3-Nitroaniline: An In-Depth Commentary
Historical Development of 3-Nitroaniline
Long before the digital age, chemists set out to map the world of aromatic amines and nitro compounds. Among these, 3-nitroaniline emerged as a distinct player in the late 19th and early 20th centuries. The compound’s appearance in scientific papers came as dyes and explosives grew crucial to industry. Chemists learned to build 3-nitroaniline through a careful process of nitration, taking ideas that shaped aniline dye culture and merging them with the explosive science blossoming in Europe. By the middle of the 20th century, this compound found roles in both the textile and pharmaceutical sectors, thanks to early pioneers chasing more than color—they aimed for new molecular skeletons to anchor further chemical reactions. That appetite for molecular diversity fueled the compound’s evolution from academic curiosity to a key industrial staple.
Product Overview
3-Nitroaniline, recognized by the formula C6H6N2O2, walks a line between raw industrial ingredient and building block for more complex synthetic efforts. Suppliers typically offer this compound as a yellow crystalline solid, useful for small-scale laboratory research and large-volume applications. The value of this compound reveals itself across a spectrum of industries: synthetic dyes, veterinary pharmaceuticals, and sometimes as an intermediate for photographic chemicals. Over countless batches and years, this material proved reliable for introducing both nitro and amino functionality into larger chemicals. Its versatility speaks to a chemical economy that never stands still.
Physical & Chemical Properties
Many who work with 3-nitroaniline find the faint yellow hue familiar, with crystals that melt at around 114–116°C. While not overly volatile, its powder can be a respiratory irritant. The compound dissolves modestly in hot water, much more willingly in common organic solvents like ethanol, acetone, and ether. Its moderate solubility matches up well with typical lab demands—enough to work into solution, resistant to loss through accidental spills. On the chemical level, the presence of nitro and amine groups controls its every reaction. That dual-functionality lets chemists steer the compound towards reduction, substitution, or coupling reactions. The electron-withdrawing nitro group sits meta to the amino group, influencing reactivity at other ring positions.
Technical Specifications & Labeling
In a lab or factory, standard material labels demand clarity: chemical name, purity, batch/lot number, hazard symbols, and supplier information. Certified batches must often exceed 97% or even 99% purity, with trace contaminants—like moisture or by-product isomers—kept minimal. Manufacturers list melting point ranges, molecular weights, and spectral data such as IR or NMR fingerprints. This content gives both reassurance and accountability, ensuring everything leaving a shipping dock meets strict performance metrics. From my own experience, meticulous labeling cuts down on costly errors, especially when dangerous or sensitive procedures depend on known composition and stability.
Preparation Method
Most commercial 3-nitroaniline follows a nitration route, starting with acetanilide or directly from aniline through controlled addition of nitric and sulfuric acids. This reaction requires precise temperature regulation—too hot, and unwanted isomers dominate; too cold, and yield drops off. Isolation calls for careful washing and crystallization. Modern operators might switch up the acids or adopt continuous-flow systems for safety and efficiency. Across the years, bench chemists often learned the hard way—quality in equals quality out, and small changes in protocol affect both yield and impurity profile. Regular analytical checks let manufacturers avoid dangerous dinitro or undesired para-substituted byproducts.
Chemical Reactions & Modifications
The chemical action around 3-nitroaniline draws from its two major groups. Reduction turns the nitro function into another amino group, allowing moves toward diamines. Electrophilic aromatic substitution on the ring remains feasible with careful selection of reagents, but reactivity drops because the nitro group’s strong deactivation. Researchers rely on diazotization to decorate the ring or form azo dyes. Each functional handle has its use—some processes count on selective transformations, while others push both groups to react in tandem. Over time, the growing library of modified 3-nitroaniline derivatives fed into dye, pigment, pharmaceutical, and agricultural chemical discovery.
Synonyms & Product Names
Aside from “3-nitroaniline,” you might see this compound under a handful of names: meta-nitroaniline, m-nitroaniline, C.I. 37130, or 1-amino-3-nitrobenzene. Each label reflects either structural placement or commercial context. The dye industry, especially, likes shorthand like “C.I.” numbers, whereas fine chemical catalogs stick to IUPAC logic. These naming quirks matter most in global trade or legacy synthetic protocols that still cite older texts.
