3-Nitrophenol: The Story Behind a Critical Chemical Compound
Historical Development
Walk through the pages of industrial chemistry, and 3-nitrophenol’s history shows how organic chemistry keeps finding new routes to push boundaries. Researchers in the late 1800s sought to harness the potential of aromatic nitro compounds for dyes and medicinal agents. They relied on direct nitration processes to synthesize ortho, para, and meta compounds from phenol, watching with fascination as the nitro group steered its way to different ring positions. Practically speaking, 3-nitrophenol’s debut stems from the growing demand during the synthetic dye boom, especially in Europe. Laboratories discovered it could stand up to harsher processing conditions, and that promise inspired further industrial scaling in the early 20th century. Scientists catalogs soon filled with notations about both its reactivity and safety quirks, shaping its continued use.
Product Overview
3-Nitrophenol crops up as a pale yellow, crystalline solid and finds steady demand in laboratories, manufacturing lines, and specialty chemical blends. This compound draws attention thanks to the nitro group snuggled into the meta position on the benzene ring—structural chemistry that leads to its unique characteristics. Chemical suppliers ship it in drums or bottles, addressing calls from agriculture, dye production, pharmaceuticals, and academic institutions. It often plays a supporting role as a building block molecule—rarely the star of the show, yet crucial for intermediates that go into larger, more complex chemical families.
Physical & Chemical Properties
3-Nitrophenol boils around 279°C and melts near 97°C, with a faint aromatic scent. While slightly soluble in cold water, it dissolves better in hot water and organic solvents like ethanol, ether, and chloroform. That nitro group draws electrons, lowering its tendency to donate hydrogen ions compared to plain phenol, and leaves it more acidic (pKa ~8.4). It appears yellow in solution, which flags its presence in analytical labs using colorimetric tests. In moisture, it doesn’t break down fast, so handling calls for careful storage. Chemically, it’s less reactive at the ortho and para positions because the nitro pulls electron density, so simple substitutions or couplings rely on activating conditions or catalysts.
Technical Specifications & Labeling
Industrial suppliers list content grades ranging from 98% to 99.5% pure, with data sheets spelling out moisture content, ash, and typical contaminant limits. They stamp containers with hazard codes, batch numbers, and expiry dates for traceability. You won’t see ambiguous descriptions—markings detail its chemical identity using CAS 554-84-7, structural formula, and regulated hazard symbols (often a yellow exclamation point and environmental warning). Specifications address physical form (crystal, powder) and any color deviations that could flag decomposition. These practices don’t just meet compliance needs; they ground quality assurance, so research and production find the consistency they bank on.
Preparation Method
Chemists make 3-nitrophenol primarily by nitrating phenol with nitric acid while holding down the temperature. This tough dance involves slow acid addition to control exothermic runaway and avoid over-nitration, followed by neutralization steps. Some labs leverage sodium arsenite or similar reducing agents to convert 3-nitrochlorobenzene back to 3-nitrophenol, usually after refinement of crude mixtures. Technological shifts have aimed to streamline recovery and limit byproducts—especially since nitrophenols turn up as trace pollutants in wastewater. Pilot plants closely monitor reaction time, temperature, and concentrations to avoid forming too much 2-nitrophenol or 4-nitrophenol since these isomers trip up downstream purification.
Chemical Reactions & Modifications
Synthetic chemists use 3-nitrophenol as a starting point for reductions, coupling reactions, and further derivatization. Its nitro group converts to an amine by hydrogenation, delivering 3-aminophenol for use in dye and pharmaceutical chemistry. Electrophilic aromatic substitutions generally skirt around its ring, so scientists might activate the oxygen or look for transition-metal catalysis to modify other positions. Conjugations with sulfonic acids, halogens, or alkyl chains help researchers build out targeted intermediates used in pigments and antimicrobials. Over the decades, protocols have emerged for careful handling during large-scale reactions, which avoids raising levels of dinitrophenolic side products that lose economic value and can up safety risks.
Synonyms & Product Names
Chemists and suppliers recognize 3-nitrophenol under names like m-nitrophenol, meta-nitrophenol, and 3-hydroxynitrobenzene. International trade references lift the titles up in registries; regulatory files mention variants for compliance or labelling. Literature reviews turn up product codes from manufacturers, and researchers often default to short forms like mNP in publications. Name confusion occasionally slowed down research or accidently mixed up isomers in procurement, which brought about push for standardized naming and cataloging across chemical companies.
