3-Nitrobenzenearsonic Acid: A Down-to-Earth Commentary on Origins, Properties, and Impact
Historical Development
Farmers and chemists have wrestled for over a century with the challenge of increasing crop yield and managing animal health. In the early 1900s, researchers started exploring organic arsenic compounds, chasing new ways to guard against parasitic infections in livestock and to boost growth. The story of 3-nitrobenzenearsonic acid, better known on farms as roxarsone, grew out of these experiments. Its development wasn’t accidental. The demand for more efficient poultry production pushed scientists to develop feed additives that delivered results. Regulatory bodies didn’t overlook its introduction; early researchers flagged both the benefits for animal health and potential worries about arsenic accumulation. These dual themes followed the compound into modern times, shaping both acceptance and skepticism among producers, regulators, and the communities where these farms operated.
Product Overview
Walk into a feed mill supplying large-scale poultry operations, and you’ll find 3-nitrobenzenearsonic acid among the specialty additives once mixed into rations. The compound’s key selling point came from its ability to promote weight gain and control specific intestinal parasites in chickens. It’s often mistaken for other forms because of the “arsenic” in the name, but this synthetic organic molecule behaves differently from elemental arsenic or inorganic salts. Decades of use in commercial agriculture fixed its reputation as a serious tool to “finish” birds quickly for market. Yet, society’s growing focus on sustainable food and chemical residues also led to phased bans and restrictions across countries as researchers highlighted persistence and environmental impact.
Physical & Chemical Properties
3-Nitrobenzenearsonic acid comes as a yellow to orange crystalline powder. Anyone who’s mixed it by hand remembers its bitter, acrid odor lingering even after gloves came off. Solubility matters on the farm, and this compound dissolves more readily in hot water than cold, which helped producers blend it effectively into feed mixes. Chemically, this molecule packs a nitro group, an arsonic acid group, and a benzene ring—a combination that shapes both its biological role and environmental persistence. Heating or acidic conditions can sometimes break it down, but under normal storage it stands up to time and doesn’t react with air or standard feed components.
Technical Specifications & Labeling
Bags of 3-nitrobenzenearsonic acid carry technical data because dosing leaves little margin for error. Industrial suppliers publish purity percentages, moisture content, and assay results—figures that don’t just live on paper. The bulk of the product must contain a high percentage of the active compound, often above 98%. Labels note the recommended inclusion rates, safe handling instructions, and regulatory warnings. The product’s shelf life also appears on documentation, with warnings about exposure to sunlight or high humidity. Certification of origin, batch numbers, and hazard statements underscore the need for robust supply chain management. Feed formulators live with these numbers because over- or under-dosing triggers both regulatory penalties and animal health disasters.
Preparation Method
The manufacture of 3-nitrobenzenearsonic acid follows a route thought up in classic organic synthesis labs. Workers start with nitrobenzene, using a sequence of reactions that attaches the arsonic acid group to the aromatic ring. The key steps involve sulfonation and arsenation, followed by neutralization and crystallization. This process doesn’t just depend on chemistry—it requires close attention to reaction temperatures, timing, and pH. Technicians learned from hard experience that tiny changes could swing yields up or down, and poor purification left behind toxic residues. Quality control starts on the production floor and tracks each batch through extensive analytical checks before anything ships out.
Chemical Reactions & Modifications
On the lab bench, 3-nitrobenzenearsonic acid resists many simple chemical attacks but changes character under reducing conditions. Treatment with common reducing agents can strip the nitro group, yielding amine or azo derivatives. Environmental scientists got interested in these transformations, looking for clues about breakdown products in soil and water. Feed compounders have also looked at tweaking the molecule to adjust activity or reduce residue risks, but these modifications often hit roads blocked by both chemistry and regulation. Nearly every change meant a fresh round of safety and bioavailability studies, limiting commercial enthusiasm for new analogues.
Synonyms & Product Names
Out in the field or in the scientific literature, the same molecule appears under a tangle of names. Farmers know roxarsone. Chemists write 4-hydroxy-3-nitrophenylarsonic acid. Sometimes “AS-286” pops up, or plain “3-Nitrobenzenearsonic acid.” Regulatory documents may refer to its CAS number or European feed additive codes. This mess of names comes in handy for tracing studies on health, toxicity, and regulations. Different industries favor different words, but anyone following debates on arsenical feed additives recognizes each of these labels as talking about the same core problem and potential.
