3-Nitro-N,N-Dimethylaniline: An In-Depth Commentary
Historical Development
Chemistry has shaped our world, often quietly. 3-Nitro-N,N-Dimethylaniline came up on the radar back in the late 1800s, right in the thick of the synthetic dye revolution. This period saw chemists racing to outdo each other, trying to crack open new colors and techniques for textiles and inks. Historical records show German dye producers kept thorough notes on aromatic amine nitrations, always searching for molecules with more brightness or sturdiness. Once synthetic dyes hit the market, natural sources lost ground quickly, and compounds such as 3-Nitro-N,N-Dimethylaniline earned a spot in the lineup. Over time, its role branched out. It didn’t stay stuck in the dye world. Rubber compounding picked it up, then later researchers in pharmaceuticals and sensors, always hunting for novel applications.
Product Overview
This aromatic amine derivative, shaped by a nitro group on the benzene ring and two methyl groups nitrogen-bound, draws attention for good reason. A lemon-yellow solid at room temperature, it lands as a specialty intermediate more than a bulk commodity. Companies dealing in organic synthons keep it as a staple item, but most regular people never hear its name. In my own years working alongside R&D chemists, 3-Nitro-N,N-Dimethylaniline usually showed up when somebody went off track from the standard catalog of amines. The small volume specialty market creates less overhead but demands greater attention to purity, handling, and custom batches.
Physical & Chemical Properties
Its molecular formula, C8H10N2O2, weighs in at about 166.18 g/mol. A melting point hanging close to 70°C gives it a certain hand-feel, soft but pressable in warmer labs. Its yellow shade comes from the electron-withdrawing nitro group, which alters the ring’s resonance. Chemists sniff at its moderate solubility in ethanol and ether, though water leaves it mostly untouched. This hydrophobicity makes sense considering the nonpolar backbone and the methyl groups’ shielding effect. I’ve noticed younger chemists often mistake its solubility for contamination, then get the “ah-ha” moment after running a TLC plate and seeing solid separation lines.
Technical Specifications & Labeling
Typical labeling practices run with no-nonsense clarity: chemical name, CAS number (99-59-2), molecular weight, batch number, and safety guidance. Bulk shipments use UN numbers and hazardous statements spelled out under GHS, red diamonds showing the health risks. Purity standards usually start above 98%, with modern labs pressing up to 99.5% for sensitive reactions. A good batch logs melting point, trace metal content, and moisture. I’ve worked in places where half a percent variance in purity cut a reaction yield by a third. That’s why specification sheets go beyond what’s mandated — one missed contaminant could sabotage an entire synthesis train.
Preparation Method
Factories and labs take dimethylaniline and treat it with nitric acid under controlled temperatures. The process skews toward “clean” nitration, gently favoring the meta-position. This route throws off a mix of isomers, but by tuning conditions — ice baths, slow acid addition, snail-paced stirring — modern setups tip the balance sharply toward the 3-nitro product. Routinely, workups follow with basification, organic solvent extraction, then recrystallization for cleanup. Chromatography tends to get saved for stubborn batches. The method hasn’t evolved radically in years, but each tech tweaks the ratios to match their scale, which could mean a beaker or a stainless steel drum.
Chemical Reactions & Modifications
3-Nitro-N,N-Dimethylaniline plays well as a starting block for further functionalization. Reductive conditions — think iron filings with acid — strip away the nitro group for various amino derivatives, which pop up in dyes and photographic reagents. The methyl arms open up sites for quaternization, useful for surfactant production. Mordant dyes need this sort of structure, with its balance between electron-donating and withdrawing groups. I’ve seen researchers hang halogens or swap in sulfonic acid chains from this core, making water-soluble molecules for analytic detection kits. Each tweak bends the UV-Vis spectrum, which matters for developers looking for dyes or detection labels that don’t get lost in the crowd.
