3-Nitrobenzoyl Chloride: Tracing the Story from Early Research to Modern Applications
Historical Development of 3-Nitrobenzoyl Chloride
Chemists started tinkering with benzoyl chlorides late in the 19th century, right as industrial chemistry picked up steam in Europe. Their efforts focused on tweaking the benzene ring to create new reagents—3-nitrobenzoyl chloride emerged as a standout from those early trials. By adding a nitro group to the meta position on the ring, researchers found fresh possibilities not available in the unsubstituted or para-substituted cousins. German dye and pharmaceutical laboratories led most early syntheses, using new sulfur and nitric acid methods. Over time, as organic synthesis became central to drug design and materials science, chemists integrated this molecule into more sophisticated reactions. Now, university teaching labs and chemical plants look at 3-nitrobenzoyl chloride as a key player for both method development and commercial end products.
Product Overview
Labs classify 3-nitrobenzoyl chloride as an aromatic acyl chloride with the formula C7H4ClNO3. The nitro group cranks up its reactivity, opening up gateways for peptide coupling and dye synthesis. White to light yellow in appearance, its pungent smell signals the need for serious ventilation and careful storage. Researchers count on it because it acts as a classic intermediate, stable in dry conditions but ready to hydrolyze if moisture shows up. Its portfolio includes uses in pharmaceuticals, agrochemical routes, and even advanced polymer studies.
Physical and Chemical Properties
Crystalline and solid at room temperature, 3-nitrobenzoyl chloride usually falls between 56–60°C for melting, depending on purity and crystal form. The structure, featuring both a nitro group and an acyl chloride, tilts the molecule toward strong electrophilicity. Its moderately low vapor pressure still puts it on the radar for inhalation hazard. Insoluble in water but easily mixing with ether, benzene, and chloroform, it serves chemists who want selective solubility. The chemical reacts violently when water enters the flask, breaking down into the parent acid and releasing hydrochloric acid gas. Its stability in sealed bottles, far from sunlight and dampness, shapes how suppliers manage and deliver it.
Technical Specifications and Labeling
Certified labs package 3-nitrobenzoyl chloride at upwards of 98% purity, with key measurements for melting point, chloride content, nitro assay, and residual solvents. The best producers document spectral data from NMR and IR scans to confirm the substitution pattern and functional groups. Transport labels display UN numbers for corrosives, hazard pictograms including “corrosive” and “health hazard,” and clear instructions for storage below 25°C, in tightly closed amber bottles. Safety Data Sheets list decomposition products, short- and long-term symptoms, and standard disposal routes. Regulatory compliance means bracketing it under Schedule II dangerous substances in some countries, restricting bulk shipments.
Preparation Method
Most labs stick with the classic path: converting 3-nitrobenzoic acid to the chloride using either thionyl chloride or phosphorus pentachloride. Thionyl chloride boils off as sulfur dioxide and hydrogen chloride, leaving behind a clean solution of the acyl chloride, which can be coaxed into crystallization. Some researchers favor oxalyl chloride for higher yields and fewer side products, though costs run higher and handling grows complex. In all cases, the methods demand anhydrous glassware, fume hoods, and careful control of addition rates—the exothermic nature escalates with larger batches, giving operators little room for error.
Chemical Reactions and Modifications
3-Nitrobenzoyl chloride acts as a potent acylating agent. With amines, it forms amides; with alcohols, it forges esters. Its nitro group also takes part in further reactions, such as reduction to amino derivatives—a step often followed in dye and pharmaceutical routes. In peptide chemistry, scientists reach for this molecule when they want a strong electron-withdrawing group to protect certain portions of a growing chain. On the industrial side, it gets plugged into nucleophilic aromatic substitution routes, turning out advanced intermediates for organic electronics. Its dual reactivity—both as a typical acyl chloride and as a nitroarene—opens up a long menu for chemists in both research and plant settings.
Synonyms and Product Names
Besides its main label, this compound pops up as 3-nitrobenzenecarbonyl chloride, meta-nitrobenzoyl chloride, and 3-NBC among catalogues from leading chemical suppliers. Each synonym links back to specific regulatory, patent, or regional naming traditions. Sometimes, it hides under less formal monikers in academic papers or chemical inventories, but most technical workflows stick with IUPAC or traditional trade terms to avoid confusion. Despite slight differences in terminology, all refer to the sharply reactive, crystalline solid built off the nitro-substituted benzene ring.
