3-Nitropyridine: From Discovery to Future Use
Historical Development
Chemists began exploring pyridine derivatives in the mid-19th century, hunting for new building blocks that might change the way society harnesses nitrogen chemistry. Pyridine’s basic ring soon attracted interest, and once researchers figured out how to add a nitro group to the ring, they saw a dramatic shift in chemical reactivity. 3-Nitropyridine entered lab notebooks long before computers started keeping track—usually as a stepping stone towards dyes, drugs, or specialty chemicals. Over the decades, improvements in nitration techniques pushed yields higher and minimized messy byproducts. Today, chemists stand on generations of passed-down protocols, able to run preparations with far fewer surprises than their predecessors.
Product Overview
3-Nitropyridine stands out as a yellow to pale-brown crystalline powder, far more than just another lab chemical. Its appeal comes from the nitro group attached to the third position on the pyridine ring, which ramps up its potential as an intermediate in pharmaceutical and agrochemical syntheses. Researchers flag it not just for what it is, but for what it helps build: active pharmaceutical ingredients, specialty dyes, and chemical ligands. It rarely stays on a shelf for long without being pulled into some new reaction sequence.
Physical & Chemical Properties
Anyone who’s spent time with 3-Nitropyridine notices a faint but distinct odor, a reminder of its pyridine backbone. Expect it to melt right around 54°C and boil roughly at 306°C, so ordinary storage conditions rarely threaten its stability. Solubility leans toward organic solvents like ethanol, ether, and chloroform, while in water, it shows only modest willingness to dissolve. Chemically, the nitro group soaks up electrons, making the ring more likely to participate in nucleophilic aromatic substitutions. This electron-withdrawing squad affects everything from its reactivity in the lab to the safety measures chemists must respect.
Technical Specifications & Labeling
Labs usually require 3-Nitropyridine to top 98% purity, given any side-products can throw off sensitive downstream reactions. Regulatory standards demand proper labeling with hazard and precautionary information, including identifiers like the CAS number 589-89-1. Packaging must remain airtight and moisture-free, a necessary precaution since degraded samples complicate reaction outcomes and waste valuable time troubleshooting. Safety Data Sheets cover everything from fire fighting instructions to accidental contact guidelines. Batch numbers and expiration dates stay linked to certificates of analysis, and reputable suppliers put transparency and traceability first.
Preparation Method
Most factories and labs synthesize 3-Nitropyridine using direct nitration of pyridine under controlled conditions. Fuming nitric acid, buffered by sulfuric acid, reacts with pyridine cooled to prevent runaway exotherms. It’s a balancing act: run temperatures too warm and you’ll over-nitrate or even destroy the ring; keep things too cold and yields trickle in. Once the reaction wraps up, chemists neutralize acids, extract the organic phase, and run purification steps like crystallization or distillation until the desired product shines through. Some innovative methods use milder nitration agents or phase-transfer catalysts to squeeze out higher selectivity, but the core approach hasn’t fundamentally changed in decades.
Chemical Reactions & Modifications
The nitro group doesn’t just sit idle—it becomes a launchpad for further manipulation. Skilled chemists often reduce the nitro group to form 3-aminopyridine, which brings in a whole new set of reaction possibilities ranging from coupling reactions to ring closures. Metallation becomes easier, thanks to the electron-deficient ring. The intermediate also undergoes halogenation, acylation, or Suzuki couplings, depending on the needs of the target molecule. Playing with conditions—temperature, solvent, catalyst—lets chemists direct reactivity toward ever more complex structures, supporting both academic curiosity and commercial applications.
Synonyms & Product Names
Search scientific catalogs and you’ll come across 3-Nitropyridine under plenty of names, each a reflection of different research backgrounds or regulatory databases. Some chemists write it as m-Nitropyridine, others stick with systematic naming like Pyridine, 3-nitro-, while commercial product lines use combinations including trade names or catalog codes. This tangle of synonyms can slow down ordering or literature reviews, making precision in communication critical across teams, sectors, and continents.
