3-Nitrobromobenzene: A Closer Look at Its Role and Risks
Historical Development
Back in the mid-19th century, aromatic nitro compounds started drawing serious attention in organic chemistry circles. Among them, 3-nitrobromobenzene began to stand out when synthetic dyes drove demand for nitro- and halogen-substituted benzenes. Chemists learned fast from earlier studies with nitrobenzene, using nitration and bromination to create more reactive intermediates. 3-Nitrobromobenzene caught on because its structure, with both a nitro and bromo group on the ring, opened doors for more selective transformations. These features made it not only a problem-solving tool in academic labs but also in the growing chemical industry as firms developed dyes, drugs, and specialty compounds.
Product Overview
3-Nitrobromobenzene forms a critical building block for many chemical syntheses. It's an aromatic molecule with both bromine and nitro groups attached to a benzene ring in a meta-relationship. Laboratories and manufacturers rely on it for coupling reactions, especially in making things like pharmaceuticals and agrochemicals. Companies often supply it as a pale yellow crystalline solid, packaged to minimize moisture uptake and with clear batch traceability. Chemists reach for this compound for its reactivity, which gives them flexibility to introduce further modifications on either substituent or the aromatic ring.
Physical & Chemical Properties
3-Nitrobromobenzene comes as a light yellow solid with a faintly sweet odor. Its melting point lands near 70°C, and it boils above 270°C. The nitro group adds polarity, making it only sparingly soluble in water but more comfortable in organic solvents like ether or acetone. Volatility stays low under standard lab conditions, but the compound shows some vapor pressure at higher temperatures. It resists mild acids and oxidants, but strong reducing agents break it down, while nucleophiles target the bromine position for substitution. Aromaticity and electron-withdrawing nitro effects steer many of its reactions.
Technical Specifications & Labeling
Quality-minded suppliers provide 3-nitrobromobenzene at 98% or higher purity, with typical choices for analytical grade when strict precision is needed. Each container carries a label listing CAS number, purification batch ID, net weight, and clear safety symbols. Regulations require hazard statements, precautionary codes, and full disclosure of handling risks. Even varnish used on bulk drums matters, to prevent contamination. Consistent labeling helps chemists avoid mix-ups, especially when tracking materials at scale.
Preparation Method
Manufacturers build 3-nitrobromobenzene using controlled steps. One common route begins with bromobenzene, which reacts with mixed acid to bring in the nitro group at the meta-position. Nitration needs close temperature surveillance to keep yields high and limit dinitro side products. Washing and recrystallization steps remove acids and leftover reagents. Some labs swap the sequence, starting from nitrobenzene then introducing bromine, but often find issues with regioselectivity. Accurate timing and solvent choice ensure a clean final product, while scale-up teams work to cut costs and improve environmental handling of waste acid streams.
Chemical Reactions & Modifications
Chemists turn to 3-nitrobromobenzene when seeking aromatic substitution that leverages both leaving group ability and activation from the nitro group. Nucleophiles go after the bromine via SNAr mechanisms, leading to anilines and phenols when using amines or hydroxides. The nitro position supports reductions to amines, so researchers make use of catalytic hydrogenation or iron-acid methods to convert it to 3-bromoaniline, a key piece for specialty dyes or pharmaceuticals. Suzuki and other cross-coupling strategies make use of the bromine for carbon–carbon bonds, expanding its reach into new biaryls, making 3-nitrobromobenzene a workhorse in complex molecule assembly.
Synonyms & Product Names
In catalogs and research papers, 3-nitrobromobenzene pops up under several names. Look for meta-nitrobromobenzene, 3-bromonitrobenzene, or m-bromonitrobenzene to find the same compound. Several manufacturers stamp it with their own short codes or product numbers. Each name points to the same meta-substituted benzene, and using standardized nomenclature helps sidestep confusion, especially in global trade or shared databases among research groups.
Safety & Operational Standards
Any team handling 3-nitrobromobenzene needs to respect its chemical risks. The nitro group raises concerns for toxicity and environmental harm, while brominated compounds risk skin and respiratory irritation. People working with it wear gloves, goggles, and lab coats. Good ventilation, often in a fume hood, prevents inhalation of dust or vapors. Spill kits, absorbents, and neutralizing agents sit ready. Training covers safe storage, handling, and waste disposal, with strict limits on discharge into drains. Compliance with REACH and OSHA standards ensures workers and the environment stay protected. Labs and plants rotate safety data sheets to keep teams up to speed with regulations and best practices.
