Looking Deeper into 4,4'-Diaminobiphenyl Sulfate: Practical Realities and Future Hurdles
Historical Development
Curiosity about 4,4'-Diaminobiphenyl sulfate didn’t spring up from nowhere. This compound, built off the backbone of biphenyl chemistry, traces back to the early days of synthetic dyes and plastics. Researchers worked their way through aromatic amine chemistry, guided mostly by experiments with aniline. In industrial history, markets pushed for stronger, more resistant materials and 4,4'-Diaminobiphenyl became a foundational intermediate. The sulfate salt was not simply an afterthought; it grew out of the need for easier handling and improved solubility during manufacture. Each time a new application cropped up—engineering plastics, specialty dyes, or chemical sensors—chemists circled back to this sulfate, tweaking it until it met the demands of the era.
Product Overview: Understanding What It Is and Why It Matters
To people unfamiliar with chemical supply chains, 4,4'-Diaminobiphenyl sulfate sounds technical—almost faceless. In labs and factories, it looks like an off-white or pale yellow powder. It doesn’t stun with beauty or surprise with mystery, but it keeps showing up where strong, high-end polymers grow into airplane wings, membranes, and filtration fibers. Engineers learned that the sulfate form gave them purer results for certain reactions. Research often points to its role in making aromatic polyamides, like Kevlar. This puts real-world value on a product most never see or recognize, except in the toughness of a well-made engineered surface.
Physical & Chemical Properties: No-Nonsense Details
You handle 4,4'-Diaminobiphenyl sulfate, and you notice quickly: it's non-volatile, stable at room temperature, dissolves in hot water, and carries a moderate melting point. This physical profile puts it among the easier-to-store intermediates, though you don’t want to let its dust hang around. Chemically, the two amine groups on each end of the biphenyl give it plenty of reactive sites, adjusted by proprietary tweaks. When chemists want to build long molecular chains, that reactivity matters—it decides whether the resulting fiber or plastic accepts further modifications, how it crosslinks, and what kind of performance it delivers under physical stress.
Technical Specifications & Labeling: Straight Talk
No one wants surprises in a process run. That’s why tight technical specs matter, both to regulatory bodies and to in-house process engineers. Standard labeling on chemical drums typically tells you concentration, identity checks, batch verifications, and contamination limits. Practically, these numbers draw the line between an acceptable lot and one that could throw off yields or trigger hazardous side reactions. End users demand validation, so compliance with labeling requirements isn’t just red tape—it grounds quality and safety in real data.
Preparation Method: Getting to the Source
Not every synthesis route gives the same purity, and that reality chases out any inclination toward shortcuts. Preparation of 4,4'-Diaminobiphenyl sulfate draws from the chemistry of biphenyl and nitro compounds. Traditional methods involve nitrating biphenyl, reducing it to the diamine, and neutralizing with sulfuric acid. Innovators still tinker with the reaction temperature, solvents, and even the type of reducing agent, searching for cleaner, lower-waste methods. Environmental trends push manufacturers to capture byproducts, cut effluent, and squeeze out as much usable chemical as possible from every kilogram of starting material.
Chemical Reactions & Modifications
Once the sulfate lands in a chemist’s hands, potential branches off in many directions. In polymer labs, the diamine component invites coupling reactions with acid chlorides to make polyamides. Some routes aim for azo dye precursors; others want to tweak the molecule for more specialty uses. By fiddling with the biphenyl cores and attaching different groups, chemists chase better thermal resistance or tuned conductivity. The backbone stays the same, but even slight changes ripple out through industries, affecting heat management in electronics or the performance of high-performance coatings.
Synonyms & Product Names: The Language of Chemistry
You might hear it called benzidine sulfate, 4,4'-Diaminodiphenyl sulfate, or diphenyl-4,4'-diammonium sulfate—sometimes more, depending on jurisdiction or manufacturer. This patchwork of naming conventions speaks less to branding and more to chemistry’s messy global village. One name pokes its head in supply regulations, another slips into a grant application, and still another anchors an old patent. Every name signals a different use-case, a unique standard, or a regulatory hurdle, so no one who works with the compound takes its labeling lightly.