Safety & Operational Standards
Handling 3-Nitropyridine calls for respect and layered protection. Toxic dusts and vapors require gloves, goggles, and good ventilation—usually a fume hood with an efficient exhaust system. Lab veterans recall stories of colleagues who brushed off minor spills, only to pay the price with headaches or skin irritation later. The nitro group also adds an element of fire risk under certain conditions. Spill procedures, proper labeling, and storage in tightly sealed containers cut down on accidental exposures. Waste disposal rules ban pouring stock solutions down the drain, since both local and federal agencies treat nitroarenes as hazardous. Safety training, up-to-date protocols, and real-world drills don’t just satisfy auditors—they protect health and reputations.
Application Area
3-Nitropyridine rarely gets top billing in end-use products, but its influence runs deep across several industries. Big pharmaceutical houses use it as a key intermediate in antihypertensive or anti-infective synthesis routes; crop science companies pull it into libraries to test new pesticide leads. Dyes, pigments, and specialty catalysts also owe part of their existence to this compound’s backbone. In university labs, 3-Nitropyridine features prominently in method development and molecular design, giving future scientists hands-on experience with challenging reagents. Anywhere that chemical innovation meets target-driven synthesis, this compound finds a role.
Research & Development
Active research tries to coax new tricks out of old molecules, and 3-Nitropyridine draws its fair share of attention. Chemists dig for greener synthetic routes that cut waste and shrink the carbon footprint, while others build catalysts that push reactivity into unexplored territory. Computational chemists model its behavior to predict new applications as drug precursors or as ligands for transition-metal complexes. Interdisciplinary teams—drawing from biology, material science, and engineering—test new methodologies using this chemical as a probe or as a starting block on the journey toward better drugs, safer agrochemicals, or more functional materials. Every improvement builds on data-driven insights, peer-reviewed studies, and careful lab work.
Toxicity Research
Past studies show 3-Nitropyridine carries risks beyond the standard chemical hazards. The most pressing concerns come from inhalation or prolonged skin contact, with symptoms ranging from respiratory irritation to headaches or dizziness. Animal model studies occasionally flag concerns for mutagenicity or environmental persistence, demanding careful handling and disposal. Regulatory agencies set exposure limits based on both acute and chronic studies, and industry standards evolve as new science emerges. Environmental scientists pay close attention to breakdown pathways in soil and water, tracking residues to avoid buildup in food chains or groundwater. Training new chemists in risk assessment and emergency response stays high on public health agendas.
Future Prospects
The future for 3-Nitropyridine looks surprisingly robust, shaped by shifting industry priorities and hungry for sustainable alternatives. Green chemistry offers real hope for safer synthesis, with milder reactants and recyclable catalysts leading the way. As pharmaceutical pipelines demand ever more diverse molecular scaffolds, regulatory work and process innovation keep pace. Machine learning and quantum simulation open new doors for prediction, reducing trial-and-error and shrinking both cost and risk. If tomorrow’s research can squeeze more functionality from each molecule, or streamline each step in manufacture, 3-Nitropyridine will stay relevant—serving as both a trusted workhorse in labs and a springboard for building the next generation of medicines and materials.
What is 3-Nitropyridine used for?
Why Chemists Value 3-Nitropyridine
3-Nitropyridine belongs to the pyridine family—a group of chemicals shaped around a six-membered ring. In the lab, this compound stands out because of its nitro group in the third position, which gives it some distinct chemical behaviors, especially compared to other nitro-substituted pyridines. Over the years, I’ve noticed that scientists, especially those in pharmaceuticals and materials science, have relied on this compound as a starting point for all kinds of projects.
Crucial Ingredient In Advanced Synthesis
Anyone who’s spent time in a synthetic chemistry lab has crossed paths with 3-Nitropyridine. This molecule doesn’t just show up by accident; researchers reach for it because its nitro group helps open new doors in making more complex molecules. Medicinal chemists use 3-Nitropyridine to build up drug molecules, especially ones that end up tackling infections, inflammation, and some cancers. That nitro group acts as a handle: scientists swap it out, reduce it, or use it as a point for further reactions, especially to make biologically active heterocycles.
There’s been a push lately for more affordable and efficient medication, with companies always looking for new routes to synthesize difficult compounds. 3-Nitropyridine fits right in here, helping companies cut down on the number of chemical steps, waste, and overall production costs. Sometimes, one version of an anti-cancer drug or antibiotics depends on one well-timed reaction involving this molecule. These are the sorts of invisible details that go unnoticed by most patients, but they can make a difference in how quickly and reliably new medicines get to those who need them.
