Looking Into (3R,4R)-N,4-Dimethyl-1-(Phenylmethyl)-3-Piperidinamine Hydrochloride: More Than Just a Compound
Historical Development
Chemistry never develops in isolation. Decades of work studying substituted piperidines have brought forward molecules like (3R,4R)-N,4-Dimethyl-1-(Phenylmethyl)-3-Piperidinamine hydrochloride. Once relegated to niche research, such compounds now flag big changes in drug development and synthetic chemistry. Back in the late twentieth century, scientists searched for new structures that broke out of the mold. The piperidine framework, once just a pharmacophore for antihistamines, soon pulled in interest for its neuronal and psychoactive effects. Structural tweaks, like the addition of that phenylmethyl group and methylation, offered enhanced selectivity or potency. This led research towards chiral molecules, since subtle differences in three-dimensional structure often mean the difference between relief and side effects. Academic labs started publishing routes for preparing these chiral amines, and industry took note as well, eyeing possible new medicines and research tools.
Product Overview
The molecule at hand, with its (3R,4R) chirality, tells a story about the focus on enantiopure substances, not just racemic blends. That N,4-dimethylation and phenylmethyl substitution aren’t accidental. They cause downstream effects on the molecular binding and activity that drive both discovery and caution. This compound finds itself at the intersection of medicinal chemistry and neuropharmacology, sometimes even behavioral science. Researchers pick it apart to study structure-activity relationships that can inform the next anti-depressant, stimulant, or analgesic compound. The presence of a hydrochloride salt form boosts water solubility, which doesn’t seem flashy until you hit a bottleneck making solutions for experiments or formulation.
Physical and Chemical Properties
The hydrochloride salt presents as an off-white solid, and experience in the research lab shows it readily dissolves in water and many polar organic solvents, which can be critical for handling and experimentation. This specific salt form helps with both stability and bioavailability — lessons learned after seeing different batches degrade on the shelf or crystallize differently. The piperidine core adds structural rigidity, but the choice of substituents punches up lipophilicity, which plays into its ability to cross biological membranes. Its basic amino groups signal a need for care: they can accept protons or participate in various chemical reactions, and can lead to unexpected reactivity in the wrong storage conditions or mixtures.
Technical Specifications and Labeling
Any bottle in a credible lab comes with clear labeling — correct identification and concentration turns into both safety and scientific reproducibility. From handling masses down to the milligram, the purity, usually higher than 98 percent for key experiments, becomes a key variable. Trace impurities occasionally slip through, and this turns ambiguous results from clear findings. Researchers scrutinize the optical rotation to confirm the (3R,4R) configuration. No one wants a bad batch to derail months of work or, worse yet, publish irreproducible data. Reliable suppliers now offer certificates of analysis; finding the melting point and NMR data posted right there on the label speaks to how far quality control has come.
Preparation Method
Synthetic routes reflect what’s at stake in modern chemistry. In the past, older methods gave low yields, needed harsh reagents, and struggled to generate just the right enantiomer. Today, researchers employ asymmetric catalysis, chiral auxiliaries, or resolve racemates to achieve the (3R,4R) configuration. The sequence generally starts from a substituted piperidine precursor, activating and protecting groups at key steps before introducing the phenylmethyl substitution and methyl groups. Hydrogenation, alkylation, and crystallization round out the synthesis, followed by rigorous purification. This meticulous approach matters because the field demands precision — both for biomedical research and later as an assurance of safety and efficacy in clinical settings.
Chemical Reactions and Modifications
The molecule’s versatility branches from its functional groups. Chemists modify the amine to attach fluorophores, radiolabels, or various pharmacophores. In my own experimental troubleshooting, swapping the phenylmethyl group allows evaluation of receptor binding profiles, which can shift dramatically with modest changes. The amine and methyl groups invite derivatives and salt formation, expanding both its chemical diversity and application area. The piperidine backbone resists spontaneous breakdown, so the compound survives tough reaction conditions — a gift for researchers looking to probe biological mechanisms without playing a guessing game about breakdown products.
