3-Nitro-4-Toluidine: A Closer Look at Its Role in Industry and Research
Historical Development
Long before 3-Nitro-4-Toluidine found its place in specialty synthetic routes, chemists sought compounds that balanced reactivity with selectivity. In the late nineteenth century, dye manufacturers started exploring nitroaromatic derivatives as color intermediates, finding that 3-Nitro-4-Toluidine delivered deeper shades and stayed stable through various processing conditions. This compound emerged out of necessity, responding to growing needs in textiles, pigments, and emerging photographic chemistry. Its adoption grew as both industrial chemistry and regulatory oversight advanced, pushing manufacturers toward chemicals with predictable, testable properties and traceable syntheses. As larger facilities learned to operate safely with aromatic amines and nitro compounds, researchers started building entire branches of organic chemistry around these building blocks. Their work continues to influence both academia and industry labs worldwide.
Product Overview
3-Nitro-4-Toluidine appears as a yellow crystalline powder that falls into the category of aromatic nitroamines. Its distinctive chemical structure makes it valuable in synthesizing azo dyes, pigment intermediates, and other organic substances that need resilience against sunlight and oxidizing agents. Unlike some earlier aromatic amines, this compound maintains a balanced profile, providing solid color stability for dye and pigment production without an extreme hazard footprint when handled responsibly. It responds well to tailored processes, meaning large manufacturers and smaller specialty labs can both use the substance in various batch and continuous-flow operations. Global chemical trade recognizes the versatility of this nitroaromatic through consistent specification sheets and purity standards, supporting its continued presence in supply chains for plastics, coatings, and analytical reagents.
Physical & Chemical Properties
The molecular formula C7H8N2O2 translates to a rigid framework: a methyl group, an amino group, and a nitro group locked onto a benzene ring. The melting point usually ranges between 138°C and 141°C, an indicator of its crystalline purity and structural integrity. In water, solubility comes low—most of the compound prefers to stay in organic solvents like ethanol, ether, and acetone, making it easy to isolate from aqueous solutions and organic syntheses. Its color holds a noticeable yellow hue, pointing to the resonance stabilization provided by its functional groups. In the lab, anyone handling this material quickly learns to respect its faint but distinct aromatic odor, and to appreciate its relative stability under dry, cool storage. Chemical reactivity stays central to its use, as both electrophilic and nucleophilic substitutions take advantage of the arrangement of functional groups on the ring.
Technical Specifications & Labeling
Producers generally list this compound with purity levels above 98%, with advanced grades tailored for specialty applications that cannot tolerate side products. Labels note both structural formulae and common identifiers such as CAS numbers, UN numbers (for transport), and EC listings for regulatory tracking. Specification sheets dive deep, covering melting points, recommended storage conditions—ideally dry, well-ventilated locations—and batch analysis data for contaminants like other toluidine isomers, moisture content, and residual solvents. Standard packaging involves sealed containers, often in dark brown bottles or lined drums for bulk transport, aiming to avoid both accidental exposure and chemical degradation. Regulatory agencies keep a watchful eye, so material safety labels typically address hazards, recommended protections, and first-aid steps in line with global safety protocols.
Preparation Method
Industrial routes to 3-Nitro-4-Toluidine follow the time-tested logic of direct aromatic substitution, starting with p-toluidine or analogous precursors. Nitration in carefully controlled acidic conditions ensures addition occurs at the meta-position relative to the amino group. Temperature control and acid concentration dictate yield and purity, with separation steps employing filtration, solvent extraction, and repeated crystallization cycles to isolate the product. For every kilogram produced, operators must balance conversion rates, minimize side reactions, and treat waste streams, signaling the need for both chemical know-how and environmental stewardship. Modern facilities employ closed-system reactors, scrubbers, and recovery units that reclaim solvents and neutralize acidic effluents, reflecting both technological progress and responsibility toward the environment and safety.
Chemical Reactions & Modifications
The platform structure of 3-Nitro-4-Toluidine opens doors to further chemical manipulation, especially valuable in research settings. Reduction transforms the nitro group to an amino function, producing diamines that underpin several advanced polymers and dyes. Diazotization prepares the way for coupling with phenols or aromatic amines, unlocking a spectrum of azo dyes with varied hues and fastness qualities. Halogenation, acetylation, and sulfonation each reveal new reactivities, expanding the list of dye intermediates or analytical reagents that can be synthesized efficiently. Chemists working with these derivatives discover both challenges and rewards, adapting old literature routes or pioneering new catalyst systems to improve yields, cut costs, and solve purification bottlenecks. Every step in the synthetic chain demands careful control of pH, reaction time, and temperature, with analytical techniques confirming the identity and purity of products before use in applied research.