Safety & Operational Standards
Anyone handling 3-nitroaniline in the workspace cannot ignore the real hazards. Dust inhalation often irritates respiratory tracts; long exposure brings risk of skin sensitization. Contact with open wounds raises the risk of systemic toxicity. Standard PPE—gloves, goggles, and often fume hoods—becomes non-negotiable, especially at scale. MSDS documents stress emergency response: in case of skin or eye contact, immediate flushing with water, and alerting medical teams for more serious exposures. Industries must store this powder in tightly closed, labelled containers in cool, ventilated rooms. I’ve seen labs lose valuable time to minor lapses in protocol—small spills or poorly closed vessels turn minor problems into costly shutdowns. Regulatory agencies like OSHA and REACH set clear benchmarks here, with penalties for ignoring safe handling.
Application Area
The colorant trade pulls in the bulk of global 3-nitroaniline production, since it acts as a precursor for both acid and basic dyes. Beyond textiles, the pharmaceutical industry explores analogs for antimicrobial and anti-inflammatory agents. Agrochemical researchers found 3-nitroaniline derivatives useful for custom herbicides and growth regulators. Photographic chemical streams depended on aromatic nitro compounds for decades as developers and stabilizers. Chemists committed to novel materials synthesis value it for accessing harder-to-reach functionalized rings. Over the past decade, its application spread through R&D in organic electronics, where aromatic nitro derivatives influence charge transport or color response.
Research & Development
Labs worldwide keep pushing new uses and safer handling of 3-nitroaniline. Medicinal chemists design new drug leads based on the core structure, looking for ways to dampen toxicity while keeping biological activity. Green chemistry efforts explore less polluting and more energy-efficient nitration pathways, using milder oxidants or ionic liquids to cut down dangerous waste streams. Teams studying high-performance organic coatings experiment with 3-nitroaniline-derived polycondensation agents, aiming for improved durability and self-healing properties. As competition ramps up, producers develop better purification columns and process sensors, reducing worker exposure and boosting manufacturing consistency. My own work with process safety engineers taught me: refining production isn’t just about output, but about preventing legacy health problems that shadowed earlier generations.
Toxicity Research
Studies over decades show 3-nitroaniline can cause acute toxicity through ingestion, skin absorption, or inhalation. Symptoms range from cyanosis and headache to convulsions at higher doses, thanks to its ability to form methemoglobin and disrupt oxygen transport. Animal testing underscores the need for exposure limits; chronic exposure triggers liver and kidney stress, while some data hint at possible mutagenic action. The compound’s environmental persistence also causes concerns—soil and aquatic tests suggest partial bioaccumulation and slow breakdown. Industrial engineers collaborate with toxicologists to monitor air and water emissions tied to 3-nitroaniline to curb dangerous levels in workplace and nearby communities. Regular medical monitoring for factory workers helps catch health effects before they escalate, a lesson hard-earned by earlier decades of less-regulated manufacturing.
Future Prospects
Today’s chemists and engineers rethink the lifecycle of 3-nitroaniline, chasing routes that balance performance and reduced impact. Interest grows around catalytic hydrogenation and bio-based alternatives for both feedstock and waste cleanup steps. Emerging application fields—such as organic solar cells or bioactive scaffold materials—demand both technical performance and proof of safety. Analytical teams deploy in-situ sensors and real-time data logging to improve quality and worker protection. Environmental regulators signal stricter controls on nitroaromatics, driving substitution efforts and better remediation strategies. The story of 3-nitroaniline points toward a future built on transparent supply chains, smarter process automation, and leadership from R&D offices that put both workers and surrounding communities at the center of progress.
What are the main uses of 3-Nitroaniline?
Looking Past the Lab Bench
3-Nitroaniline sounds like something you'd only spot on a cluttered chemistry shelf, but this yellowish compound pulls weight in several areas. I first heard about it during a college project. My team tried to synthesize azo dyes for textiles, and our instructor said, “3-Nitroaniline isn’t just a reagent on paper—it winds up in everyday colors you see around you.” Turns out, he was right. Take a look at a brightly dyed cotton shirt or a vivid paint on a classroom wall. There’s a chance 3-nitroaniline played a role.
Big Splash in Dyes and Pigments
One of the main jobs for 3-nitroaniline is as a starting material for dyes. That world—textile, inks, certain plastics—leans on complex molecules to create shades that last. This compound contains both nitro and amine groups, which lets it react with lots of other chemicals. Factories often rely on it to make azo dyes, which bring out that saturated yellow, orange, or red in natural and synthetic fibers.
It doesn’t stop there. Pigment makers take advantage of 3-nitroaniline for manufacturing colors used in plastics, rubbers, and coatings. It's less about being flashy and more about reliability. These colors hold up in sunlight, stay bright after washing, and meet regulations set for public safety. The textile industry doesn’t gamble with the stuff that lands on our skin, so safety and testing back up its use.
