3-Nitrobenzenesulfonyl Chloride: A Perspective on Science and Industry
Historical Development
Long before modern chemistry labs filled with machines and computer screens, scientists searching for new synthetic routes stumbled upon the utility of aromatic sulfonyl chlorides. The nitro-substituted variant grew out of need for better reagents in both academic and industrial research. Early 20th century chemistry journals mention 3-nitrobenzenesulfonyl chloride as a prized intermediate, especially when workhorse sulfonylations demanded more specificity than inherited with plain benzenesulfonyl chloride. The compound migrated from glass-packed laboratories to chemical plants, underscoring its value in the toolkit for making dyes, pharmaceuticals, and agrochemicals.
Product Overview
3-Nitrobenzenesulfonyl chloride stands out with a yellowish hue, signaling its electron-rich aromatic ring balanced by both nitro and sulfonyl chloride groups. Unlike many specialty reagents, it remains stable enough for storage but reacts briskly in synthetic steps. Chemists who spend days seeking cleaner, one-pot strategies often praise its predictable behavior and the strong leaving group built into its structure.
Physical & Chemical Properties
With a molecular formula of C6H4ClNO4S and a molar mass just under 237 grams per mole, 3-nitrobenzenesulfonyl chloride remains solid at room temperature—a fine powder that often clumps in open air. Its melting point ticks up to about 93-95°C, offering enough heat stability for most bench chemistry but melting easily during scale-up. A strong odor escapes opened containers, matched by the warning burn if it meets unprotected skin. Water readily hydrolyzes the sulfonyl chloride, so dry glassware becomes a habit, not an afterthought. Solubility favors organic solvents like dichloromethane or ethyl acetate, skipping water for obvious reasons.
Technical Specifications & Labeling
Clear labeling protects both the handler’s health and experimental accuracy. Bottles bear hazard symbols under the Globally Harmonized System (GHS): corrosive and harmful, sometimes with an oxidizer sign depending on supplier lot analysis. Purity runs above 98% for research-grade material, with GC or HPLC showing minor impurities from side reactions. Each batch might come with an accompanying analysis for trace moisture, residual toluene sulfonyls, or breakdown compounds.
Preparation Method
Synthesis usually relies on nitration of benzenesulfonyl chloride, a step that demands temperature control to suppress overreaction and parallel ring substitution. Commercial makers often streamline the process: start with m-nitrobenzene, then introduce chlorosulfonic acid, heating slowly for smooth conversion. After extracting with organic solvents and repeated crystallization, workers isolate a product ready for both shelf and scale. Even small changes in temperature or acid concentration shift yields or cause sulfonation at unwanted positions. In smaller labs, the process echoes this route, swapped for round-bottom flasks and ice-bath cooling.
Chemical Reactions & Modifications
3-Nitrobenzenesulfonyl chloride opens up wide possibilities for both protection strategies and molecular rearrangements. Its main trick involves sulfonylation of amines and alcohols. The nitro group tunes electronic properties so acylation proceeds with control—chemists crafting sulfonamides and esters have come to rely on it. It acts as more than a reagent; it guides the pace and outcome of substitution in aromatic rings, crucial for fine-tuning dyes and pharmaceutical precursors. Nucleophilic displacement with amines gives solid yields, and the easy extrusion of the sulfonyl group under mild reduction sets up further transformations, sometimes saving days in a synthetic sequence.
Synonyms & Product Names
Common alternative names turn up in catalogs and journals: “3-Nitrobenzenesulfonic acid chloride”, “m-Nitrobenzenesulfonyl chloride”, or “NSC 11110”. Older chemistry books sometimes call it μ-nitrobenzenesulfonyl chloride, but the “m-” prefix has stuck as naming standards sharpened. European suppliers occasionally use “Nitrobenzene-1-sulfonyl chloride”, a reminder that product research benefits from checking CAS numbers to avoid confusion.
Safety & Operational Standards
Safety practice keeps this compound from becoming a liability in the lab. The sulfonyl chloride group’s reactivity means splash-proof goggles are mandatory. Nitrated aromatic compounds can irritate skin or lungs. Fume hoods, gloves of at least nitrile make a difference, especially during weighing or transfer. Storage away from heat or water, in tightly sealed bottles, prevents both decomposition and hazardous fumes. Labs that routinely work with this material keep their MSDS on hand, and waste disposal separates it into organic chlorinated bins, not the general waste stream. Fire hazards climb if traces of flammable solvents remain—thus, process engineers often double-check for clean, dry product before packing.