Applications In Agrochemicals And Materials
I’ve also seen 3-Nitropyridine used in the development of crop protection agents—herbicides, fungicides, and insecticides. Companies work hard to develop new molecules that will help farmers grow food more efficiently, especially with rising populations and climate pressure. Because of its electronic features, chemists use this nitro-pyridine to build up compounds that break down safely in nature but effectively stop pests.
Beyond the farm and pharmacy, materials scientists have started looking closer at specialized pyridines for electronics and polymers. Fine-tuning the structure by incorporating a nitro group often changes how a molecule interacts with light or conducts electricity. This is crucial as sustainable technology depends more on organic materials, whether for sensors, solar cells, or advanced coatings.
Turning Challenges Into Opportunities
Any chemist will admit that working with nitro-pyridines means handling safety concerns, especially concerning toxicity and environmental risks. Exposure can irritate the skin and mucous membranes, and improper disposal risks polluting groundwater. Growing up near a chemical plant, I saw firsthand how failure to manage waste led to real environmental concerns and community pushback.
This is where companies and academic labs come in. Embracing safer protocols, recycling solvents, and using greener reducing agents all help. Research into bio-based and low-impact synthesis methods is slowly making its way into commercial practice. Regulatory bodies pay close attention to these chemicals for a reason, and firms that stay ahead of environmental standards avoid fines and build better community trust.
Looking Ahead
While 3-Nitropyridine might sound like just another line item on a lab inventory, its uses ripple out across modern science and manufacturing. Its true value comes to life not just in what it helps create, but in how labs and companies choose to handle and innovate around it. Open dialogue between chemists, policy makers, and the wider public sets the stage for advances that respect both human needs and our environment.
What is the chemical formula and molecular weight of 3-Nitropyridine?
Breaking Down 3-Nitropyridine
Every lab wizard, from student to seasoned chemist, knows the power of understanding a compound at the ground level. 3-Nitropyridine, a pale yellow solid, often lives in glass bottles tucked on back shelves where researchers reach for it in organic synthesis. The chemical formula—C5H4N2O2—looks basic, but the story it tells goes beyond the letters. Each atom plays a part: five carbons hold the backbone, four hydrogens fill in the edges, two nitrogens shape both the ring and the nitro group, and two oxygens lend strong reactivity.
Molecular Weight and Its Role
Molecular weight doesn’t just make for nice math. At 124.10 g/mol, every calculation for a reaction, every scale-up from a microgram to a kilogram, leans on that number. In one of my first internships, a miscalculation of molar mass for a similar compound led to an embarrassing mess: too little product, a lot of unanswered questions. Getting molecular weights right saves time, money, and sanity—the heart of a solid laboratory process.
Why Chemists Care About Structure
The nitro group at the third position on the pyridine ring steers where this molecule fits best. For chemical synthesis, that nitro group acts as a strong electron-withdrawing agent, helping guide further modifications on the ring. In pharmaceutical research, building blocks like these open up routes to new drugs. Researchers in crop science use compounds like 3-Nitropyridine to shape selective herbicides. Having the right chemical formula lets chemists track every atom’s journey from start to finish.
Risks and Safety Measures
Nitroaromatics don't go down easy. Even in small trials, lab safety matters. Exposure can irritate the skin, lungs, or eyes, and nobody wants to land in trouble from a carelessly-opened vial. The formula might look innocent, but every person who has spilled even a milligram of a nitro compound knows the headaches it delivers. Proper gloves, fume hoods, and careful handling should always stand between a researcher and any risk.
Building Knowledge Through Consistency
I learned early on that chemical formulas and weights form the foundation of accurate research. Once you make peace with details like molecular weight, you unlock consistency in your work. This principle applies to more than just advanced labs. Even students in early organic chemistry classes benefit from treating every measurement as a critical step. Textbooks will list the numbers and structures, but nothing beats hands-on experience with safe handling and double-checking every calculation.