Safety & Operational Standards: Why Precaution Isn’t Optional
Ignoring reputation can hurt—benzidine and its salts stirred health concerns decades ago. Exposure carries risks of skin sensitization and concerns over carcinogenicity. Regulations today require careful storage, protective gear, and strict monitoring of workplace atmosphere. Processes run with exhaust management and sealed systems, not because bureaucracy demands it, but because the risks aren’t abstract. Even a small spill means decontamination and documentation, not a quick mop-up. Compliance with handling standards is not just legal; it keeps workers safe from consequences that materialize gradually, sometimes years down the road.
Application Area: More Than Just a Building Block
Most attention goes to the role in tough polymers—aramids, engineering plastics, advanced resins—but uses branch out further. Some research groups examine its value in analytical reagents, such as colorimetric sensors, while others rebuild the molecule for specialized dyes. On a broader front, trace detection methods for environmental or biological monitoring sometimes use derivatives, catching pollutants, or marking proteins. Not every application becomes a mainstream product, but every attempt teaches new lessons about performance and risk.
Research & Development: Where Questions Drive Solutions
Research on 4,4'-Diaminobiphenyl sulfate doesn’t slow down. Teams work to improve synthesis efficiency, lower costs, limit waste, and find greener processes. Others probe the structure for tweaks that could open new application doors, looking beyond the known boundaries. I’ve seen grant proposals and university theses that chase biocompatibility or new analytical uses, digging deeper into mechanistic chemistry the textbooks often overlook. This continual push advances both science and industry, not as some abstract quest, but as a direct response to competitive pressures, regulatory shifts, and environmental imperatives.
Toxicity Research: Not a Forgotten Issue
Carcinogenicity studies from the 20th century still hang over this chemistry. Detailed animal tests and epidemiological work connect exposures with health risks, especially in industrial settings. Regulatory agencies restrict its use in consumer products and require supply chain transparency. More recent work asks if alternatives or modifications can reduce hazards, or if encapsulation can fence in the risks. I’ve watched safety teams refuse to compromise, insisting on monitoring protocols, secondary containment, and periodic medical testing for anyone who spends time near the material. Industry doesn’t forget these lessons, and neither should anyone who uses the compound.
Future Prospects: Hard Choices Ahead
The years ahead will decide if 4,4'-Diaminobiphenyl sulfate keeps its foothold in specialty manufacturing, or if new materials push it aside. Industry searches for drop-in replacements that sidestep toxicity while holding onto durability and process reliability. Regulations evolve, sometimes faster than industrial workflows can pivot, creating real tension between innovation and compliance. I see room for green chemistry to reinvent this niche—biobased alternatives, better catalysts, smarter waste treatment technologies. At the same time, legacy applications don’t shut down overnight. Producers, regulators, and researchers tread carefully, aware that improving safety and sustainability is more marathon than sprint. Only by acknowledging hard tradeoffs and sharing best practices does the sector move forward.
What is 4,4'-Diaminobiphenyl Sulfate used for?
Niche Chemical with Big Impacts
4,4'-Diaminobiphenyl sulfate sounds like a term cooked up in a university lab, yet it quietly shapes some of the things we take for granted. The compound, also known as benzidine sulfate, has played a key role in the creation of industrial dyes. While benzidine on its own raises red flags due to serious health risks, especially cancer, the sulfate form stands as a workhorse in dye manufacturing—helping textiles, leather, and paper come alive with bright, deep colors.
Inside the Dye Industry
Factories put this compound to work mainly to produce azo dyes. These synthetic colors don’t fade easily and keep fabrics vibrant through many washes. I’ve watched the way a bolt of fabric moves through dye baths, soaking up color that’s stable thanks to those chemicals. Many classic reds, blues, and blacks owe their durability to the presence of benzidine-based chemistry.
Colorfastness isn’t just about fashion. Hospitals need crisp, color-coded linens that rarely dull. School uniforms, workwear, and heavy-use fabrics each count on strong color to do their jobs daily. Creating this reliability requires precise chemistry, which is where 4,4'-Diaminobiphenyl sulfate comes in. Its molecular structure supports reactions that give birth to these tough dyes.
Health and Environmental Challenges
It’s impossible to discuss this chemical without bringing up the risks. Scientists and regulators flagged benzidine compounds years ago for causing bladder cancer. Handling or breathing in the dust from benzidine derivatives puts workers at risk. I’ve stood on factory floors, watched safety teams suit up in protective gear, and seen the strict protocols. Companies have to keep everything air-tight to keep exposure low, and they must handle all waste with care.
