3-Octanone: More Than Just a Molecule
Historical Development
The journey of 3-Octanone traces back over a century, appearing as chemists began dissecting the complex odors in plants, fruits, and some animal fats. This compound, recognized for its strong, mushroom-like scent, first grabbed attention from flavorists exploring the building blocks behind what makes food irresistible. Early references pop up in research journals of the early 1900s, a period hungry for answers about the chemicals creating distinctive flavors and aromas in nature. By the middle of the twentieth century, food scientists started refining methods to produce and isolate 3-Octanone, anchoring it in the world of commercial food additives and fragrance chemistry.
Product Overview
3-Octanone carries a reputation for lending creamy, earthy, and somewhat fruity notes to flavor and fragrance formulations. Inside every bottle, producers expect a pale liquid, free from color and overpowering synthetic off-odors. It’s a staple in food production, perfumery, and even in industrial cleaning agents, thanks to its robust and recognizable aroma that lifts other components without bulldozing nuanced scents. Many flavor chemists keep a stash close at hand, relying on it as both an ingredient and a reference point.
Physical & Chemical Properties
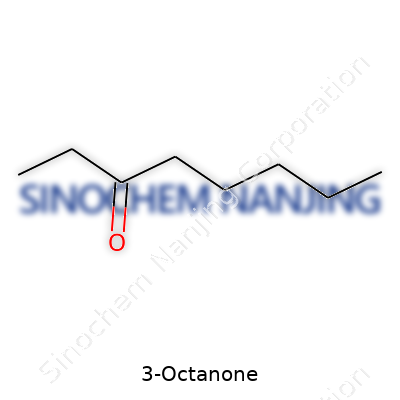
Tilting a vial of 3-Octanone, you’re looking at a clear oily liquid. Its boiling point typically rests at around 172°C, with a density close to 0.82 g/cm³. It has a reasonably low vapor pressure, making it less likely to fill a room with scent at room temperature, yet a little warmth draws out its signature aroma. Its molecular structure—C8H16O—comes packed with a straight eight-carbon chain and a ketone group set off at the third position. Solubility stays low in water but ramps up in ethanol, ether, and oils, which explains its value in flavor delivery and perfume blending.
Technical Specifications & Labeling
Buyers expect 3-Octanone by its purity, typically 98% and above for food and fragrance grade. Impurities can wreck a blend’s subtlety, so suppliers and users alike invest in gas chromatography to catch unwanted side-products. Most bottles ship with hazard labeling under the Globally Harmonized System, warning about flammability and eye irritation risks. Labels give concentration, batch number, and sometimes application recommendations. It’s not just bureaucracy—these details pin down quality and traceability, especially in food and pharmaceutical use where recalls mean big headaches.
Preparation Method
Most large-scale production starts from castor oil derivatives or uses octanol as a precursor. Synthetic routes often use controlled oxidation or even catalytic dehydrogenation, with copper-based catalysts helping convert alcohols to ketones. Green chemistry fans still look for cheaper, more sustainable routes, using enzymes or whole microbial cells to mimic pathways found in mushrooms, giving a nod to the compound’s roots in natural flavor creation.
Chemical Reactions & Modifications
Scientists exploring new flavors or industrial chemicals often tweak 3-Octanone using reduction, Grignard additions, and halogenation. Reduction turns it into 3-octanol—a molecule valuable in its own right as a flavorant. Reactions with amines produce imines, unlocking new notes for fragrance chemists. Some researchers anchor it to polymers, chasing slow-release flavor bursts in food coatings or air fresheners. Each modification spins out new possibilities for how this workhorse compound performs in commercial products.
Synonyms & Product Names
You’ll hear 3-Octanone called ethyl amyl ketone, octan-3-one, or simply octanone, depending on who’s talking. In the flavor and fragrance industry, “mushroom ketone” pops up, especially on ingredient lists written for buyers rather than bench chemists. Catalogs sometimes list it as FEMA No. 2807 or under flavor codes issued by the Joint FAO/WHO Expert Committee on Food Additives (JECFA), helping buyers trace regulatory status across markets.
