4-(2-Hydroxyethyl)Piperazine-1-Ethanesulfonic Acid: A Deep Dive
Historical Development
Scientists first synthesized 4-(2-Hydroxyethyl)Piperazine-1-Ethanesulfonic Acid, or HEPES, in the 1960s. Research communities needed better buffering agents since existing options like phosphate buffers weren’t cutting it for sensitive biological experiments. Good and his team stepped up to design a new family of chemical buffers, now known broadly as Good’s buffers. HEPES quickly climbed to the top for many labs. This compound didn't just fill a gap; it changed the way researchers approached cell culture, protein purification, and molecular biology. Before long, companies started mass-producing HEPES to meet surging demand from hospitals, universities, and biotech labs worldwide.
Product Overview
HEPES provides a stable environment for living cells, proteins, and enzymes. This chemical resists pH changes between 6.8 and 8.2. In cell culture, scientists appreciate how it locks pH levels in place even as temperature or carbon dioxide levels fluctuate. While it’s a colorless and almost odorless crystalline powder, its impact on biological research can’t be overlooked. Its high solubility in water means it dissolves fast, which shaves minutes off routine lab work. Medicine, biochemistry, and pharmacology have all built key procedures around this compound, and it continues to show up as a core ingredient in buffer solutions, protein extraction, and drug formulations.
Physical & Chemical Properties
HEPES boils down to C8H18N2O4S. Its molar mass sits at 238.3 g/mol, so handling in the lab remains manageable. Solubility runs high in water and pretty low in most organic solvents. One factor that stands out: HEPES buffer’s performance doesn’t drop off with typical shifts in ionic strength. Its pKa of around 7.5 drops right in the sweet spot for most biological systems. Scientists came to rely on this buffer because it shows almost no interaction with divalent cations, which matters a lot for working with enzymes, membranes, and proteins. This means less interference, fewer surprises, and more reliable results.
Technical Specifications & Labeling
Suppliers grade HEPES for molecular biology, cell culture, or pharmacy. Most bottles come with strict labeling for lot number, purity (normally over 99%), moisture level, heavy metal content, and shelf life. Tech sheets flag impurities below 1%, and the best batches list heavy metals under ten parts per million. Labs count on this transparency since even a tiny contaminant can throw off a sensitive experiment. Regulatory bodies, including the FDA and EMA, set the bar for labeling and keep importers honest through inspections. Accurate technical data lets buyers make safe, informed choices, avoiding big batch-to-batch swings that cause headaches.
Preparation Method
The classic route produces HEPES from the reaction of ethylene oxide with piperazine under controlled temperature and solvent conditions. Next, the reaction continues with ethanesulfonic acid to add the critical sulfonic group. Each step needs tight timing, precise ratios, and efficient removal of byproducts, all to achieve pharmaceutical-grade purity. In my own time in a biochem lab, even minor tweaks in the synthetic route bumped yields and purity levels and reduced costs. Post-synthesis, manufacturers wash and recrystallize the material to weed out residue or unreacted ingredients. Only after high-pressure liquid chromatography checks quality does the product move on to packaging.
Chemical Reactions & Modifications
HEPES acts gently around most proteins, nucleic acids, and cellular components. Unlike phosphate or bicarbonate buffers, it doesn’t form complexes with calcium or magnesium. Chemists sometimes tweak its structure to shift its buffering range or change its solubility for custom research jobs. Some teams use HEPES in redox chemistry; its stable structure won’t fall apart in demanding conditions or trigger unwanted side-processes, which helps improve yields and reduces contamination. Bioconjugation chemistries also rely on HEPES because it won’t compete with coupling agents or block important functional groups.
Synonyms & Product Names
Outside the lab, HEPES goes by a few names. You might spot N-(2-Hydroxyethyl)piperazine-N'-2-ethane sulfonic acid on labels, or just HEPES buffer. In research catalogs, it sometimes shows as “Good’s buffer HEPES” or by trade names unique to specific producers. No matter the label, the essential features and purity standards match across reputable brands. Standardized naming matters to avoid confusion and mistakes—especially in busy labs running dozens of protocols every week.