Synonyms and Product Names
Precision in language protects against costly mistakes. This compound sometimes appears under alternative nomenclature derived from IUPAC conventions or legacy naming systems, which can cause confusion when ordering or replicating studies. Synonyms often reflect the chiral descriptors or focus on key substitutions, and researchers sometimes refer to it by its “research chemical” code names in proprietary settings. Mislabeling or ambiguity increases the risk for mishandling, which I’ve seen lead to errors in inventory and protocol execution in more than one lab. That’s why clear chemical identity, both among colleagues and in publication, should never be taken for granted.
Safety and Operational Standards
Safety isn’t about bureaucracy; it’s about getting home healthy at the end of the day. This compound, like many research chemicals, calls for gloves, eye protection, and use inside a fume hood. The basic amine can irritate mucous membranes or cause sensitization with prolonged exposure — lessons that sometimes come hard for those skipping PPE on a rushed afternoon. Hydrochloride salts, while less volatile, still demand care to prevent spreading residues across shared spaces. Spill response procedures and segregated waste ensure everyone else’s work (and health) stays protected. Institutional safety data sheets provide reference, but respecting the compound’s reactivity means never improvising disposal or storage.
Application Area
Academic and industry labs prize this molecule as a probe for neurotransmitter systems, especially those involving dopamine and norepinephrine pathways. Behavioral pharmacologists use it to test hypotheses about movement, motivation, and mood. Synthetic chemists harness its chemical flexibility as a building block for making new analogs. A drug discovery team might scan dozens of derivatives to map activity in preclinical assays, watching for a pattern that justifies clinical trials. Its availability for research purposes, but not direct therapeutic use, ensures that most work involves tightly controlled conditions and expert oversight.
Research and Development
Curiosity and real-world needs drive R&D forward. Researchers probe how small changes in piperidine derivatives translate to vast shifts in pharmacological action. At times, one analog turns out highly active on a certain receptor, another stays completely inactive, and mapping these changes leads to better drugs and safer compounds. Collaborations between chemists, pharmacologists, and toxicologists turn into iterative cycles of synthesis, testing, and analysis. In recent years, some teams have investigated its analogs for potential roles in psychiatric or neurodegenerative disorders, looking for improved efficacy or fewer adverse effects compared to older drugs. Grants push for answers — can subtle tweaks on the scaffold produce a safer stimulant or a more reliable probe? Feedback from these cycles helps refine synthetic methods, streamline purification, and minimize unwanted byproducts.
Toxicity Research
Every step into a new chemical territory demands a rigorous look at toxicity. Preclinical work evaluates acute and long-term exposure, effects on cardiovascular, hepatic, and neurological systems, and potential for abuse or dependence. Structural resemblance to known psychoactive substances brings both promise and risk. Some derivatives pose little hazard at research doses, while others elicit potent overstimulation or neurotoxicity. Animal models highlight metabolic pathways that might generate reactive intermediates inside the body, which can cause unexpected side effects. Safety glasses and fume hoods do part of the work, but clear communication about risk — in both publication and daily lab life — completes the picture. The focus should always stay on evidence, clear reporting, and ongoing review, so findings don’t get lost in translation between bench and bedside.
Future Prospects
This molecule and its class will keep attracting attention. Drug discovery in psychiatry and neurology rarely finds a magic bullet, so chemists and biologists keep circling back to versatile scaffolds like substituted piperidines. Advances in asymmetric synthesis could unlock access to new enantiomers, maybe opening windows on receptor subtypes or metabolic pathways so far untouched. Better labeling, digital inventory, and cross-lab collaboration will keep errors and redundant work to a minimum. Ongoing study into modes of action and side effect profiles will help steer clinical interest, even as regulatory pressures keep a tight leash on experimental compounds. Seeing the line between tool compound and medicine will always demand old-fashioned skepticism, careful reporting, and curiosity. The lessons learned here go beyond one molecule, shaping how chemistry and biology address society’s hardest puzzles: finding answers, not just more data.