Application Area
Demand for 3-nitrobenzenesulfonyl chloride spans industries that touch everyday life. Dyes no longer fade so quickly after chemists deploy the compound to append stable groups onto complex backbones. Pharmaceutical manufacturers build anti-infective agents and enzyme inhibitors starting with sulfonylation reactions—particularly since the nitro group sometimes acts as scaffolding for further elaboration. Pesticides owe some effectiveness to modifications on nitrobenzene rings, their synthetic complexity often anchored by a single, reliable reagent such as this one. Even in electronics, advanced materials depend on high-performance intermediates where tightly-defined specifications sharpen the final product. College labs demonstrate nucleophilic aromatic substitution using it, teaching new chemists valuable lessons in both mechanism and safety.
Research & Development
The push for greener processes and safer reagents motivates chemists to revisit even trusted workhorses. 3-Nitrobenzenesulfonyl chloride finds its role tested in solvent-less reactions and flow chemistry setups, where minimizing waste or boosting selectivity makes a difference in both environmental footprint and cost. Researchers report on alternative sulfonation catalysts or efforts to replace hazardous chlorosulfonic acid with milder surrogates. Surface chemistry studies examine how controlled sulfonylation tweaks polymer properties, opening pathways to responsive coatings or smart films. Even niche studies explore the impact of electron-withdrawing groups on downstream catalytic steps, an area that brings together computational and synthetic chemistry in collaborative work.
Toxicity Research
Toxicologists consider both acute and chronic exposure in animal models. Data point to moderate skin and eye irritation, with inhalation risks rising during large-scale handling. Studies show the nitro group’s persistence complicates degradation in aquatic environments, prompting monitoring of industrial effluent streams. No significant mutagenicity appeared in several bacterial assays, but prudent researchers avoid direct contact and limit environmental release. Regulations in the EU and US reflect these findings, requiring clear documentation and exposure tracking for both workers and waste processors.
Future Prospects
Chemistry moves forward every year as new artificial intelligence models, green synthetic protocols, and stricter environmental rules shape industrial decision-making. 3-Nitrobenzenesulfonyl chloride faces pressure to meet these standards without sacrificing performance. Makers invest in process optimization to cut down on waste and boost yield, while researchers look for ways to tether the compound to bio-resins for easier recovery. Its role as a versatile intermediate looks secure, especially given the expanding set of fine chemicals built on “designer” benzene rings. With ongoing work in continuous flow reactors and low-impact solvents, its story remains one of adaptation and technical curiosity, never standing still in a crowded chemical landscape.
What is the chemical formula of 3-Nitrobenzenesulfonyl Chloride?
Getting Down to the Formula
3-Nitrobenzenesulfonyl chloride might sound intimidating, but its chemical formula keeps things pretty straightforward: C6H4(NO2)SO2Cl. Breaking it down, the benzene ring (C6H4) forms the backbone. Add a nitro group (NO2) at the third position on the ring, then tack on a sulfonyl chloride group (SO2Cl). It's not just a collection of elements on a page—this arrangement shapes how the substance acts in a lab and out in the world.
Experience with Synthesis and Safety
Working with organic chemistry in both academic and industrial labs, you notice some chemicals command extra respect. Substances like 3-nitrobenzenesulfonyl chloride come with real-world consequences. The sulfonyl chloride group makes it reactive, so it demands careful handling and the right protective gear. I once watched a classmate skip goggles during a demonstration involving a sulfonyl chloride. A splash sent everyone scrambling and left a cautionary tale. Proper ventilation and protocols keep people safe, even when experiments get routine.
Why Structure Shapes Function
This compound has a structure that suits a niche but essential job. The nitro group on the ring not only marks its position, it shifts the electronic properties across the whole molecule. That tweak ramp ups its reactivity, especially when building new molecules for research or pharmaceuticals. It’s not about making things more complex for the sake of it. This very formula allows chemists to attach groups exactly where needed, opening paths to create antibiotics, dyes, or specialty reagents. The value of precision gets clear when a slight shift—like a different position for that nitro group—results in completely different chemistry.