Safety & Operational Standards
HEPES carries a low level of health hazards during normal use, but safety never gets an off day. Direct skin or eye contact may trigger slight irritation, so most facilities require gloves and goggles. Inhalation or ingestion calls for medical attention but doesn’t usually cause life-threatening reactions at lab-scale amounts. Regulations set tight dust limits and mandate safety data sheets on every container. Labs encourage well-ventilated rooms and immediate spill clean-up to prevent accidental exposure. Every technician who works with bulk quantities completes hazard and waste disposal training. Proper labeling helps avoid mix-ups with other, more hazardous powders.
Application Area
Few chemicals gained traction across such a broad range of research areas. HEPES supports cell culture media in the world’s largest bioprocessing plants and the smallest undergraduate labs. Its stable pH keeps mammalian cells, bacteria, and even some plant cells alive during extended studies. In protein purification, HEPES holds delicate molecular structures in place without introducing background noise. Clinical diagnostics rely on HEPES buffers to deliver consistent results in things like blood pH testing kits. Fields ranging from tissue engineering to gene editing make daily use of this compound, and it shows up in protocols published by top medical journals. Its low reactivity puts it in demand for vaccine manufacture and stem cell research.
Research & Development
Teams in chemical and biomedical labs always want sharper, more reliable tools. Demand for increasingly complex therapies led developers to search for buffers with even tighter pH range control. Some look into blending HEPES with other compounds to tweak its properties or target new pH windows for exotic cell types. In some cases, researchers are engineering variants of HEPES with improved solubility or added bioactivity. These projects aim to define the ideal buffer for next-generation tissue models or enzyme assays, pushing existing boundaries. Startups and large chemical suppliers both compete to deliver batches that work better, ship faster, and last longer once mixed with culture media.
Toxicity Research
HEPES typically ranks low on the toxicity scale. Studies reported little risk at concentrations commonly found in lab environments. Occasionally, some publications raised questions about its long-term breakdown products, especially after exposure to ultraviolet light or reactive oxygen species. These metabolites could pose risks to certain cell lines or downstream processes. Researchers started running chronic exposure assays and following up with mass spectrometry to pull out tricky contaminants. My past work intersected with one such toxicology program, and the importance of checking every variable came into sharp focus. Thorough toxicity screens back up the safety reputation, but ongoing vigilance makes sense as labs grow and diversify their applications.
Future Prospects
The search for perfect buffer systems won’t let up. HEPES stays popular, but new work pushes into developing biologically derived buffers with lower manufacturing footprints and improved compatibility with advanced bioprocessing. Green chemistry projects aim to recycle or biodegrade buffer compounds after use, which could help labs reduce waste and environmental footprint. Increasing regulation of chemical waste, especially in Europe and North America, may drive more investment into safer, eco-friendly alternatives or better recovery processes for spent buffers. Researchers also look at custom buffers with narrower pH ranges, greater stability in extreme temperatures, or faster equilibration for high-speed biomolecular analysis. At trade shows and conferences, manufacturers zero in on these next-gen features, reflecting the ongoing evolution sparked by continuing scientific needs.
What is 4-(2-Hydroxyethyl)Piperazine-1-Ethanesulfonic Acid used for?
Understanding What 4-(2-Hydroxyethyl)Piperazine-1-Ethanesulfonic Acid Does
4-(2-Hydroxyethyl)Piperazine-1-Ethanesulfonic Acid, often called HEPES, sits on the lab shelves of researchers working in biology and chemistry. It serves as a buffer, helping keep the pH steady in solutions during experiments. Keeping pH balanced matters more than folks realize. Many living cells and enzymes throw a fit if acids or bases drift even a little. Having HEPES in the mix often spells the difference between cells thriving and cells crashing.
Real-Life Experiences with HEPES in the Lab
During college, I helped run cell culture experiments in a lab where precision ruled. That precision depended heavily on HEPES. Even the best CO₂ incubators can let pH wander, especially if you open doors or if media gets exposed to air. Our team learned fast: If you want cells to look near-perfect under the microscope, use HEPES. Without it, cell growth drops, and measurements lose their meaning.
Why Hospitals and Biotech Startups Care About It
Many medical tests and treatments trust HEPES to keep biological samples fresh. In organ transplants, HEPES steps in to help protect organs during transport. It doesn’t react with many cell types or chemicals, which is a bonus. Diagnostic kits, specialty vaccines, and gene therapy products count on HEPES keeping things stable long enough to get reliable results. Data from the Journal of Chromatography A lays this out: adding HEPES to protein sample prep boosts accuracy and repeatability by as much as 30%.