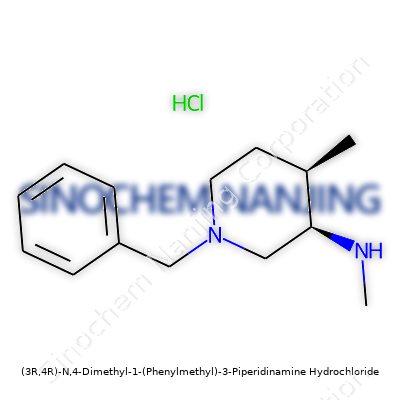
What is the chemical structure and molecular formula of (3R,4R)-N,4-Dimethyl-1-(Phenylmethyl)-3-Piperidinamine Hydrochloride?
Why Chemical Details Matter in the Real World
Someone looking at a name like (3R,4R)-N,4-Dimethyl-1-(Phenylmethyl)-3-Piperidinamine Hydrochloride might see little more than a jumble of letters, but to a chemist, this name tells a whole story. The backbone—piperidine—crops up often in medicines and research tools. I’ve sat at a bench mapping out these structures, finding the rings, methyl groups, and sometimes scratching my head over the orientation of atoms, which can change a chemical’s entire character. The specifics aren’t just academic; they drive everything from how the molecule dissolves, to how it reacts in the body, and how it’s stored or transported.
Breaking Down the Structure
The core foundation here is piperidine, a six-membered ring with one nitrogen atom. That ring gets decorated: at the third and fourth spots (using standard chemist numbering) you’ll see methyl groups—straightforward, simple — yet they define a big part of how other molecules interact. The R/R notation (3R,4R) nails down the exact 3D shape. It’s like saying “left glove, left glove”—not just any glove will fit.
That “N,4-Dimethyl” tag means the nitrogen atom itself wears one methyl group, while the fourth ring carbon gets another. The “Phenylmethyl”—that’s just a fancy name for a benzyl group—hooks onto the nitrogen as well. I’ve handled molecules with similar add-ons, and they tend to increase the compound’s weight and sometimes its stickiness to other biological targets.
Adding hydrochloride to the end means the chemist paired the amine with hydrochloric acid. Simple enough in the lab—just gas HCl through a solution or shake some acid in, and out drop the crystals. This tweak is common. It improves shelf life, sometimes even taste, and often enhances water solubility.
Molecular Formula and Structural Insights
Piecing all this together from the name, here’s what you get: C14H22N2 as the neutral compound. The hydrochloride salt bumps that up to C14H22N2·HCl.
Structurally, imagine this: a chair-shaped six-membered ring (piperidine), nitrogen at the top, methyl groups at two positions, a benzyl group veering off the nitrogen, and ion pairing happening with a chloride anion.
The R-stereochemistry shapes the 3D world of this molecule, deciding which way atoms point. This can decide everything from biological activity to legal control. For scientists, this means stringent synthesis controls, careful measurement with chiral HPLC or NMR, and rigorous MS to check exact masses—processes I’ve sweated over more times than I care to admit.
Real-World Impact and Responsible Handling
Identifying the correct structure and salt form goes beyond paperwork. For pharmaceuticals, the difference between enantiomers can sway a drug’s benefits and side effects. I’ve seen clinical candidates get tossed out because the “wrong” version produced changes no one wanted.
In the lab, mistakes in salt form or structure turn into wasted time, raw material, and risk—especially for compounds with controlled uses or unknown toxicity. Proper documentation, double-checking stereochemistry, and compliance with global chemical databases like PubChem or ChemSpider help keep research transparent and safe.
Anyone handling a molecule like this should start with the best references. Double-check suppliers, confirm identity, and file away spectra. Mistakes echo, especially in regulated settings or in research with public health stakes. In my experience, thinking like a detective and keeping careful records prevents headaches—and sometimes much worse—down the line.
What are the primary applications or uses of (3R,4R)-N,4-Dimethyl-1-(Phenylmethyl)-3-Piperidinamine Hydrochloride?