Safety and Operational Standards
3-Nitrobenzoyl chloride requires careful handling because it reacts with moisture and produces highly corrosive acids on contact. In my own experience prepping similar reagents, it pays to double-check PPE, with butyl gloves and face shields as routine kit. Always keep water sources locked out of handling rooms, and bag up any contaminated waste in double-sealed containers. Respiratory risk remains top of mind—one accidental spill can stink up a whole corridor and kick off a fire drill or demand an evacuation. Regular training on spill kits, eyewashes, and remote ventilation makes all the difference in keeping lab accidents minor. Supply chains look for suppliers certified under ISO chemical safety standards, plus GHS-compliant labeling. Disposal routes enforce neutralization with cold, dilute base before consigning materials to hazardous waste incineration facilities.
Application Area
Pharmaceutical developers leverage 3-nitrobenzoyl chloride as a building block for beta-lactam antibiotics and custom amide-linked drugs. Agrochemical scientists loop it into formulations for herbicides and advanced insecticides, counting on the reactivity of both the acyl chloride and nitro groups. In materials science, it helps produce monomers and specialty dyes that absorb in the visible spectrum. In my research group, attaching the 3-nitrobenzoyl moiety to small-molecule scaffolds gave us a tunable handle to boost solubility and tweak electronic properties. Bioanalytical circles use it as a derivatizing agent for amino acids and peptides, improving detection in mass spectrometry. Its widespread industrial uptake mostly reflects the value of combining a high-activity acyl chloride with a robust electron-withdrawing group.
Research and Development
Recent journal literature around 3-nitrobenzoyl chloride rallies around sustainable synthesis and safer scaling. Green chemistry labs try swapping thionyl chloride for less hazardous reagents and look into continuous flow systems for better accuracy and efficiency. Medicinal chemistry projects harness its unique features for drug conjugates targeting resistant infections. Polymer chemists test its performance in functional monomer preparations, chasing better flexibility and colorfastness in specialty plastics. And data science teams keep the molecule on virtual shelves, feeding its structure into AI models for predictive reactivity and environmental fate. Having worked with teams pushing reaction selectivity, I’ve learned that the real power comes from blending computational design with hands-on optimization—tweaking conditions by the hour pays off after weeks of trial runs.
Toxicity Research
Toxicology studies reveal that 3-nitrobenzoyl chloride poses significant risks: inhalation triggers swelling of respiratory tissue, and skin contact can leave blisters or chronic dermatitis. Acute exposure wastes no time in causing eye injuries. Chronic effects receive less publicity but include the formation of methemoglobin in blood and possible liver impacts, as reported in animal models. Ecotoxicologists chart out its behavior in water systems, tracking how rapid hydrolysis to 3-nitrobenzoic acid might disrupt local aquatic life. Governments review updated hazard data regularly, imposing stricter handling limits and storage protocols. In the lab, we always treat this substance as inherently dangerous until every trace gets washed down with neutralizers and confirmed clear by swab tests. No shortcuts exist—an ounce of prevention and thorough post-use cleaning turns hazardous work into routine practice.
Future Prospects
Interest in 3-nitrobenzoyl chloride keeps growing with the rise of custom organic synthesis and green chemistry initiatives. Companies invest in cleaner production rigs, aiming to phase out the worst byproducts and automate most transfer steps. Its adaptability for specialty pharmaceuticals and custom polymers points toward an era where each new variant gets a full safety and environmental audit before rollout. Computer-aided process control continues to eat away at human error, while predictive modeling helps dial in ideal conditions before chemists crack open the reagent cabinet. As more nations revise their chemical laws, the field heads for a world where tech meets vigilant stewardship—each new experiment checked against both immediate results and long-term responsibilities.
What is the chemical formula of 3-Nitrobenzoyl Chloride?
Breakdown of Composition
Chemistry books might list 3-Nitrobenzoyl chloride next to dozens of similarly named compounds, but the basics stand out once you dig in. This compound builds from the benzene ring, which forms the backbone for a huge slice of organic chemistry. In 3-nitrobenzoyl chloride, a nitro group attaches to the third carbon of that aromatic ring, while a carbonyl group linked to a chlorine atom (the acyl chloride part) anchors its identity. Its chemical formula spells out C7H4ClNO3.
Why Structure Matters in the Real World
Every lab technician dealing with organic synthesis learns that the placement of just one group alters more than a textbook page. By adding a nitro group in the meta position—three carbons away from the carbonyl chloride—chemists can coax out differences in reactivity. This comes into play big time in pharmaceutical or dye manufacture. The formula tells you more than just the quantity of each atom. It hints at how this compound will act under certain lab conditions, especially with nucleophiles.