Safety & Operational Standards
Safe handling never stops mattering with arsenic compounds. Workers at formulation plants keep to strict industrial hygiene—dust masks, gloves, and protective clothing stand as standard kit. Regulatory agencies keep manuals of safe operation and emergency spill procedures, and most companies drill these regularly. Storage matters too: dry, cool, and away from incompatible chemicals. Waste disposal rules reflect the lingering environmental risks, requiring specialized landfills or incinerators. Human exposure, even at low levels, draws scrutiny, and any sign of health issues sparks investigation. Proper record-keeping and access control limit risks both to employees and the broader ecosystem.
Application Area
In the heyday of its use, 3-nitrobenzenearsonic acid surged through poultry and swine operations, where it played a dual role in disease control and growth promotion. Intensive animal production benefited from faster “finishing” times, and the drug often lined up alongside other feed antibiotics. Some aquaculture and rabbit operations experimented too. In each case, the main draw stayed the same: healthier animals and improved feed conversion rates. Regulations started pulling the plug on these uses, but the legacy remains—trace residues appear in older facilities and sometimes in irrigation run-off. The environmental questions nobody could ignore have left their mark on management practices in animal agriculture.
Research & Development
Scientists chase answers to lingering questions about both the compound’s fate and the alternatives that might someday replace it. Researchers dig into soil samples, water run-off, and chicken tissue, probing for the paths arsenic residues follow as they cycle through the environment. Some look at genetic impacts in poultry and resistance patterns in intestinal parasites. Other teams focus on detection methods, seeking faster, cheaper assays for regulatory enforcement. Less common—yet growing—are efforts in biotechnology labs to develop less toxic growth promoters, sparked by the environmental debates around arsenic-laden feed. Real innovation means looking at nutrition, animal welfare, and sustainability in new ways.
Toxicity Research
Toxicologists and epidemiologists pay close attention to the transformation of 3-nitrobenzenearsonic acid once it enters the animal and the environment. Studies show that, inside the gut, the compound can convert to inorganic arsenic forms, which pose well-documented health risks. Chronic exposure, even at low levels, has been linked to higher cancer rates and developmental issues in both animals and humans. Tracking actual exposure is tricky, given the variable breakdown rates in different soil and water conditions. Regulatory agencies in the United States, European Union, and Asia continue to set and revise maximum contaminant levels based on these studies. Community health advocates trace arsenic levels around major production sites as part of ongoing health monitoring.
Future Prospects
Sitting at a crossroads of policy, science, and agriculture, the story of 3-nitrobenzenearsonic acid remains unfinished. Regulatory bans in many countries push the feed industry to look hard at replacements that deliver growth promotion and disease control without the baggage of arsenic. Biotech firms and nutritionists are testing compounds that claim lower toxicity and fewer ecological side effects. Pressure mounts from consumers demanding traceability and safety in food products. Regions without strict bans still rely on traditional practices, but global food supply chains build pressure for harmonized standards. The big question for tomorrow is how to balance production needs with public health and environmental responsibility—one that takes more than single-molecule fixes, but the full measure of science, policy, and practical farming know-how.
What is 3-Nitrobenzenearsonic Acid used for?
The Role in Animal Feed Additives
Farm life easily takes its toll on animals, especially poultry. Over the past decades, 3-nitrobenzenearsonic acid walked a straight path into animal feed, specifically for chickens and turkeys. This compound, known in the feed trade as “3-Nitro” or “Roxarsone,” promised to encourage steady weight gain and combat common gut infections. This approach helped farmers deliver more chicken to grocery shelves, letting them keep costs predictable and flocks healthy.
Scientists took this compound from the chemistry table and put it into practice across numerous industrial farms. The idea seemed solid: birds stayed healthier, flocks reached market weight on schedule, and losses dropped. To people who have seen the day-to-day grind of livestock, the reliability offered by this additive looked like a small miracle.