Synonyms & Product Names
Literature and commercial catalogs identify 3-Nitro-4-Toluidine under a variety of names: p-Toluidine, 3-nitro-; 4-Methyl-3-nitroaniline; and 3-Nitro-p-toluidine often headline the list. European chemicals regulations use EC identifiers, while Asian markets refer to established regional nomenclature. Commercial product listings might mention color index numbers when highlighting its role in the azo dye supply chain, creating a bridge between raw material suppliers and end users seeking specific technical attributes. Tracking synonyms ensures compliance with safety and trade regulations, especially as companies expand across borders and intersect with new market requirements.
Safety & Operational Standards
No responsible discussion of 3-Nitro-4-Toluidine overlooks the risks that come with handling aromatic nitro compounds. Regulatory data classify this substance as potentially hazardous, particularly with chronic contact or inhalation of dust and fumes. Industry best practice sets up closed handling, strong ventilation, and routine monitoring to prevent accidental exposures. Protective gear—gloves, goggles, masks—remains non-negotiable, especially during weighing, blending, or transfer operations. Facilities emphasize ongoing safety training, responding both to evolving standards and lessons learned from past incidents. Emergency protocols and spill cleanup guidelines form part of every material transfer or audit, showing that chemical safety goes hand in hand with technical progress and environmental stewardship. Workers and supervisors pay close attention to regulatory updates from agencies like OSHA, ECHA, or local equivalents, favoring a proactive stance toward both personal safety and environmental responsibility.
Application Area
3-Nitro-4-Toluidine drives innovation most strongly in color chemistry. Dye manufacturers use it as a robust intermediate for producing bright, long-lasting azo pigments applied in textiles, plastics, and printing inks. The compound also appears in smaller quantities for specialty polymers, organic electronics research, and pigment synthesis for modern paints and coatings. Analysts sometimes rely on its chemical profile for reagent formulations, especially where selectivity in detection or quantification matters. In regions where regulations restrict certain aromatic amines, 3-Nitro-4-Toluidine’s performance and record of safe handling with proper controls create space for responsible use and ongoing development. Specialized departments inside larger chemical firms track both product performance and market requirements, positioning the substance for applications that reward high-purity inputs and stable long-term supply.
Research & Development
R&D teams do not rest on legacy chemistry alone, regularly probing new process routes and product ideas involving 3-Nitro-4-Toluidine. Researchers refine synthetic pathways, reduce byproduct streams, and adapt green chemistry principles for both economic and environmental gain. Academia tackles reaction mechanism studies, photochemical evaluations, and functionalization strategies—pushing the boundaries of what this compound offers. Collaborations between universities and industry sometimes bring bench-scale ideas to industrial pilot lines, translating theoretical findings into scalable, market-driven products. Analytical chemists track trace impurities and develop robust testing methods, supporting both process safety and end-use reliability. The result: a dynamic field where tradition gives way to innovation and continuous learning.
Toxicity Research
No chemical with an aromatic nitro group escapes rigorous scrutiny when it comes to health effects. Toxicologists study both acute and chronic impacts, exploring respiratory, dermal, and systemic toxicities using validated models. Occupational exposure limits stand as products of both lab studies and real-world industrial data, anchoring workplace practices to sound science. Data suggests that long-term handling can bring risks—methemoglobinemia, skin sensitization, and potential carcinogenic effects—calling for strict controls and routine medical checkups for workers. Environmental impact also stays on the radar, as breakdown products and waste streams require treatment before discharge. Modern toxicology increasingly draws on big data and computational methods to predict risks, supporting policy makers and industry leaders in updating guidelines and outreach materials. These findings reach the shop floor through revised labels, updated safety data sheets, and targeted worker training.
