4,4'-Dihydroxybenzophenone: In-Depth Commentary
Historical Development
Chemists started to pay attention to 4,4'-Dihydroxybenzophenone around the middle of the 20th century, drawn in by its ability to absorb ultraviolet light and stabilize polymers. Companies racing to develop stronger, longer-lasting plastics searched for molecular solutions at a time when sunlight damage was shortening the lifespan of paints, coatings, and synthetic materials. Laboratories in Europe and North America pressed forward with new aromatic ketones, and the molecule’s core—two hydroxyl groups attached to a benzophenone backbone—showed promise. Over time, industrial demand grew, especially once plastic manufacturers realized just how much it could delay yellowing and surface cracking in consumer products exposed to sunlight.
Product Overview
Synthesized as a fine, pale-yellow crystalline powder, 4,4'-Dihydroxybenzophenone became popular for its dual action: absorbing UV and providing chemical stability in challenging environments. Polymer producers turned to it as a crucial additive for transparent plastics, often aiming to extend product shelf life. It found a home in many everyday objects: credit cards, transparent bottles, car coatings—even sensitive scientific equipment. The chemical’s reach expanded further as companies explored it in sunscreen formulations and cosmetic products designed to fend off sun damage, though later discussions raised concerns about skin penetration and hormonal activity. Demand flowed from sectors as wide-reaching as construction, electronics, textiles, and personal care, every industry looking for that edge against UV degradation.
Physical & Chemical Properties
Looking closely at it in a lab, 4,4'-Dihydroxybenzophenone presents low odor, melts at around 210°C, and dissolves modestly in alcohols and most organic solvents but hardly budges in water. Its molecular mass sits at about 214.2 grams per mole. Two phenolic hydroxyl groups define its chemistry, joined by a carbonyl bridge between two benzene rings—this arrangement grants remarkable light absorption in the near-UV range. The powder resists most acids but reacts with strong bases, forming salts that sometimes serve as intermediates for further modifications. Not just stable under normal handling, it handles heat and mild chemical environments without breaking down, a reason it succeeded in harsh industrial processes.
Technical Specifications & Labeling
Regulations govern purity thresholds here. Producers document minimal residual solvents, set impurity caps below 0.5%, and often list melting point, particle size, heavy metals, and even country of origin. Some brands supply it with Certificates of Analysis, detailing batch-by-batch composition. Labels mark all safety hazards, include GHS pictograms, and feature precautionary handling guidelines. Product data sheets highlight recommended dosages in plastics, usually falling below 0.5% by weight, to meet both performance and safety needs. Professionals in regulated environments—especially those mixing it into food packaging or personal care—review these labels closely to stay compliant with global chemical laws.
Preparation Method
Factories prepare it using a Friedel-Crafts acylation, joining hydroquinone—a common phenol derivative—with benzoyl chloride under acidic conditions, often with aluminum chloride as the catalyst. This route directs the carbonyl group into the correct spot, forming a symmetrical structure. After the initial union, chemists neutralize, wash, and recrystallize to achieve high purity. Advances in process control and filtration raised yields and limited waste products, benefiting both supply reliability and environmental profiles. Efforts continue to tweak solvents and recycle catalysts, since green chemistry and regulatory shifts put pressure on producers to limit hazardous byproducts.
Chemical Reactions & Modifications
Because both phenolic groups remain reactive, chemists can modify 4,4'-Dihydroxybenzophenone in several ways: etherification to make UV-absorbing polymers, esterification for more lipophilic derivatives, or electrophilic aromatic substitution to add functional groups for specialized applications. Epoxy resins benefit when these modified molecules integrate into the backbone, making coatings and electronics more durable. Research groups experiment with functionalizing the benzene rings to create brand new additives, exploring whether slight tweaks bring even greater resistance to photooxidation and color change over time.
Synonyms & Product Names
This molecule appears under many guises, and recognizing them cuts confusion. Some call it bis(4-hydroxyphenyl) ketone, benzene-1,4-diol benzophenone, or BP-4. Commercial brands give it names that hint at its role—UVINUL A, Escalol 557, and similar. It pops up in patents with systematic IUPAC names as well, and regulatory registries, such as the EINECS or TSCA inventory, keep listings under all these variations to streamline import and export approvals.