Synonyms & Product Names
Over time, different fields tacked on their own labels, which can catch people off guard. ‘3-Nitro-N,N-Dimethylaniline’ headlines regulatory papers, but ‘Meta-Nitro-dimethylaniline’ and ‘N,N-Dimethyl-m-nitroaniline’ pop up in catalogs. German suppliers sometimes list ‘m-Nitro-N,N-Dimethylanilin,’ while trade labels shave things down to numbers, like ‘C.I. 20220’ within the color index. I’ve seen mixups where an order gets delayed over naming confusion, especially across borders. A quick cross-check on CAS numbers usually solves it, though not always if suppliers fudge lot specs to match foreign regulations.
Safety & Operational Standards
Anyone in the lab or plant who’s worked with aromatic nitro compounds hears warnings for a reason. 3-Nitro-N,N-Dimethylaniline irritates skin and eyes, and long exposure can undercut blood’s oxygen carrying power (methemoglobinemia). Regulatory bodies such as OSHA and ECHA stress gloves, goggles, and careful engineering controls. Fume hoods aren’t optional — nitro aromatics volatilize without drama, and repeated low-level exposure creeps up on you. Lab coats go into separate laundry. Spill kits stay on standby. Safety Data Sheets (SDS) lay out reactivity issues, and strong oxidizers or reducing agents get flagged as high-risk partners. Disposal plans usually mean high-temp incineration. From my own experience, health and safety teams take special note of the nitro group’s toxicity, running regular air and skin monitoring in settings with higher throughput. In the last decade, more labs embraced glovebox systems for this very class, for the extra measure of isolation and control.
Application Area
The traditional anchor point sits with synthetic dyes, lending backbone to azo compounds for textiles, inks, and occasionally some stubborn ceramics. Rubber processing industries use it as a secondary accelerator, boosting vulcanization reactions and speeding up curing cycles. More recent years have seen innovations in electronics, where this molecule and its cousins become sensitive intermediates in organic light-emitting diodes or molecular sensors. In analytical chemistry, it can turn up in trace detection of formaldehyde, playing a role in colorimetric assays. Occasionally, even agricultural chemical manufacturers run a line off the backbone, hunting for new growth regulators or pesticide intermediates. The versatility never quite breaks into volume commodities — but the range keeps it in demand among specialists.
Research & Development
Current research pushes 3-Nitro-N,N-Dimethylaniline toward tailored photochemical and electrochemical applications. Many groups work on dye-sensitized solar cell efficiency by modifying the aromatic ring. Others target more selective analytical probes, piggybacking on the color shifts that small chemical tweaks produce. Catalogs of new organometallic complexes start from this molecule, exploring catalysis for green chemistry. I remember a recent professional conference where a team showed nitro-aniline derivatives shaving energy loss in OLED displays by channeling electron flows. The relatively cheap cost and known safety profile help keep this compound front-of-mind for university research teams. Some groups now focus on “greener” synthesis pathways, using aqueous media or enzymatic routes to cut down hazardous waste, which broadens its appeal to labs mindful of environmental stewardship.
Toxicity Research
Most available studies point to tissue-level toxicity through disruption of red blood cell function, especially at higher dosages or poor ventilation. Animal models show methemoglobinemia and organ impacts with chronic exposure. Regulatory filings with ECHA and TSCA both highlight it as a hazardous substance, subject to workplace monitoring and strict controls on waste. Epidemiological studies still lack large sample sizes, but a handful of industrial reports flag increased absenteeism among workers in facilities lacking modern controls. The IARC doesn’t currently categorize it as a proven human carcinogen, but some metabolic byproducts bear structural resemblance to known toxics. One research group has tracked mutagenic activity in bacterial assays, keeping the regulatory focus sharp and driving pressure for alternative synthesis and improved PPE protocols. My time supporting industrial hygiene trials proves no firm takes these warnings lightly. Routine blood checks and restricted access policies remain standard, showing hard lessons learned from past carelessness.