Future Prospects
The outlook for 3-Nitro-4-Toluidine links directly to shifting industry needs and evolving regulatory landscapes. Demand for brighter, more resilient dyes and pigments keeps the compound in demand, as specialty applications—from medical diagnostics to smart coatings—stretch performance requirements and purity standards. Regulatory agencies tighten oversight in response to new safety findings, prompting suppliers to innovate both product formulations and containment methods. Researchers seek to expand green synthesis approaches, aiming for production cycles with reduced environmental impact and higher yield-to-waste ratios. As digitization and data-driven control techniques reach more chemical plants, both producers and end users weigh risk-benefit profiles with increasing precision. Success in this space favors those who blend technical expertise with transparency, bringing clear data to all stakeholders and adapting quickly to both market shifts and policy changes. The future of 3-Nitro-4-Toluidine likely depends on this combination of responsible innovation, cooperative regulation, and open scientific exchange.
What is 3-Nitro-4-Toluidine used for?
A Hidden Ingredient in Color and More
The world rarely pays much attention to factory chemicals until a crisis unfolds. But many dyes that color our clothes and home goods start their journey with building blocks like 3-Nitro-4-Toluidine. This yellowish crystal, smelling faintly of something sharp, goes straight into the production of azo dyes. These dyes add shade to everything from cotton T-shirts to wool carpets. Without it, textile mills would struggle to reproduce vibrant reds, oranges, yellows, and browns.
Connecting Dots with History and Chemistry
This compound caught my interest after I spent time researching our clothing's supply chain. I noticed how most manufacturers in Asia, especially in India and China, listed "toluidine" derivatives early in their process descriptions. They take 3-Nitro-4-Toluidine and react it with other chemicals to create colorants that hold up well through washes and sunlight. In the late 1800s, when scientists hunted for new shades and better consistency, they landed on this substance for its reliability. Over a century later, the recipe hasn't changed much because it still works.
Why This Matters Beyond Color
Azo dyes don't only show up in jeans. Look to printing inks on product packaging and gloss in children's art supplies, and the connection becomes clear. Many people spend their days around objects full of dye residues. This is where close regulation matters. Several years ago, the European Union started cracking down on dyes made from chemicals like 3-Nitro-4-Toluidine, especially if traces remained in finished goods. Researchers identified possible health effects, sometimes linking exposure to skin irritation or long-term risks. Small textile companies got caught by surprise, unprepared to swap out tried-and-true stuff.
In my own work sorting through product recalls, I've learned to scan ingredient lists and lab results for aromatic amines. A little vigilance can prevent batches of banned fabric from entering markets where health authorities watch for trouble. In some cases, importers needed to test every shipment just to keep customs officials from confiscating an entire order.
Focusing on Solutions that Work
The industry has a real challenge on its hands. Manufacturers depend on familiar chemistry, but regulations keep tightening. Some switch to new dye intermediates, but they can face new supply risks or unknown side effects. Others install multi-step purification systems to remove leftover 3-Nitro-4-Toluidine before products leave the factory floor.
A smart fix lies in supporting research into safer alternatives for dyes and intermediates, along with investment in cleaner manufacturing. Brands that demand clean supply chains push suppliers to evolve faster. Consumer awareness also matters. People with allergies or sensitive skin often look for certified textiles labeled as free from restricted amines.
The Bottom Line
3-Nitro-4-Toluidine sits quietly behind products we touch every day. Its safety profile prompts real scrutiny, shaping regulations and driving innovation in chemistry labs. Attention to transparency, smart regulation, and open communication between buyers and producers holds the key to safer products and fewer industry headaches.
What are the safety precautions when handling 3-Nitro-4-Toluidine?
What 3-Nitro-4-Toluidine Is and Why It Deserves Respect
3-Nitro-4-Toluidine tends to show up in the manufacture of dyes, agricultural products, and specialty chemicals. This pinkish-yellow crystal isn’t something you’d want to treat lightly. It carries some real dangers if ignored—a risk I learned up close in my college chemistry lab days, watching someone try to clean a flask without gloves. A chemical burn usually leaves a lasting impression.
Direct Contact Risks
This compound irritates skin and eyes. Fumes from heating or grinding can sting the throat, set off coughing, and sometimes trigger headaches. Touching it with bare hands—even for a second—can make skin red and itchy. I have seen careless moments turn laboratories into chaos, so a strict glove rule isn’t just bureaucratic red tape—it’s self-preservation. Nitrile gloves block most organic chemicals. Don’t forget safety goggles, either, because an accidental splash near your face can mean an emergency trip to the eyewash station.