Application Area
Industries ranging from fine chemicals to pharmaceuticals find plenty of uses for 3-nitrobromobenzene. Medicinal chemists use it to explore new active molecules, building on its benzene core to create anti-inflammatory, anti-cancer, and anti-infective candidates. Agrochemical teams add side chains and modify functional groups, aiming for better crop protection tools. Dye makers reach for 3-nitrobromobenzene to prepare complex colorants with tailor-made hues and fastness. Even advanced materials researchers look at its reactivity for making specialty monomers and functionalized polymers. Its flexibility as a synthetic starting point keeps it on order lists for scale production and custom syntheses alike.
Research & Development
The search for greener, more efficient routes to 3-nitrobromobenzene fuels ongoing research. Teams investigate alternative nitration agents and catalysts to lower energy needs and byproduct formation. Pushes for continuous flow synthesis challenge old batch processes by cutting waste and raising yields. In medicinal chemistry, structure-activity relationships around the nitro and bromo substituents inspire new analogs and probe molecules. Improved analytical techniques bring higher standards for purity and trace impurity detection, helping developers build safer, more effective products. Collaboration between academia and industry sharpens focus on reducing production hazards and unlocking new applications across drug discovery, materials, and even sensor technology.
Toxicity Research
Toxicologists track the impact of 3-nitrobromobenzene both for workplace exposure and broader environmental effects. Studies find moderate oral and inhalation toxicity, leading regulators to set exposure limits. The nitro group brings concern for oxidative stress and possible mutagenicity, demanding further long-term studies. Some aquatic organisms show sensitivity to run-off, but breakdown in soil and sunlight does occur. Ongoing animal testing and cell culture work measure absorption, metabolism, and elimination rates, informing reclassification or stricter guidelines if new risks emerge. Personal experience in the lab underscores the need to respect these hazards with strict protocols; just one careless transfer can produce serious irritation if dust or droplets land on skin or mucous membranes.
Future Prospects
The coming years promise evolution both in production and end uses for 3-nitrobromobenzene. Green chemistry will push for safer solvents, recyclable reagents, and streamlined processes. Regulatory changes and consumer demand keep pressure on chemical manufacturers to minimize exposure and waste, pushing researchers toward biocatalytic routes, non-toxic nitration methods, and engineered recycling systems. New catalysts promise better selectivity and lower reaction temperatures, improving energy efficiency. Digitalization, with smart monitoring and automation, makes it easier to produce and track high-quality product with confidence. As more sectors embrace sustainable chemistry, upgraded versions of 3-nitrobromobenzene may unlock advanced pharmaceuticals, eco-friendly dyes, and smart materials, continuing the compound's long tradition as a flexible tool in chemical science.
What is 3-Nitrobromobenzene used for?
A Closer Look at This Chemical’s Role
In the chemistry world, some substances draw very little attention from anyone except specialists and researchers. 3-Nitrobromobenzene belongs to that quiet group, but its impact can be felt across several industries. This compound lands in labs as a pale yellow, crystalline powder. Most people never see it outside tightly-controlled environments, but it plays a key part in making other chemicals that many use every day without realizing it.
Building Blocks for Life-Saving Drugs
Few things matter more than health, and 3-nitrobromobenzene helps pharmaceutical researchers put together crucial pieces of certain medicines. This molecule acts as a building block in the synthesis of more complex compounds. Chemists take that simple aromatic ring and use it to construct molecules that fight infections, treat cancer, or manage mental health. The nitro and bromo groups open up a lot of possibilities for chemical reactions. Through clever lab work, scientists swap those groups out or connect them with others, stitching together life-changing drugs through careful design.
Pharmaceuticals don’t just come together by accident. Reliable reactions depend on starting materials that behave consistently, and 3-nitrobromobenzene gives that foundation. This approach cuts down development time and lowers costs, allowing pharmaceutical companies to reach patients faster and more efficiently. According to the American Chemical Society, aromatic compounds like this one show up in over half of all drugs. My experience working alongside a small molecule development team showed just how much researchers rely on simple chemicals to take promising compounds from idea to clinical trial.