Future Prospects
3-Nitro-N,N-Dimethylaniline enters the future balanced between traditional demand and new scientific curiosity. Dye and pigment markets continue to rely on its reliability and cost-effectiveness, though the search for less hazardous substitutes never stops. Electronics and photonics see more action, with labs experimenting in fields from optoelectronics to organic semiconductors, especially as performance-to-price ratios tip in favor of older intermediates. More researchers probe biological derivatization, testing safer analogs for biosensor applications. Regulatory tightening, especially around toxicity and environmental fate, nudges producers toward inventive “green” pathways, which inspires both technology upgrades and cross-industry collaborations. From the stories of chemical industry veterans, nothing stays fixed forever – 3-Nitro-N,N-Dimethylaniline may shift from dye essential to niche catalyst or high-tech sensor backbone, shaped by how nimbly the field responds to health, safety, and environmental calls. The next decade might reroute its major uses entirely, fueled by the stubborn pace of both regulation and invention.
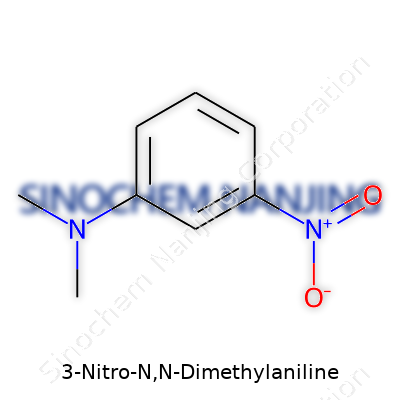
What is the chemical structure of 3-Nitro-N,N-Dimethylaniline?
Why It Matters
3-Nitro-N,N-dimethylaniline pops up in many chemistry discussions. Its structure packs a punch both in the lab and in real-world applications, showing a creative blend of organic chemistry fundamentals. You find it listed in the IUPAC registry as N,N-dimethyl-3-nitroaniline. This compound highlights the fascinating way small tweaks to a benzene ring can significantly influence chemical behavior and usefulness.
Understanding the Chemical Build
The backbone of this molecule is a benzene ring, a flat hexagonal structure made of six carbon atoms. What transforms it from plain benzene to 3-nitro-N,N-dimethylaniline is the arrangement of three main groups. At one position, a nitro group (-NO2) clings to the third carbon. At another, an aniline group features nitrogen, which connects to two methyl groups (–N(CH3)2), sitting at the first carbon. Chemists call these groups "substituents," and their arrangement defines the character of the molecule. The '3' in the name means the nitro group attaches to the third carbon from the dimethylaniline location. This specific placement changes how the molecule interacts, both with other chemicals and in real-life environments.
My Take from the Lab Bench
Having worked with aromatic amines and nitroaromatics during my time at the university, I remember the sharp smell of aniline derivatives and how sticky the world of nitration can become. With 3-nitro-N,N-dimethylaniline, the nitro group draws electrons away from the ring, giving the molecule an edge in reactivity. The dimethylamino bit pumps in extra electron density on its end, balancing out the effect. Each functional group has its own story, much like every cook adding their touch to a stew. Chemists rely on these pushes and pulls to shape everything from dyes to pharmaceuticals.
Why Chemists and Industries Care
3-Nitro-N,N-dimethylaniline lands squarely in the world of colorants. Its structure makes it suited for synthesizing certain dyes and pigments that last in textiles and plastics. The same molecular traits help researchers trace reactivity trends in organic synthesis, letting them build complex molecules step-by-step. This dual personality, having a nitro group to provide stability and a dialkylamino group to inject basicity, boosts its versatility.
Safety and Environmental Impact
The nitro group behaves like a double-edged sword: it adds useful properties, but it also flags the compound for toxicological risks. Too many aromatic amines have left a long shadow in worker health records. Making or handling molecules like 3-nitro-N,N-dimethylaniline calls for solid protocols. Fume hoods, gloves, and careful labeling matter in the lab. If mishandled, compounds with these substituents pose risks to skin and wildlife.
Finding Safer Paths
Sustainable chemistry doesn’t aim to simply replace useful molecules, but to tweak them gently for reduced risk. Newer research looks at alternative groups that keep color-fastness in dyes, with lower toxicity. Some teams turn to biocatalysis, using enzymes as molecular editors instead of harsh nitration reagents. Open dialogue between chemical manufacturers, regulatory agencies, and health experts helps steer research toward better safety without losing performance.