Breathing and Air Quality
Powder form brings inhalation dangers. It doesn’t take much for airborne dust to float, especially if someone dumps some in a weighing dish. The compound’s fine particles can slip right into the lungs, and from there, the body has a harder time getting rid of them. End result: respiratory distress ranging from a nasty cough to something severe like chemical pneumonitis. I always double-checked lab hoods and made sure air flows, drawing away dust and fumes before ever opening the bottle. If a fume hood isn’t available, a half-face respirator loaded with organic vapor cartridges cuts risk dramatically.
Proper Storage Means Fewer Accidents
I’ve yet to see anything escalate faster than a bottle of reactive chemicals left in sunlight or near sources of ignition. 3-Nitro-4-Toluidine reacts with strong oxidizers and doesn’t mix well with open flames or sparks. Storing it in a cool, dark place away from acids or bases prevents surprise reactions—and nobody wants to clean up after one of those. Marking containers in bold, permanent labels helps everyone know the hazards inside at a glance.
Disposal: Not Just Tossed in the Bin
I saw someone once dump a beaker down the drain, thinking water would just dilute everything away. The result? A nasty stench, a clog, and a hefty reprimand from our safety officer. 3-Nitro-4-Toluidine falls under hazardous waste. Collection in labeled, spill-proof containers keeps it out of waterways and away from the hands of the untrained. Most cities provide collection programs for this kind of waste. Rinsing beakers directly in the sink just pushes the problem elsewhere—it never really goes away.
Training Makes the Biggest Difference
None of these precautions work if people don’t respect them. Regular hands-on practice, not just reading safety data sheets, keeps everyone alert. Signs, quick briefings before work, and sharing stories of past mishaps keep the risks real. I remember getting burned once by assuming “just this once” wouldn’t hurt. After that, every person on my team took refresher training. Sharing real examples and mistakes leads to a team that watches out for each other and follows safety culture for the long haul.
What is the chemical formula and molecular weight of 3-Nitro-4-Toluidine?
Looking Closer at 3-Nitro-4-Toluidine
People in research and industry often come across tricky names like 3-Nitro-4-Toluidine. The name might sound like something out of a dry chemistry class, but this compound tells a story about the way we piece together molecules. In everyday labs, the details of a chemical’s structure carry more weight than most folks think, especially when safety, utility, and research rely on precision.
Chemical Formula and Molecular Details
3-Nitro-4-Toluidine goes by the chemical formula C7H8N2O2. That isn’t just a jumble of letters and numbers; it’s the foundation of how scientists and manufacturers understand its possibilities and risks. The molecular weight clocks in at 152.15 g/mol. Here, every atom counts — even a small tweak could mean a major difference in how this molecule reacts under heat, light, or pressure.
Why The Formula Matters in Real-World Use
In practical settings, knowing the exact formula and weight isn’t some academic trivia. It lets folks calculate dosages in synthesis, figure out safe handling, or predict how the substance might act if released into the environment. For me, handling compounds with precision meant not only trusting what the supplier wrote but double-checking the molecular weight during calculations. Inaccuracies can upend reaction yields and mess with purity in the end product, which may lead to more waste and higher costs.
Health, Safety, and Environmental Impact
The structure of 3-Nitro-4-Toluidine includes both a nitro group and an amino group attached to a benzene ring. Chemists know that nitro compounds sometimes pose toxicity concerns, both for the handler and for ecosystems. I remember working in a facility where small leaks or spills of related chemicals pushed the environmental team to rethink containment procedures. Whether it’s the nitro group or the aromatic ring, overlooking these details can lead to groundwater trouble or air quality complaints from the surrounding community.
Responsible Use and Research
Down the line, solutions don’t just hinge on following safety labels. Researchers and industry workers do better when they foster a habit of thorough cross-checking — reviewing SDS sheets, keeping up with published toxicity data, and running extra calculations if anything looks odd. Groups like the American Chemical Society and government bodies have spent decades pushing for transparent chemical registries and clear labeling for exactly this reason. The work isn’t just about ticking boxes on compliance forms. It’s about building trust among colleagues, customers, and neighbors who rely on safe and ethical chemical handling.
Moving Toward Safer Chemistry
Better chemistry starts at the bench. Double-checking formulas like C7H8N2O2, weighing precisely, and understanding functional groups can protect everyone involved. Every person in a lab, from the greenest technician to the head of research, has a stake in getting the details right — not just to avoid mistakes, but because behind every chemical lies a network of people who depend on safe decisions.