Safety & Operational Standards
Practical work with 4,4'-Dihydroxybenzophenone involves careful handling. Direct dust contact can irritate eyes or skin, so gloves and glasses stay standard in most facilities. Airborne levels above a few milligrams per cubic meter may call for extra ventilation or dust masks, even though acute toxicity rates remain low. The industry keeps inventories within REACH and TSCA compliance, and new developments in occupational safety encourage real-time air monitoring in larger plants. Waste laws steer disposal away from water outflows: authorities treat processes with special scrutiny in regions prone to discharge, and companies feel increasing pressure to report waste treatment details in sustainability reports. Responsible use and worker health have improved as awareness and training broadened over the past decades.
Application Area
Manufacturers rely on it most when they need trusted UV absorption in transparent plastics—polycarbonate, polyethylene terephthalate, and some acrylics top the list. Its use in packaging protects food and medical goods from light-induced spoilage and material breakdown. Electronics suppliers choose it for optical fibers and LCD screens, since even a hint of UV can degrade transmission or discolor housing. Automotive paints, wood varnishes, and even premium sunglasses all feature it as a shield against sun-driven fading. Formulators in personal care steer it into sunscreens and lotions, but growing scrutiny of environmental and human impact encourages safer alternatives where possible. The chemical’s long presence in plastics places it at the core of durability solutions in consumer goods.
Research & Development
A chunk of current research aims to address unwanted side effects—environmental accumulation, hormone mimicry, and long-term stability in recycled materials. Academic centers investigate molecular analogs with better breakdown profiles, so-called “green” UV stabilizers meant for easier downstream disposal and less bioactivity. Industrial labs test combinations: smaller doses blended with other stabilizers or antioxidants, trying for the same performance with lower overall chemical loading. Research funding often flows toward improving compatibility with newer bioplastics and recyclates, as circular economy policies reshape production targets. Collaborative efforts between the chemical industry and independent toxicologists have created consortiums to share data and preempt regulatory surprises.
Toxicity Research
Over the years, toxicological studies indicated that acute exposures hardly trigger health emergencies, but questions linger about chronic, low-level exposures—especially from persistent environmental build-up. Cell and animal testing shows some weak estrogenic activity, sparking fierce debate among health agencies about allowed levels in consumer items. National authorities in Europe and North America regularly review safety thresholds, keeping closer tabs on uses in children’s products and foods. Monitoring programs check soil and water, aware that traces have been found near manufacturing sites. These findings encourage better filtration at plants and motivate companies to look for safer, less bioactive substitutes, especially in sectors where vulnerable groups may encounter residues daily.
Future Prospects
The market trajectory of 4,4'-Dihydroxybenzophenone remains tied to the global debate over chemical safety versus material performance. Plastic and coatings makers rely on its proven function, though regulatory changes continue to raise development risks. With nations ramping up push for safe-by-design additives, demand for safer derivatives—similar performance, lower toxicity and persistence—keeps building momentum. The drive for bioplastic-compatible stabilizers creates space for startups and research groups, but established players invest heavily to adapt legacy products to fit into a more environmentally conscious industry landscape. Progress in analytical chemistry and toxicology will keep shaping rules, and only real, peer-reviewed data—a pillar of scientific trust—will guide industry decisions about where this versatile molecule belongs in tomorrow’s materials.
What is 4,4'-Dihydroxybenzophenone used for?
A Familiar Face in Everyday Plastics
Open the pantry and pick up a plastic water bottle, or glance at the dashboard during a drive. Bright colors and slick finishes meet the eye, but few folks stop to think about what keeps these plastics looking sharp year after year. I’ve handled my fair share of plastic storage bins that sat in the sun and faded to a dull pink in just a season. Companies turn to 4,4'-Dihydroxybenzophenone for a solution. This compound helps plastics resist damage from sunlight, especially from UV rays that break down materials over time. Without it, durable goods lose their color, turn brittle, and head fast toward the landfill.
Why the Polymer Industry Relies on This Compound
Durability isn’t just about looks. In my older home, sunlight streaming in through the window had yellowed blinds and turned garden tools brittle. The backbone of many transparent plastics, including polycarbonate and polyester, bends and cracks easily without strong UV protection. By adding 4,4'-Dihydroxybenzophenone, manufacturers make products like eyewear lenses, food packaging, and electronics casings hold up against constant sun exposure.