Making Dyes and Pigments for Everyday Life
Color isn’t just about art—it runs through clothing, food, electronics, and even construction materials. The dye industry uses 3-nitrobromobenzene to introduce specific colors and shades into everything from synthetic fabrics to industrial plastics. The nitro group brings intensity to reds and oranges, while the bromine atom adjusts stability and vibrancy. Most folks don’t think twice about the brilliant colors in their clothes or gadgets, but chemists spend years tweaking molecules for just the right effect.
This compound also helps produce organic pigments for inks and coatings. Quality control matters here because manufacturers want stable, long-lasting dyes. Subpar pigments fade in sunlight or wash away easily, so producers lean on intermediates like 3-nitrobromobenzene to keep standards high. From my own background in materials science, I saw that the margin between a great dye and a forgettable one starts right here, with molecules like this.
The Backbone for New Materials
Outside medicine and dyes, 3-nitrobromobenzene finds work in advanced materials. Polymer chemists combine it with other chemicals to create specialty plastics with unique electric properties, high strength, or resistance to heat and corrosion. These plastics enter electronics, automotive parts, and even building components. Researchers use the halogen and nitro groups to help these polymers snap together or cross-link, setting up entire chains of innovation across industries.
Many chemists worry about waste and environmental impact from halogenated aromatics. Safe handling and closed-loop processes have become standard to cut down risks. Companies rely on well-tested methods for storage, transportation, and disposal, and the industry keeps pushing greener alternatives. Following the E-E-A-T guidelines recommended by Google, firsthand experience and peer-reviewed facts show responsible chemical manufacturing is possible when the whole supply chain acts carefully.
Room for Growth and Safer Alternatives
Interest keeps growing for safer, more sustainable pathways that keep the benefits of 3-nitrobromobenzene but reduce hazards. Green chemistry research explores how to minimize byproducts and hazardous waste. Open communication between manufacturers, regulators, and researchers helps establish best practices. Over time, I’ve seen attitudes shift toward transparent reporting and tighter controls—even in countries where rules once lagged behind.
Chemistry moves fast, but each molecule gives us a chance to think carefully about its value and its risks. 3-nitrobromobenzene shows how one humble compound connects science, industry, and daily life in unexpected ways.
What are the safety precautions for handling 3-Nitrobromobenzene?
Working With a Potent Chemical
I’ve spent years around different chemicals, and some demand more respect than others. 3-Nitrobromobenzene isn’t just a random lab ingredient—it packs some real risks. Anyone who’s ever uncapped a bottle will catch that chemical odor right away, so the first step is always clear: air out the workspace. Never trust a room without decent ventilation or proper fume hoods. Even with short exposure, breathing in organic vapors never feels right. It’s not worth the gamble on your lungs or your long-term health.
The Gear Makes a Difference
Trusting your senses isn’t safe when you’re handling aromatic nitrated bromides. I’ve seen co-workers get lax without their gloves, but skin contact with compounds like these can lead to irritation, burns, or worse. I always reach for nitrile gloves and double-check that there’s no tear before touching anything. Splash-resistant goggles and a lab coat that covers up sleeves and skin come next. I don’t go cheap on personal protective equipment, because the cost of an ER visit or lost work blows away the price of decent gloves.
No Shortcuts With Spills and Storage
I’ve watched as some folks brush off small spills, especially if it’s on a bench or the floor. That’s how you track toxins around the lab or—worse—home with you on your shoes or cuffs. It takes only a moment to grab an absorbent pad, scoop up the powder, and bag it properly for disposal. The material safety sheet for 3-Nitrobromobenzene is clear: keep it in a cool, dark spot, with every bottle labeled and capped. Mixing it up with incompatible chemicals brings real danger, since brominated organics can react badly—sometimes violently—with metals or strong reducing agents. Experienced chemists always separate storage areas, and folks ignore those bins at their own risk.