Mistakes can get costly. Subbing a para or ortho nitro group for meta can wreck a planned reaction. More than once I’ve seen a student grab the wrong bottle, thinking all benzoyl chlorides react the same. Next thing you know, product yields dip and lab managers frown. Getting the formula and naming right gives clarity. Manufacturers rely on this accuracy to stick to safety protocols, especially since acyl chlorides react dramatically with water, often producing toxic or corrosive byproducts.
Connections to Industry and Research
C7H4ClNO3 doesn’t exist only on the chalkboard. Fine chemical producers use it to introduce the 3-nitrobenzoyl group into peptides or other organic molecules. These reactions help produce targeted drugs or test new materials. Its formula gets checked meticulously because impurities, whether starting from the wrong isomer or slipping up in synthesis, can disrupt the end product and sometimes trigger regulatory headaches.
Academic research leans heavily on precision. An undergraduate project can stop dead for days over a formula mix-up. I remember a year when our group tried to design a compound library for protein inhibition studies. We needed acyl chlorides with specific nitro positions. Each one had to match the formula and structure exactly. Skipping proper checks, a couple of samples came contaminated—not even visible until later analysis. That delayed our project and forced us to repeat expensive steps.
Solutions Going Forward
Lab techs and students shouldn’t just memorize formulas. Hands-on activities, like building models and tracing each atom’s location, help lock in how structure drives function. In a fast-paced lab, even seasoned researchers benefit from quick verification routines—checking both chemical formula and IUPAC name before weighing or mixing. Digital inventory systems with built-in alerts can reduce human error. Regulatory bodies provide guidelines for labeling, storage, and disposal, helping keep the workplace safe and product quality high.
Reliable chemical databases make all the difference. Scientists working under pressure can verify structures, understand hazards, and avoid mix-ups. Sharing practical stories like these during safety briefings adds real value, making the formula more than just numbers and letters on a page.
What are the common applications of 3-Nitrobenzoyl Chloride?
What Sets 3-Nitrobenzoyl Chloride Apart
3-Nitrobenzoyl chloride stands out as a well-regarded reagent in the world of organic synthesis. You often find this compound in places where scientists need to build more complex molecules from simple blocks. It has a nitro group and an acyl chloride function, both of which lead the way for making all sorts of chemical transformations possible.
Driving Progress in Pharmaceuticals
The pharmaceutical industry counts on intermediates like 3-nitrobenzoyl chloride. Lab chemists use it to attach nitrobenzoyl groups to amino acids or nucleosides, which then serve as building blocks for drug candidates. This molecule helps with the synthesis of peptides and other small organic compounds, offering key properties that researchers look for in medicinal chemistry. Sometimes you see it helping to mask a free amine group during protection-deprotection strategies, which improves selectivity and final yields of active substances. Painkillers, beta-lactam antibiotics, and even some anti-inflammatory drugs may trace a step in their process back to reagents just like this one.
Broad Role in Chemical Research
Beyond drug development, labs rely on 3-nitrobenzoyl chloride for a variety of synthetic routes. If you step into a university or chemical research firm, you will see graduate students and seasoned professionals working with such reagents to explore new reactions or produce specialty compounds. Analytical chemists sometimes use it to form derivatives for high performance liquid chromatography (HPLC), which helps them analyze amino acids more easily. During my research days, I used this technique to separate complex mixtures and verify amino acid compositions in samples taken from food or feed.
Spotlight on Agrochemicals and Dyes
This compound finds itself in the process flows of agrochemical manufacturing. You get herbicides and pesticide intermediates from reactions that involve 3-nitrobenzoyl chloride. Farmers may never hear of it, but chemists making crop protection products depend on such intermediates to build molecules that keep harvests strong.
The dye and pigment sector also benefits from its aromatic structure and electron-withdrawing nitro group. Synthesizing yellow or orange azo dyes and other pigments involves these building blocks. Makers of materials used in textile dyeing or even coloring plastics know that specialty chemicals provide the hues and fastness consumers ask for.
Potential Risks and a Call for Safer Practices
With reactive chemicals like 3-nitrobenzoyl chloride, the question of health and environmental risks comes up quickly. It reacts with water to release hydrochloric acid, which can threaten workers if proper ventilation and personal protection do not stand in place. Regulations in Europe, the U.S., and several Asian countries now mandate closed systems or improved labeling. At an old workplace, we faced an incident after someone handled acyl chlorides without a face shield—simple steps in training and availability of protective equipment would prevent much of the harm.