Health, Safety, and Environmental Risks
The shine wore off as evidence stacked up. Studies started flagging arsenic residues in both poultry meat and the environment. Arsenic in any form isn’t something you want in your diet or drinking water. While this specific form of arsenic proved less toxic than others, years of bird droppings and farm runoff pushed these residues into fields, then creeks, and eventually water tables.
Food safety regulators took this information seriously. The United States Food and Drug Administration (FDA) ran its own tests and found low but measurable levels of inorganic arsenic in meat from 3-Nitro-fed birds. While the immediate risk looked small, the idea of years of daily arsenic exposure—especially on a population level—raises real health flags. Heart disease, cancer, and other conditions hang in the balance when it comes to even small levels of arsenic.
Regulatory Response and Safer Alternatives
In response, regulatory shifts started picking up pace. Beginning in 2011, the main manufacturer in North America voluntarily pulled the product from the market. Many countries have since banned or heavily limited the use of arsenical feed additives out of concern for both human and environmental health.
Experience on the farm didn’t lie—removing growth promoters required farmers to sharpen up their management. Better biosecurity, cleaner housing, more precise nutrition, and vaccines now do much of the heavy lifting that “3-Nitro” once shouldered. Investments in these areas cost more upfront, but they reduce long-term risks, protect natural resources, and keep consumer trust on stable ground.
The Future of Animal Farming
The story of 3-nitrobenzenearsonic acid isn’t just about chemistry textbooks or regulatory boards. It’s about food, sustainability, and the choices we make for future generations. Keeping our farms productive without shortcuts that come back to haunt us means leaning into transparency, innovation, and a real openness to critique and improvement. Farmers, scientists, regulators, and eaters all share this load.
What are the safety precautions when handling 3-Nitrobenzenearsonic Acid?
Understanding the Dangers
3-Nitrobenzenearsonic acid already sounds like something to stay wary of, and experience backs that instinct. This compound often finds its place in research labs or old agricultural setups. The reality is, it lands solidly among substances carrying significant hazards. The arsenic alone gives pause. It holds a reputation for toxicity—chronic exposure links up with nerve trouble, kidney damage, and even heightened cancer risks. Toss nitro groups into the mix and you start stacking potential health impacts like irritation or worse. I learned early during my own time in university chemistry labs: no shortcut ever pays off when real poison is sitting on the bench.
PPE: The Barrier Between You and Harm
Lab coats should be buttoned, glove cuffs pulled tight. Here, nitrile gloves work best—better resistance to chemicals than old-school latex. Eyes stay protected with wrap-around goggles, not just regular specs. Don’t let bare skin risk it; closed-toe shoes and long pants are just common sense when handling reagents like this. Let’s be honest—any seasoned lab worker knows nobody wants the story of a splashed glove or stained sleeve to become personal.
Managing Dust and Fumes
Dust drifts, fumes linger—tiny exposures snowball over time. I always run the fume hood fan, regardless of how “quick” the procedure. Lab ventilation fans hum for a reason. A whiff isn’t harmless when it’s an arsonic acid. The room never smells sweet. Using local exhaust and keeping containers sealed goes far beyond ticking boxes on a safety checklist. It means breathing easy at home later, too.
Storage Realities
Find 3-nitrobenzenearsonic acid a sealed, cool, ventilated spot, nowhere near flammable solvents or strong reducing agents. I’ve seen people stash dangerous chemicals under fume hoods—bad practice. Isolate toxins and label everything. Label fading? Replace it. Not knowing what sits on the shelf causes headaches, spills, and sometimes worse.
Dealing with Spills and Exposure
Accidents will happen, even with best intentions. Spill kits should live within reach, including absorbents and neutralizing agents—no hunting across the building. Skin contact, even a splash, means water—lots of it. Eyes? The eyewash station beats tap water every time. Safety showers save more than just the skin, and I’d rather feel cold and wet than regretful for life.
Waste: Out of Sight, Not Out of Mind
It feels tempting just to rinse glassware down the drain, but toxins like this don’t vanish; they return in groundwater or rivers. Collection bottles get labeled and handed to hazardous waste specialists. Environmental risks extend past the workplace. The old hands I worked with always treated chemical waste seriously, and their methods stuck with me.