Making Sense of the Details
Knowledge of how specific groups like nitro and dimethylamino adjust the benzene’s behavior stays relevant for years. Seeing both the utility and the risks sharpens the decision-making process for labs and industry. Better understanding starts with looking closely at molecules like 3-nitro-N,N-dimethylaniline and weighing their value against their hazards in the real world.
What are the common applications of 3-Nitro-N,N-Dimethylaniline?
Why Chemists Keep Eyeing This Molecule
3-Nitro-N,N-Dimethylaniline pops up in research labs and factory floors more often than most people guess. Its quirky structure makes it valuable for producing dyes, medicines, and laboratory tools. As someone who tracks industrial chemical trends, I’ve seen how a single ingredient like this one can kickstart both innovation and big debates about safety and sustainability.
Dye Industry: Coloring Our Everyday World
If you've ever tossed on a brightly colored shirt or seen diagrams in your old chemistry textbook, chances are you’ve run across molecules made with 3-Nitro-N,N-Dimethylaniline. Dye makers rely on this substance because it reacts easily with other chemicals to form vivid pigments. The textile world can’t look away from it, since it delivers lasting color that doesn’t give up after a few washes. Photographic color development—before digital took over—also leaned heavily on similar aromatic amines for sharp, stable color prints.
This isn’t just about making things look nice. Textile companies wrestle daily with strict rules about colorfastness and environmental discharge. 3-Nitro-N,N-Dimethylaniline answers both these challenges: It makes dyes that stick and can be structured to reduce harmful byproducts. Yet, it’s not magic; wastewater treatment tech must still keep up to prevent toxic lapses.
Pharmaceuticals and Research: Fueling Discovery With Reactions
Medicinal chemists don’t talk much publicly about their recipes, but this compound slips into early experimental medicines as a starting point for more complex drugs. It provides a backbone for building up molecules that may fight disease. Pharmaceutical researchers choose it because the nitro and dimethylamino groups react predictably, laying the groundwork for synthesizing more potent structures.
Every time a new painkiller or antibiotic gets discovered, the path often passes through plain-looking chemicals like this one. Laboratory syntheses use 3-Nitro-N,N-Dimethylaniline to create intermediates, dyes for drug testing, and sometimes fluorescent markers that help track molecular action inside living systems. Years ago, I watched a friend run hours of tests hoping to nudge such molecules into new directions. The ultimate impact? New knowledge about how diseases progress and sometimes, brand-new treatments.
Analytical Chemistry: Right Tool, Reliable Outcome
Many labs keep a stash of 3-Nitro-N,N-Dimethylaniline for colorimetric tests. When you add it to certain samples and see a color shift, it tells you something critical about what’s inside. These methods help scientists track minute changes—whether in blood, water, or industrial oils. Companies count on these low-tech but trustworthy visual tests, especially in areas where high-end instruments just won’t work.
Pushing for Safer, Greener Solutions
It’s easy to get hooked on solutions that work. Yet, every time someone pours a beaker of 3-Nitro-N,N-Dimethylaniline, safety goggles should go on. Aromatic amines have long histories, and some carry real risks. Exposure through skin or inhalation poses hazards, so solid storage rules and personal protective equipment matter. Companies now investigate substitutes with fewer health or environmental risks, but the story is ongoing. We can’t swap everything overnight, but each new chemical regulation and green guideline draws us closer to safer manufacturing lines.
In the real world, change starts one small step at a time. Engineers, supply managers, and scientists work together, reviewing safety data sheets, tracking spills, and teaching new staff how to measure and handle chemicals like this one. Good teamwork and clear communication usually carry more weight than any shiny lab discovery alone. From the dyes in our clothes to the medicines in our cabinets, 3-Nitro-N,N-Dimethylaniline continues making a quiet but real difference—reminding us that even invisible ingredients deserve respect and scrutiny.
What are the safety precautions for handling 3-Nitro-N,N-Dimethylaniline?