Real-World Importance
Moving past the dry details, people rely on these reactive molecules to spur new discoveries. 3-Nitrobenzenesulfonyl chloride helps chemists add protection to amine groups, offering a stepping stone in the creation of acids, drugs or advanced polymers. Processes like peptide synthesis wouldn't run as smoothly without it. It’s not about being on a shelf for show. Its reactivity, courtesy of the SO2Cl group, plays into steps that streamline entire production lines.
Safer Chemistry, Smarter Regulation
The more I see in this field, the clearer it is: safety and transparency matter as much as the research itself. Details about toxicity, handling, and environmental impact can’t get buried. Sulfonyl chlorides release hydrochloric acid on contact with water, creating health risks. Companies need tight controls, from storage to disposal. Proper documentation—clear labels, up-to-date Material Safety Data Sheets—keeps both new and experienced chemists alert.
Steps Toward Sustainable Use
There’s movement in green chemistry to find substitutes that cut down on toxic byproducts. Engineers and chemists are tweaking procedures to recycle reagents or even swap in milder alternatives. Sharing results, even from small lab-scale successes, can nudge the whole field toward safer and more sustainable practices.
Looking Ahead
The world of chemistry keeps evolving, and it starts with deep knowledge about even a single formula like C6H4(NO2)SO2Cl. Putting that knowledge into safe, smart use makes a difference across research, industry, and most importantly, in day-to-day lives.
What are the main applications or uses of 3-Nitrobenzenesulfonyl Chloride?
Inside the Chemical Toolbox
Walk into any mid-sized organic chemistry lab, and there’s a good chance you’ll spot a bottle of 3-Nitrobenzenesulfonyl chloride somewhere on the shelf. For all its unwieldy name, this compound proves its mettle as a workhorse when researchers want precise chemical transformations. It’s not some rare specialty reagent sold only by a single niche company; it regularly shows up in books and journal articles because it brings flexibility to synthetic chemistry, especially during tough steps of drug and fine chemical production.
Amino Group Protection
Any chemist who has tried building a complex organic molecule knows how tricky it can be to keep one part of a molecule quiet while changing another. 3-Nitrobenzenesulfonyl chloride steps in here, especially if you’re working with amines. It quickly reacts with them to form sulfonamides, which means that chemists can “protect” the amine part, do their magic on the rest of the molecule, and then gently remove the protection when they’re ready. In my own studies, I reached for this sulfonyl chloride to avoid harsh conditions that would destroy fragile pieces elsewhere in the structure. This selective reactivity saves countless hours and money during pharmaceutical and agricultural chemical development.
Making New Drugs Possible
The world of drug creation relies heavily on building blocks. 3-Nitrobenzenesulfonyl chloride helps craft those pieces. It shows up not just for protection steps, but also for constructing harder-to-build drug intermediates, especially those containing nitrogen. During industrial projects, process chemists often turn to this compound when they need to whip up sulfonamide drugs or antibiotics. It brings both speed and reliability, which counts for a lot when regulatory deadlines loom.
Boosting Diagnostic Reagents and Dyes
Labs don’t just invent medicines—they churn out new dyes and diagnostic agents, too. In these fields, 3-Nitrobenzenesulfonyl chloride helps attach color-giving or light-responsive pieces onto molecules. Some reagents used to flag certain biomolecules in blood tests depend on this chemical to make sure they light up when needed. Industrial research has unlocked new imaging agents for cancer detection, built off the backbone of compounds first prepared with the help of this sulfonyl chloride. That means better test accuracy and earlier health warnings.
Toward Greener Chemistry
With every chemical, questions about safety and waste follow close behind. 3-Nitrobenzenesulfonyl chloride’s production and use generate concerns because of its reactivity and the potential hazards of chlorinated byproducts. Research groups and manufacturers have started using new containment and recycling methods to capture as much of the material as possible and neutralize runoff before it reaches the environment. As more industrial labs shift to less toxic or recyclable materials, companies investing in greener approaches—such as using alternative solvents or developing one-pot reactions—help reduce risk for both workers and the planet.