How should 3-Nitro-4-Toluidine be stored?
Everyday Risks Call for Clear-Cut Action
If you're around chemical labs, you've probably crossed paths with substances that can knock your socks off — figuratively and literally. 3-Nitro-4-Toluidine fits squarely into this bucket. Its hazards aren't just a technical detail tucked away in safety data sheets. It can trigger fires, release toxic fumes, or damage your health if handled like a bag of sugar. I’ve seen labs suffer costly clean-up because storage got lax. A single mistake multiplies into a web of headaches for staff and the environment.
Why Storage Details Save More Than Money
Some folks may want an all-in-one rulebook, but with 3-Nitro-4-Toluidine, the facts stay simple: let it stray from a controlled environment, and you invite trouble. This compound shows sensitivity to heat, friction, and rough handling. Shelf placement matters. Store it in tightly sealed containers built from glass or compatible plastics. If you’re using metal storage, make sure it lines up with manufacturer recommendations, since certain metals help speed up breakdown and can spark dangerous reactions.
You don’t want a leaky cap or corroded container. Traces of moisture seep in, increasing decomposition risk. Fumes may build up, triggering pressure inside the bottle. Pressure leads to leaks—then exposure, inhalation, or even an explosion. Sticking each bottle into an explosion-proof safety cabinet makes a real difference. Labs with strict routines on this front rarely deal with fire alarms or painful incident reports.
It’s About Health Too
Long before we talk about disaster-scale fires, there’s another danger: health. I’ve known colleagues who got careless, thinking gloves and goggles gave them a free pass. Shortcuts led to skin irritation, eye damage, and nasty coughs no one could trace to anywhere else. Studies say repeat exposure ups cancer risk. Fume hoods work wonders here, but only if people use them every single time.
Keep It Cool, Keep It Dry
There’s always pressure to store supplies closer to prep benches for quick work. This shortcut doesn't work with something like 3-Nitro-4-Toluidine. Heat sources build up — furnaces, radiators, even sunny windows can bump up temps well past safe thresholds. The golden rule: cool, dry, and dark. Even moderate sunlight can break down chemicals faster than folks expect.
Never stack more than you need. Overstocked shelves buckle. Stuff tumbles. Spills become a team-wide event. Limit access to staff who understand the risks. Each extra trip in and out ramps up potential for accidents. Clear labels help, but so does a team that checks inventory and storage conditions weekly.
Fix What's Broken—Fast
Waiting for a squeaky cabinet to get even worse lands you in trouble fast. Fixing loose lids, swapping cracked containers, and mopping up any spills right away turns big risks into minor blips. Keep sturdy spill kits in the storage area. Practice using them. Many people freeze the first time a powder spills or a minor leak appears, so drills make a huge impact.
Practical Steps, Real Safety
Safe storage of 3-Nitro-4-Toluidine sounds like a hassle. It’s far cheaper and safer than dealing with emergency crews or lab shutdowns. The best labs I’ve worked with treat their chemical storage like a living thing, checking, organizing, and updating routines as projects change. That mindset saves money, protects people, and keeps regulators off your back. Responsible practices make all the difference, whether you’re running a massive facility or just a single bench in a teaching lab.
Is 3-Nitro-4-Toluidine hazardous to health or the environment?
Understanding 3-Nitro-4-Toluidine
I’ve spent years writing about industrial chemicals and their impact, so seeing 3-Nitro-4-Toluidine on an ingredient list always gives me pause. This substance works its way into manufacturing, especially in dye and pigment production. It carries a chemical structure that lets it interact closely with living tissue and ecosystems, sometimes in ways that raise alarm bells.
Health Risks From Direct Exposure
Most folks rarely cross paths with 3-Nitro-4-Toluidine in their daily lives. Inside factories, though, it's different. Inhaling dust or vapors or touching contaminated surfaces can introduce this compound through the skin or lungs. According to safety data, workers exposed to this chemical face risks like skin irritation, breathing trouble, and, with enough contact, even harmful effects on the blood. Lab research links similar chemicals to changes in red blood cells, leading to issues like methemoglobinemia. That’s a condition where blood struggles to carry oxygen properly, and it can sneak up on people without proper protection.