Understanding the Risks
3-Nitro-N,N-Dimethylaniline isn’t something you want to treat lightly. It’s a strong chemical, often used in dyes and pigments, which means it can find its way into all kinds of labs and industries. Working with this kind of compound always brings me back to my days prepping chemical stocks at a university lab. There, the main lesson was always “respect the risk.” Touching, inhaling, or even mixing this kind of aromatic amine without the right precautions can easily lead to harmful effects—skin burns, eye injuries, and breathing troubles. Scientific literature and safety data sheets both say: protect yourself at all times.
Main Precautions Needed
Protection starts at the door. The moment I got near any raw chemicals, gloves went on. Nitrile or neoprene gloves stand up better against aromatic amines. Even if hands feel clean, this chemical can soak through skin fast. Full-length lab coats—ones you wash separately from street clothes—add another layer of defense. Chemical splash goggles are non-negotiable; regular specs won’t cut it. In my own experience, one careless moment can mean a splash that takes weeks to recover from.
Fume hoods become a best friend. Even small amounts can give off vapors that hit the lungs or eyes. Always using chemical fume hoods, and checking the airflow before starting, goes a long way to reduce problems. The fume hood keeps vapors out of the face and the room, something safety officers hammer home in their walkthroughs. Emergency eyewash stations and showers must stay accessible—don’t stack boxes or store gear in the way, no matter how cramped the lab feels.
Storage and Spills
Keep 3-Nitro-N,N-Dimethylaniline in sealed, labeled containers. Storage on a low, solid shelf prevents falls and leaks. Segregating from acids and oxidizers is a must, since dangerous reactions happen fast with the wrong mix. I’ve seen plenty who ignore the shelf guide labels, but that shortcut isn’t worth the risk. Even a minor spill brings chaos: vapors around the room, contaminated rags, and real danger to janitors or visitors.
Spill kits with absorbent pads, neutralizers, and proper disposal bags should stay on hand—all staff should practice how to use them. Even if a mentor or supervisor says a little spill is “no big deal,” trust training over bravado. For larger releases, get clear: clear the space, call authorities, and shut doors to contain fumes.
Health and Hygiene
Lunch breaks and lab work need strict separation. Washing hands with soap, not just water, protects from chemicals hiding beneath fingernails. I’ve met colleagues who skipped this and dealt with burns and rashes later. Changing gloves after any chemical contact, and never touching phones or door handles with the same gloves, keeps the work area safer for all.
Proper disposal counts for as much as careful handling. Used gloves, pipette tips, rags, and empty bottles all get their own hazardous waste bin, never standard trash. Hazmat disposal rules, set by environmental authorities, protect not just the lab, but neighboring communities too. Even if it feels inconvenient, strict procedures support the health of everyone involved.
Improving Safety Culture
Labs and workshops handling 3-Nitro-N,N-Dimethylaniline should run regular refresher drills. Reading safety data, updating risk assessments, and holding frank conversations about close calls build trust and improve habits. Encouraging team members to speak up about unsafe practices—without fear of blame—leads to faster problem-solving. Open communication about accidents teaches future generations better methods and shows that safety isn’t an obstacle, but a foundation for real progress.
How should 3-Nitro-N,N-Dimethylaniline be stored?
The Real Risks Behind Lab Chemicals
Anyone who has spent time in the lab knows the scene: cluttered shelves, glass jars, colored powders, and the sharp smell of solvents. Chemicals like 3-Nitro-N,N-Dimethylaniline add another layer of risk. Many would never pick it out of a lineup, but it’s more than just another compound on a shelf. Mishandling a chemical with toxic and potentially volatile properties turns a laboratory from a place of discovery into a hazard zone.
Why Proper Storage Prevents Accidents
3-Nitro-N,N-Dimethylaniline finds use in industrial dye making and research. Most people never hear about it outside of chemistry circles, but improper storage leaves the wider community vulnerable. A sealed, cool, well-ventilated, and dry space keeps it stable. High humidity and direct sunlight speed up decomposition or convert it into even more dangerous byproducts. Over time, some chemicals degrade and corrode their containers, and a leaky bottle means skin contact or fumes escaping into the air. I’ve seen labs forced out of commission for weeks because of a single chemical spill spreading through an old wooden floor. None of this is theoretical. Chemical accidents force evacuations, rack up cleanup bills, and result in real injuries.