Building Habits, Not Just Rules
Safety grows from habits and stories more than from printed signs. Every moment saved by skipping a step can bring years of harm. Share close calls openly in the lab. Talk mistakes. Encourage a culture where everyone double-checks PPE and asks if someone missed a glove or forgot their goggles. That’s the real shield against accidents—shared vigilance, not just a safety manual gathering dust.
What is the chemical structure and formula of 3-Nitrobenzenearsonic Acid?
Chemical Makeup of 3-Nitrobenzenearsonic Acid
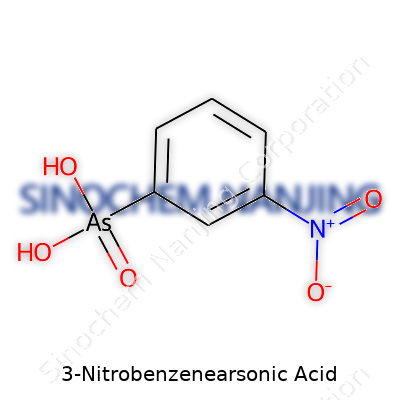
Chemistry rarely gets the appreciation it deserves outside laboratories and classrooms. One compound that’s been on the lips of more researchers than dinner-table guests is 3-nitrobenzenearsonic acid. Its chemical formula is C6H6AsNO6. Shaped by a six-carbon benzene ring, the structure includes an arsonic acid group (–AsO3H2) and a nitro group (–NO2).
Swapping simple words for what happens inside a lab, think of a benzene ring set up with one arsonic acid group attached at the first carbon. Move over to the third spot on the ring, and you’ll see the nitro group. Chemists write it down as 3-nitrobenzenearsonic acid, but the real story unfolds in its structure: each group sends the ring in different directions for reactivity and application.
Why Bother with the Details?
There’s something personal about digging into molecular architecture. As a student who wrestled with organic chemistry in late-night study sessions, those diagrams often seemed cryptic. But behind every molecule lies a purpose. The combination of arsenic and nitro groups sounds a warning bell. In the past, livestock feed additives explored forms of benzenearsonic acids for their growth-promoting effects. This stuff never belonged in your kitchen, but its grip on agriculture stuck around long enough to warrant scrutiny.
The nitro group compounds—often flagged for their strong electron-withdrawing properties—typically increase the compound’s reactivity. Life experience teaches that increased reactivity sometimes brings risks. When I read discussions about environmental persistence, I remember the stories that unveil in polluted water or soil near animal farms. Compounds like this, with stable aromatic rings and arsenic connections, don’t just vanish after a rainstorm. They can end up in food chains, echoing concerns raised by toxicologists and environmentalists.
Real-World Troubles and Paths Forward
When turning over the soil of a chemical like 3-nitrobenzenearsonic acid, people tend to ask about its risks. Toxicity profiles for arsenic compounds spell out the health stories we wish to avoid—neurological harm, cancer risks, water contamination. Agencies like the EPA have dug through these facts and set limits for compounds with arsenic, and that vigilance is necessary. History’s grip on arsenic’s reputation doesn’t fade quickly. Used carelessly, compounds like these have left scars worldwide, from mining to medicine.
Regulation handles part of the puzzle, but that’s not the end. Science pushes for greener alternatives. Labs, funded by public and private money, chase after molecules with the same utility but lower toxicity. Knowledge, in my view, builds on mistakes. Since high-school chemistry, I learned no substance stands apart from context. Today, transparency in chemical use matters as much as creativity in synthesis or analysis.
Trust, Transparency, and Responsible Use
With this compound, and any new one on the horizon, open discussion between scientists, regulators, consumers, and farmers drives better choices. Reliable studies let people judge risks without guesswork. Sharing structures, formulas, risks, and benefits openly shapes how industries work and how communities stay healthy. Every molecular story ties back to trust in science and the choices made on the ground.
Understanding compounds like 3-nitrobenzenearsonic acid isn't just a matter for researchers. It’s about how much safety people deserve and how science has to adjust when risks outweigh rewards. Honest discussions push the field (and all of us) forward.
How should 3-Nitrobenzenearsonic Acid be stored?