Green chemistry brings hope by pushing chemists to invent milder alternatives or reduce the use of hazardous reagents altogether. Newer methods sometimes turn to enzymatic processes or safer acylating agents, though adoption lags behind tradition. For the moment, thorough safety routines, routine monitoring, and informed handling stand as the best barriers against harm, while researchers keep looking for better ways to get the chemistry done without so much risk.
How should 3-Nitrobenzoyl Chloride be stored and handled safely?
The Real Dangers Behind a Small Bottle
3-Nitrobenzoyl chloride isn’t the kind of chemical someone should toss in a supply closet and forget. It looks like a harmless white to pale-yellow solid, but one whiff or a splash on the skin can ruin someone’s day—or much worse. The fumes irritate the nose and lungs, and if it spills on hands, it can burn deep. I once worked in a lab where someone fumbled a container, ended up with a chemical burn, and we all learned there’s no room for shortcuts with chemicals like this.
Choosing the Right Storage Spot
A shelf above the family fridge won’t cut it. This kind of chemical works best when it sits in a cool, dry spot with good air flow. Moisture in the air breaks down the compound pretty quickly, releasing corrosive hydrochloric acid. I’ve walked into labs that figured regular cabinets were good enough, only to see rusted hinges and blistered paint where vapors escaped. That’s a signal for chemical-resistant cabinets. A dedicated, tightly-sealed storage container helps keep fumes inside and air outside. Keep it away from doors and walkways. You want a spill to happen in a place with quick access to showers and eye washes, not where folks are rushing past.
Keeping the Air Clean
Fume hoods aren’t fancy extras—they’ll save your lungs. Pouring or measuring 3-nitrobenzoyl chloride without proper ventilation fills a room with biting fumes, and those don’t just evaporate. Some colleagues installed exhaust fans above their benchtops and thought that was enough. The fumes settled anyway, leaving a yellowish residue that corroded their equipment. Sealed systems, fitted fume hoods, or glove boxes help keep the work safe. I always check the ventilation before popping open any bottle, not after.
Personal Protective Gear Matters
Goggles and gloves aren’t negotiable. Friends of mine who cut corners believed safety glasses alone would protect them, only to end up blinking through burning tears from vapor exposure. Nitrile or neoprene gloves hold up well, but thin latex doesn’t. Lab coats help too, but sleeves should always be snapped tight; loose cuffs catch and spread splashes. After using this compound, always wash exposed skin, even if nothing feels wrong. Sometimes irritation sneaks up hours later.
Handling Spills and Waste
No matter how careful someone acts, spills happen. Around this chemical, having a neutralizing agent close by, like sodium bicarbonate, turns a bigger crisis into a minor cleanup. Always soak up spills with absorbent pads designed for acids, not kitchen towels or paper napkins. Once used, label the waste clearly and send it off for professional disposal. A local waste handler once told me stories of mystery drums leaking at landfill sites—all because someone didn’t label or seal a chemical like this properly. Nobody wants their mistake to show up in groundwater.
Keep Training and Communication Going
Relying on old habits or assumptions slowly turns a workplace dangerous. I’ve seen teams who updated each other on storage locations and emergency plans after every shipment. Those teams had fewer injuries and never panicked during spills. Regular training and direct communication about hazards and procedures keep everyone sharp, especially with potent chemicals.
Every Step Matters
Protecting people and property around reactive compounds, like 3-nitrobenzoyl chloride, never happens by accident. It takes clear planning, strong discipline, and the collective wisdom of every technician in the room. One person’s shortcut can put everyone at risk, so every box ticked on the storage and handling checklist ends up defending someone’s health.
What are the potential hazards associated with 3-Nitrobenzoyl Chloride?
Why It Matters
Chemists and lab workers run into dozens of unfamiliar names on labels every day, but 3-nitrobenzoyl chloride tends to stick out with some real safety concerns. This chemical gets a lot of use making dyes, pharmaceuticals, and specialty materials, and ends up in labs because it reacts so easily. The thing is, its reactivity can spell trouble for anybody who comes in contact with it without the right know-how.