Beyond the MSDS Sheet
Reading a Material Safety Data Sheet (MSDS) gives important guidelines, but the real world doesn’t always match the tidy scenarios those instructions picture. Chemicals with nitro groups, such as this one, have a reputation for being flammable and for forming unstable residues. Staff sometimes get complacent, thinking, “nothing happened last month, so nothing will happen next month.” I knew a technician who stacked reagent bottles on top of each other to save space; one day, a small earthquake knocked the shelf over. Luckily, no one got hurt, but the mess cost weeks of work and thousands of dollars. These stories stick because they remind everyone why chemical safety means respect, not just red tape.
Keeping a Chemical in Its Place
Storing 3-Nitro-N,N-Dimethylaniline in sealed containers made from glass or compatible plastic gives it the lowest chance of leaking or reacting. Secondary containment trays make cleanup easier if a bottle breaks or leaks. Put it on lower shelves to reduce breakage from drops. Avoid keeping it near acids, bases, or oxidizers — mixing the wrong chemicals by accident creates unpredictable reactions. Label everything with handling instructions and warning symbols. Review inventory often. Outdated or abandoned bottles build up and lose labels, leading to confusion and risk when someone finally tries to handle them.
Culture of Lab Safety
Lab safety goes beyond rules on a wall. It needs real habits. Each person in the lab should feel empowered to question whether chemicals sit in the right place or whether a container shows signs of wear. Regular audits catch mistakes before they snowball. New staff need hands-on training, not just a stack of policies. Disposal of old or unused chemicals deserves equal attention. Hazardous waste programs exist for a reason—shoving bottles in the back of a storage cabinet avoids the immediate problem but only makes the danger worse over time.
Moving Forward
3-Nitro-N,N-Dimethylaniline might seem obscure, but the principles behind safe storage speak to any chemical in a lab. Respect for chemicals prevents injuries, protects careers, and saves institutions from disaster. News headlines only tell the worst stories, but every lab worker has a tale of a close call. These close calls teach vigilance, not complacency. Safe storage is about people, not just property. Practical habits, clear communication, and careful oversight keep everyone safer—and the science moving forward.
Is 3-Nitro-N,N-Dimethylaniline hazardous to health or the environment?
What Is 3-Nitro-N,N-Dimethylaniline?
3-Nitro-N,N-Dimethylaniline pops up in labs and sometimes in dye manufacturing. Many folks who’ve worked in a lab, myself included, have at least heard its name fly around. It has that telltale pale-yellow look, dissolves in many solvents, and smells a bit sharp. But the thing that sticks out isn’t its color or scent — it's the reputation it carries when the topic swings to health and the environment.
Human Health Risks
Getting close to this compound, especially in workplace settings, brings certain concerns. Scientists flagged it years ago because of the nitro group, which isn’t a friendly neighbor when it comes to human health. Chemicals built this way sometimes mess with blood cells, sometimes even cause methemoglobinemia, which means the blood can’t carry oxygen the way it should. Symptoms can sneak up as headaches, dizziness, or worse. I’ve had colleagues who worked around similar nitro-aromatics, and the rule in the lab—gloves always, fume hood always—is there for a reason. Breathing it in or getting it on your skin can get risky fast.
Safety data from both the EPA and scientific journals agree: carelessness burns. Long stretches of exposure (even low doses) can add up, sometimes causing longer-term troubles, including possible impacts on liver function. The compound also likes to stick around in fat tissue, which doesn't flush out quickly. It isn’t labeled as a proven human carcinogen, but toxicology reports haven’t given it a free pass. Lab animals shown regular doses have shown cell changes that worry toxicologists.
Environmental Impacts
Ever spilled a little of this stuff on your bench or watched the waste bottle fill up? That’s when you wonder what happens after it leaves your sight. 3-Nitro-N,N-Dimethylaniline doesn’t break down in the environment with any sort of speed. Research shows its structure resists bacterial breakdown in waterways or soil, letting it linger much longer than many other organics. Fish and other aquatic life aren’t fans. Even tiny amounts in water can cause harm. There’s also a risk of bioaccumulation, which means each step up the food chain sees bigger concentrations, not smaller.