Safety & Operational Standards
Handling 3-nitrophenol requires a healthy dose of respect. The solid and dust forms can irritate skin, eyes, and airways, so personal protective equipment remains non-negotiable in both academic and industrial labs. This compound gets flagged on Safety Data Sheets for toxicity, requiring gloves made from nitrile or neoprene, protective eyewear, and proper ventilation. Spills call for immediate containment and cleanup with absorbent materials that avoid release into drains. Waste disposal aligns with local hazardous regulations, and storage setups keep drums away from sunlight and moisture to block decomposition. Regulatory agencies monitor environmental release, since nitrophenols survive wastewater treatment and can accumulate in aquatic systems. Factories stress training for emergency response teams, and periodic audits check that safety protocols stack up against evolving standards.
Application Area
3-Nitrophenol finds its place woven into several manufacturing and research sectors. Dye and pigment industries lean on it for producing intermediates resulting in stable, vivid hues. Agriculture leans toward its role in herbicide synthesis (notably in 3,5-dinitrophenol production). Pharmaceutical outlets employ it as a building block for certain pain-relief and anti-inflammatory agents. Analytical chemists reach for it as a colorimetric marker to test enzyme activity or water pollutants, exploiting its distinct absorption peak. You’ll find it less in consumer-facing products and more hidden in the supply chains that feed everyday materials—cosmetics, inks, specialty polymers, and molecular probes.
Research & Development
Innovation drives forward as researchers dig deeper into how 3-nitrophenol can transform molecular scaffolds and spark new application fields. Analytical advances now trace ultra-low concentrations of nitrophenol residues in crops and water, aiding regulatory cleanup standards and ensuring food safety. Enzymatic degradation studies chase ways to speed up nitrophenol breakdown using engineered bacteria or catalytic reactors, hoping to shrink environmental footprints. Synthetic chemists explore functionalized nitrophenols as starting points for cutting-edge optoelectronic materials, bringing new energy to polymer science and light-harvesting devices. Investment in green chemistry keeps growing—one eye on safer nitration processes and novel reagents that reduce hazardous waste.
Toxicity Research
Research over decades underscores 3-nitrophenol’s moderate-to-high toxicity. Lab tests found that animals exposed to high doses show tremors, methemoglobinemia (which disrupts oxygen carrying in blood), and organ damage. Wildlife studies point to aquatic invertebrates as particularly sensitive, flagging long-term bioaccumulation risks. Occupational health data add that chronic exposure, even at low levels, can damage kidney and liver cells and stress the immune system. Environmental monitoring programs track its levels in urban water tables, aiming to balance economic activity and health outcomes by pushing lower emission limits. For me, knowing about these findings underlines the value of training all staff about chemical hygiene and spill prevention—not as window dressing, but to keep real-world risk under control.
Future Prospects
Growth in specialty chemicals and rising attention to green chemistry gives 3-nitrophenol a complex future. Its role as a chemical intermediate won’t vanish—markets for azo dyes, pharmaceuticals, and functionalized polymers keep on expanding, especially in growing economies. At the same time, environmental scrutiny puts pressure on process improvements, cleaner nitration techniques, and efficient waste treatment. Research into bio-based nitrophenol production signals a shift: fermentation technology and enzyme-driven synthesis could lessen reliance on harsh acids. Regulatory limits look set to tighten across the globe, driving investments in closed-loop systems and sustainable alternatives. All of this leans on multidisciplinary teams—engineers, chemists, and environmental scientists—coming together to find practical solutions. From working alongside process engineers in a pilot plant, I’ve learned that pressures for efficiency, safety, and sustainability land on everyone’s desk, and each incremental improvement counts for both business and community well-being.
What is 3-Nitrophenol used for?
What Brings 3-Nitrophenol Into the Spotlight?
3-Nitrophenol isn’t something most people come across in their daily routine. This yellowish powder pops up in labs and factories much more than in households, but what it touches feels closer to home than most realize. My first brush with this compound came during a college chemistry project. Our professor handed out a sample in a thick glass bottle, warning us about its strong, bitter odor and the need for gloves. For something with almost no fame among non-chemists, 3-Nitrophenol plays a background role in several big industries.