Finding Smarter Solutions
Newer synthetic routes now use 3-Nitrobenzenesulfonyl chloride more efficiently, squeezing out better yields with less waste. Some teams design closed-loop systems where leftovers can serve as feedstock, trimming costs and emissions. Smarter automation and safety protocols protect lab workers from accidental exposure. Above all, clear labeling and education about best practices matter most. My own experience taught me early on to respect chemicals that “do a job with bite”—and this one definitely falls into that category. Investing in proper storage, handling, and green disposal stands as the best bet for keeping its benefits while limiting hazards.
How should 3-Nitrobenzenesulfonyl Chloride be stored and handled safely?
Why Chemical Safety Matters
I remember standing in a cramped research lab about a decade ago, watching a seasoned chemist talk about chemical burns from simple mistakes. He described the sting that followed poor handling of chemicals similar to 3-Nitrobenzenesulfonyl Chloride—hazardous and stubbornly reactive. The room full of fresh graduates, myself included, listened closely. Those stories stay with you, especially when storing and handling anything that can create danger from carelessness.
Understanding the Risks
3-Nitrobenzenesulfonyl Chloride stands out for its reactivity, especially with moisture. Contact with even a small amount of water brings about the release of toxic gases like hydrogen chloride. Inhale those fumes and you’re facing potential burns to airways, skin irritation, and worse if you don’t get out fast. The substance also irritates the eyes and skin, so hurrying through any task spells disaster. In an era focused on occupational well-being, nobody should overlook these risks.
Based on my years around chemical stockrooms, complacency sets the stage for emergencies. Over the past five years, chemical industry reports have highlighted a handful of accidents each year tied directly to improper storage or handling of sulfonyl compounds. All could have been prevented.
Storage: Lessons from Straightforward Mistakes
I once saw a perfectly labeled jar of 3-Nitrobenzenesulfonyl Chloride go cloudy on the shelf after a careless lid replacement. Humidity from the air did more harm than a spilled drop. This stuff belongs in air-tight containers, away from both water and strong bases. Dry, cool, and well-ventilated shelves or cabinets mark the line between safety and a chemical mess. Use containers made from materials that do not react with the chemical itself—glass or high-quality plastic with snug seals get the job done.
Do not keep this chemical near acids, strong oxidizers, or anything combustible. Mixing leads to violent reactions or risky fumes. Every reputable safety guide points this out for good reason, as real-world accidents show.
Safe Handling: Techniques You Can Trust
The safest hands wear gloves—nitrile, not bare skin. Eye protection remains non-negotiable. Lab coats ought to cover arms, not just hang off the shoulders. That’s what we settled on during my own lab work. Running the fume hood, even if the task seems simple, keeps lingering vapors out of your lungs. Never skip this step.
Only work with 3-Nitrobenzenesulfonyl Chloride using proper containment: trays that catch drips, clean and dry surfaces, and tidy up before and after. Spill kits don’t collect dust—they get used. Handle with small amounts and plan the procedure from start to finish, minimizing open time.
If exposure occurs—rinsing with water right away makes the difference. Move to fresh air if fumes build up. I saw a quick rinse prevent a nasty burn once; people remember that lesson and share it with newcomers again and again.
Building a Culture of Care
Anyone working with chemicals like this owes it to coworkers and themselves to follow tested methods. Companies succeed not by cutting corners, but by learning from those who came before. Safety training shouldn’t be rushed or skipped. Doing things the right way protects health, keeps projects on track, and avoids costly incidents.
Smart choices—sealed containers, the right gear, deliberate steps—turn risky compounds into manageable tools. Managers encouraging that mindset, and giving people the resources to do things right, create safer workplaces for everyone.
What is the purity or grade of 3-Nitrobenzenesulfonyl Chloride typically offered?
Practical Considerations in the Lab
Anyone who’s worked with specialty chemicals in a lab understands purity is more than just a number on a spec sheet. Using 3-Nitrobenzenesulfonyl Chloride—often called NsCl—puts this lesson into clear focus. For synthetic chemists, the fine print matters; a reagent loaded with impurities can sabotage a reaction or contaminate a delicate product. Purity makes the difference between a successful synthesis and a week’s work down the drain. For research and industrial users alike, most suppliers list 98% to 99% as the typical grade. Suppliers rarely offer anything below this range, since the compound often goes into high-sensitivity reactions, like peptide synthesis or fine chemical intermediates.