Journeys Down the Pharmaceutical Route
Anyone who has spent time flicking through research journals or chemical supplier catalogs will spot 3-nitroaniline as a “key intermediate.” Drug development is one of those fields where even tiny changes to a molecule can mean something works or fails in the human body. Chemists use 3-nitroaniline to build more complex compounds, some of which end up as active ingredients in pain pills or antibiotics. It offers a simple scaffold to stack on different side chains, making it valuable for medicinal chemistry.
There’s always a big push for safer processes in drug production. Using 3-nitroaniline, with its track record in controlled reactions, helps researchers avoid dangerous byproducts. I’ve seen regulators increasingly press manufacturers not just for proof that drugs work, but that nothing toxic lingered in the process.
Specialty Chemical Synthesis
Sometimes you work with niche chemicals that have hidden impacts. 3-Nitroaniline’s properties let it react in creative ways, giving access to antioxidants, corrosion inhibitors, and specialty materials. Companies making engine oils, adhesives, or brake fluids look for molecules that keep machines running smoothly. Often, teams engineer backbones derived from 3-nitroaniline, aiming for answers when heat or pressure would break simpler stuff down.
Safety Matters and Paths Forward
This chemical isn’t something to handle without care. It’s toxic by inhalation or skin contact, and some countries track its use closely. There’s always that question: can we find greener or safer alternatives while keeping performance high? Research groups keep exploring less hazardous routes. Some start with other amines or pursue bio-based sources. Banning well-known intermediates can slow down progress, so most experts call for stricter handling and better worker training.
From a practical angle, industries shift toward closed systems and recycling solvents where 3-nitroaniline appears, not just to cut costs but to protect people and the environment. This approach has already made many workplaces safer in the past decade.
In my experience, the story of 3-nitroaniline speaks to chemistry’s central trade-off. Use the right tools to get the job done—just never lose sight of what’s safe for both people and the planet.
What is the chemical formula and structure of 3-Nitroaniline?
Breaking Down 3-Nitroaniline
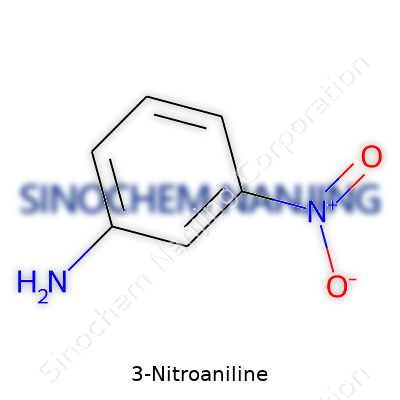
3-Nitroaniline draws attention for its role in making dyes, pigments, and chemicals that are hard to miss in everyday life. This compound’s name already spills much of its story—an aniline backbone paired up with a nitro group. In simple terms, this molecule has a benzene ring showing off two attachments: an amino group (-NH2) at the first spot, and a nitro group (-NO2) at the third, or meta, position. Its chemical formula is C6H6N2O2.
Peeking at the Structure
The benzene ring provides a familiar six-carbon skeleton that hosts an amino group at position one and a nitro group three carbons around the ring. Imagine the ring as a clock. Placing the amino group at "12 o’clock" puts the nitro group at "3 o’clock." This relative positioning shapes several physical and chemical traits. For many lab workers and chemistry students, sketching this out serves as one of the first exercises in organic chemistry—drawing each group linked to the carbon ring and realizing the subtle difference a few carbons’ distance can make.
Why Where the Groups Go Matters
Position makes all the difference. The nitro group in the meta position changes how the molecule behaves compared to its ortho and para cousins. This meta layout affects electron flow through the ring, which steers reactivity, color, and safety risks. For example, the meta position slows certain kinds of reaction, especially those involving the amino group, by pulling electron density away from other reactive spots.
Industrial and Academic Importance
Labs across the globe use 3-Nitroaniline to build azo dyes and pharmaceutical ingredients. Specialty pigments in industries like textiles or printing depend on this starting material. Its color, usually a bright yellow, comes straight from the way its structure messes with light. While people might not give much thought to molecules behind pigments or drugs, this chemistry keeps businesses running and classrooms busy.
On a personal level, I spent some of my graduate years wrangling aniline derivatives, learning quickly to respect their strong smells and persistent yellow stains. Breathing their fumes is best avoided, and good gloves are necessary. The compound offers an introduction to tricky organic syntheses, showing beginners what it means to guide reactions by moving groups around a benzene ring.