This chemical absorbs ultraviolet light, stopping it from passing through and breaking chemical bonds in the plastic. Think about how sunblock works for skin—this molecule does a similar job, providing a shield that keeps products sound for longer.
Reaching Beyond Plastics
The influence of 4,4'-Dihydroxybenzophenone doesn’t end with polymers. In my kitchen drawer, there are receipts and packaging labels printed with inks that keep their contrast far longer than those from a decade ago. Printers and ink makers use this compound to stabilize colors. Certain adhesives and coatings also count on its stability, especially where sunlight or fluorescent lighting pose a risk of fading or weakening.
Personal care products sometimes borrow this molecule too. Health Canada and the U.S. FDA have reviewed its use as a UV blocker in lotions and creams, especially those meant for short-term, topical use. Sunscreen products need a punchy UV filter to protect the skin from burns and aging. This chemical lends its strength, but health authorities keep its concentration well below harmful levels.
Safe Use and Environmental Considerations
Working with chemicals day-to-day, I learned that no substance comes without tradeoffs. Studies have pointed to the persistence of 4,4'-Dihydroxybenzophenone in the environment. This molecule sometimes makes its way out of manufacturing plants and into storm drains, where fish and plants pick it up. The European Chemicals Agency lists it under substances that may require restriction, and some watchdog groups have urged closer scrutiny.
The solution starts with tighter production controls. Companies can invest in better filtration systems and recycling protocols, stopping leaks and spills before they reach waterways. Strong labeling and worker education also limit the amount that enters the air or soil during use.
Innovation and Future Directions
As demand for greener products grows, chemists and engineers push for alternatives with quicker breakdown rates. Some plant-based UV absorbers are on the horizon, showing promise in both lab tests and early commercial runs. Yet, finding a substitute with the same power, cost, and safety as 4,4'-Dihydroxybenzophenone is no small task.
Through science and teamwork, the goal stays clear: keep the benefits while shrinking the downsides. Shoppers can help by choosing recyclable plastics, safe sunscreens, and supporting brands that use cleaner processes.
Is 4,4'-Dihydroxybenzophenone safe to handle?
What’s in a Chemical?
Working with chemicals every day changes how you look at safety. 4,4'-Dihydroxybenzophenone, known in labs for its ability to absorb ultraviolet light, crops up in all sorts of products—from sunscreens to plastics. On paper, it doesn’t look especially scary, but real safety depends on where and how you meet it.
Digging into the Facts
Many labs and manufacturers value this chemical for its protective qualities. Researchers say it helps stop UV rays from breaking down other chemicals or materials. This kind of property brings a sense of double-edged usefulness—good in sunscreen, not so good if it makes its way into rivers or lakes.
Reading through the chemical’s safety sheet, a few red flags pop up. Some studies, especially those looking at exposure in animal models, have found signs of skin and eye irritation. Potential long-term effects spark even more debate. Data show the chemical can disrupt hormone levels in animals under certain test conditions. The European Chemicals Agency tracks it closely, flagging it for possible endocrine-disrupting effects. Even if you trust typical consumer use stays well below those study levels, it’s still worth paying attention to.
Personal Experience in the Lab
I spent years in academic labs running synthesis with dozens of similar aromatic molecules. Gloves, goggles, and lab coats weren’t mere suggestions. After a day handling powders like 4,4'-Dihydroxybenzophenone, the dust clinging to every surface made me grateful for the simple act of washing up. Minor rashes and headaches sometimes happened to colleagues who got careless about protective gear.
Once you lose trust in your gloves by ignoring a tiny cut, you remember every weird molecule sticks with you. I watched a peer skip the face shield, splash a few granules in his eye, and spend the afternoon at employee health.
Looking at the Big Picture
Unlike harsher substances, this compound doesn’t trigger alarms with a single whiff or gentle touch. Still, that doesn’t mean a free pass. Manufacturers regulate the amount used in popular items like coatings, sunscreens, and adhesives. Researchers and safety authorities stay alert for hints of health risk, especially for people handling the raw chemical before it gets diluted or combined with other materials.