Key Places You’ll Find 3-Nitrophenol
Spend time in a pesticide plant and you’ll see the value of intermediate compounds like 3-Nitrophenol. Companies rely on it for making parathion, a powerful insecticide. Without such intermediates, many crop-protecting chemicals wouldn’t exist, and global crop yields would take a hit. Farmers feed billions, so improvements and efficiency in the pesticide chain matter.
Dye manufacturers value 3-Nitrophenol, too. It serves as a starting point for certain azo dyes, widely used in textiles and printing inks. Bright colors on everyday products start far from the shop floor—often in chemical plants stirring mixtures most people would never want to smell or spill.
Pharmaceutical labs sometimes harness this compound when hunting for new drugs. Its molecular structure offers researchers a building block for synthesizing medicines that fight headaches, pain, or even infections. The irony strikes me every time: the road to many modern conveniences runs through harsh, risky chemicals carefully handled by trained professionals.
The Upsides and the Risks
Science loves 3-Nitrophenol for its predictability in reactions. Product consistency helps keep costs in line, which matters for companies and, ultimately, for families paying for groceries or medical bills. Over the years, chemical synthesis using 3-Nitrophenol has helped level the playing field between rich and poor regions by keeping essential agricultural and healthcare products cheaper.
On the other hand, this isn’t an eco-friendly compound. Spills and careless disposal have harmed rivers and soil. I’ve seen reports from rural areas where water tests showed alarming nitro compound levels after a factory accident. These chemicals don’t just vanish; they stick around in the environment, building up and stressing plants, animals, and people. Chronic exposure carries real risks, from respiratory trouble to more severe health conditions.
Practical Steps for Balance
It doesn’t make sense to just stop using 3-Nitrophenol overnight. The world still relies on its applications. Careful management—this is where industry leaders and regulators should focus energy. Modern plants now invest in closed systems, thorough employee training, and on-site chemical treatment. I’ve visited factories where teams use sensors for early detection of leaks. These changes help, but enforcement gaps still exist in some countries.
For communities near chemical plants, outreach programs and routine health screenings could spot early signs of trouble. Farmers should get clear directions about safe pesticide usage, storage, and alternatives. Supporting research into greener chemistry paves the way for next-generation compounds that do the job with less risk. Just as I learned in that college lab, a strong respect for chemical power—matched with thoughtful safety protocols—gives society the benefits without gambling on our health or our environment.
What is the chemical structure of 3-Nitrophenol?
Understanding the Molecule
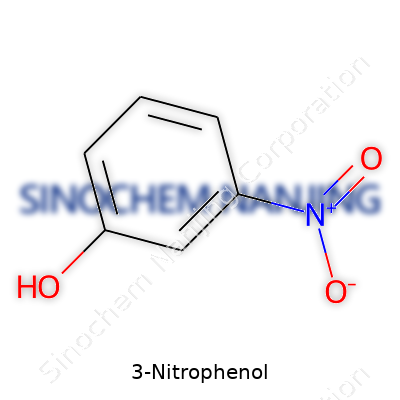
Maybe you’ve seen the name 3-Nitrophenol pop up in a textbook or a chemistry lab. It sounds technical until you look at its structure. Here’s what you actually find: this molecule starts with a benzene ring, which is a classic six-carbon ring you learn about early on in chemistry. Each corner of the ring holds a carbon atom, with alternating double bonds between them. Now, let’s get a bit more specific. Attached to this ring sits a hydroxyl group (that’s –OH) on the first carbon, making it a phenol. Then comes a nitro group (–NO2) fixed to the third carbon over from where that –OH holds on to the ring. That positions the molecule as a “meta” derivative: if you followed your finger around the ring from the –OH, the nitro group would be one carbon away from being directly across.
Why the Structure Matters
It’s easy to gloss over these details, but the exact position of each group on a ring transforms what the molecule does in the real world. If you switch the nitro group nearer to the –OH or further away, the way the molecule interacts with other compounds, its solubility, and even its color can all change. I remember my first advanced organic chemistry course: working with nitrophenols, everyone in the lab soon realized how small changes to a compound’s structure could spark big differences in reactivity.
3-Nitrophenol’s layout plays a big role in its properties. The –NO2 group pulls electrons away from the ring because it’s strongly electronegative. The presence of the –OH group adds acidity, especially since the nitro group sits not next door, but one position further off. This “meta” setup shifts the equilibrium in a way that makes 3-Nitrophenol more acidic than regular phenol, but not as much as its cousin, 4-Nitrophenol.