Risks and Safer Handling
Exposure brings health worries, especially when handled in bulk. The nitro group means risk of toxic effects if these compounds are inhaled or absorbed. Businesses and educators who use this chemical should enforce hood use, gloves, and regulatory guidance found in material safety data sheets. Outdated storage or labeling practices heighten accident risk, prompting calls for regular audits and training. Anyone working there sees how small changes in procedure can keep people from harm. Proper disposal is just as crucial, with environmental agencies watching for nitroaromatic waste in water and soil.
Paths Toward Safer Chemistry
Institutions seek new routes for making dyes and drugs with fewer hazards or greener byproducts. Research backs up using less hazardous reagents or smaller amounts when possible, and that kind of progress often grows from lessons learned handling legacy chemicals like 3-Nitroaniline. Grad students, technicians, and supervisors alike learn to respect not only the chemistry but the real-life risks, thereby nudging companies and schools closer toward safer routines and better environmental outcomes.
Is 3-Nitroaniline hazardous or toxic?
Looking Beyond the Lab Bench
Walk through any industrial chemical storage and you’ll likely see containers marked 3-Nitroaniline tucked between dyes and intermediates. It’s got the scientific flare in the name, but underneath, this powder isn’t just another number on a safety sheet. In college, I sat through more than one seminar on aromatic amines, and 3-Nitroaniline always grabbed attention for a reason: it’s not something you take lightly, no matter how comfortable you get around glassware.
Diving Into the Hazards
Most folks dealing with 3-Nitroaniline know its yellowish crystals show up in manufacturing dyes, and sometimes in lab experiments. The trouble starts with skin contact or even inhaling the dust. This compound can irritate eyes and skin, cause headaches, dizziness, and even a dangerous condition called methemoglobinemia, which lowers the blood’s ability to carry oxygen. I remember a peer handling the powder without proper gloves; his hand broke out in a rash, and he wound up at campus health, rattled. You don’t expect something as ordinary as a dye additive to turn serious, but it happens.
Fact-Checking Toxicity
Toxicology data shows 3-Nitroaniline doesn’t require truckload-size exposures to make people sick. The lethal dose in lab rats, for example, sits around 100-200 mg per kilogram of body weight — certainly not a friendly stat. People exposed to large amounts risk kidney and liver damage. Even lower exposures over time can leave you tired beyond belief, gasping for breath, and wondering what’s off. No one wants to find themselves in an ER because they took a shortcut scooping out powder.
Bigger Environmental Concerns
3-Nitroaniline doesn’t just stop at personal health; it shows up in wastewater and seeps into soil from industrial runoff, especially in areas heavy with textile production. Fish and aquatic invertebrates don’t fare well with it either. The compound isn’t quick to break down, so ecosystems pay a long-term price. I grew up a block away from a small stream that ran next to a dye plant. After a few big storms, dead fish surfaced for days. It wasn’t until later we connected the dots to chemicals like this one washing out of pipes.
What Real Safety Looks Like
Working with 3-Nitroaniline calls for more than a dust mask and a shrug. Proper gloves, goggles, and decent ventilation should be non-negotiable. Good facilities invest in chemical fume hoods and emergency wash stations. Regular training on how to handle spills or accidental contact makes a world of difference. I’ve seen old labs where people skipped these basics, figuring nothing bad would ever happen to them. The odds catch up faster than you think.
Pushing for Smarter Regulation and Alternatives
Some countries already restrict how and where 3-Nitroaniline is used, flagging it as hazardous waste. This kind of oversight protects both workers and the land around factories. Companies looking to do better swap in less toxic substitutes whenever possible. I’ve met engineers who spend years tweaking dye formulations to ditch nitro compounds altogether. Upgrading processes and keeping waste out of waterways gives communities peace of mind.
Bottom Line
Anyone using or living near where 3-Nitroaniline circulates benefits from knowing what’s at stake. Whether you wear a lab coat or live downstream from a factory, clear warnings and strong safety practices mean fewer ER visits and cleaner rivers. No shortcut—no matter how small—beats understanding the risk and respecting what this chemical can do.
What are the storage and handling precautions for 3-Nitroaniline?
Understanding 3-Nitroaniline
3-Nitroaniline shows up as a pale yellow solid with a faint, slightly sweet odor. People working in labs or factories usually see it as a chemical intermediate, especially in dyes and pharmaceuticals. The hazards aren’t always obvious at first glance, which is why handling it without strong safety habits opens doors to unnecessary risks. The most pressing dangers often connect to its toxicity, potential to irritate skin and eyes, and flammable dust if conditions turn wrong.