Inhalation and Breathing Risks
I still remember my first time opening a fresh bottle in the lab. Even with a fume hood humming, the sharp stinging scent lingered fast and strong. The vapor irritates quickly – eyes water, nose burns, throat gets scratchy. Usually, just a whiff is enough to know you need to stay cautious. The real risk comes if someone forgets to keep things ventilated. Over time, exposure leads to coughing, shortness of breath, and in high doses, lasting lung issues. I’ve had colleagues who needed a trip to the nurse because they underestimated how much vapor would escape during transfer.
Skin and Eye Problems
Gloves and safety goggles aren’t just a formality with 3-nitrobenzoyl chloride. A splash on skin burns pretty fast, leaving redness and blisters. The corrosive bite wrecks tissue layers before you can even start flushing the area, and if it gets in your eyes, the risk of lasting damage goes way up. I always double-check my protective equipment before handling it, after seeing how fast a missed droplet can turn into a medical emergency.
Reactivity Dangers
Most accidents happen when people rush. 3-nitrobenzoyl chloride mixes violently with water and alcohols, shooting out hydrochloric acid gas. Even tiny amounts of water on surfaces or tools can set off that reaction. In one mishap, an intern in the department grabbed a wet spatula, leading to an immediate cloud of acid vapor. It doesn’t take much for a minor mistake to turn serious.
Long-Term Exposure and Sensitization
There’s growing evidence that long-term, low-level exposure causes issues many don’t spot right away. Cases of asthma and severe allergic reactions have popped up in seasoned chemists who worked around this chemical for years. I’ve seen people become sensitized, starting with minor irritation, only to find even trace amounts set off full symptoms later.
What Works to Reduce Risk
Vigilance lowers the odds of getting hurt. Fume hoods, proper gloves, and splash-resistant goggles beat shortcuts every time. If a spill happens, quick cleanup by trained staff with neutralizing agents stops the spread fast. Labels and training stick in memory – “Do not breathe fumes, corrosive, avoid contact with water” isn’t just a classroom warning. I encourage colleagues to run safety drills once a month, even if it feels repetitive. The more comfortable people get with correct procedures, the less likely they’ll freeze up during real incidents.
Storage matters too. Keep 3-nitrobenzoyl chloride sealed tight, away from damp air and incompatible chemicals. Accidents drop when inventory doesn’t sit in reach of busy traffic. Disposal by professionals at certified facilities takes chemicals out of regular waste streams. Teaching good habits to students and new workers, along with regular reviews, keeps lessons fresh.
Looking Ahead
Research continues on safer alternatives and improved personal protective gear for working with this compound. A smart approach combines respect for the chemical’s hazards with practical steps – the kinds I’ve learned are worth following every single lab shift.
What is the purity and CAS number of 3-Nitrobenzoyl Chloride available for purchase?
Spotting Purity Standards in Chemical Purchases
Most labs, whether in pharmaceuticals, materials science, or university research, put chemical purity at the very top of their shopping list. For 3-Nitrobenzoyl Chloride, nearly every supplier will advertise purity of at least 98%. Some offer 99% for projects demanding tighter controls. That extra percent sometimes costs more, but for sensitive organic reactions or analytical studies, it often pays off in fewer impurities and better reproducibility. As a general practice, experienced researchers buy higher-purity chemicals when the process has narrow tolerances or where trace contaminants can create headaches down the line.
Published certificates of analysis—these arrive with reputable shipments—will list not just purity but also known impurity levels. I’ve dealt with several catalogs over the years, and the small differences between 98% and 99% have big effects in end results, especially in pharmaceutical intermediates. Less-pure materials often carry leftover moisture or traces of by-products like unreacted acid chlorides or nitrobenzoic acid, which throw off yields or complicate purification steps. Nothing slows down a day like running chromatography three or four extra times because the starting batch wasn’t clean enough.
Tracking Down the CAS Number
Every chemical picked up from the shelf or ordered online gets tracked by its CAS number—sort of like having a unique fingerprint for molecules. For 3-Nitrobenzoyl Chloride, that number is 121-24-6. This string shows up on invoices, bottles, and MSDS, helping everyone in the chain know exactly what’s being handled. Mixing up numbers puts projects, safety, and compliance at risk. In my time at a university chemistry storeroom, clear CAS labeling cut confusion, especially with similar-sounding aromatics and benzoyl compounds sitting a row apart.
Sourcing chemicals under the right CAS builds more trust between buyer and seller. It keeps communications clear, especially across international orders, where product names or trade labels have regional differences. CAS 121-24-6 links directly to 3-Nitrobenzoyl Chloride in chemical registries, giving access to all the hazard information, regulatory flags, and proper disposal instructions a lab may need. This small step protects not just experiments, but staff and downstream users.