Many countries, including the United States and European nations, have heavily restricted or outright banned the use of benzidine-based dyes for consumer goods. Textile plants in regions with less regulation sometimes ignore these warnings, leading to dangerous working conditions and environmental dumping. Rivers near unregulated dye plants can turn unnatural shades, and water testing often reveals traces of toxic chemicals. Local fishermen feel the pinch when fish stocks drop. Folks in nearby villages worry about long-term health problems.
Finding Safer Paths Forward
The story of 4,4'-Diaminobiphenyl sulfate spotlights a wider truth: Chemistry walks a fine line between progress and risk. Researchers know this, so today’s chemists spend long nights searching for alternative molecules. The goal is to match the brilliant color—or even improve it—without the cancer risk. Some new dyes use plant-based compounds, while others rely on safer synthetic routes. Regulatory agencies encourage this switch by fast-tracking approvals for less hazardous ingredients and funding research.
Transitioning entire supply chains isn’t easy. Factories need clear rules, technical support, and funding if they’re switching away from legacy chemicals. Collaborative industry groups and academic labs help companies move toward green chemistry. I’ve seen small dye-makers link up with universities to test different formulas. Success stories give hope, showing that strong color and safety can go hand-in-hand. Shifting away from dangerous substances like 4,4'-Diaminobiphenyl sulfate builds trust, protects workers, and keeps local environments alive for the next generation.
What are the safety precautions when handling 4,4'-Diaminobiphenyl Sulfate?
Respecting the Risks
Anyone who has ever spent time in a chemical lab knows you can’t afford to get careless, especially with compounds like 4,4'-Diaminobiphenyl Sulfate. You feel it in your gut: this isn’t baking soda. This substance can cause real harm if you ignore basic safety practices. A single misstep—touching your face after handling the powder, breathing dust, or skipping eye protection—makes trouble show up fast.
Gloves and Gear Matter
You need more than just a lab coat. Splash-proof goggles aren’t just for show; a tiny particle can burn or even blind. Nitrile gloves shield skin from direct contact—latex doesn’t always cut it. Chemical labs love to throw disposable gloves everywhere, but with this compound, change them often. Tears and pinholes happen, and you won’t always see them. Respirators or at least a fume hood should always be running. Even trace exposure over time can harm the lungs.
Clean Workspace, Clear Head
Messy benches invite disaster. Dedicated trays, lined with absorbent paper, keep accidental spills from spreading. After every session, any leftover powder needs safe disposal, paired with a wipe-down using damp cloths—never dry sweeping, which stirs up dust. Some folks assume regular soap works, but decontamination stations with proper solvents are safer. Cold water works better than hot, because hot opens the pores, letting chemicals in faster.
Storage Can't Be an Afterthought
Strong chemicals like this demand sturdy containers, sealed and labeled. Open containers crowding shelves never stood well in my experience; one small jar cracked in a storage fridge once, and we spent hours in PPE cleaning up, fearing for our skin and breathing. Keep the stuff in a cool, dry cupboard far from acids, bases, and oxidizers—it reacts badly to many common chemicals.
Emergency Plans Make a Difference
Imagine chemical splashes or breathing in powder, and you see the need for training that sticks. Quick access to eyewash stations and showers saves people from serious injuries. It seems like a hassle, but practicing those drills is worth everyone’s time. Add clear, laminated spill response charts on the wall—nobody remembers a protocol perfectly when adrenaline floods in. Keep a spill kit nearby, not stuffed in a closet. This isn’t just about washing hands; it’s about knowing exactly what to do in a worst-case scenario.
Why It Matters
Long-term health risks from 4,4'-Diaminobiphenyl Sulfate aren't hype. Chronic exposure shows links to cancer and organ damage. The research isn’t always front-page news, but peer-reviewed studies over decades back it up. A friend who worked in dye production ignored mask protocols, and later faced breathing trouble nobody could fix. Lessons learned the hard way matter more than any warning label.