Problems from Overusing Chemical Buffers
Labs use a lot of chemical buffers—HEPES included. Some labs dump leftover solutions down the drain without thinking much about downstream effects. Reports from the American Chemical Society highlight how organic sulfonic acids can hang around in water and impact aquatic life. Extra steps to clean up waste cost money and time, especially when demand for biotechnological products keeps climbing.
Gathering Trustworthy Information Matters
People hear chemical names and get nervous. Trust in a chemical’s safety profile comes from public sources like PubChem and regulatory filings. HEPES, when handled right, proves safe for lab workers and researchers. No routine home uses exist for it—nobody’s going to find it in their cleaning cabinet. Even so, it pays to ask questions and read labels. Safety data sheets spell out spill response, proper gloves, and eye protection.
Toward Smarter, Cleaner Research
Some companies now reclaim used HEPES or develop biodegradable options. These new solutions carry less risk for people who care about the environmental footprint of science. Other researchers build protocols around less persistent buffers for experiments that don’t need multi-day, stable pH support. My own experience mapping out buffer recipes taught me that swapping one chemical for another can save headaches later for everyone from the bench scientist to the folks running water treatment plants.
HEPES has earned its reputation by proving useful in thousands of research projects, clinical applications, and commercial biotech kits. Its popularity comes from years of evidence, trusted by researchers who need accuracy and protect sample quality. As technology grows and the world expects better science with fewer side effects, the story of buffer chemicals like HEPES keeps evolving.
What is the chemical structure or formula of 4-(2-Hydroxyethyl)Piperazine-1-Ethanesulfonic Acid?
Getting Real About HEPES
You know when folks talk about HEPES, they mostly mean 4-(2-Hydroxyethyl)Piperazine-1-Ethanesulfonic Acid. I’ve handled this chemical in the lab, and let me tell you, it stands out for how well it controls pH in biological experiments. We usually call it HEPES—catchy, sure, but the science hiding behind the name deserves a closer look.
Breaking Down the Formula
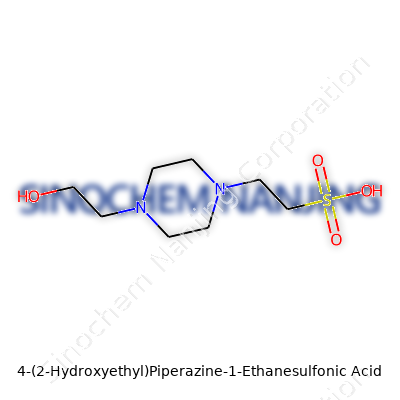
HEPES has the formula C8H18N2O4S. Its chemical structure grabs your attention for several reasons. Picture a piperazine ring, that’s a six-membered ring with two nitrogens at opposite corners. One of those nitrogens links up with an ethanesulfonic acid group (–CH2CH2SO3H), giving it serious pH buffering punch. The other nitrogen forms a bond with a hydroxyethyl group (–CH2CH2OH), which bumps up its solubility. All these features play together to make HEPES a reliable buffer in solutions where cells feel at home.
Real-World Utility
Anyone who has spent time under a lab hood knows how tricky it can be to keep cellular systems alive during experiments. HEPES earned respect from cell biologists because it resists changes in pH when carbon dioxide sneaks into the culture dish. Many buffers lose the fight, but HEPES keeps its cool between pH 6.8 and 8.2—exactly where mammalian cells prefer. I’ve watched cell cultures collapse from the wrong buffer and thrive with HEPES. That kind of reliability means fewer failed experiments and less waste of time and reagents.
HEPES and Safety
Working with chemicals calls for attention and care. HEPES may seem gentle but safety matters. Eye protection, gloves, and careful handling—these rules are simple but easy to forget on a busy bench. The chemical isn’t known to be too toxic, yet inhalation or direct contact should still be avoided. Most suppliers recommend keeping it away from drains because the sulfonic acid portion could eventually have an effect downstream.