Safety & Operational Standards
Handling 3-Octanone means donning gloves and goggles. Spills call for ventilated spaces and containment to keep vapors under control—it’s flammable, but it also slips through cracks if left unchecked. Regulatory bodies including OSHA and the European Chemicals Agency stress proper training for those handling large quantities. Safety data sheets spell out risk management strategies, and established producers rarely cut corners, knowing that breach of protocols can cost licenses and reputations.
Application Area
Food technologists drop 3-Octanone into dairy flavors, savory snacks, and plant-based meat analogues, searching for ways to pull genuine umami out of bland formulations. Perfumers blend it into high-end colognes or luxury candles, aiming to add earthiness that’s both sophisticated and approachable. In research labs, it steps in as a standard for testing gas detection methods or calibrating sensors designed for food quality control. There’s even a spot for it in pest control, where its natural occurrence in fungal volatiles steers behavioral studies of insects.
Research & Development
Ongoing efforts look for biocatalytic processes that could cut costs and improve yields, as well as ways to modify 3-Octanone’s profile for wider appeal. Startups and large firms alike run pilot fermentations using genetically modified yeast or mushroom cultures, aiming to produce the molecule without chemical synthesis at scale. Some academic labs dive into its interactions with taste and smell receptors in humans, peeling back layers of how flavors come together and how they shape consumer choices in crowded supermarkets.
Toxicity Research
Toxicologists have dug into the acute and chronic effects of 3-Octanone for decades, particularly since it ends up in foods and personal care products. Animal studies put its oral LD50 at relatively high levels—over 2000 mg/kg in rats—suggesting low acute toxicity. Some irritation pops up with heavy exposure, mainly to skin and eyes. Breakdown in the body tends to follow regular metabolic paths for simple ketones, with little sign of persistent or cumulative hazard at realistic use levels. Researchers still push for better long-term studies to cement its reputation as safe, especially as demand grows worldwide.
Future Prospects
The next wave for 3-Octanone looks full of opportunity and challenge. As consumer hunger for natural and sustainable flavors deepens, companies invest in bio-based synthesis and supply chain improvements to meet strict environmental standards. Growth in plant-based and functional foods relies on compounds that can boost sensory profiles—and here, 3-Octanone stands out. Its chemistry gives food designers more room to experiment with textures and authentic flavors that keep shoppers coming back. The fragrance world, not content with yesterday’s designs, continues to carve out new scent spaces using this earthy backbone. If research brings faster, cleaner production and confident safety data, it’s set to keep a strong spot across industries from fine dining to green chemistry.
What is 3-Octanone used for?
What Is 3-Octanone?
3-Octanone sounds like something that belongs in a high school chemistry class, but it turns up in places most of us would never expect. It’s a natural compound—scientists call it a ketone—found in mushrooms, dairy products, and even some fruits. What makes it interesting isn’t just where it comes from, but what people do with it.
The Power of Scent and Flavor
Most folks don’t know this, but 3-octanone plays a key role in the flavor and fragrance world. One whiff of fresh mushrooms, and you’re smelling this compound. Food manufacturers use it to recreate that earthy, mushroom-like aroma in processed foods. It’s a behind-the-scenes helper, giving plant-based meats or instant noodles a more realistic taste. I’ve tasted my fair share of vegetarian foods, and when they get that savory depth right, chemistry is doing some heavy lifting. It’s 3-octanone helping every bite taste a bit closer to real food.
Flavors Beyond the Plate
Perfumers also lean on 3-octanone because it brings a green, slightly floral scent. That splash of “freshness” in a new cologne often traces back to compounds like this. Fragrance makers rely on it to add character and complexity to candles, lotions, or even air fresheners. Stores know customers pick up those subtle notes, even if they can’t name what they’re smelling.