Why You Should Care About All This
Understanding 3-Nitrophenol begins with its structure, but that’s just the start. In the real world, 3-Nitrophenol pops up in the breakdown of pesticides and dyes. It also lands on the list of possible contaminants in water. If you’ve worked in environmental chemistry or even checked municipal water reports, grasping which isomer you’re dealing with goes way beyond the classroom. For example, during water testing, the lab has to distinguish between 2-, 3-, and 4-Nitrophenol. Each isomer behaves differently, both in water and in living systems.
Growing up in the farm belt, I watched local labs racing to handle water safety concerns after heavy rains. Nitrated aromatic compounds can slip through the soil and wind up in wells. This pushed testing to the forefront. By knowing the specific structure, analysts identify the source and decide the best cleanup methods. Activated carbon tackles certain isomers more efficiently, and advanced oxidation works better for others. So these small details—like where the NO2 group sits—set the stage for real environmental action.
A Closer Look at Solutions
Detailed chemical knowledge pays off. Lab teams can select the right adsorption technology. Engineers designing treatment plants choose the best combination of oxidation steps. Regulators set limits based on toxicity profiles that stem from, you guessed it, chemical structure. One molecule, arranged just slightly differently, can require a whole new approach.
3-Nitrophenol is more than a molecule with a hard-to-pronounce name. Its structure signals how we handle safety, environmental responsibility, and even industry regulation. Learning those details early on gave me a deeper respect for how much hangs on getting the chemistry right.
Is 3-Nitrophenol hazardous or toxic?
Understanding the Chemical
3-Nitrophenol shows up as a yellow crystalline powder and comes from the family of nitrophenol compounds. It features in various chemical manufacturing processes—think dyes, pesticides, and pharmaceuticals. Handling any industrial chemical, including this one, brings up concerns about safety, both for people working directly with it and for the wider environment.
Health and Safety Risks
Breathing in dust or vapors from 3-nitrophenol can irritate the nose and throat pretty fast. Eyes and skin contact often spark redness or burning. High or repeated exposure may move beyond minor irritations. I’ve read research pointing out how some workers developed more serious health issues after prolonged periods without enough protective gear. Some developed headaches, dizziness, and in rare situations, signs of toxicity in the liver or kidneys.
The chemical earns its “toxic” label due to how it behaves in the body. According to data from the Agency for Toxic Substances and Disease Registry (ATSDR), 3-nitrophenol breaks down into compounds that strain organs tasked with clearing toxins. This can make a bad situation worse if someone is already sensitive or exposed to other dangerous substances on the job.
Environmental Impact
Factories dumping nitrophenol-laced waste into water sources can spark chain reactions in local ecosystems. Fish, frogs, and other aquatic animals deal with the chemical by building up the substance in their tissues. This creates ripple effects through the food chain, especially since nitrophenols reduce oxygen levels in water and add stress to already fragile habitats.
Cleanup work takes serious effort. Espoo’s regional river watchdogs in Finland once reported sharp drops in fish populations after 3-nitrophenol leaks. Restoration stretched over years—as a reminder of how one mishap can ripple through a larger system.
Why This Matters
In my time volunteering with safety groups, I’ve noticed too many factories that still cut corners on chemical handling. Outdated gear or poor ventilation systems raise the risk of workplace exposure. Some workers have told me they never saw clear warning signs or got proper training. Many safety organizations agree that lowering risk starts with strong training and personal protective equipment that fits the job.
People who live near chemical plants worry just as much. Many stories surface from neighbors who notice odd smells in the air or strange discoloration in local streams. Data from the European Chemicals Agency (ECHA) links nitrophenol leaks to soil and water pollution. The problem grows if cleanup drags out and more people get exposed.
Finding Better Solutions
Real progress comes from swapping in safer chemicals wherever possible. In cases where 3-nitrophenol remains essential, better containment practices and smarter waste management curb accidental releases. Remote monitoring and regular health checks play a huge role. A simple air quality monitor set up inside a plant can flag unexpected spikes. Health screenings help catch early signs in workers before things get out of hand.
Stronger rules and faster reporting also shape safer habits. The industries that invest in both worker wellbeing and environmental protection tend to avoid disasters and lawsuits. My experience with teams at responsible companies proves that “just enough” safety rarely cuts it. Plenty of success stories show how honest risk assessment and tight procedures drive both productivity and safety at the same time.