Potential Solutions for Common Challenges
Mix-ups and mistakes crop up in busy labs. To stay ahead, digital inventory tools track exact chemical identities and weights. Open data platforms from organizations like PubChem or the National Institutes of Health help verify every number and every letter. Cloud-based calculators, clear labeling, and updated standard operating procedures lower the chances of accidents or formula confusion. In short, clear steps and reliable references keep the chemistry moving forward without getting tripped up over basics like C5H4N2O2 or 124.10 g/mol.
Importance in the Wider World
On the surface, 3-Nitropyridine may seem like just another line in a chemical catalog. Behind that unassuming formula is a key to countless experiments, innovations, and discoveries across fields. If you value reliability in research and the safety that keeps teams healthy, paying attention to fundamentals like these pays off every single day.
Is 3-Nitropyridine hazardous or toxic?
Looking Closely at 3-Nitropyridine
3-Nitropyridine often pops up in labs and chemical plants. Its bright yellow crystals may look harmless, but chemicals like this usually come with risks. Safety data makes clear that 3-Nitropyridine is not something to handle carelessly. The skin can become irritated after contact. Inhaling dust or fumes brings on coughing, throat discomfort, and sometimes more serious lung problems. This matches my experience working around similar nitro compounds, where tight protocols keep people safe for good reasons.
Toxic Properties and Real-World Hazards
This substance packs toxicity, both in contact and if it gets into the body. Reports in the literature point out symptoms like headaches, dizziness, nausea, and sometimes vomiting. Long exposure can even mess with the blood’s oxygen-carrying ability by forming methemoglobin, which starves tissues of oxygen. Long-term effects on humans aren’t totally understood yet, but pyridine derivatives have not earned a reputation for being gentle. If dust or fumes fill the air, the risk goes up fast for anyone nearby, including workers and emergency responders.
Environmental Concerns and Cleanup
Chemicals like 3-Nitropyridine don’t just stay put after a spill. They can travel into the ground or water and pose a hazard for fish and other wildlife. Pyridine rings resist breaking down in the environment, so once this stuff escapes a controlled setting, the trouble sticks around. One lab spill in my old workplace shut down a floor for days because clean-up took much longer than expected. Special gear—respirators, gloves, eye protection—became mandatory until surfaces tested clean.
Handling, Storage, and Protection
Working with 3-Nitropyridine means double-checking safety steps every time. Labeling, sealed containers, and using chemical hoods make all the difference. Everyone likes to think nasty accidents only happen elsewhere, but short-cuts make things worse. Chemical burns, lung injuries, and environmental cleanup grow far easier to prevent than to fix. Simple actions—wearing gloves, inspecting transfer equipment, and planning for spills—save time, money, and sometimes lives, compared to rushing or skipping steps under pressure.
Trusted Safety Data
Resources like the European Chemicals Agency (ECHA) and PubChem lay out hazards in plain words. For 3-Nitropyridine, the documentation calls out acute toxic effects, environmental concerns, and even the potential for mutagenicity. These agencies base warnings on lab tests and real incidents, so ignoring their guidance invites trouble. My years in research remind me: the risks in the paperwork usually show up in the real world if ignored.
Solutions for Safer Use
Managing the dangers of 3-Nitropyridine is possible. Good design keeps storage locked up and lab ventilation high. Training workers to handle spills safely and dispose of waste properly proves the most effective shield. Manufacturers and researchers should keep safer substitutes in mind for sensitive uses. Until then, sticking with safety rules doesn’t just check a box—it keeps people healthy and makes sure we avoid cleaning up even bigger messes after the fact.
How should 3-Nitropyridine be stored?
Understanding the Risks
3-Nitropyridine isn’t a chemical to stash on any random shelf. This compound, used in labs and industries for synthesizing pharmaceuticals and other fine chemicals, comes with its own set of hazards. It carries warning signs: toxicity, skin irritation, and the potential to catch fire if handled carelessly. In my lab days, I saw even experienced chemists treat it with a little extra respect. Nobody enjoys emergency cleanup or a trip to the health clinic over sloppy storage decisions.