Farming and Protection
I grew up surrounded by gardens, and pests were always part of the deal. People sometimes use 3-octanone as a signaling chemical to protect plants. Scientists have seen that some plants release it to warn neighbors about insects. In labs, they test it for new crop protection solutions, aiming for pest repellent products that don’t rely on harsh chemicals.
Safety and Trust
Consumers care about what goes into their food. Studies from regulatory agencies like the FDA and European Food Safety Authority show that 3-octanone at typical levels doesn’t set off health alarms. The flavor industry has rules and reviews in place, so companies can’t just toss in whatever they want. People working in flavor research have spent decades testing safety to keep toxic risks far below concern.
Chasing Better Solutions
Modern eaters ask strong questions about ingredients. They want to know where things come from and if those ingredients do any harm. Marketers and developers can earn trust by focusing on transparency. It helps to explain why 3-octanone winds up in snacks, smoothies, or candles. Smart labeling and honest brand stories give everyone more control and fewer surprises.
Where Things Go From Here
Plant-based foods keep trending up, with even fast-food chains tossing out new options each year. That means more work for people finding natural flavor boosters. The push for greener farming will likely bring 3-octanone into new research for biopesticides. If brands stay honest about sourcing and uses, the relationship between chemistry and daily life only gets stronger. For something so small and simple, this molecule keeps showing up and making a real difference in our kitchens and beyond.
What are the physical and chemical properties of 3-Octanone?
Getting to Know 3-Octanone
3-Octanone has a straightforward structure: eight carbon atoms, with a carbonyl group sitting on the third carbon. It looks unassuming, but even simple organic molecules like this one leave a big mark in food, fragrance, and science.
Physical Properties in Everyday Terms
Pour a bit of 3-Octanone into a glass vial and you’ll find a clear, colorless liquid. The smell is striking—fruity, with hints of mushrooms. That aroma means a lot for chefs and flavorists. This liquid tips the scales at about 0.82 grams per cubic centimeter, so it's lighter than water and floats if you mix the two.
A thermometer in a vial of 3-Octanone will nudge up to 172°C before it boils away. It evaporates faster than water, but it does not turn into gas at room temperature, so it lingers on surfaces and in the air, making its scent stubborn. Touch a drop to your skin and it slides smoothly before vanishing—not greasy and not sticky. It's a bit like a light oil in texture, but much more volatile.
Solubility separates it from many other familiar kitchen or cosmetic chemicals. Drop it in water and most of it stays apart, but pour it in alcohol or ether and it blends well. This solves problems in the mixing of perfumes and food flavorings, where water isn't always the best solvent.
Chemical Behavior
3-Octanone includes a ketone group, which means its most important chemical site is the carbonyl carbon. This carbon is hungry for electrons, drawing in attacks from nucleophiles in organic reactions. Years in labs have made it clear: 3-Octanone will react with strong reducing agents like sodium borohydride, converting its ketone into an alcohol—a staple transformation in any organic chemistry toolkit.
Oxygen makes 3-Octanone unstable over time. Exposing it to air and light encourages slow oxidation, breaking it down, sometimes turning it into smelly acids or even shorter, sharp-smelling molecules. Storage in sealed, shaded bottles slows this process, so manufacturers and labs tuck it away from sunlight.
At home, the molecule’s low reactivity means it’s not hazardous for short-term contact—but inhaling large amounts or drinking it causes irritation or worse, so safety data sheets recommend gloves and ventilation. In a lab setting, chemistry teachers use examples like 3-Octanone to show both dangers and smart handling.
Why This Molecule Draws Attention
3-Octanone isn’t just some obscure compound. In truffles, this molecule pumps out much of that earthy aroma chefs call “umami.” Food scientists add it in tiny amounts to butter, baked goods, and even imitation cheese for depth of flavor. Beyond food, perfumers use it to bring a creamy, green-earth nuance to luxury fragrances.
Its secret lies in that pleasing scent, paired with a profile of low toxicity and reliable shelf life (when stored cool and dark). The chemical world values molecules that deliver punchy smells or tastes without loads of risk—and 3-Octanone lands in that sweet spot.