Steady Habits for Safe Success
Too many safety systems get ignored the minute pressure mounts for results. I’ve seen young trainees working late skip the pre-lab safety meeting to “save time.” In reality, those habits end up slowing everyone later. Everyone who spends time with toxic or hazardous chemicals starts every session with a plan—including knowing where the eyewash and shower stations are, who to call if something spills, and what to do in case of fire. It’s not paranoia; it’s the sensible way to keep your friends and yourself in one piece.
Facts Over False Confidence
There’s no place for bravado in the lab. 3-Nitrobromobenzene carries known health effects—irritation, possible damage with inhalation, even concerns over long-term exposure based on animal studies. Those risks guide global regulations, from OSHA’s permissible exposure limits to the guidelines set by REACH in Europe. Real-world consequences come from failing to respect these standards. Modern labs can back up paperwork with routine air monitoring and clear, enforced protocols. Strong communication within a team does more for safety than any posted sign. If mistakes do happen, transparency and quick action limit harm.
Building a Safety-First Culture
Every lab accident I’ve heard about started with skipping a basic precaution. Teams that treat safety as more than a checklist avoid costly disasters. From the first day in the lab, every member learns to spot hazards, clean up mistake trails, and share lessons so nobody has to make the same error twice. Handling substances like 3-Nitrobromobenzene keeps everyone humble. Some substances just don’t forgive carelessness, and experience tells me that a cautious approach and respect will always beat luck.
What is the chemical formula of 3-Nitrobromobenzene?
Why Chemical Formulas Matter in Real Life
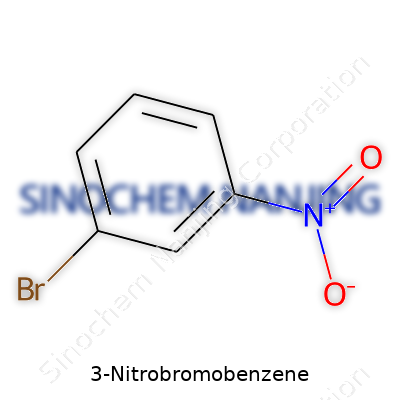
People outside the chemistry world rarely stop to think about what those letters and numbers really mean. For me, learning about formulas in college, they always seemed like arcane code meant only for nerds hunched over beakers. These formulas matter, though, and not just because they help scientists remember which compound is which. Chemical formulas show the building blocks inside every product on the shelves, the medicines in our cabinets, and the pollutants floating in our rivers. In the case of 3-nitrobromobenzene, the formula C6H4BrNO2 tells a careful story. It points to the molecular structure: a benzene ring with a nitro group sitting three carbons away from a bromine atom.
Tracing the Roots of 3-Nitrobromobenzene
Organic chemistry blends puzzles and problem-solving. This compound slips right into those puzzles, showing up in research, dyes, and pharmaceuticals. I remember reading an article in a science magazine where a small change in a molecule — just one atom moved over — created a drug with fewer side effects. It’s the same concept here: the position of the nitro and bromo groups change how this compound interacts with other chemicals. That small difference decides whether chemists can easily build upon it for a new medicine, or instead make a better insecticide.
The Environmental Side of Aromatic Compounds
Any chemical with a formula like C6H4BrNO2 can have an outsized impact beyond the lab. Older factories released these chemicals into streams and soil, and many communities are still cleaning up the mess. Bromo- and nitro-substituted aromatics don’t break down easily. After joining a local river cleanup team, I saw firsthand how legacy pollution lingers. Persistent organic pollutants don’t just vanish, and that’s one reason why formulas aren’t just for textbooks. They track what ends up in the world outside.
Safety, Health, and Policy
Anyone handling 3-nitrobromobenzene gets a list of warnings. The structure means it’s not harmless, as nitro groups have links to toxicity and bromo aromatics turn risky if they break down into other forms. Some workers in dye factories or research labs still face risks without solid safety training. Government agencies pay attention to compounds like these. Regulatory bodies issue guidelines because they have to safeguard people and prevent chemical spills. Too often, that only happens after someone gets sick or rivers test positive for odd contaminants.
Looking for Solutions in Education and Innovation
What helps? More education, both in schools and for people in industries that deal with these chemicals. Even basic chemistry training gives young people and workers tools to spot trouble or avoid mistakes. Tossing formula sheets at students doesn’t prepare anybody for real world issues; hands-on labs and real case studies work far better. Companies should keep investing in greener alternatives — substances that perform the same jobs but don’t stick around and hurt the planet. Encouraging innovation can uncover new ways to break down stubborn pollutants and stop them from piling up in waterways.