Better Solutions for Everyone
Smart labs invest in better ventilation, compulsory safety courses, and routine inspections. Personal responsibility helps, but workplaces also need policies with teeth. Fines or written warnings push people to take safety seriously. Supervisors can lead by example, putting on their own respirators and gloves, not just demanding others do it. Transparent reporting of incidents builds trust and keeps danger out in the open, where people can address it.
Straight Talk for Safer Handling
The best workplaces treat safety as a shared job. If you see shortcuts around safety with 4,4'-Diaminobiphenyl Sulfate, call it out. People shouldn’t need a badge or fancy title to demand better. The science is solid, the risks are real, and only practical habits protect you.
What is the chemical formula and molecular weight of 4,4'-Diaminobiphenyl Sulfate?
Digging into the Chemistry
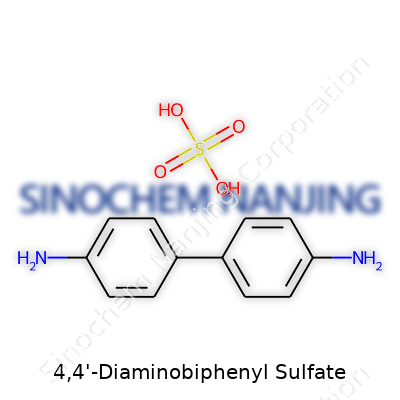
4,4'-Diaminobiphenyl sulfate stands out in chemical discussions, especially for those who’ve worked around dyes or polymers. The chemical formula is C12H14N2O4S. It’s made by treating 4,4'-diaminobiphenyl, which has two amine groups on each benzene ring, with sulfuric acid, leading to a salt that teams up with sulfate. Looking at the numbers, this compound’s molecular weight comes in at 282.32 g/mol. These aren’t just numbers on paper; knowing the formula and weight affects real-world applications, from how labs measure out reactants to safety rules that keep people alert in handling sensitive materials.
Connections to Industry and Health
People who work in chemical plants or research often cross paths with 4,4'-diaminobiphenyl compounds, sometimes without fully realizing the risks. This molecule’s close relatives—like benzidine—have a pretty grim reputation because of their link to bladder cancer, something confirmed more than once in epidemiological studies and safety advisories. Handling the sulfate salt might seem safer, but it pays not to let your guard down. Working in a lab where even small spills or airborne dust could matter drove home for me the need for constant vigilance, gloves, and solid ventilation.
Why Accuracy Matters in Measurements
Take it from someone who once reweighed a batch three times to get the calculations right: the molecular weight guides how much compound to use so reactions don’t go sideways. If the weight is off, experiments fail and time gets wasted. In industry, accuracy affects manufacturing efficiency, costs, and safety records. Training young chemists to cross-check these numbers isn’t just busywork. It prevents dangerous mistakes, cuts down on waste, and keeps production running smoothly. Science doesn’t reward guesswork, especially with chemicals that require respect.
Health and Environmental Watchpoints
Once these chemicals enter the world, concern ramps up about how they interact with both bodies and the environment. Sulfate salts often dissolve in water, raising questions about water treatment and runoff. Communities close to chemical facilities have seen firsthand what happens when vigilance slips: contaminated groundwater, regulatory fines, health scares. The legacy of similar aromatic amines has already shaped tough environmental standards and strict exposure limits. Regulators don’t coddle these chemicals, nor should they. Everyone in the supply chain—lab techs, plant managers, waste handlers—must take personal accountability for storage, labeling, and spill protocols.
Solutions That Make a Difference
It helps when facilities use proper containment—closed systems, fume hoods, and clear labeling do more than check a box. Consistent training in hazard communication and accessible Material Safety Data Sheets (MSDS) turn knowledge into daily practice. Engineers and scientists looking to replace traditional aromatic amines with safer alternatives should follow-through, not just for regulatory compliance but for the coworker standing next to them. Research into greener, less toxic cousins of compounds like 4,4'-diaminobiphenyl sulfate is ongoing. That push for innovation, combined with persistent caution and transparency, keeps chemistry vital and responsible.
How should 4,4'-Diaminobiphenyl Sulfate be stored?
Everyday Lab Safety Starts with Smart Storage
Anyone who’s worked in a lab with organic compounds knows the risks that come with complacency. I remember the first time I saw a storage shelf bow under the weight of poorly labeled bottles—a lesson burned in my memory about why storage really matters. 4,4'-Diaminobiphenyl sulfate sits among those substances you shouldn’t take lightly, both for health and for workplace safety.