Addressing Challenges
Handling 3-Octanone safely means understanding the importance of proper storage, careful mixing, and solid labeling to prevent misuse. In busy kitchens or labs, investments in storage cabinets and employee training protect health while keeping quality steady. Recycling or safe disposal is another hurdle, best solved by joining local hazardous waste programs instead of pouring leftovers down the drain.
For companies pushing for cleaner, greener ingredients, sourcing or even bio-synthesizing 3-Octanone from renewable feedstocks can shrink its environmental impact. Picking the right supplier or method makes a difference, especially with tight regulations and watchful eyes on ingredient transparency in food and personal care.
Is 3-Octanone safe to handle and use?
Getting Familiar With 3-Octanone
3-Octanone pops up in the food world, found in mushrooms, fruits, and some plant oils. It’s a colorless liquid with a faint, sweet, mushroom-like odor, which explains its popularity as a flavor ingredient and scent. Manufacturers see this chemical in everything from perfumes and cleaning products to snacks and baked goods. So while it may sound like a scientific oddity, most folks brush up against it through everyday products.
Investigating Safety in the Workplace
Working with chemicals requires clear information and honest self-awareness. 3-Octanone can irritate skin, eyes, and the respiratory tract if used carelessly. Safety data from its material safety data sheet (MSDS) recommends protective gloves and eye shields. Recent studies suggest ingestion in large doses irritates the digestive system in lab animals. Luckily, routine handling in flavoring labs and perfumeries rarely approaches those levels.
Airborne exposure tends to be the most likely hazard, given the chemical's volatile nature. Breathing in concentrated vapors may cause headaches or dizziness. Using it in a well-ventilated room or under a fume hood makes a real difference. There’s a direct link between proper ventilation and a safe workday here.
Direct Experience and Practical Lessons
I’ve spent time in both kitchens and small chemistry labs, and most issues come from skipping simple steps. Not checking that a work area is aired out, not reading fresh gloves’ expiry dates, or bringing coffee to the bench. The safety culture gets built one habit at a time. Nobody regrets putting on goggles or catching a spill early with a layer of paper towels. Relying on instinct—sniffing chemicals or testing with bare hands—trips up even seasoned workers.
There’s also a temptation to treat “nature-identical” molecules as completely harmless. Yet, toxicology doesn’t care if a substance comes from a lab or a mushroom. The truth sits in the dosage and how the material gets handled. A tablespoon poured into a scent diffuser in a tight space will get noticed fast, in much the same way burning toast fills a kitchen. At lower levels, controlled by volume and timing, most flavorists finish their recipes without a hitch.
What the Science Says
Organizations like the Joint FAO/WHO Expert Committee on Food Additives review safety data for flavor compounds. 3-Octanone falls within their list of substances considered safe in the minuscule quantities used for flavoring. The U.S. Food and Drug Administration gives it “Generally Recognized as Safe” status for food use, based on decades of toxicological evidence. Still, this endorsement covers low concentrations eaten now and then—not industrial mishaps or improper use.
How to Handle Safer, Smarter
Better storage prevents accidents. Chemicals like 3-Octanone should stay in leak-proof containers, labeled with strong tape and plain language. Keep them out of reach from kids and pets—treat them as you would cleaning agents or paint thinners. A quick clean-up kit (absorbent cloth, mask, gloves) kept handy will save time and panic during spills. If any splashes land on your skin or in your eye, go straight to the sink—don’t wait for tingling to fade. Awareness matters more than experience.
Reading up on a material’s safety sheet is step one. Trusting your nose, wearing cheap PPE, and checking that no one’s eating near the chemicals remain good advice. Staying cautious keeps work simple, and that’s a lesson anyone who’s ever dealt with irritation or nausea after a careless day in the lab won’t forget. 3-Octanone works fine for most people when handled with care.
How is 3-Octanone synthesized or produced?