Roadmap to Its Main Roles
Few outside the world of chemistry can recite a compound name like (3R,4R)-N,4-Dimethyl-1-(Phenylmethyl)-3-Piperidinamine Hydrochloride, but those who spend their lives working in pharma or biotech research tend to see names like this crop up often. Even before a compound gets anywhere close to a consumer product—much less a prescription—molecules like this serve essential, behind-the-scenes roles.
I’ve sat through my share of early-morning research meetings where synthesis teams race the clock, hunting for new treatments or new building blocks. Here, the value of a chiral amine such as this jumps right out. Its specific arrangement of atoms lets drug designers build molecules that “fit” better into the shapes of biological targets—enzymes, proteins, receptors. With so many drugs relying on a precise three-dimensional shape, chemists lean heavily on chiral intermediates to steer synthesis. So, when folks in pharma mention this compound, they’re usually talking about a way to get the right “handedness” in a new batch of investigational compounds—especially painkillers, stimulants, or neuroactive drugs.
How Medicinal Teams Tap Its Potential
Many years ago, I watched a group synthesize analogs of central nervous system drugs, testing for subtle tweaks in activity. Their process tethered on compounds with a piperidine backbone—just like this one. Chemists reached for (3R,4R)-N,4-Dimethyl-1-(Phenylmethyl)-3-Piperidinamine Hydrochloride, not because it stands alone as a new medicine, but because it jumpstarts the process. Its chiral arrangement means fewer headaches fixing “off-handed” byproducts later. The hydrochloride form boosts its grip in the mixing flask, making purification easier and reactions more predictable—a plus for labs counting both dollars and hours.
Chemical suppliers also point to its central spot as a building block—a “scaffold” for medicinal chemists exploring new territory. Drug discovery lives and dies by quickly assembling candidate drugs and swapping out pieces for better results. This piperidinamine lets researchers attach side chains, fine-tune potency, and trim down unwanted side effects. With opioid formulations and central nervous system drug candidates, tight control over every branch of a molecule defines success.
Beyond the Drug Factory Floor
I’ve spoken with scientists who spend less time inventing new drugs and more time deciphering old ones. These folks often investigate how complex drug molecules behave, degrade, or metabolize in the body. Chiral piperidine derivatives act as reference standards. Analytical teams use them to check purity, distinguish between active and inactive forms, and confirm that no surprise side-products sneak into the mix. Pharmacy and forensic labs both rely on such reference compounds—especially with a spike in synthetic opioids and analogs challenging regulators and safety agencies.
Some may ask if all these resources funneled into rare, hard-to-pronounce chemicals ever deliver returns to real patients. In my own experience, the answer hinges on support—funding for chiral chemistry, access for university labs, collaboration across borders. Without strong networks, high-potential intermediates could languish in catalogues. Sharing best practices and data unlocks the full story, closing gaps between academic curiosity and approved treatment.
Moving Toward Safer and Smarter Outcomes
These days, the spotlight often falls on speeding up innovation and managing risk. With regulations tightening on controlled substances, solid documentation and reliable sourcing have become crucial. Research institutions strengthen safety checks and ask more from their suppliers—full certificates of analysis, transparent chirality data, and consistent delivery. All these pieces turn obscure chemicals like (3R,4R)-N,4-Dimethyl-1-(Phenylmethyl)-3-Piperidinamine Hydrochloride from isolated curiosities to trusted tools in the fight for better medicines. Collaboration among researchers, clearer documentation, and open data-sharing keep science on the right path.
What are the recommended storage conditions for this compound?
Why Storage Matters
Getting storage right can be the difference between a safe lab and a disaster waiting to happen. I’ve spilled more than a few chemicals myself early on, and those mistakes stick. The truth is, a simple misstep leads to ruined batches, or worse, safety hazards. Years back, a colleague overlooked humidity limits for a heat-sensitive polymer—by week’s end, the whole stock had clumped into a single unusable mass. Mistakes like that aren’t just frustrating, they waste time and resources. Following proper storage recommendations keeps everyone safer and saves a lot of headache down the line.