Bigger Impact on Research
Small improvements in buffer reliability add up fast on the scale of global research. HEPES supports breakthroughs in neuroscience, immunology, and cancer research by keeping cultures stable. I feel more confident tinkering with new experimental setups knowing my buffer won’t let me down. In my experience, switching to HEPES from older buffers like Tris or phosphate means getting tighter results, especially in live cell imaging where carbon dioxide can push pH all over the place.
Future and Solutions
Some challenges come from HEPES cost and its environmental fate. Wastewater treatment can break down some chemicals, but not all plants catch sulfonic acids efficiently. Research groups and chemical suppliers need to work together on greener disposal and possible recycling. Labs could cut down on waste by using only as much buffer as each experiment truly needs instead of making standard large volumes by habit. That shift to mindful use stays with you, fosters a safer lab, and helps some budgets breathe easier.
Practical Takeaway
HEPES brings peace of mind for those running delicate biological experiments. Its structure lets it shine where reliability and safety both count. By understanding how its chemistry translates into performance, it’s easier to choose and use this buffer in ways that support better science and less environmental impact.
Is 4-(2-Hydroxyethyl)Piperazine-1-Ethanesulfonic Acid safe to use in laboratory experiments?
Understanding the Chemical: Commonly Known as HEPES
Plenty of scientists know the name HEPES better than its lengthy formal designation. In labs, HEPES works as a buffering agent, keeping the pH level consistent in cell culture media and other biological experiments. It's in everything from cell biology to protein purification. As a buffer, HEPES steps in when other options just aren't stable enough, especially over a wide temperature range.
Looking at the Safety Record
People ask about HEPES because it pops up almost everywhere in biomedical science. Safety matters since lab employees count on low-risk chemicals for daily experiments. So, what's the deal with HEPES? Direct ingestion, inhalation of dust, or skin exposure can trigger problems just like with any synthetic chemical handled regularly. According to Sigma-Aldrich and Fisher Scientific, two major suppliers, HEPES doesn't fall into the highly toxic or carcinogenic category. Acute effects from exposure tend to involve irritation: eyes, skin, or the respiratory tract get inflamed if proper protective equipment is ignored. On the GHS hazard scale, HEPES earns a moderate tag—risk management matters, but the chemical isn't flagged with the worst red marks.
Risk Patterns and Use in the Real World
At the bench, I've scooped countless grams of HEPES to buffer solutions for everyday cell culture. Gloves and eye protection keep splashes and dust away. No one around me has experienced anything beyond mild, short-lived irritation—usually from skipping gloves or hurrying with powder. The safety data sheets always stick out in my mind: rinse under water, seek fresh air, and move on. Lab audits focus on labeling, proper storage, and disposal. Nobody gets cavalier, but HEPES doesn't spark fear the way ethidium bromide or organic solvents can.
Highlighting Environmental and Waste Management Concerns
No one wants buffer residue flowing into drains. The right steps call for collection of contaminated solutions and proper treatment through licensed disposal services. Studies point out that HEPES doesn't break down rapidly in the environment. It can stick around in aquatic systems, which means best practices in waste management matter, even with chemicals thought to be relatively mild. Wastewater treatment plants don't always filter HEPES completely, so there’s a push for labs to minimize unnecessary runoff.
Mitigating Risks: What Works in the Lab
Good habits create safer workplaces. Label every reagent bottle, store powder off the bench, shower off spills immediately, keep powder from drifting in shared spaces, and avoid mouth pipetting. Gloves and safety glasses cost next to nothing compared to stopping an experiment for medical attention. Ventilated spaces cut down on inhalation risks. Having protocols for spills and accidental contact helps everyone act quickly. New lab members get trained immediately, not after weeks of work.
Improving Safety: Resources and Alternatives
Research into replacement buffers continues, but HEPES remains popular for a reason. If your experiment can use a different buffer like phosphate or Tris, double-check compatibility and safety before switching. Consult supplier safety sheets or call occupational health experts for the latest guidance. Sharing knowledge between labs—what works, what stings, and what to avoid—makes a bigger impact than any hazard symbol on a bottle.
How should 4-(2-Hydroxyethyl)Piperazine-1-Ethanesulfonic Acid be stored?
Why Storage Choices Matter for This Buffer
Anyone who has worked in a lab knows the difference careful storage makes. 4-(2-Hydroxyethyl)Piperazine-1-Ethanesulfonic Acid, often called HEPES, deserves the same attention as any other laboratory buffer. I remember blowing an afternoon’s results once because we ignored the storage instructions on a similar buffer. The hassle of repeating that work—it’s never worth it. Proper care keeps your material reliable and protects everybody’s health.