Safe Storage: Keeping Trouble at Bay
Poor storage practices tend to cause more problems than most people think. I remember seeing a barely labeled container on a lab’s back shelf, forgotten and left close to an old heater. Poor separation from incompatible materials like strong oxidizers or acids can turn an ordinary workday into an emergency.
Best results come from a cool, dry space, away from direct sunlight and ignition sources. Fire safety in chemical labs puts a premium on minimizing dust buildup and avoiding materials that create sparks. A properly fitted, tightly sealed container goes a long way—liquid-resistant, clearly labeled, and firmly kept on a shelf marked for organic compounds. Specialized storage cabinets rated for toxins and combustibles should stay clean, organized, and accessible only to trained workers. Good ventilation inside storage rooms helps remove vapors and keeps air fresh.
Handling: Routine, But Never Casual
People start cutting corners after they’ve handled something a hundred times. With 3-nitroaniline, skin contact or inhalation bring on symptoms like headaches, dizziness, and in the worst cases, methemoglobinemia—a disruption in blood’s ability to carry oxygen. Chemical-resistant gloves, goggles, and lab coats don’t win style points, but they save skin and lungs from exposure. Many mishaps in my own early lab days usually came from distractions or not double-checking personal protective equipment.
Fume hoods matter more than people admit. Even a quick weigh-out can release particles or small amounts of vapor. Vacuum systems should have filters and catch-traps that get checked often, not once every few months. Have safety showers and eyewash stations no more than 10 seconds from most bench spaces.
Simple acts like washing hands before eating, drinking, or leaving work areas also go a long way. Eating near toxic chemicals undermines any other safety measure in place.
Disposal and Spill Response
Messy spills don’t wait for a convenient time. Fast response lowers injury and contamination. Spilled 3-nitroaniline must get swept up with non-sparking tools, then scooped into a compatible waste drum. Contaminated material belongs in marked hazardous waste. Avoid pouring anything down the drain, since municipal water systems aren’t meant to handle these chemicals and violations invite legal trouble and damage to the environment.
Waste, including gloves or wipes, should get treated as hazardous until processed for chemical disposal. Workers need access to up-to-date spill kits and training in emergency procedures, not just the theory, but the muscle memory to act right away without panic.
The Big Picture
Every step in storing and using 3-nitroaniline comes down to respect for both the chemical and everyone in the workplace. Clear labeling, strict personal protective equipment and up-front planning handle most threats before they turn serious. Regular training, updated material safety data sheets, and a culture of watchfulness prove more useful than any single piece of equipment alone. Factoring safety into the daily routine turns caution into second nature, not just a list of rules taped to a wall.
What is the solubility of 3-Nitroaniline in water and other solvents?
Meet 3-Nitroaniline: A Tricky Customer in the Lab
Plenty of chemists, pharmacy students, and even art conservators brush up against aromatic amines. 3-Nitroaniline, with its yellowish tinge and distinctive odor, stands out because it rarely plays nice with water. It looks humble at first glance, but its solubility quirks deserve attention. If you’re trying to purify, analyze, or synthesize with this compound, knowing where it dissolves well can save you time and headaches.
Water: Not a Friendly Neighbor
Anyone who’s tried to stir a spoonful into water appreciates the struggle. Across most standard temperature conditions, 3-nitroaniline dissolves at a rate hovering around 0.7 grams per liter. For reference, common table salt clocks in at over 350 grams per liter. That difference isn’t trivial—it affects everything from analytical recoveries to the design of wastewater treatment systems. High schoolers prepping for a chemistry olympiad and senior researchers in pharmaceutical QC labs both need to understand this: Don’t count on water to do your dissolving for you if you’re working with 3-nitroaniline.
I remember once in a student project, my team tried to recrystallize a mixed sample using water. We expected to see clean, beautiful crystals by morning. Instead, we ended up sifting through a sandy mess. Maybe it’s a rite of passage in the chemistry world—learning to respect the molecule rather than the textbook protocol.
Solvents That Get Along Better
Many have better luck turning to organic solvents. Ethanol, for instance, does a decent job. Its polarity and ability to hydrogen bond with both the -NH2 and -NO2 groups tip the balance in favor of dissolution. At room temperature, you might get over 20 grams per liter—enough to make most extractions and sample preparations manageable. Acetone, another lab staple, matches or even surpasses ethanol for solubility, allowing experiments to move forward quickly. Ether and chloroform can mobilize 3-nitroaniline as well, painting a picture: it’s the non-aqueous solvents that make life easier.