Outside the lab, most of us never meet this molecule head-on. Sunscreen formulas, for example, use only trace amounts. In the workplace, those who pour, weigh, or mix it should treat it with real respect. Safety goggles, gloves, lab coats, and solid ventilation offer straightforward defenses. Regular training supports safe habits and helps everyone spot trouble before it turns into an emergency.
Room for Improvement
Better labeling and easy-to-read instructions help staff understand what they’re dealing with. Digital tools such as regular exposure tracking and real-time monitoring keep staff safer, reducing the risk of sneaky low-level exposure that sometimes gets ignored.
Safer alternatives exist for some uses, but switching out a working molecule takes research and investment. Public transparency about chemical safety, especially for people outside research or industry, helps everyone make smarter choices.
Small steps go far—wash up after use, clean surfaces, and share experiences. Building a solid safety culture starts with simple habits, not just rules on paper.
What are the storage conditions for 4,4'-Dihydroxybenzophenone?
The Real World of Chemical Storage
Anyone handling chemicals long enough learns that storing them right matters just as much as the ways we use them. 4,4'-Dihydroxybenzophenone—often called a UV absorber in plastics and coatings—serves a valuable job. It's not hard to store if you follow the basics. This material comes as a white to off-white powder. Its uses drive demand, but safe use begins with common-sense storage.
Why Dry and Cool Matter
Heat and moisture turn chemicals ugly faster than most realize. 4,4'-Dihydroxybenzophenone keeps its strength if it sits in a dry spot below 25°C. Even sealed containers let small amounts of air creep in over time, and moisture from humid air finds a way through. If things get damp, clumping or yellowing becomes a headache—and that changes how the material works. Too much heat doesn’t ruin this compound right away, but it puts stress on the powder and invites chemical breakdown. Many labs and factories forget this when they stack bags in warehouses without airflow during the summer.
Original Packaging: Not Just a Suggestion
Original packaging often gets tossed aside after the first scoop. That turns out to be a mistake. These cartons or drums block out most light, and they use tight-fitting lids with gaskets. Resealing means less contact with humidity and air, which keeps the powder free-flowing and usable. If you pour off what you need and close up right away, you lower the risk of contamination or moisture damage. Companies like BASF and Sigma Aldrich recommend this method for a reason—they know what long-term storage looks like.
Avoiding Cross-Contamination and Spills
Storage shouldn’t happen near acids, oxidizers, or food prep areas. Cross-contamination with reactive substances can trigger slow reactions in storage—or worse, chemical spills that spoil both the 4,4'-Dihydroxybenzophenone and other products. Segregating chemicals isn’t just a best practice; it prevents clean-up nightmares.
Light Sensitivity and Labeling
Even if the powder tolerates light better than some dyes, direct sunlight can don’t do it any favors. Light sometimes fades the color or even changes a small part of the chemical structure over months. A dark corner or an opaque bin solves this. Good labs and production floors label everything—date received, lot number, and expiration. That’s real quality control, not just paperwork.
The Human Element and Safety Basics
I’ve seen people toss powders into whatever closet happens to be open. Gloves, goggles, and basic dust control can go forgotten. 4,4'-Dihydroxybenzophenone dust won’t melt a glove, but repeated exposure builds up, and safety standards exist for a reason. Simple rules—no eating, drinking, or open flames near storage—stop careless accidents before they start.
Pest Control and Shelf Stability
Rodents and insects sometimes make homes in seldom-checked storerooms, chewing through bags and contaminating contents. Keeping chemicals off damp floors and away from walls cuts down on this risk. Regular checks catch spills or damaged containers before they snowball.
Investing in Storage Pays Off
Secure storage with proper signage and restricted access keeps untrained hands away. People make mistakes—clear instructions and locked cabinets go a long way. Investing in sturdy racks and simple climate control prevents costly losses and wasted product. A clean, organized space with the right conditions brings peace of mind—no drama, just steady work.
Can 4,4'-Dihydroxybenzophenone be used in cosmetics?
The Role of 4,4'-Dihydroxybenzophenone
Cosmetics sit closer to the skin than most daily products, which means recognizing every ingredient matters. 4,4'-Dihydroxybenzophenone, a chemical that blocks UV rays, appears in some beauty formulas like sunscreens, face creams, and hair products. Its job is simple: protect contents from sunlight, stop fading, and give your skin an extra shield from ultraviolet harm. More than a few brands include it for stability and preservation—they want their creams to last longer and look the same on the shelf as the day they left the lab.