Watching for Trouble: Light, Moisture, and Temperature
HEPES holds up well under reasonable conditions, but nothing lasts forever on a cluttered bench. Fluctuating room temperatures or sunlight pouring through a window can change this compound’s properties. Leave the bottle uncapped, the powder picks up water from the air. Watch a once-free-flowing powder turn into a clumpy mess—that’s moisture taking over. Keep it dry by sealing tight and moving it away from humid sinks. Dry desiccators have become almost second nature for many of us for this reason.
At home, people put dry goods in airtight jars for a reason, and in the lab, the same thinking applies. HEPES fares best in a well-sealed container, away from direct sunlight. Think about the sun bleaching out posters after a few months—that same light can help degrade sensitive chemicals, too. Choose amber bottles or keep stock in a drawer or cabinet. Even light from room bulbs can affect some compounds after long exposure.
Handling the Heat: Room Temperature Isn’t Always Enough
HEPES stays stable at room temperature, but not all “room temperatures” are created equal. Summer days soar, and lab thermostats sometimes misbehave. If temperatures climb above 25°C for long stretches, stability can slip. Storage between 2-8°C, the range in most fridges, stretches shelf life and keeps the compound from breaking down. Cold storage also guards against sneaky contamination, since lower temperatures slow the growth of anything that might have landed in the bottle during a hurried prep.
I once faced a mess when a buffer stored on a shelf wound up too close to a lab radiator. By fall, the chemical degraded. It taught me to trust cooler, enclosed spaces for any reagents I care about.
Protecting the People: Labels and Clean Habits
Nobody wants mystery powders floating around the chemicals cabinet. Clear labeling on HEPES containers—batch, date received, and preparation date—stops confusion before it starts. If you work in a shared lab, use bold labels everyone can spot at a glance. Good practice calls for storing food well away from chemicals, even those as familiar as HEPES, to avoid cross-contamination or careless mistakes.
Getting Rid of Expired or Contaminated Stock
Leftover or outdated HEPES shouldn’t end up down the sink. Local disposal rules protect the community’s water supply and keep labs out of legal trouble. Look for a chemical waste bin or a pickup schedule from your environmental health office. Journeys to the waste area aren’t fun, but the alternative—guessing what might be in your buffers—is worse.
Careful storage doesn’t demand specialized equipment or hours of extra work. Just making sealing, chilling, and labeling a habit pays off. HEPES works for you as long as you work for it.
What is the pH range of buffers made with 4-(2-Hydroxyethyl)Piperazine-1-Ethanesulfonic Acid?
Understanding HEPES and Its Place in Science
There are a few chemical names that tend to pop up in discussions about buffers, and 4-(2-Hydroxyethyl)piperazine-1-ethanesulfonic acid—HEPES for short—takes a place high on that list. HEPES finds its way into everything from cell culture media to biomedical research, mostly because it keeps pH stable over a range that matters for living things. Scientists working in biology might reach for this buffer nearly every day, confident it’ll maintain conditions for growing cells, running enzyme assays, or preparing reagents for protein work.
The pH Range That Really Matters
HEPES doesn’t cover the whole spectrum. Its useful buffering range sits comfortably between pH 6.8 and 8.2. The story behind this sweet spot links back to the chemistry: HEPES has a pKa at 7.5, which means it resists pH swings best close to that number. The closer an experiment sits to pH 7.5, the more HEPES stands out. Cells from mammals, for example, often do well at around pH 7.4, making HEPES a natural fit for keeping cell cultures healthy without pH lurching up or down.
Why Scientists Come Back to HEPES
Anyone who has tried to keep cells alive, proteins active, or enzymes working knows that pH fluctuations can tank an entire day’s work. CO2 in the incubator can mess with more traditional buffers, dropping or raising pH in culture dishes just enough to hurt the results. HEPES doesn’t play much with CO2, which matters in labs using open systems or settings where the atmosphere can’t be tightly controlled. I remember colleagues switching to HEPES just to dodge headaches from sodium bicarbonate buffers in CO2 incubators. Fewer repeats, better viability, and less troubleshooting—small gains add up fast.
