be Random
Stacking bottles on a sunlit shelf or over a heat vent shortens the shelf life of nearly any solvent, and 4,4-Dimethyl-1,3-Dioxane is no exception. Ask anyone who's needed to toss a yellowed or odd-smelling batch. A metal cabinet with a working lock, grounded for static, solves many issues. Forgetting about flammable liquid rules, or mixing bottles with acids or oxidizers, can turn a small fire into a police report.
Fire codes say clear: solvents in one space, acids or oxidizers somewhere else. Insurance adjusters and fire inspectors visiting after an incident tend to point at mixed shelves—no one wants to be on the business end of that conversation.
Lessons from Daily Practice
In real-world labs, corners get cut. A quick errand, a swapped bottle, and mistakes pile up. My own early days in a shared workspace taught me quick: taking shortcuts with storage delivers headaches, not time savings. A small sign marking locker content, a logbook for opening and closing stock, and dedicated shelves make accidents rare.
Fact: 4,4-Dimethyl-1,3-Dioxane’s flash point sits around 41°C—much lower than boiling water. If stored wrong, one spark turns a minor spill into a bigger emergency.
Moving Toward Consistent Safety
We learn fastest from the close calls: a sticky cap, lingering smells, a brown patch on a shelf. These signs nudge us toward better controls. Storage stays safe by combining habits: labeled, dated, checked regularly, always behind closed doors, away from incompatible chemicals. A little focus turns storage from a chore into peace of mind, for yourself and everyone sharing your workspace.
Is 4,4-Dimethyl-1,3-Dioxane hazardous to health or the environment?
Looking Beyond the Chemical Formula
Plenty of chemicals pass through our lives without much notice, often hiding in products we barely give a second thought. 4,4-Dimethyl-1,3-Dioxane might not spring to mind for most, yet it plays a role in some industrial and laboratory settings. Understanding its potential risks to people and the planet isn’t just for chemists or regulators—it matters to anyone who cares about air, water, and what eventually trickles into our homes.
Tracing Where Exposure Can Happen
Labs, manufacturing, chemical processing—all places where 4,4-dimethyl-1,3-dioxane might show up. Workers face the primary risk, handling it during synthesis or use. Even with protective gear, spills or leaks can lead to skin contact or inhalation. Waste streams may release small quantities to the environment. Regular folks likely never purposefully touch the stuff, but runoff and improper disposal can carry it to community water or soil.
Health Considerations
Studies on chemicals like this always leave room for more answers than we have. Toxicology data on 4,4-dimethyl-1,3-dioxane doesn’t fill shelves. What’s documented in scientific literature often comes from laboratory animals, showing irritation if inhaled or if it touches skin. No clear connection to cancer has surfaced, but the lack of data means we can’t sweep aside long-term risk.
Chemical cousin 1,4-dioxane has earned its reputation for being persistent in water and linked to cancer in animal studies. Regulatory agencies flag it as risky, making its relatives worthy of a hard look. Sometimes similar structures point to similar traits, so caution feels justified.
Environmental Footprint
Persistence creates headaches. If a synthetic chemical doesn’t easily break down, it can stick around in groundwater or soil, crossing into streams and eventually showing up in drinking water. The 1,3-dioxane skeleton in 4,4-dimethyl-1,3-dioxane doesn’t guarantee it will behave the same as 1,4-dioxane, but environmental scientists keep a close eye on these molecular structures.
Microbes in nature usually do the heavy lifting, degrading many compounds before they pile up to concerning levels. Yet, just as 1,4-dioxane resists biodegradation, structurally similar compounds could show similar stubbornness. Water treatment plants rarely catch up, letting small quantities slip by. The impact could build as traces persist from spill or poor disposal.
Finding Smarter Solutions
It’s hard to demand action without all the answers. Still, companies can step up by choosing safer alternatives or limiting releases into air and water. Improved handling and tight storage keep risky chemicals far from water sources and hands. Routine monitoring in workplaces plus regular testing around industrial sites add another layer of safety.
Regulators may not have written the book on 4,4-dimethyl-1,3-dioxane, but the patterns matter. Looking at chemicals with similar backbones offers lessons worth listening to—err on the side of caution, especially with persistent compounds. Greater transparency from industries using specialty chemicals helps watchdog groups and local communities keep tabs.
Why It Matters
A single compound rarely makes breaking news unless disaster strikes. Still, the smaller choices—better safety at work, conscientious disposal, robust pollution controls—keep those disasters from happening. Choosing to shine a light now, before problems stack up, protects both health and the environment. Informed vigilance, rather than blind trust, makes the difference.
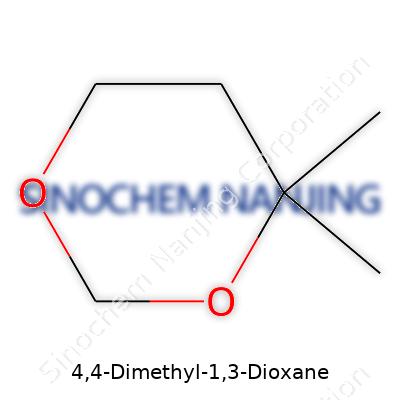
| Names | |
| Preferred IUPAC name | 2,2-Dimethyl-1,3-dioxane |
| Other names |
Dimethyldioxane p,p-Dimethyldioxane |
| Pronunciation | /ˈfɔːr.fɔːr.daɪˈmɛθɪl.wʌn.θri.daɪˈɒkseɪn/ |
| Identifiers | |
| CAS Number | [646-06-0] |
| Beilstein Reference | 1201301 |
| ChEBI | CHEBI:34419 |
| ChEMBL | CHEMBL137111 |
| ChemSpider | 75161 |
| DrugBank | DB14096 |
| ECHA InfoCard | ECHA InfoCard: 100_008_708 |
| EC Number | 203-642-4 |
| Gmelin Reference | 102890 |
| KEGG | C06465 |
| MeSH | D015203 |
| PubChem CID | 10841 |
| RTECS number | JH8630000 |
| UNII | N11LDI4S18 |
| UN number | UN3166 |
| CompTox Dashboard (EPA) | DTXSID1037393 |
| Properties | |
| Chemical formula | C6H12O2 |
| Molar mass | 118.18 g/mol |
| Appearance | Colorless liquid |
| Odor | pleasant odor |
| Density | 0.93 g/cm³ |
| Solubility in water | insoluble |
| log P | 0.63 |
| Vapor pressure | 0.8 mmHg (20 °C) |
| Acidity (pKa) | 15.84 |
| Basicity (pKb) | pKb = 6.07 |
| Refractive index (nD) | nD 1.410 |
| Viscosity | 1.184 mPa·s (20 °C) |
| Dipole moment | 1.96 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 409.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -404.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3860 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H319 |
| Precautionary statements | P210, P261, P264, P271, P301+P312, P304+P340, P312, P330, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | 71°C |
| Autoignition temperature | 215 °C |
| Lethal dose or concentration | LD50 (oral, rat): > 5000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 > 5000 mg/kg |
| NIOSH | FH2100000 |
| PEL (Permissible) | Not established |
| Related compounds | |
| Related compounds |
Acetal 1,3-Dioxane Paraldehyde Trimethylene glycol Ethylene glycol 1,3-Propanediol |