Challenges and Solutions in Buying Chemicals Online
Shopping for 3-Nitrobenzoyl Chloride online looks effortless until you hit a wall with incomplete information or questionable suppliers. Some websites display little beyond a product name, and you might have to dig on technical sheets for purity details or that crucial CAS number. Always ask for up-to-date COAs and double-check supplier reputation. Inconsistent data waste time, money, and sometimes put users at risk if a mistake leads to exposures or failed syntheses. Authentic vendors, in contrast, provide full data up front—it’s part of their transparency and legal compliance.
Safety extends beyond just buying. The same chemical under the same CAS can behave differently if made too impurely or stored for months in humid stockrooms. Good habits: only buy as much as needed, check container integrity on arrival, and rotate stocks regularly, just as you would with perishables in a fridge. Proactive safety also means reviewing and communicating hazards found under the CAS profile from sources like PubChem or Sigma-Aldrich’s database. Even seasoned professionals revisit these resources, keeping everyone aware not just of theoretical risk but practical control procedures in handling benzoyl chlorides.
Responsible Buying and Lab Practice
Knowledge of purity levels and understanding the correct CAS number isn’t dry bureaucracy; it stands as practical and sometimes life-saving routine. Labs that practice transparency and rigorous documentation foster not just better science but safer, more reliable work environments. As chemical safety culture grows globally, sharing clear and accurate information forms the basics for allyship between industries, suppliers, and researchers—a habit from which everyone benefits.
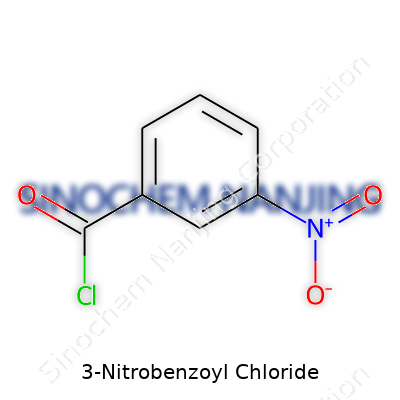
| Names | |
| Preferred IUPAC name | 3-nitrobenzoyl chloride |
| Other names |
m-Nitrobenzoyl chloride 3-Nitrobenzoic acid chloride m-Nitrobenzenecarbonyl chloride |
| Pronunciation | /ˈnaɪtroʊ.bɛnˌzɔɪl ˈklɔːr.aɪd/ |
| Identifiers | |
| CAS Number | 121-90-4 |
| Beilstein Reference | 583110 |
| ChEBI | CHEBI:28151 |
| ChEMBL | CHEMBL15404 |
| ChemSpider | 121604 |
| DrugBank | DB08641 |
| ECHA InfoCard | ECHA InfoCard: 100.007.474 |
| EC Number | 211-625-2 |
| Gmelin Reference | 7894 |
| KEGG | C19263 |
| MeSH | D014748 |
| PubChem CID | 74984 |
| RTECS number | CU5950000 |
| UNII | 660D8U366K |
| UN number | UN1575 |
| Properties | |
| Chemical formula | C7H4ClNO3 |
| Molar mass | 169.56 g/mol |
| Appearance | White to light yellow powder |
| Odor | Pungent |
| Density | 1.44 g/cm3 |
| Solubility in water | Reacts with water |
| log P | 1.96 |
| Vapor pressure | 0.17 mmHg (25°C) |
| Acidity (pKa) | 6.8 |
| Basicity (pKb) | 12.50 |
| Magnetic susceptibility (χ) | -51.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.5890 |
| Viscosity | 1.45 cP (20°C) |
| Dipole moment | 2.89 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 340.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -72.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -492.4 kJ·mol⁻¹ |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07, GHS08 |
| Pictograms | GHS05, GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H314, H317, H332, H334, H335 |
| Precautionary statements | P261, P264, P271, P272, P280, P301+P330+P331, P302+P352, P304+P340, P305+P351+P338, P310, P312, P330, P363 |
| NFPA 704 (fire diamond) | 3-1-0-OX |
| Flash point | 112 °C |
| Autoignition temperature | 225 °C |
| Lethal dose or concentration | LD50 oral rat 2340 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 325 mg/kg |
| NIOSH | NA0452000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 1 mg/m³ |
| Related compounds | |
| Related compounds |
Benzoyl chloride 2-Nitrobenzoyl chloride 4-Nitrobenzoyl chloride 3-Nitrobenzoic acid 3-Nitrobenzamide |