Why High Purity Matters
Small differences in grade can cause big headaches. Sulfonyl chlorides react aggressively, especially with nucleophiles or moisture. If the chemical arrives tainted—organic byproducts, excess moisture, or leftover solvents—these hitchhikers can change the product profile or lower yields. Even a little contamination interferes. Lab veterans know a cross-linked mess in a flask often comes down to a few tenths of a percent of impurity. A 98% grade lets you trust the results and troubleshoot only the experimental factors that truly matter.
Manufacturer Practices and Reasoning Behind Grades
Producers tend to stick with 98% and up for one key reason: demand. Academic labs and pharmaceutical industries order small lots for precision work. No one wants to account for unpredictable contamination, so anything called “analytical reagent” or “high purity” comes through at that 98-99% mark. This grade supports most synthesis protocols where NsCl acts as a protecting group or sulfonylation agent.
Bulk producers could, in theory, offer technical grades with lower purity for non-critical industrial uses. In practice, the cost saving isn’t worth the risk. Handling costs, storage stability, and crushing health hazards from the dust push them to stick with the standard.
Quality Assurance and Verification
Nobody likes surprises at the bench. Suppliers usually supply a certificate of analysis (CoA) detailing how they arrive at the purity figure—usually by melting point, titration, or HPLC. This isn’t just for show. A credible supplier knows labs run their own spot checks and expect results to fall into line with the paperwork. Chain of trust forms between supplier and scientist, and the industry rides on it.
Common Issues and Ways Forward
Purity alone doesn’t solve everything. Some shipments still run into trouble from poor packing, accidental exposure to light or humidity, or prolonged storage. Problems like caking, color change, or clumpy crystals should trigger quality control alarms before you ever crack the bottle open. When a batch falls short, it’s tempting to blame the handler or shipping firm. Still, chemical firms gain their reputations by facing complaints head on—free replacement, technical support, or batch recalls stand as common fixes. Suppliers who keep the lines open with customers tend to prevent small problems from snowballing.
Finding the Best Partner
Experience in the lab shows that sticking with trusted suppliers is worth the premium. Fast responses to CoA requests, transparency in production, and willingness to answer tough questions go a long way. There’s no gold standard enforced by law for NsCl purity, but the trust built between chemist and supplier matches the demands of the modern lab. Choosing wisely means fewer surprises and better science.
Are there any important safety or hazard precautions for 3-Nitrobenzenesulfonyl Chloride?
Working with the Real Risks
If you have ever worked with reagents that seem harsh, you realize fast that some compounds ask for extra respect. 3-Nitrobenzenesulfonyl chloride is one of those chemicals that drives the point home. You open the bottle and pick up a sharp odor right away. People usually grab this compound for sulfonation work or as a protecting group, but it brings plenty of hazards along for the ride.
Skin, Eyes, and Breathing: Handle with Caution
Even the briefest exposure to this compound on your skin can sting. I once splashed a few droplets on my hand by rushing during a transfer. The next five minutes turned into an exercise in flushing hands and feeling your skin itch. The irritation lasts. If it hits your eyes, you risk something far worse: burns, damage, or worse if you can’t rinse fast. Old-timers in organic chemistry always say never to mess around with inadequate PPE. Goggles and gloves matter far more with this stuff than with gentler chemicals.
The fumes tell their own story. 3-Nitrobenzenesulfonyl chloride sends off a sharp, pungent vapor that will burn your throat and nose in no time. Some colleagues try working with it in open air just to “make a quick weigh,” but a hood saves you from that cough and headache. Regular gloves, not the thin disposable kind, reduce risk. I prefer thick nitrile or even double gloving in tough cases. Lab coats and, ideally, a splash shield become necessities rather than nice-to-haves.
Fire and Reactivity: Don’t Play with Ignition
Let’s not forget the reactivity. Like many sulfonyl chlorides, this chemical doesn’t behave well with water. It can bubble up, producing hydrochloric acid and heat. Any spill near a wet surface creates a cloud of corrosive vapor and heat that speeds up damage. And if you ever wondered about fires, the compound itself doesn’t ignite easily, but it gives off noxious gases if burned. This includes chlorine, sulfur oxides, and nitrogen oxides—pretty much a worst-case mix you don’t want to breathe. Planning the cleanup for even a small spill means neutralizing agents and plenty of ventilation.