Why Respect This Chemical?
This compound shows up in specialty chemical manufacturing and advanced research labs. 4,4'-Diaminobiphenyl itself draws regulatory attention. Its sulfate form brings some of the same concerns, mainly around toxicity and possible carcinogenic outcomes from chronic exposure. The safety data sheets don’t mince words about protective measures—skin contact, inhalation, accidental spills each pose health risks. Many scientists have learned too late that lax storage creates long-term problems, not only for individuals but for the environment that surrounds the lab.
Understanding the Properties—And the Dangers
4,4'-Diaminobiphenyl sulfate’s crystalline form looks harmless, but it doesn’t take much to release dust into the air. Fine particles linger, sticking to workstations and, if mishandled, contaminate clothing and skin. Humidity and heat speed up degradation or release vapors. Improper storage also leads to accidental reactions—especially if it ends up near acids or strong oxidizers. Quick access should never override safe handling principles.
Building a Sensible Storage Routine
In my own experience, the simplest routines often create the safest environments. A designated storage area cuts down on confusion. I’ve seen labs try to save money with DIY shelving, but factory-sealed containers kept on chemically resistant shelves offer peace of mind and meet compliance. High shelves introduce risks during retrieval, so eye-level or waist-level storage cuts down on accidents. Cheaping out on safety backsplashes, spill trays, or clear labelling often ends in regret. Labels need to shout out hazards. Double containment keeps leaks in check.
Environmental Controls Make a Difference
Temperature swings damage a lot of chemicals—sulfates included. Avoiding direct sunlight, maintaining room temperature, and keeping areas dry slow down decomposition and stop unwanted reactions. I’ve worked in places where people stored chemicals near windows “for convenience”—resulting in bottles sweating, cracking, and sometimes leaking. Ventilated storage, preferably with HEPA filtration, minimizes the build-up of chemical dust and fumes—especially important for materials that don’t take well to humid or stagnant air. Keeping incompatible chemicals apart avoids drama from accidental mixing. Clear separation lines, both in charts and in physical space, stop many a crisis before it starts.
Prep for Spills—Don’t Just Cross Your Fingers
Spill kits, gloves, eyewash stations, and emergency contact charts need to sit close by. Regular inventory checks catch problems before they escalate. I’ve watched seasoned chemists run “dry runs”—walking through emergency spill or exposure scenarios—turning what feels like overkill into lifesaving reflexes when mistakes happen. If something spills, a fast response minimizes risk and reduces cleanup headaches in the long run.
Raising the Bar for Future Lab Workers
It always falls on individuals to set a standard for safe chemical storage. Building habits around solid protocols, retraining staff on the basics, and routinely checking for violations have real impacts. 4,4'-Diaminobiphenyl sulfate won’t forgive shortcuts. Responsible storage shows respect not just for the science but for health and legal obligations. Real vigilance—plus reliable supplies, clearly marked containers, and smart placement—keep risks to a minimum and set up the next shift for success. That’s experience talking—not just policy.
What are the potential health hazards of 4,4'-Diaminobiphenyl Sulfate?
Understanding What’s at Stake
4,4'-Diaminobiphenyl Sulfate plays a part in the production of dyes and polymers. Workers in chemical plants, textile factories, or labs stand closest to its potential effects. People outside those circles rarely hear about the compound, but exposure can reach into the neighborhood air, especially near older facilities. Health doesn’t exist in a vacuum. What lands in the lungs or on the skin can follow a person home, affecting families along the way.
Physical Harm: Not Just a Minor Irritation
Skin contact with 4,4'-Diaminobiphenyl Sulfate often leads to rashes or burns. Eyes feel the sting even faster, causing redness, swelling, and sometimes blurry vision. I’ve seen factory employees who shrugged off the warning signs, only to end up seeking medical care for what looked like “just a little irritation” at first. Even a mask or gloves might not stop accidental splashes. The soreness sticks around long after clocking out.
Internal Damage: A Hidden Attack
Breathing dust or vapors brings another, sneakier risk. The compound doesn’t always show its hand immediately. Over weeks or months, workers report chest tightness or a persistent cough. Over time, substances like these, classified as aromatic amines, have shown an ability to interfere with the body at the cellular level. Cancer risk rises. Research on its close chemical relatives backs this up: studies from the U.S. National Toxicology Program connect prolonged aromatic amine exposure to an elevated rate of bladder and liver cancers.