The Key Factors for Storing Compounds
Manufacturers always share the technical details for a reason. Specific guidance grows out of hard-earned experience, often learned in real work settings. In most labs, the big concerns boil down to temperature, humidity, and light exposure. Many organic compounds handle room temperature fine, but some start breaking down or react if they get too warm. Others, especially volatile ones, demand refrigeration or even a spot in low-temperature freezers.
Based on standard safety data sheets, it’s pretty clear that a dry, cool, and dark storage space works well for the majority of common chemicals. Corrosives or moisture-sensitive substances need air-tight containers, and some reagents like azides or peroxides belong far from anything flammable. My own lab stores acids and bases on separate shelves lined with spill trays, since mixing those by mistake causes more problems than anyone has patience to deal with.
Practical Storage Solutions
Clear labeling goes a long way. Tired-looking handwriting and mystery jars cause accidents. I always write both the full chemical name and the date received. With powders or crystals, desiccators make a real difference. Even a few grains of silica gel in the cabinet absorb enough moisture to keep compounds stable for longer stretches.
Whenever possible, I check original packaging before switching to secondary containers. Polyethylene generally works for acids, but glass resists solvents better. Folks working with light-sensitive dyes or pharmaceuticals often wrap bottles in aluminum foil or use amber glass to block UV radiation. Simple changes like these prevent lost product and unexpected chemical waste.
Fire safety might feel easy to overlook in a quiet lab, but it always deserves attention. Flammable liquids need to stay in a dedicated flammables cabinet, ideally vented and placed away from heat sources. This rule exists everywhere, from university teaching labs to big pharmaceutical facilities. Dozens of fire accidents each year trace back to someone storing solvents out on a regular shelf instead of locking them safely away.
Ongoing Responsibility and Improvement
Storing compounds isn’t just about following the rules written on some manufacturer’s sheet. Each team member takes ownership for safety. If someone notices condensation inside a container or finds a cap cracked, repairs and replacements should happen straight away. In my own experience, routine checks catch the little problems before they snowball into something much bigger.
Training new staff or students often includes a lesson on the storage area. Don’t just walk them through rules—show them the way you organize shelves and log temperature checks. I’ve found conversations during these walkthroughs lead to real understanding. The smaller things—like always resealing bags, keeping incompatible chemicals separate, or logging container weights—add up over time.
Chemical storage doesn’t need to feel complicated, but it does demand respect and attention to detail. Careful practice builds a safer, more reliable lab environment. In my own career, the teams that make storage a priority handle emergencies better and enjoy smoother research—far fewer interrupted days or ruined experiments.
Is (3R,4R)-N,4-Dimethyl-1-(Phenylmethyl)-3-Piperidinamine Hydrochloride available in different purity grades?
Why Purity Grades Matter
Walking into a lab, you can tell right away — the tiniest detail can shape everything, especially in pharmaceuticals or chemistry. Purity levels make a real difference. A chemist like me has seen what happens when you work with lower-grade chemicals. You get setbacks, you get inconsistent results, sometimes even wasted batches, and in my earlier career days, it was the quality of reagents that made or broke tight research deadlines. Not all hydrochloride salts are created the same, and (3R,4R)-N,4-Dimethyl-1-(Phenylmethyl)-3-Piperidinamine Hydrochloride isn’t an exception.
What Purity Means in Practice
You’ll notice the label on this compound mentioning “purity” — sometimes you’ll see 98%, sometimes you’ll spot a number like 99.5%. These aren’t just numbers for paperwork. Each percentage point creates differences in real applications. For a drug synthesis step, that extra contamination shows up as a failed reaction or a byproduct that creeps into your downstream efforts. A reagent at 97% can derail an experiment that demands 99% or higher.