The Nature of 3-Nitrobenzenearsonic Acid
3-Nitrobenzenearsonic acid may not ring a bell for folks outside of a chemistry lab, but it’s shown up in animal nutrition research and industrial applications. The substance blends organic chemistry and arsenic—a pairing that brings both usefulness and real caution. This stuff doesn’t belong anywhere near kitchen shelves or general stockrooms. People who work with it should respect its hazards as much as its uses.
What Makes Storage Critical
I remember working in a research building where labels carried more weight than any warning sign. Some chemicals look innocent—white powders that sit in glass jars for months. A mistake in handling isn’t always loud or dramatic, but a lapse in judgment with something containing arsenic can have lasting consequences. 3-Nitrobenzenearsonic acid releases hazardous dust and fumes if mishandled, and long-term exposure spells trouble for the lungs, liver, or skin—even at low doses.
Regulatory agencies, like OSHA and the EU’s REACH program, set strict storage protocols for chemicals such as this one. These aren’t based on bureaucracy for the sake of it; they reflect real-world incidents where lives and environments suffered due to sloppy handling.
Down-to-Earth Storage Guidelines
It’s tempting to pick any empty shelf and slap a “Hazardous” sticker on a bottle, but that’s not enough for something with arsenic. The best place for 3-nitrobenzenearsonic acid is a dedicated chemical storage cabinet—preferably one built for toxic substances, featuring lockable doors and spill trays. No food, drinks, or unrelated items should share this space, since cross-contamination can happen just from a poorly sealed container.
Every chemical reacts differently to light, temperature, and humidity. I’ve found that heat can speed up decay and reactions, while moisture invites clumping or dangerous transformations. This compound does well in a cool, dry spot, shielded from sunlight and direct heat—think of a temperature range that matches a well-air-conditioned office, not a drafty warehouse corner. Sealing the lid tightly after each use keeps escape routes closed, which matters a lot with small, invisible particles.
Lab managers sometimes forget the importance of inventory logs and safety sheets, especially with low-turnover materials. These records become lifesavers during spills, accidents, or inspections, helping trace where things went wrong. Everyone with access to hazardous compounds should know emergency procedures, from eyewash station locations to what cleanup kit matches the chemical on the shelf.
Reducing Risk and Finding Solutions
Personal experience says that relying on one person to remember all storage details doesn’t work. Regular safety audits—unannounced and thorough—often reveal an expired jar or a misfiled reagent that could cause a mishap. Secure labels in bold, legible print and secondary containers for transportation cut down on risks, especially for junior staff or those new to the chemical.
Training new workers in real-life scenarios—not just paperwork—changed how our lab approached safety. Practicing what to do in a spill with harmless powder mimics helped build muscle memory. This kind of preparation, along with a strong focus on clean storage, keeps not only the chemical contained but also the people and spaces safe.
No shortcut replaces a strong routine. Good habits, clear documentation, and reliable equipment shape the difference between safe storage and disaster. For something like 3-nitrobenzenearsonic acid, safe habits are the only choice.
What are the possible health hazards associated with 3-Nitrobenzenearsonic Acid?
What Is 3-Nitrobenzenearsonic Acid?
3-Nitrobenzenearsonic acid, sometimes called Roxarsone, pops up mostly in animal feed, especially for poultry. It has a complicated reputation. For some years, farmers have used it to boost animal growth and make meat look pinker. The chemical has roots going back to the 1940s, before folks had much information about long-term risks.
Risks Linked To Arsenic Compounds
My time around old farm equipment and dusty feed bins taught me not to underestimate any chemical with arsenic in its name. Organic arsenic compounds, including this one, don’t linger in headlines as much as lead or mercury, but trouble often comes quietly. The human body struggles to deal with even small traces of arsenic. Over time, it builds up in organs like the liver and skin, opening the door to a bunch of health problems.
Studies from the National Institutes of Health have linked arsenic exposure with cancers of the bladder, lung, and skin. Chronic low-level exposure plays a role in heart problems and diabetes too. Kids, pregnant women, and older adults face even bigger risks because their bodies process toxins differently or less efficiently. Some scientists raised alarms after finding higher arsenic levels in people living near large-scale poultry operations.
Occupational Exposure & Environmental Concerns
Anybody handling raw 3-Nitrobenzenearsonic acid or feed laced with it faces real risks. Skin contact can lead to redness, peeling, and rashes. Inhaling the dust stings the throat and nose, causing headaches, dizziness, and breathing issues. Workers need solid training and personal protective gear, but in my experience, farm workers sometimes receive bare-minimum instruction or outdated gear. Even a short gap in safety can leave a lasting mark.