Temperature, Light, and Air: The Storage Trifecta
Keep 3-Nitropyridine in a cool, dry spot. Heat can spark unwanted reactions, so avoid placing it near heat sources or direct sunlight. Fluctuating temperatures can make containers sweat, which can mix moisture with your chemicals—never a good combination. I remember one storage cabinet in a university lab that nearly ignited a debate over where to put reactive nitrated compounds. Cool and stable won out, and for good reason.
Air exposure spells trouble for some chemicals, and 3-Nitropyridine is no exception. It absorbs moisture from the air, turning clumpy and sometimes degrading in quality. Sealed containers aren’t just a suggestion—they’re essential. Seal it up, and you avoid breathing in fumes that could make your day a lot worse. Make sure the cap fits tight, and use containers made from glass or high-quality plastic designed for chemicals.
Labeling and Segregation Make a Difference
No one enjoys an accidental mix-up, especially when working with substances that don’t play nice together. Label every bottle and jar, not just with the name, but with date of receipt and expiration. When I first started stocking chemical storage, I underestimated how quickly labels faded or peeled. Long-lasting, chemical-resistant labels solve the problem.
Put 3-Nitropyridine away from acids, strong bases, and reducing agents. These combinations increase risk for hazardous reactions. Look up storage compatibilities before placing anything on the shelf—it’s worth a few extra minutes. Most labs use color-coded shelves or dedicated cabinets for this purpose, cutting down on accidents and keeping compliance checklists short.
PPE and Emergency Plans
Access rarely means ‘help yourself’ in any chemical storage room—controls matter. Limit access to trained personnel. Stock the area with personal protective equipment: gloves, goggles, and lab coats make a big difference. I always kept gloves nearby and insisted everyone used them. Eye wash stations and fire extinguishers should be within reach, not two hallways over.
Spill kits tailored for organic solvents are a must. If a bottle breaks or leaks, a tuned-up response kit can save hundreds in lost stock and protect people from harm. Allocate time for regular inspections. If you spot corrosion on caps or containers, replace them. No one wants a leaky mess at the back of the cupboard.
Follow Legal and Environmental Rules
Local regulations oversee chemical storage, and for good reason. Proper documentation and inventories help during audits and keep emergency responders in the loop. Safe storage isn’t just about compliance—it’s about protecting people and property from real harm. Responsible chemical management gives any lab or facility a better reputation and keeps it running smoothly.
What are the physical properties of 3-Nitropyridine?
The Nature of 3-Nitropyridine
If you’ve ever leafed through a textbook from an organic chemistry course, pyridines probably make an appearance somewhere early on. Add in a nitro group and you get something entirely different—3-nitropyridine, a pale yellow crystalline solid with a sharp, bitter odor. At room temperature, it keeps its form and won’t melt until the thermometer climbs to about 107-110°C. In a world where even a few degrees' difference can change an entire manufacturing process, those few extra ticks matter.
Solubility and Behavior in Everyday Conditions
Drop some 3-nitropyridine into water—nothing much happens. Water barely touches it, because its solubility in water is low. Heads turn, though, if the sample meets ethanol, diethyl ether, or chloroform. It dissolves right in, and that gives chemists more room to work with during extractions and preparations. The choice of solvent decides reaction yields and even how safe the entire workflow turns out to be.
Volatility and Handling
Boiling points tell stories. At 273°C, this compound isn’t jumping into the vapor phase under normal conditions. That puts some ease on storage and transportation; it isn’t drifting away invisibly from a half-closed container. In a workplace, the lower volatility means less risk of sudden inhalation. Anyone who’s ever caught a lungful of solvent fumes in a lab will appreciate the reduced risk here. Still, the characteristic odor serves as a gentle warning—don’t stick your face too close.
Color, Form, and Purity
The yellow tint in 3-nitropyridine isn’t just cosmetic. Color often tells on purity, and consistent, sharp crystallization signals that the batch hasn’t taken up any contaminants. For research or pharmaceutical applications, knowing that the powder in your flask really is what the label says drives trust in results. Inconsistent hues or caked, off-color material tell you something’s wrong. When labs buy this chemical, they’ll often check color and melting point before use.
Molecular Structure and Impact on Properties
The nitro group at the 3-position does more than change the name. It changes everything from polarity to how the molecule handles heat and light. Strong electron-withdrawing means the molecule acts differently in reactions compared to plain pyridine. As someone working on organic synthesis, you notice this shift in reactivity and plan routes accordingly. It also helps explain why the compound is sensitive to strong bases and reducing agents, opening up safety considerations during chemical transformations.