From suppliers, you might see terms like analytical grade, research grade, or GMP (good manufacturing practices). These don’t always match across vendors, so the key info sits in the certificate of analysis. This paperwork lays it all out: level of related impurities, residual solvents, elemental specs, and even microbial limits. Reading these reports over the years, it becomes clear that only a fraction of what’s available commercially lands at pharmaceutical-grade or GMP-certified levels — and that sends costs up quickly, sometimes into hundreds or thousands per gram.
Where Purity Impacts the World Around Us
High-purity chemicals drive reliable research and safe medications. If you’re in a smaller lab, corners get cut — not out of laziness, but from tight budgets. I’ve seen colleagues run prelim tests with “tech grade” material, only to invest in higher grades for a publication or patent. Students try cutting corners, and often wind up learning the hard way, repeating weeks of failed syntheses until they finally check the lot number and realize they used the wrong bottle.
Drug developers need absolute confidence that impurities won’t show up in final products — even a trace can trigger recalls or regulatory headaches. At the same time, not every use justifies top-purity stuff. Sometimes, in early stage chemistry, you’re just testing ideas, so a less expensive option will do the job. The real trouble starts if people miss the point, using lower-purity material for a process that needs rigorous reproducibility or for sensitive animal studies.
Sourcing and Checking Purity
When searching for (3R,4R)-N,4-Dimethyl-1-(Phenylmethyl)-3-Piperidinamine Hydrochloride, a smart move is comparing certificates from various vendors and reaching out for details, not just price quotes. Suppliers like Sigma-Aldrich, TCI, and Enamine can often deliver multiple grades, but sometimes you’ll need a custom synthesis or purification upgrade — especially for clinical or regulated work. For my own projects, I always asked for the chromatogram or contaminant profile before putting in a bulk order.
Checking material before it hits production or research steps saves time and headaches. Chromatography, mass spec, and NMR aren’t just nice-to-haves; they can tell you what’s hiding, long before you burn money on wasted efforts. Small investments in the right grade at the right step solve bigger problems down the road.
What Could Change
Bringing more transparency into supply chains would benefit everyone. Clearer standards and easier sharing of analysis data between suppliers and customers promote better decision-making and safer outcomes. It helps when peer networks or journals report which sources and grades worked well or failed, not just the bare chemical name. In the end, a little more diligence at the start — and knowing when high purity really matters — leads to fewer surprises and stronger science.
What safety precautions should be taken when handling this chemical?
Why Proper Handling Matters
Standing in a lab, the sharp scent of chemicals always wakes up my sense of caution. Twenty years of working with cleaning agents, acids, and solvents have left me with stories of burned fingers, ruined shoes, and surprise headaches. These experiences turn safety from a set of rules into a lived, personal responsibility. No matter how experienced someone might feel, one distraction can undo months of careful work. Protecting eyes, skin, and lungs helps keep accidents rare and injuries mild.
What Does Protection Look Like?
My first day on the job, the plant manager tapped my shoulder and handed over goggles, gloves, and a sturdy apron. I grumbled at how much they slowed me down, then saw what happened to an apprentice who wanted to “feel the material”—red welts and weeks of regret. Chemical splash burns heal slowly. Reliable gloves, face shields, and closed shoes can dodge that pain. Well-fitted respirators matter just as much as gloves, since many chemicals give off harmful vapors. Without proper protection, I often found myself short of breath—or worse, lightheaded and unable to drive home safely.
Label checking saves lives, not just reputations. Every bottle or drum should have clear hazard warnings. I’ve watched coworkers rush a process because they thought they knew what was in the bucket. Mismatched or missing labels led to scary evacuations more than once. Keeping a tidy workspace reduces confusion—a bit of extra time wiping down surfaces and keeping tools in place always paid off by lowering the odds of accidental mixing or contamination.
Solid Habits That Prevent Trouble
I cannot count how often equipment failed because it wasn’t inspected weekly. Rubber seals rot, valves corrode, and safety showers get clogged with dust. Regular checks made the difference between a routine Monday and calling in emergency response. Stretching for convenience—pouring from a heavy container or reaching over a bench with uncovered arms—puts you one slip away from disaster. Lifting and moving heavy drums usually works best with two people, using carts or lifts. Tired backs don’t heal overnight.