What are the storage conditions for 3-Nitrophenol?
Knowing the Risks: Properties of 3-Nitrophenol
3-Nitrophenol doesn’t draw much attention outside the lab, but anyone who's spent time working with chemicals knows that a single misstep with a compound like this can cause headaches for both staff and the environment. As a pale yellow solid, it holds a punch both in handling and in the consequences of neglect. It can irritate the skin and eyes, and dust inhalation leads to respiratory problems. It isn’t just about keeping things tidy; it’s about keeping people safe and maintaining a clean workspace. I've watched poorly stored chemicals turn storerooms into hazards overnight. A wet shelf, unsecured jar, or a forgotten open lid—these aren't small mistakes. They’re invitations for accidents.
Storing 3-Nitrophenol – Not Optional, Just Sensible
3-Nitrophenol calls for cool, dry, and well-ventilated storage. Humidity is its enemy, so running a dehumidifier or keeping desiccants nearby pays off. Any leak will let this compound seep into the air or react with moisture, increasing toxicity risks in the lab. Forget high shelves where broken containers fall; place 3-nitrophenol on a sturdy, reachable surface. Temperature swings can break down the compound, so keep it away from heat sources like radiators or direct sunlight. In my experience, even a single sunny window can turn a shelf into an unpredictable zone. Cotton lab coats and flimsy cork stoppers won’t cut it—get serious with tight-sealing containers, preferably glass or high-quality plastic with secure lids.
Labeling and Compatibility
Label everything clearly. In fast-paced environments, poorly labeled jars get mixed up, leading to cross-contamination or worse. I once saw a mislabeled bottle spark a near-disaster with incompatible chemicals. 3-Nitrophenol should stay away from strong reducing agents and bases. Grouping chemicals out of habit or convenience can turn dangerous in a hurry, so storage logs and segregation are more than box-ticking exercises—they save resources and sometimes lives. Use secondary containers—spill trays keep things manageable if something leaks.
Legal and Environmental Responsibility
Laws vary by region, but most safety codes demand strict segregation and regular inventory checks. The Occupational Safety and Health Administration (OSHA) lays out guidelines for handling hazardous materials, and the Environmental Protection Agency (EPA) monitors disposal and spill response. Ignoring these agencies risks more than fines. In one incident, I watched local authorities shut down an entire wing after a single chemical leak. That mess could have been avoided with proper storage practices.
Simple Steps, Big Impact
Every lab worker has seen shortcuts—plastic wrap instead of lids, chemicals stored above eye level, cluttered shelves. These habits pose serious risks with 3-nitrophenol. Set up a routine check of storage areas and keep a clear record of every bottle. If humidity creeps up, consider automated monitoring to avoid surprises. Staff training—often rushed—is essential; hands-on refreshers do more than online quizzes. People remember what they've practiced, and it only takes one careless moment to spark a serious problem.
Building Better Habits Starts Small
Putting safety first with 3-nitrophenol storage isn't complicated. Make space for chemicals, buy the right containers, and resist the urge to cut corners. The investment pays off in fewer spills, healthier workspaces, and fewer run-ins with compliance officers. Every year brings new stories of mishandling and regret—often with common chemicals. It never has to get that far.
How is 3-Nitrophenol synthesized?
Inside the Lab: Making 3-Nitrophenol
Chemists often face tight constraints with time, safety, and cost. When making something like 3-nitrophenol, each choice in the lab has an effect on research goals and the demands from industries that depend on that chemical. 3-nitrophenol, a yellow crystalline compound, shows up in applications from pharmaceutical building blocks to dye intermediates. The standard way to make it uses a process called nitration, which chemists have refined over decades for both accuracy and yield.
Working with Phenol and Nitration Methods
Synthesis usually starts with phenol. By pouring a mix of concentrated nitric and sulfuric acids—what chemists call "mixed acid"—onto phenol, a reaction kicks off that introduces a nitro group onto the benzene ring. The position on the ring matters. This process doesn’t play favorites, so the result brings about two products: 2-nitrophenol and 3-nitrophenol. Boiling the reaction mixture helps. The "ortho" product (2-nitrophenol) forms more readily, but by adjusting temperatures and acid concentrations, the yield of the "meta" product, 3-nitrophenol, increases.