Farm runoff spreads this chemical into water and soil. Over the years, small leaks and spills add up. The real shame comes when these arsenic residues end up in groundwater, wells, or nearby streams, reaching people outside the industry. Animals, too, suffer silent effects: lowered immune systems and strange growth patterns.
Monitoring, Substitution, and Better Practices
Health authorities like the World Health Organization and the U.S. Food & Drug Administration have started limiting or phasing out use of organic arsenic compounds in livestock. That’s a positive move, but older stocks sometimes pop up at smaller or unregulated farms. Monitoring programs, regular environmental testing, and better education can go a long way in closing these loopholes.
Substitutes exist. Alternatives such as probiotics and approved minerals can help boost animal growth and help farmers avoid chemicals that carry hidden costs down the line. Part of that switch comes from consumer demand, but it also depends on strong outreach and fair prices so small producers aren’t left out.
Personal Choices & Community Roles
People in rural communities deserve fair warning when potential hazards like 3-Nitrobenzenearsonic acid circulate nearby. Local news and agricultural extension offices can play a big role—at least in my part of the world, neighbor-to-neighbor information spreads faster than government bulletins. Knowing what’s in your water and what’s drifting in the dust makes a difference. Health comes down to more than lab numbers and regulatory limits—it’s also about looking out for each other and keeping the conversation going until better habits take root.
| Names | |
| Preferred IUPAC name | 3-Nitrobenzenearsonic acid |
| Other names |
3-Nitroarsonic acid m-Nitrobenzenearsonic acid m-Nitrobenzene arsonic acid 3-Nitrophenyl arsonic acid |
| Pronunciation | /ˌtraɪˌnaɪtroʊˌbɛnˈziːnˌɑːrˈsɒnɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | 98-88-4 |
| Beilstein Reference | 2039470 |
| ChEBI | CHEBI:52232 |
| ChEMBL | CHEMBL3212438 |
| ChemSpider | 20594 |
| DrugBank | DB14000 |
| ECHA InfoCard | 100.004.107 |
| EC Number | 205-571-1 |
| Gmelin Reference | 95274 |
| KEGG | C18606 |
| MeSH | D009634 |
| PubChem CID | 10464 |
| RTECS number | CH1600000 |
| UNII | 86Q358L86E |
| UN number | UN1660 |
| Properties | |
| Chemical formula | C6H6AsNO5 |
| Molar mass | 247.07 g/mol |
| Appearance | White to light yellow crystalline powder |
| Odor | Odorless |
| Density | 1.8 g/cm3 |
| Solubility in water | Soluble in water |
| log P | -0.1 |
| Vapor pressure | 2.14 x 10^-7 mmHg (at 25°C) |
| Acidity (pKa) | 1.63 |
| Basicity (pKb) | 3.88 |
| Magnetic susceptibility (χ) | -70.7·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.6600 |
| Dipole moment | 2.23 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 204.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -229.6 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -1261.6 kJ/mol |
| Pharmacology | |
| ATC code | QH04BA01 |
| Hazards | |
| Main hazards | Toxic if swallowed. Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS07, GHS08 |
| Pictograms | GHS07,GHS09 |
| Signal word | Danger |
| Hazard statements | H302, H312, H332, H351 |
| Precautionary statements | P201, P202, P260, P264, P270, P273, P280, P308+P313, P314, P391, P405, P501 |
| NFPA 704 (fire diamond) | 3-3-2-oxidizer |
| Explosive limits | Lower: 0.13%, Upper: 1.9% |
| Lethal dose or concentration | LD50 oral rat 4100 mg/kg |
| LD50 (median dose) | LD50 (median dose): 930 mg/kg (rat, oral) |
| NIOSH | CY9625000 |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | 0.002 mg/m3 |
| IDLH (Immediate danger) | IDLH: 25 mg/m³ |
| Related compounds | |
| Related compounds |
Arsanilic acid p-Aminophenylarsonic acid Roxarsone Nitrobenzene 3-Nitrobenzoic acid |