Understanding 3-Octanone’s Place in Industry
3-Octanone stands out in flavors, fragrances, and even pharmaceutical work. It gives off a fruity, mossy aroma that you’ll notice in perfumes and some foods. Because its scent profile holds such value, chemists and manufacturers call for accurate methods to make this compound. I see people in labs use everything from biology-driven approaches to traditional chemical reactions to deliver a product that fits both safety standards and large-scale needs.
Classic Chemical Routes to 3-Octanone
One of the most direct methods draws from basic organic chemistry. You start with compounds containing an eight-carbon backbone, like octanol or octanoic acid. Through oxidation—for example, with the right mix of copper catalysts and heat—the reaction removes hydrogen atoms from octanol, turning this alcohol into a ketone. In many university labs, this straightforward oxidation yields a lab-grade product, but industrial plants often focus on efficiency and cost by investing in larger vessels and more controlled environments. Experience tells me that precise temperature control and careful choice of solvents help get a cleaner product, which matters for both flavor work and downstream applications.
Green Chemistry and Biocatalysis
Producing 3-Octanone just with chemicals has its limits. Chemical oxidants can generate waste. As environmental rules get stricter, companies have started using microbes and enzymes to run similar reactions. For example, specific strains of yeast or bacteria break down fatty acids or alcohols found in plant oils. Given enough time and the right conditions, these microorganisms turn simple raw materials into 3-Octanone with far fewer unwanted byproducts. The biocatalytic route keeps hazardous chemicals out of the equation, which personally gives me more peace of mind as both a consumer and someone who’s worked in quality control. Plus, it aligns with the demand for "natural" ingredients in foods and scents.
Challenges in Scaling Up
Lab-scale prep works well for research and boutique batches, but scaling up introduces its own headaches. Too much heat or oxygen in a reactor can create side products, some of which bring off-notes or reduce overall yield. Process engineers need to watch carefully for “runaway” reactions—something I’ve seen leave behind thick, hard-to-clean residues if you’re not vigilant. Tight process monitoring helps, such as using chromatography to check that the bulk of the product matches purity standards laid down by groups like ISO or the FDA.
Seeking Balance: Purity, Safety, and Efficiency
Achieving the desired aroma and chemical reliability relies not just on perfecting the core reaction but also on refining the final steps. After the main conversion, producers often turn to distillation and recrystallization to get rid of impurities. Steam distillation, for instance, can help separate 3-Octanone from water-soluble contaminants and certain other byproducts. Working with these techniques in a real production setting, you learn that the cleanliness of the starting materials, quality of equipment, and adherence to Good Manufacturing Practices keep the final product safe for use in food and body care.
Looking Forward
Demand for 3-Octanone continues to climb as industries move towards safer, greener, and more reliable raw materials. Finding ways to synthesize it in cleaner ways—a combination of better chemical methods and smarter use of nature’s catalysts—can help secure both the safety of workers and end users. In my experience, open communication between researchers, production managers, and quality inspectors remains vital for meeting these goals while keeping production costs realistic.
What industries utilize 3-Octanone in their products?
Flavors and Fragrances
Walk into a high-end perfume shop or crack open a scented candle, and it’s easy to forget all the chemistry that goes into creating those aromas. 3-Octanone, a colorless liquid with a fresh, mushroomy scent, quietly plays a big part behind the scenes. Perfume makers lean on this molecule to add a soft, earthy note to their products. Manufacturers know that consumers expect nature-inspired fragrances. 3-Octanone comes straight out of the playbook for giving that earthy, forest-floor undertone associated with luxury. In food manufacturing, this compound helps to round out the flavors of processed foods—cheese, butter, and even some baked goods—delivering a subtle, natural mushroom aroma that feels genuine, not artificial. Companies designing these flavor formulations aren’t just interested in taste; a familiar smell reassures people that a product is wholesome and appealing.