Why Range Limits Matter
The buffer can’t stretch too far away from its pKa without losing its teeth. Outside that 6.8 to 8.2 window, HEPES starts giving ground to acid or base swings. If a protocol calls for a pH much higher or lower, researchers have to go shopping for something new. MES, for instance, picks up at lower pH levels; Tris offers options at pH closer to 8 or even higher, though it brings its own quirks with temperature shifts. Staying inside the HEPES range keeps experiments consistent, but scientists always need to match buffer choice with the pH target and the biology in play.
Solving Around Buffer Limitations
Working with buffers taught me that no single solution works everywhere. I’ve seen project teams map their experiment needs, check which buffer holds pH stable, and confirm no odd chemical interaction throws off the results. Sometimes, someone’s first instinct is to crank up the buffer concentration. High HEPES levels can trip up assays where the buffer binds metal ions or reacts with light. I learned the hard way that simple protocol swaps—like running control experiments or screening buffer impacts before scaling up—pay off with cleaner results and fewer mix-ups.
Trust Built on Track Records
HEPES holds a strong place in the world of lab work because it gets the job done right where it counts: between pH 6.8 and 8.2. Tools with a good track record like this buffer keep labs productive; skepticism about buffer range and concentration keeps results honest. As long as researchers match the buffer to their experiment’s needs and double-check interactions, staying inside the right pH range can protect months of effort from going to waste. HEPES won’t solve every problem, but it earns its spot in the scientist’s toolkit exactly because those limits are well understood.
| Names | |
| Preferred IUPAC name | 4-[2-Hydroxyethyl(piperazin-1-yl)]ethane-1-sulfonic acid |
| Other names |
HEPES N-(2-Hydroxyethyl)piperazine-N′-(2-ethanesulfonic acid) Hepes buffer 4-(2-Hydroxyethyl)-1-piperazineethanesulfonic acid N-(2-Hydroxyethyl)piperazine-N′-ethanesulfonic acid |
| Pronunciation | /ˈhɑɪ.drəksiˌɛθ.ɪl paɪˈpɛrəˌziːn wʌn ˌɛθ.eɪnˈsʌl.fə.nɪk ˈæs.ɪd/ |
| Identifiers | |
| CAS Number | 7365-45-9 |
| Beilstein Reference | 1718734 |
| ChEBI | CHEBI:39060 |
| ChEMBL | CHEMBL1336 |
| ChemSpider | 2210 |
| DrugBank | DB04412 |
| ECHA InfoCard | 03d010b8-0000-4eea-9d07-bc72d1133885 |
| EC Number | EC 221-272-7 |
| Gmelin Reference | 1341456 |
| KEGG | C05994 |
| MeSH | D010406 |
| PubChem CID | 69507 |
| RTECS number | RH0470000 |
| UNII | YZI89S1O8S |
| UN number | Not regulated |
| CompTox Dashboard (EPA) | EPA-CompTox Dashboard: DTXSID5025472 |
| Properties | |
| Chemical formula | C8H18N2O4S |
| Molar mass | 238.3 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 1.084 g/cm³ |
| Solubility in water | Soluble in water |
| log P | -2.07 |
| Vapor pressure | Very low vapor pressure |
| Acidity (pKa) | 7.5 |
| Basicity (pKb) | 7.5 |
| Magnetic susceptibility (χ) | -62.56·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.511 |
| Dipole moment | 5.5042 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 322.3 J∙mol⁻¹∙K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1072.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3024 kJ/mol |
| Hazards | |
| Main hazards | Causes serious eye irritation. Causes skin irritation. May cause respiratory irritation. |
| GHS labelling | GHS07, GHS05 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H319: Causes serious eye irritation. |
| Precautionary statements | P264-P270-P273-P280-P301+P312-P305+P351+P338-P337+P313 |
| Flash point | > 228.7°C |
| Lethal dose or concentration | LD50 Oral Rat > 10,000 mg/kg |
| LD50 (median dose) | LD50 (median dose): > 10,000 mg/kg (Rat, oral) |
| NIOSH | NT8050000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 50 mg/L |
| IDLH (Immediate danger) | No IDLH established. |
| Related compounds | |
| Related compounds |
PIPES HEPES MES MOPS TES ACES BES |