Battle to Track and Limit Exposure
Factories using this chemical must follow strict rules—air quality checks, training, medical monitoring. But safety often boils down to company culture. I’ve spoken with line supervisors who take short cuts to save a few dollars. Management sometimes skips regular air monitoring or lets protective gear run low. In those settings, risk trickles down fast. Unions play a critical role here, pushing back when corners get cut and test results get buried.
Environmental Fallout Reaches Communities
Wastewater runoff doesn’t respect property lines. Towns near industrial plants have reported chemical odors in wells and streams, especially after heavy rain. Young children and older adults carry extra risk, since their bodies deal with toxins less efficiently. Cases from the past—think Love Canal—prove that ignoring possible contamination courts disaster for entire neighborhoods. Regulators must keep a close eye on storage tanks, discharge points, and waste transfer systems.
Seeking Safer Solutions
Switching to less hazardous chemicals often hits roadblocks. Cost, tradition, or a lack of awareness can slow things down. Still, smart companies have found ways to substitute safer alternatives without hurting profits. Sharing those stories—how lower-risk dyes or plastics keep both workers and local families out of danger—could inspire others. External oversight helps too. Transparent reporting of spills, poisoning cases, and air quality levels keeps everyone honest.
Real change starts from the ground up: staff training, strong labor voices, community involvement, tough inspections. Protecting people from the hidden dangers of 4,4'-Diaminobiphenyl Sulfate can't rest on paperwork alone. Every life around the chemical deserves that promise.
| Names | |
| Preferred IUPAC name | Benzenamine-4,4'-diamine,sulfuric acid (1:1) |
| Other names |
Benzidine sulfate Benzidine monosulfate |
| Pronunciation | /ˈfɔːr.fɔːr.daɪˌæmɪnoʊ.baɪˈfiː.nɪl ˈsʌl.feɪt/ |
| Identifiers | |
| CAS Number | 531-85-1 |
| Beilstein Reference | 22261 |
| ChEBI | CHEBI:38747 |
| ChEMBL | CHEMBL1201012 |
| ChemSpider | 23413 |
| DrugBank | DB13702 |
| ECHA InfoCard | 03e6536f-164b-4123-a69a-3ef1d4532c21 |
| EC Number | '2524-66-7' |
| Gmelin Reference | 113060 |
| KEGG | C14372 |
| MeSH | D08IDM7Z9F |
| PubChem CID | 129715689 |
| RTECS number | DA6475000 |
| UNII | V1B40762B7 |
| UN number | UN3438 |
| Properties | |
| Chemical formula | (C12H12N2)·H2SO4 |
| Molar mass | 316.37 g/mol |
| Appearance | Off-white to light brown powder |
| Odor | Odorless |
| Density | 1.29 g/cm³ |
| Solubility in water | Soluble in water |
| log P | -1.6 |
| Acidity (pKa) | 11.02 |
| Basicity (pKb) | 3.54 |
| Magnetic susceptibility (χ) | -86.0e-6 cm³/mol |
| Refractive index (nD) | 1.668 |
| Viscosity | Viscous liquid |
| Dipole moment | 3.55 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 341.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -872.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | –7592 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin irritation, causes serious eye irritation, may cause an allergic skin reaction, suspected of causing cancer. |
| GHS labelling | GHS02, GHS05, GHS06, GHS08 |
| Pictograms | GHS07,GHS08 |
| Signal word | Danger |
| Hazard statements | H302, H319, H351, H373 |
| Precautionary statements | P261, P280, P305+P351+P338, P309+P311 |
| NFPA 704 (fire diamond) | 2-1-0 |
| Lethal dose or concentration | LD50 oral rat 400 mg/kg |
| LD50 (median dose) | LD50 (median dose): 309 mg/kg (Oral, Rat) |
| NIOSH | UY9275000 |
| PEL (Permissible) | 0.1 mg/m3 |
| REL (Recommended) | 0.002 mg/m³ |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Benzidine Biphenyl 4-Aminobiphenyl Benzidine dihydrochloride Benzidine dihydrosulfate |