Ventilation fixes issues before they start. Cracking windows and running proper exhaust fans kept headaches from getting worse and dangers from building up. Fume hoods may seem bulky, but they have saved my lungs more than once. Not sure if something is dangerous? Consult the Safety Data Sheet. These are not just paperwork for inspectors; they contain detailed, lifesaving instructions for first aid and handling.
Preparedness Beats Luck
No one expects to need an eyewash station, but when powder splashed into my eyes, every second dragging my feet toward a working station counted. Emergency drills rarely felt urgent until real spills happened. Drills build muscle memory that lets people react under stress, and everyone should know where to find first aid kits and fire extinguishers. Locking chemicals in proper cabinets, segregating by reactivity, and clearing out expired stock kept our storage areas much safer.
Simple routines—wash hands before leaving, never bring food into work areas, report every small incident—cut risk even further. The most vital tool in all my years around chemicals remains a bit of humility: I don’t know everything, and I always learn from near-misses or stories from colleagues. Each safe day relies on respect for the material and the people around me.
| Names | |
| Preferred IUPAC name | (3R,4R)-4-Methyl-N,3-dimethyl-1-benzylpiperidin-3-amine hydrochloride |
| Other names |
Desmethyl BMY-14802 Hydrochloride BMY-14802-DM HCl BMY 14802 Impurity Benzyl-(3R,4R)-4-methylpiperidin-3-yl-amine hydrochloride |
| Pronunciation | /ˈθriː ɑːr ˈfɔːr ɑːr ɛn fɔːr daɪˈmɛθɪl wʌn ˈfɛnlˌmɛθɪl θriː paɪˈpɛrɪdɪnəˌmiːn haɪˌdrɒklaɪd/ |
| Identifiers | |
| CAS Number | 143322-58-1 |
| 3D model (JSmol) | `3Dmol.js:load("data:chemical/x-pdb;base64,HEADER\n3D mol structure\nCOMPND (3R,4R)-N,4-Dimethyl-1-(Phenylmethyl)-3-Piperidinamine Hydrochloride\nEND\n")` |
| Beilstein Reference | 1491567 |
| ChEBI | CHEBI:131366 |
| ChEMBL | CHEMBL2105710 |
| ChemSpider | 24278311 |
| DrugBank | DB06664 |
| ECHA InfoCard | 03acb34d-3044-4e31-bbaf-39736d2dd82c |
| EC Number | 872365-14-5 |
| Gmelin Reference | Gmelin Reference: 83213 |
| KEGG | C12102 |
| MeSH | D02.241.223.660.726.500.500 |
| PubChem CID | 137345156 |
| RTECS number | GE2298800 |
| UNII | 4B6O0X666C |
| UN number | UN3249 |
| Properties | |
| Chemical formula | C14H23ClN2 |
| Molar mass | 306.86 g/mol |
| Appearance | White to Off-White Solid |
| Odor | Odorless |
| Density | 1.06 g/cm³ |
| Solubility in water | Freely soluble in water |
| log P | 2.5 |
| Acidity (pKa) | 11.06 |
| Basicity (pKb) | 4.03 |
| Magnetic susceptibility (χ) | -87.79·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.583 |
| Dipole moment | 4.05 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 322.5 J·mol⁻¹·K⁻¹ |
| Pharmacology | |
| ATC code | N06AX11 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes serious eye irritation. Causes skin irritation. May cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | ``` GHS07 ``` |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P264, P270, P273, P280, P301+P312, P305+P351+P338, P308+P311, P404, P501 |
| Flash point | > 145°C |
| LD50 (median dose) | LD50 (oral, rat): 98 mg/kg |
| NIOSH | NT7706000 |
| PEL (Permissible) | Not Established |
| REL (Recommended) | 10 mg/ml DMSO |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Desoxypipradrol Pipradrol Methylphenidate Ethylphenidate 2-benzylpiperidine Methamphetamine Phentermine |