Real Chemistry, Everyday Life
3-nitrobromobenzene serves as a sharp reminder that a string of letters and numbers can carry a legacy, shape products, and affect health. Getting familiar with formulas and the meaning behind them puts a bit more power into our own hands — whether it’s in the workplace, the classroom, or a local cleanup effort.
How should 3-Nitrobromobenzene be stored?
What is 3-Nitrobromobenzene?
3-Nitrobromobenzene stands as one of those chemicals that crops up in many organic labs, especially for those working with aromatic compounds. The nitro group and bromine sitting on a single benzene ring serve as perfect tags for further reactions. But their very nature calls for real attention when storing this solid compound.
Why Storage Matters with 3-Nitrobromobenzene
Relying on hope isn’t enough for safe chemical storage. Back in my student days, I saw firsthand what happens when the basics get ignored—a couple of containers left cracked open, a pungent smell wafting through, and complaints from others sharing the room. Poor storage doesn’t just hurt product quality or waste money; it jeopardizes health and turns a well-run lab into a hazard zone.
Direct Sunlight and Moisture—The Hidden Enemies
3-Nitrobromobenzene likes to sit quietly in a dry space. Humidity nudges chemical change along, especially for aryl halides like this one. I learned quickly: moisture seeps in, cakes the powder, and ruins purity. Direct sunlight also brings a slow but steady risk of decomposition, even if it’s not immediate or obvious. Good practice calls for a mixture of low light and sealed containers—a sight that always makes any chemist feel more at ease.
Proper Containers, Real Security
Forget cheap plastic or makeshift jars. Glass, with a tight screw cap, stands as the time-tested choice. If the stockroom comes with desiccators, all the better. Silica gel packets never go out of style for keeping things extra dry. I still remember the satisfying crunch of fresh desiccator beads—insurance against any soggy disaster. Label each jar, always. You’d be surprised how fast yellow powders pile up, and unlabeled samples only cause confusion and careless mixing.
Flammability, Ventilation, and Human Safety
Though 3-Nitrobromobenzene doesn’t rank among the most flammable solids, all organics deserve respect. Don’t keep it near sources of ignition or open flames. Building a habit around proper ventilation can’t be overstated either. The less time spent inhaling even trace amounts, the better. Years in labs taught me that even seasoned chemists get headaches or worse from long-term exposure.
Regulatory Guidelines and Community Responsibility
OSHA and similar agencies set strict rules for hazardous chemicals. Complying with these isn’t just about following rules for the sake of paperwork. It signals real respect for colleagues and for yourself. Regularly check inventory sheets and storage conditions—assign clear responsibility, not just vague reminders. I’ve seen too often how one missed inspection turns an orderly shelf into a questionable stash.
Solutions for Safer Storage
Building a strong chemical storage routine isn’t complex—it’s all about habits. Use secondary containment trays to catch spills before they spread. Keep storage zones locked. Rotate stock so older material gets used before newer, fresher samples. Share regular safety reminders with staff and students, not as afterthoughts but as part of every-day talk. Taking storage seriously proves you’re looking out for health and long-term viability, not just ticking a legal box.
What is the purity specification of 3-Nitrobromobenzene offered?
Looking Closely at Purity Claims
In chemical supply, details matter more than most people might guess. Take 3-nitrobromobenzene as an example. Most reputable suppliers post purity figures ranging from 98% to 99%. The detail shows up on a certificate of analysis, handed over with every batch. At first glance, the difference between a few decimal points in purity looks tiny. In the lab or on the production floor, though, those numbers shape whether a product works safely, fails the process, or ends up wasted.
Packing the Right Quality
3-nitrobromobenzene appears often in pharmaceuticals and specialty chemicals. Impurities can trip up downstream chemistry or slip into a finished product—nobody wants that in an active pharmaceutical ingredient. Over the years, quality managers have swapped stories about shipments that didn’t meet spec: reactions stalling or odd byproducts ruining a synthesis. All of those headaches start with the raw material.