In a classroom years ago, I made this compound in a cold fume hood, clinging to safety goggles and gloves. Even a whiff of the acids reminded me that chemistry isn’t forgiving. Practical problems pop up: excessive heat risks runaway reactions, while low temperatures bring poor nitro yields. Getting the right balance is about more than just the final product. It's about keeping people safe and reducing the mess left behind.
Making Clean Separations and Purity Matter
After the nitration finishes, you face another challenge. Nitration dumps an unwanted mix of ortho and meta isomers together. Separating them can soak up time. Chemists often lean on steam distillation since 2-nitrophenol comes out as a vapor while 3-nitrophenol stays behind in the flask. This trick capitalizes on the different volatilities of isomers. It works most of the time, but further purification—maybe crystallization from water—brings the product to high purity.
In a real lab, purity is not just about looking good on a certificate of analysis. For pharmaceutical research, even trace contamination can throw off biological tests or lead to mistakes in scaling up production. Keeping control of the process and spot-checking every batch reduces risk and ensures results that researchers can trust.
Environmental and Safety Impacts
Bringing up harsh acids and nitration means you can't ignore the risks. Waste acid mixes harshly with water and hurts the ecosystem if released untreated. Labs lean on neutralization using bases, then careful disposal. Green chemistry researchers look for cleaner routes—maybe via enzymatic methods or milder oxidants. These alternatives remain in the testing phase, but there's a strong push for them. Regulations in many countries get tighter each year, pushing for lower emissions and safer workplaces.
Where to Go from Here
3-nitrophenol synthesis sits at the crossroads of chemistry skill and practical problem-solving. Safer, cleaner methods are in demand. Until then, chemists must rely on careful technique, strong safety habits, and continued process tweaks to keep quality high and teams safe. Companies supporting pharmaceutical research, dye synthesis, and agrochemical work count on this reliability every day. Seeing this process unfold up close, even with its risks, you learn respect for what responsible chemistry can do.
| Names | |
| Preferred IUPAC name | 3-nitrobenzenol |
| Other names |
m-Nitrophenol Meta-nitrophenol 3-Hydroxynitrobenzene |
| Pronunciation | /ˌθriːˌnaɪ.trəʊˈfiː.nɒl/ |
| Identifiers | |
| CAS Number | 554-84-7 |
| Beilstein Reference | 2059615 |
| ChEBI | CHEBI:15948 |
| ChEMBL | CHEMBL15481 |
| ChemSpider | 6996 |
| DrugBank | DB02637 |
| ECHA InfoCard | 100.013.039 |
| EC Number | 200-242-2 |
| Gmelin Reference | 81516 |
| KEGG | C01531 |
| MeSH | D010370 |
| PubChem CID | 7418 |
| RTECS number | SN3150000 |
| UNII | 4NS9410F7A |
| UN number | UN1663 |
| CompTox Dashboard (EPA) | DTXSID1022912 |
| Properties | |
| Chemical formula | C6H5NO3 |
| Molar mass | 139.11 g/mol |
| Appearance | Pale yellow crystalline solid |
| Odor | Odorless |
| Density | 1.48 g/cm³ |
| Solubility in water | Moderately soluble |
| log P | 1.91 |
| Vapor pressure | 0.001 mmHg (25 °C) |
| Acidity (pKa) | 8.36 |
| Basicity (pKb) | 10.95 |
| Magnetic susceptibility (χ) | -70.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.617 |
| Viscosity | 1.34 mPa·s (25 °C) |
| Dipole moment | 3.60 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 110.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -34.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2219 kJ·mol⁻¹ |
| Hazards | |
| Main hazards | Toxic if swallowed, harmful if inhaled, causes skin and eye irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS06,GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H332, H400 |
| Precautionary statements | P261, P280, P305+P351+P338, P312, P337+P313 |
| NFPA 704 (fire diamond) | 2-2-0-AOX |
| Flash point | 113°C |
| Autoignition temperature | 540 °C |
| Lethal dose or concentration | LD50 oral rat 282 mg/kg |
| LD50 (median dose) | LD50 (median dose): 325 mg/kg (oral, rat) |
| NIOSH | SN 35000 |
| PEL (Permissible) | PEL: 5 mg/m3 |
| REL (Recommended) | 'Standard Laboratory Chemicals' |
| IDLH (Immediate danger) | 100 mg/m3 |
| Related compounds | |
| Related compounds |
Phenol 2-Nitrophenol 4-Nitrophenol 3-Aminophenol Nitrobenzene |