Health Concerns and Regulatory Guidance
Safety draws plenty of attention, and for good reason. 4,4'-Dihydroxybenzophenone, known as benzophenone-2 (BP-2), falls under scrutiny. Research published in journals like Environmental Science & Technology shows that BP-2 has acted as an endocrine disruptor in animal tests, suggesting it can affect hormone systems. The European Chemicals Agency (ECHA) classifies BP-2 as a substance of very high concern. The United States FDA and Health Canada haven’t banned it outright, but they limit concentrations in both sunscreens and daily cosmetics. The European Union takes a stricter approach, restricting use in body products because of those hormone concerns.
Why Everyday Choices Matter
Mainstream advice says to protect your skin from UV rays—premature aging and cancers are real worries. BP-2 gets the job done, but at what price? For someone like me who’s seen dry patches flare up after trying new face masks, ingredient lists matter. People with sensitive skin or hormonal conditions often track every compound. The connection between repeated, long-term exposure and hormone disruption hasn’t produced clear numbers for humans. Still, seeing rising studies in peer-reviewed journals brings home that even trace levels could build up. A 2020 study found traces of BP-2 in water sources, showing these chemicals don’t just vanish—they follow us down the drain.
How Brands React and What Consumers Can Do
Brands keep their ear to the ground—consumer pressure already shifted many toward paraben-free, phthalate-free ranges. If enough buyers steer clear of BP-2, cosmetics companies will take note. Certifications like “EU compliant” or “reef safe” reflect growing awareness, rewarding companies using newer, less risky preservatives and UV blockers like zinc oxide or titanium dioxide.
Staying informed doesn’t mean hunting down every chemical at home. It comes down to reading labels—any ingredient ending in “benzophenone” deserves a second look. The Campaign for Safe Cosmetics and similar watchdogs regularly update lists of ingredients to question. Dermatologists encourage patch testing and following up on persistent rashes, as individual sensitivity varies.
Dropping 4,4'-Dihydroxybenzophenone from your routine takes a bit of label reading and a willingness to switch brands. But each purchase shapes the industry. Regulation might not move fast, but collective choice always sparks change. For those who value peace of mind and skin comfort, looking for alternatives puts control back in your hands.
What is the chemical structure and formula of 4,4'-Dihydroxybenzophenone?
Chemical Structure Behind the Name
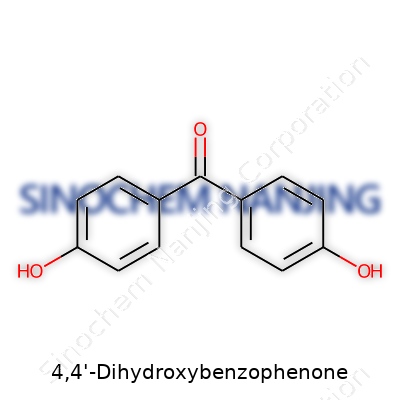
4,4'-Dihydroxybenzophenone carries the formula C13H10O3, which stands out in the family of benzophenones. The compound displays two benzene rings and a central carbonyl group—quite a common frame for molecules with UV-absorbing qualities. Here, the “4,4'-dihydroxy” signals two hydroxy groups, each attached to the fourth carbon of either ring. The IUPAC name, 4,4'-dihydroxybenzophenone, points straight to its symmetry: a benzophenone scaffold with hydroxy groups in mirror positions.
A Closer Look at the Skeleton
The skeletal structure features a central C=O group joining two phenyl rings at the 1-position of each ring. Hydroxyl groups reside at the para position on both rings. Looking at the molecule, this configuration promotes stability and a consistent way of absorbing high-energy ultraviolet radiation. From my own time in the polymer industry, seeing bottles yellow under sunlight brought home how useful these sorts of structures can be in commercial stabilizers for plastics and coatings. If someone flipped over a shampoo bottle in the store and caught “benzophenone-3” on the label, they were looking at a cousin to this compound, all supporting the battle to keep products from breaking down in the sun.