Safe Storage and Disposal Build Real Credibility
I’ve seen labs make the mistake of storing this stuff in regular cabinets next to acids and bases. 3-Nitrobenzenesulfonyl chloride needs cool, dry, airtight containers and segregation from water, alcohols, and amines. An accidental mixture leads to fast, hazardous reactions that no one wants to clean up. You don’t keep it out for long periods, and every container gets a fresh date and hazard sticker at my bench.
Waste handling goes beyond tossing it in the organic bin. Check with environmental health staff before you think about disposal—this isn’t a drain disposal candidate, ever. Solutions with strong nucleophiles or bases break it down, but you want real chemical waste management for the leftovers because the breakdown products have their own risks.
Training and Habit: The Best Preventive Tool
Years in research teach you that staying safe doesn’t come from the label, but from habit. My team sets aside time every month to walk through handling procedures for these potent reagents. No one assumes the new person knows everything, and every spill, near-miss, and fume hood failure gets logged and discussed. The more you see how easy it is for things to go wrong, the steadier your technique becomes. If you work with 3-nitrobenzenesulfonyl chloride, you owe it to your crew and your own health to give it the respect it demands—every single time.
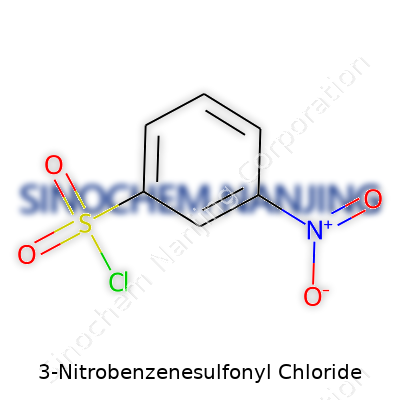
| Names | |
| Preferred IUPAC name | 3-nitrobenzenesulfonyl chloride |
| Other names |
m-Nitrobenzenesulfonyl chloride 3-Nitrobenzenesulphonyl chloride m-Nosyl chloride 3-Nitrobenzenesulfonic acid chloride NSC 16271 |
| Pronunciation | /ˈnʌɪtrəʊˌbɛnˌziːnˌsʌlˈfəʊnɪl ˈklɔːraɪd/ |
| Identifiers | |
| CAS Number | 636-95-3 |
| 3D model (JSmol) | `3D:JSMOL:::ClS(=O)(=O)c1cccc([N+](=O)[O-])c1` |
| Beilstein Reference | 1918987 |
| ChEBI | CHEBI:52187 |
| ChEMBL | CHEMBL3214349 |
| ChemSpider | 79514 |
| DrugBank | DB14055 |
| ECHA InfoCard | 100.015.913 |
| EC Number | 221-002-6 |
| Gmelin Reference | 60487 |
| KEGG | C19268 |
| MeSH | D016227 |
| PubChem CID | 72660 |
| RTECS number | DB8575000 |
| UNII | 7435FI02RB |
| UN number | UN2811 |
| Properties | |
| Chemical formula | C6H4ClNO4S |
| Molar mass | 255.63 g/mol |
| Appearance | White to pale yellow crystalline powder |
| Odor | Odorless |
| Density | 1.64 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 0.67 |
| Vapor pressure | 0.00026 mmHg (25 °C) |
| Acidity (pKa) | -6.5 |
| Basicity (pKb) | 6.92 |
| Magnetic susceptibility (χ) | -49.3×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.611 |
| Viscosity | Viscous liquid |
| Dipole moment | 3.61 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 356.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -53.72 kcal/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1058.8 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin burns and eye damage, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | Hazard statements: H302, H314, H317, H332 |
| Precautionary statements | Precautionary statements: P261, P280, P305+P351+P338, P304+P340, P310 |
| NFPA 704 (fire diamond) | 3-Nitrobenzenesulfonyl Chloride: 2-2-1 |
| Flash point | 120°C (closed cup) |
| Autoignition temperature | Autoignition temperature: 400 °C |
| Lethal dose or concentration | LD₅₀ (oral, rat): 636 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 500 mg/kg |
| NIOSH | TB6650000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 10 mg/m³ |
| Related compounds | |
| Related compounds |
Benzenesulfonyl chloride 4-Nitrobenzenesulfonyl chloride Methanesulfonyl chloride Tosyl chloride 3-Nitrobenzenesulfonic acid |