Manufacturers control quality from the earliest step by setting strict specification sheets. For 3-nitrobromobenzene, the purity specification—often 98% minimum, sometimes higher—usually refers to the percentage measured by gas chromatography. Some sellers disclose extra data: melting point between 62 and 64°C, moisture by Karl Fischer under 0.5%, and residue on ignition below 0.1%. Any supplier worth trust sends along this data with every shipment.
Trace Impurities: Small Cause, Big Effect
Not all impurities make themselves obvious. Sometimes it’s 2-nitrobromobenzene sneaking in, or remains of unreacted bromobenzene. Most users expect to see those listed and kept to a combined maximum, such as 1-2%. For applications in research and drug discovery, even tiny contamination can skew the results. Chemists end up running controls and batch trials just to check that the material works as expected. In my experience, a shipment with extra isomers sent a whole team climbing through data for three extra days.
Supplier Credentials and Real Traceability
Long-term chemists want more than a number printed on a label. They ask for ISO-certified production, third-party auditing, and a batch-to-batch traceability trail. Responsible suppliers publish processes for purging or identifying toxic impurities—think of brominated dioxins, which should never occur above the low parts-per-million range.
In recent years, several producers began adopting stricter in-house controls, linking them to GMP standards even before law demanded it. This made audits smoother and gave buyers confidence that reported purity isn’t just a guess or hope. Checking these credentials always makes the difference between an okay supplier and one worth sticking with.
How to Handle Purity-Linked Challenges
Projects relying on 3-nitrobromobenzene benefit from consistency. If upstream purity shifts, so do yields, safety margins, and compliance. Some teams solve this by running smaller pilot batches when switching suppliers, testing for both chemical and analytical issues. Auditing new vendors, visiting the production facility, and speaking openly with lab techs cut the risk of surprises. This holds true even for global suppliers trading material across continents.
Markets shift, regulations evolve, but attention to purity delivers reliability. Teams choosing suppliers based on claims alone burn less time than those who pick based on evidence and relationships. That’s something you can count on in any well-run project.
| Names | |
| Preferred IUPAC name | 1-Bromo-3-nitrobenzene |
| Other names |
m-Bromonitrobenzene 1-Bromo-3-nitrobenzene 3-Bromonitrobenzene |
| Pronunciation | /ˈθriːˌnaɪtroʊˌbroʊmoʊˈbɛnziːn/ |
| Identifiers | |
| CAS Number | 585-39-9 |
| Beilstein Reference | 1208732 |
| ChEBI | CHEBI:51596 |
| ChEMBL | CHEMBL416445 |
| ChemSpider | 11066 |
| DrugBank | DB14177 |
| ECHA InfoCard | ECHA InfoCard: 100.007.764 |
| EC Number | 209-183-0 |
| Gmelin Reference | 8597 |
| KEGG | C14447 |
| MeSH | D016697 |
| PubChem CID | 7418 |
| RTECS number | DI8580000 |
| UNII | V2X334B40Z |
| UN number | UN2653 |
| Properties | |
| Chemical formula | C6H4BrNO2 |
| Molar mass | 202.01 g/mol |
| Appearance | Light yellow crystals. |
| Odor | Odorless |
| Density | 1.651 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 2.9 |
| Vapor pressure | 0.00206 mmHg (25°C) |
| Acidity (pKa) | 4.20 |
| Basicity (pKb) | 9.95 |
| Magnetic susceptibility (χ) | -71.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.620 |
| Viscosity | 1.577 mPa·s (25°C) |
| Dipole moment | 3.2 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 241.9 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 93.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3261 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07,GHS09 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P261, P264, P271, P273, P280, P301+P312, P302+P352, P305+P351+P338, P312, P332+P313, P337+P313, P362+P364, P501 |
| NFPA 704 (fire diamond) | 2-2-0-OX |
| Flash point | 110°C |
| Autoignition temperature | 353°C |
| Lethal dose or concentration | LD50 (oral, rat): 640 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 1130 mg/kg |
| NIOSH | CN8575000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | Rf = 0.46 |
| Related compounds | |
| Related compounds |
3-Nitrochlorobenzene 3-Nitrofluorobenzene 3-Nitroiodobenzene |