Storage Challenges and Real-World Risks
Like most nitroaromatics, some caution goes a long way. Prolonged exposure to sunlight or open air isn’t good, since it can degrade or discolor the solid. Storing in tightly sealed amber bottles, away from sparks and excessive heat, isn’t just a guideline; it can prevent degradation or accidental ignition. 3-nitropyridine isn’t explosive like some of its relatives, but the nitro group always adds a dash of unpredictability if someone’s careless with flame or incompatible chemicals. Labs who’ve experienced near-misses with unstable compounds learn quickly—the hard way—that storage and labeling can never be afterthoughts.
The Takeaway for Chemists and Industry
Understanding a chemical’s physical properties shapes every step from storage to application. Chemists, project managers, and safety experts build these facts into routines, not just as trivia, but as tools to work smarter. Pay attention to solubility, boiling and melting points, and even color. In fields where margins are thin and safety always hangs in the balance, these details aren’t just technical—they’re practical foundations.
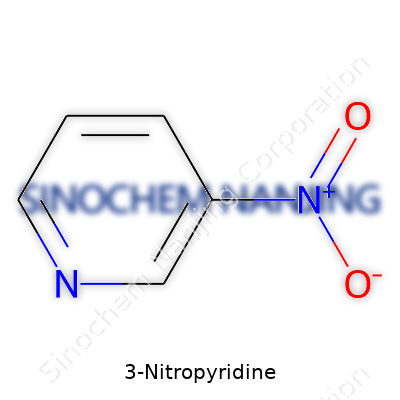
| Names | |
| Preferred IUPAC name | 3-nitropyridine |
| Other names |
3-Nitropyridine 3-nitropyridin nitro-3-pyridine pyridine, 3-nitro- |
| Pronunciation | /ˌθriːˌnaɪ.trəʊ.pɪˈrɪ.dɪn/ |
| Identifiers | |
| CAS Number | 589-08-2 |
| Beilstein Reference | 110928 |
| ChEBI | CHEBI:34244 |
| ChEMBL | CHEMBL320230 |
| ChemSpider | 7928 |
| DrugBank | DB03260 |
| ECHA InfoCard | ECHA InfoCard: 100.007.853 |
| EC Number | 210-189-7 |
| Gmelin Reference | Gmelin Reference: 73082 |
| KEGG | C01748 |
| MeSH | D016692 |
| PubChem CID | 10172 |
| RTECS number | UY7350000 |
| UNII | Y7W025366D |
| UN number | 1665 |
| Properties | |
| Chemical formula | C5H4N2O2 |
| Molar mass | Molar mass of 3-Nitropyridine: 124.10 g/mol |
| Appearance | Pale yellow to yellow crystalline solid |
| Odor | Odorless |
| Density | 1.295 g/cm³ |
| Solubility in water | soluble |
| log P | 0.18 |
| Vapor pressure | 0.0167 mmHg (25 °C) |
| Acidity (pKa) | 2.03 |
| Basicity (pKb) | 8.80 |
| Magnetic susceptibility (χ) | -41.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.552 |
| Viscosity | 0.972 mPa·s (20 °C) |
| Dipole moment | 2.92 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 146.2 J mol⁻¹ K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 92.1 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1600 kJ·mol⁻¹ |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07,GHS02 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P264, P270, P280, P301+P312, P330, P501 |
| NFPA 704 (fire diamond) | 2-3-1 |
| Flash point | 99°C |
| Autoignition temperature | 180 °C |
| Explosive limits | Explosive limits: 1.7–8.0% |
| Lethal dose or concentration | LD50 oral rat 200 mg/kg |
| LD50 (median dose) | LD50 (median dose) of 3-Nitropyridine is "100 mg/kg (rat, oral) |
| NIOSH | SN 3850000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 3-Nitropyridine: Not established |
| REL (Recommended) | 10-30°C |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
2-Nitropyridine 4-Nitropyridine Pyridine 3-Aminopyridine 3-Pyridinesulfonic acid |