Why It Matters Beyond the Lab
4,4'-Dihydroxybenzophenone does not keep to research shelves. Its ability to trap damaging UV light turns up in the creation of sunscreens, hair sprays, and transparent plastics that stay clear and strong outside. For industrial chemists, the structure’s particular aromatic rings—linked by a carbonyl and decked out with hydroxy groups—make it especially good at soaking up the wavelengths that would otherwise crack, fade, or weaken polymers. Research led by the American Chemical Society pinpoints how these hydroxy additions drastically improve UV absorption compared to plain benzophenone, giving manufacturers a solid reason to reach for the 4,4’-dihydroxy variant.
Environmental scientists have raised flags, though, about where these molecules end up. Once a product washes down the drain, residues land in water streams and persist imperfectly broken, stirring debate around their safety. It’s tough to ignore stories of trace benzophenones showing up in fish tissue. The issue comes down to balance: enjoying reliable UV protection but also keeping an eye on ecological impact. Regulations today already limit concentrations in consumer goods, but the search goes on for safer, biodegradable alternatives—using plant-derived scaffolds or tweaking chemical side groups to nudge break-down in sunlight. Green chemistry groups now run trials with polyphenols and modified natural products, all looking for a better answer that keeps rivers and people safe.
Solutions and Future Direction
From my time as a formulator, I learned that switching to cleaner alternatives carries its own hurdles—cost, shelf life, transparency, and performance under real sunlight. The trick depends on teamwork across chemists, regulatory experts, and manufacturers. Transparency in sourcing, using advanced analytics to track breakdown in the wild, and offering public data about environmental effects will help rebuild trust. Tackling pollution problems linked to established UV blockers points toward adopting safety-minded innovations, but also getting everyone from community activists to large manufacturers on board. That’s an effort everyone has a stake in, not just a technical fix sitting in a test tube.
Key Facts to Remember
Formula: C13H10O3Structure: Two benzene rings, both with para hydroxy groups, connected by a carbonyl (C=O) group at the 1-position.
4,4'-Dihydroxybenzophenone stands as more than a mouthful on a data sheet; its structure shapes durability for everything from clear water bottles to sunscreen, underlining the real-world importance of thoughtful molecular design.
| Names | |
| Pronunciation | /ˌfɔː(r).fɔː(r)ˌdaɪˌhaɪˈdrɒksiˌbɛnzoʊˈfiːnoʊn/ |
| Identifiers | |
| CAS Number | 611-99-4 |
| Beilstein Reference | 1206609 |
| ChEBI | CHEBI:34718 |
| ChEMBL | CHEMBL1426 |
| ChemSpider | 12023 |
| DrugBank | DB14087 |
| ECHA InfoCard | 03edeac8-f578-4147-bc8d-42b7fd3465c2 |
| EC Number | 201-381-2 |
| Gmelin Reference | 81444 |
| KEGG | C10641 |
| MeSH | D004210 |
| PubChem CID | 6928 |
| RTECS number | DO4375000 |
| UNII | 6X8YC6J0E9 |
| UN number | UN3077 |
| Properties | |
| Chemical formula | C13H10O3 |
| Molar mass | 214.22 g/mol |
| Appearance | Pale yellow powder |
| Odor | Odorless |
| Density | 1.38 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 1.88 |
| Vapor pressure | 3.2 x 10^-6 mmHg (25°C) |
| Acidity (pKa) | 7.30 |
| Basicity (pKb) | 8.17 |
| Magnetic susceptibility (χ) | -63.0e-6 cm³/mol |
| Refractive index (nD) | 1.652 |
| Viscosity | 2.14 cP (25°C) |
| Dipole moment | 4.05 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 143.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -361.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2631 kJ/mol |
| Pharmacology | |
| ATC code | D01AE24 |
| Hazards | |
| Main hazards | Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS07, GHS08 |
| Pictograms | GHS07,GHS09 |
| Signal word | Warning |
| Hazard statements | H315, H319, H335 |
| Flash point | 146 °C |
| Autoignition temperature | 500 °C |
| Lethal dose or concentration | LD50 (oral, rat): 3,240 mg/kg |
| LD50 (median dose) | LD50 (median dose): >2000 mg/kg (rat, oral) |
| NIOSH | DJ4800000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 10 mg/m³ |
| IDLH (Immediate danger) | Unknown |