Agriculture and Crop Protection
Farmers and agricultural scientists don’t always get credit for their use of biochemistry, but they’re quick to embrace a new tool if it shows promise. 3-Octanone is one of those tools. Some researchers have discovered its use for its antifungal properties and its ability to stunt the growth of certain crop pathogens. While large scale use remains limited, ongoing research points toward a future where this compound could serve as a softer alternative to harsher chemical pesticides. Finding gentler crop protection is essential these days—especially as folks grow wary about residues left on food and the health of soil microorganisms. Moving toward ingredients like 3-Octanone reflects the kind of innovation driven by both farmer experience and scientific trials.
Cleaning and Industrial Uses
Industrial cleaners and some specialty degreasers rely on 3-Octanone’s solvent abilities. Smells matter in janitorial work too. People demand clean, but they also want spaces to smell fresh, not like a chemical soup. By adding 3-Octanone, formulators give cleaning products a more pleasant aroma with no overpowering edge. My experience working in an auto shop taught me that even seasoned mechanics appreciate products that don’t make the workspace unbearable. A subtle, less intrusive odor can make everyone’s job easier. Today, cleaning product engineers are always on the lookout for ingredients that can clean well and keep things user-friendly.
Pest Control Techniques
Integrated pest management relies heavily on setting traps and repellents. Scientists studying pest behavior have used 3-Octanone due to its nature as a semiochemical—a compound certain insects recognize and avoid. In mushroom farms or storage areas where pests put livelihoods at risk, small adjustments like using the right scent can mean the difference between a lost crop and a healthy yield. Success in pest control frequently comes down to such clever tweaks, combining natural signals like 3-Octanone with strategic placement, without having to depend entirely on synthetic pesticides. The more we listen to what actually works—whether from a lab or from growers in the field—the more we get solutions that last.
Looking Ahead
Sourcing ingredients like 3-Octanone responsibly matters. Labs can now isolate or synthesize it with a better environmental profile than the older, more resource-intensive chemical processes. As industries steadily push toward products that feel natural and leave less of a mark, demand for molecules that come from both lab innovation and nature grows. Collaboration between companies, scientists, and manufacturers drives better products, with 3-Octanone showing up in places people often overlook—from the snacks in the pantry to the polish on the countertop. Healthy skepticism about additives isn’t going away, but the benefit of molecules like this can move conversations forward.
| Names | |
| Preferred IUPAC name | octan-3-one |
| Pronunciation | /ˈoʊkˌteɪn.oʊn/ |
| Identifiers | |
| CAS Number | 106-68-3 |
| Beilstein Reference | 1206823 |
| ChEBI | CHEBI:37814 |
| ChEMBL | CHEMBL33053 |
| ChemSpider | 6711 |
| DrugBank | DB01793 |
| ECHA InfoCard | 100.003.274 |
| EC Number | 203-982-0 |
| Gmelin Reference | 60455 |
| KEGG | C01811 |
| MeSH | D000067615 |
| PubChem CID | 8093 |
| RTECS number | RH7545000 |
| UNII | YN1R53J1RU |
| UN number | UN2810 |
| Properties | |
| Chemical formula | C8H16O |
| Molar mass | 128.22 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | fruity; herbaceous; mushroom |
| Density | 0.827 g/mL at 25 °C (lit.) |
| Solubility in water | 1.1 g/L (20 °C) |
| log P | 2.7 |
| Vapor pressure | 0.46 mmHg (at 25 °C) |
| Acidity (pKa) | 20.05 |
| Basicity (pKb) | pKb > 14 |
| Magnetic susceptibility (χ) | -7.59e-6 cm³/mol |
| Refractive index (nD) | 1.423 |
| Viscosity | 2.7 mPa·s (25 °C) |
| Dipole moment | 2.75 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 409.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -301.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4541.1 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS06, GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H315, H319 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P271, P280, P301+P310, P303+P361+P353, P304+P340, P305+P351+P338, P312, P331, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | 79°C |
| Autoignition temperature | 205 °C |
| Explosive limits | Explosive limits: 1.1–7% |
| Lethal dose or concentration | LD50 oral rat 1280 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 3200 mg/kg |
| NIOSH | RN122-00-9 |