Plenty of process folks out there rely on dimethylformamide (DMF) or dimethyl sulfoxide (DMSO) if the reaction or analysis absolutely demands high solubility. DMF can dissolve many aromatic amines in impressive concentrations. Of course, that comes with its own set of safety headaches, including strong odors and skin irritation. DMSO brings rapid penetration through organic layers—but also through your own skin, so gloves stay on.
Why Solubility Matters Beyond the Flask
The real world isn’t just about lab reports. Knowing where 3-nitroaniline dissolves guides safe cleanup after spills, plans for wastewater management, and how much of a residue might get left behind on glassware. Environmental scientists pay attention, too. If a spill happens, water won’t move much of the material—contamination tends to stick around unless solvents enter the scene. That can affect local aquatic environments. Regulators actually set rigorous standards because nitro-aniline derivatives show up as persistent organic pollutants in water, especially when industrial discharge sidesteps best practices.
Solving the Problem
Anyone recovering or disposing of 3-nitroaniline—whether in industry or a teaching lab—should invest in strong extraction protocols. Dilute acid washes can protonate the amine, making it more water-soluble for easier cleanup. For synthesis, pick a solvent with both a high safety tolerance and an ability to dissolve the material well enough for your scale of work. Chemical inventories should clearly flag the poor water solubility for anyone training new staff. Mistakes happen less often when the right warnings stand front and center.
Experience and the facts both point to this: solubility doesn’t just stay a concern for midterms and textbooks. It’s a deciding factor in how well chemistry gets done, whether it’s a small bench-top run or a massive production batch. Anyone serious about safety or success needs to check the numbers on their solvents first.
| Names | |
| Preferred IUPAC name | 3-nitrobenzenamine |
| Other names |
m-Nitroaniline 3-Nitrobenzenamine Meta-nitroaniline 1-Amino-3-nitrobenzene |
| Pronunciation | /ˌʌɪ.trəʊ.əˈnɪl.iːn/ |
| Identifiers | |
| CAS Number | 99-09-2 |
| Beilstein Reference | 1209224 |
| ChEBI | CHEBI:50145 |
| ChEMBL | CHEMBL14218 |
| ChemSpider | 5500 |
| DrugBank | DB04194 |
| ECHA InfoCard | 100.010.419 |
| EC Number | 202-870-9 |
| Gmelin Reference | Gmelin Reference: 81756 |
| KEGG | C06587 |
| MeSH | D000774 |
| PubChem CID | 7418 |
| RTECS number | NL4375000 |
| UNII | 52V5238K52 |
| UN number | UN1661 |
| Properties | |
| Chemical formula | C6H6N2O2 |
| Molar mass | 138.13 g/mol |
| Appearance | Yellow crystalline solid |
| Odor | Odorless |
| Density | 1.29 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 1.47 |
| Vapor pressure | 0.000045 mmHg (25 °C) |
| Acidity (pKa) | pKa = 2.47 |
| Basicity (pKb) | 9.38 |
| Magnetic susceptibility (χ) | -49.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.653 |
| Viscosity | 1.35 cP (25°C) |
| Dipole moment | 3.96 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 110.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -33.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1924 kJ·mol⁻¹ |
| Hazards | |
| Main hazards | Harmful if swallowed, toxic in contact with skin, causes skin and eye irritation, may cause methemoglobinemia. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS06,GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H317, H334, H341, H351, H410 |
| Precautionary statements | P261, P264, P270, P271, P301+P312, P304+P340, P312, P330, P405, P501 |
| NFPA 704 (fire diamond) | Health: 2, Flammability: 1, Instability: 1, Special: - |
| Flash point | 181°C |
| Autoignition temperature | 520°C |
| Lethal dose or concentration | LD50 oral rat 1750 mg/kg |
| LD50 (median dose) | LD50 (median dose): 300 mg/kg (oral, rat) |
| NIOSH | SN 15700 |
| PEL (Permissible) | Not established |
| REL (Recommended) | REL (Recommended Exposure Limit) of 3-Nitroaniline is "0.1 mg/m3". |
| IDLH (Immediate danger) | 100 mg/m3 |
| Related compounds | |
| Related compounds |
aniline 2-nitroaniline 4-nitroaniline 3-aminophenol 3-nitrobenzoic acid |