Regulation and Workplace Controls
It’s tempting to dismiss stories of chemical exposure as overblown, but real-world data proves that regulations shape safer workplaces. OSHA and the EPA list a bunch of rules that force companies to limit release and to use protective gear. I’ve seen places that slack on these rules—nobody wins in those setups, neither the workers nor the company. The cost of a ventilation system or regular health checks pales beside the cost of a chemical accident or exposure lawsuit. Plus the personal cost of health problems never quite goes away.
Reducing the Risks
Handling 3-Nitro-N,N-Dimethylaniline safely starts with good habits—gloves every time, goggles and lab coats, clear labels, and a culture of safety in the workplace. It’s not hard to treat chemical waste right, but some places still dump it down the drain or let it evaporate. Those decisions always roll downhill into bigger problems, hurting water supplies or the lungs of folks nearby. Encouraging regular training and audits keeps standards high.
Research into greener chemistry gives hope. Swapping out hazardous compounds with safer substitutes in dyes and research shouldn’t be some distant dream. Companies who push for these changes tend to see fewer accidents, less bad press, and healthier people at every level. From the lab bench to the riverbank, the choices made matter.
| Names | |
| Preferred IUPAC name | N,N-dimethyl-3-nitroaniline |
| Other names |
3-Nitro-N,N-dimethylaniline m-Nitro-N,N-dimethylaniline N,N-Dimethyl-3-nitroaniline 3-Nitro-N,N-dimethylanilin |
| Pronunciation | /ˈnaɪtroʊ ɛn ɛn daɪˈmɛθəlˌænɪliːn/ |
| Identifiers | |
| CAS Number | 121-87-9 |
| Beilstein Reference | 605682 |
| ChEBI | CHEBI:62908 |
| ChEMBL | CHEMBL429116 |
| ChemSpider | 63159 |
| DrugBank | DB08312 |
| ECHA InfoCard | ECHA InfoCard: 100.007.375 |
| EC Number | 202-206-5 |
| Gmelin Reference | 78143 |
| KEGG | C14230 |
| MeSH | D015912 |
| PubChem CID | 87522 |
| RTECS number | DJ9275000 |
| UNII | V3X0F7541T |
| UN number | 2810 |
| CompTox Dashboard (EPA) | 6F8W3T08RF |
| Properties | |
| Chemical formula | C8H10N2O2 |
| Molar mass | 166.19 g/mol |
| Appearance | Yellow liquid |
| Odor | Aromatic |
| Density | 1.16 g/cm³ |
| Solubility in water | Soluble |
| log P | 1.9 |
| Vapor pressure | 0.00844 mmHg at 25 °C |
| Acidity (pKa) | 5.15 |
| Basicity (pKb) | 8.31 |
| Magnetic susceptibility (χ) | -60.2·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.579 |
| Viscosity | 2.19 cP (25°C) |
| Dipole moment | 3.56 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 200.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 167.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2361.3 kJ/mol |
| Hazards | |
| Main hazards | Toxic if swallowed, in contact with skin or if inhaled; causes skin irritation; causes serious eye irritation; may cause respiratory irritation. |
| GHS labelling | GHS02, GHS07, GHS09 |
| Pictograms | GHS02,GHS06 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H335, H341, H351 |
| Precautionary statements | P210, P261, P264, P271, P280, P301+P312, P304+P340, P305+P351+P338, P312, P337+P313, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-1 |
| Flash point | 58 °C (closed cup) |
| Explosive limits | Lower: 0.9%, Upper: 6.0% |
| Lethal dose or concentration | Rat oral LD₅₀: 523 mg/kg |
| LD50 (median dose) | Mouse oral LD50: 595 mg/kg |
| NIOSH | SN4025000 |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | 0.5 mg/m³ |
| IDLH (Immediate danger) | 50 mg/m3 |
| Related compounds | |
| Related compounds |
N,N-Dimethylaniline 2-Nitro-N,N-Dimethylaniline 4-Nitro-N,N-Dimethylaniline 3-Nitroaniline 3-Nitrotoluene |