Potential for Chronic Effects
Over time, chemical exposure gets trickier. Prolonged or repeated contact, especially in poorly ventilated spaces, can raise the risk further. 3-Nitro-4-Toluidine belongs to a group where several members show signs of being cancer-causing. While direct long-term studies are limited, the chemical structure resembles other known carcinogens. If history teaches anything, it’s that these risks tend to come out after years, not months.
Environmental Hazards
Factories must pay attention to waste streams, especially those involving 3-Nitro-4-Toluidine. If dumped or spilled, it sticks around in soil and water. Studies from European environmental agencies highlight how such chemicals can linger, affecting aquatic life and soil organisms. Fish exposed to even small concentrations can show stress or developmental issues. Microbes that help break down waste in the ground work less efficiently with this chemical in the mix. There’s a real chain reaction—damage spreads from the smallest life forms up the food web.
Fact-Based Red Flags and Solutions
Accidents and poor disposal practices make headlines for a reason. In the US and Europe, regulations already treat compounds like this one with heavy gloves. Factories need to handle it with the same care reserved for old-school pollutants, using sealed systems, protective clothing, and strong ventilation. Workers get routine training on safe handling and emergency response. Water treatment plants must filter out these chemicals before discharging anything back into rivers and streams.
People in chemistry and manufacturing roles need to push for process changes, opting for safer alternatives when possible. Green chemistry helps steer companies away from substances that pose long-term harm. Regular monitoring and transparent reporting also keep the broader community in the loop. Health and environment agencies publish guidelines for acceptable limits, but there’s room for improvement—especially in developing countries where oversight can lag.
Final Thoughts
No chemical is completely safe, but some demand extra respect. 3-Nitro-4-Toluidine sits in a category that should never be handled lightly. It’s a good example of why experience, vigilance, and stronger safety cultures matter. Better workplace training, stricter disposal, and smarter regulations won’t remove all risk, but they can save lives and protect local environments from lasting scars.
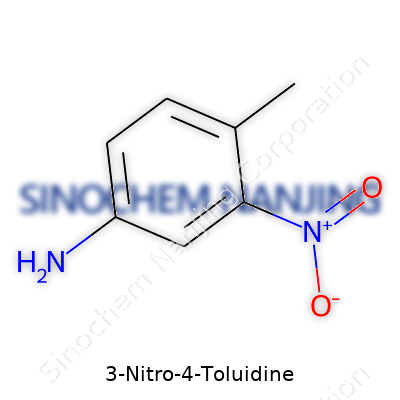
| Names | |
| Preferred IUPAC name | 4-Methyl-2-nitroaniline |
| Other names |
3-Nitro-p-toluidine 4-Methyl-3-nitroaniline 3-Nitro-4-methylaniline p-Toluidine, 3-nitro- 2-Methyl-5-nitroaniline |
| Pronunciation | /ˈnaɪtrəʊ fɔː ˈtɒljʊiːdiːn/ |
| Identifiers | |
| CAS Number | 89-62-3 |
| 3D model (JSmol) | `3d8c71c7aa4d0b9b7e6cf042d15a6c43` |
| Beilstein Reference | 1409070 |
| ChEBI | CHEBI:38460 |
| ChEMBL | CHEMBL517309 |
| ChemSpider | 17419 |
| DrugBank | DB13813 |
| ECHA InfoCard | EC Number: 202-204-7 |
| EC Number | 202-029-7 |
| Gmelin Reference | 82260 |
| KEGG | C06505 |
| MeSH | D009596 |
| PubChem CID | 8666 |
| RTECS number | XZ3150000 |
| UNII | 5U8BB66OZ7 |
| UN number | UN1663 |
| Properties | |
| Chemical formula | C7H8N2O2 |
| Molar mass | 137.14 g/mol |
| Appearance | Orange powder |
| Odor | Odorless |
| Density | 1.26 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 1.98 |
| Vapor pressure | 1.52E-3 mmHg at 25°C |
| Acidity (pKa) | 4.19 |
| Basicity (pKb) | 11.18 |
| Magnetic susceptibility (χ) | -73.8·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.654 |
| Viscosity | Low viscosity liquid |
| Dipole moment | 3.97 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 151.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -12.2 kJ/mol |
| Hazards | |
| Main hazards | Toxic if swallowed. Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. Suspected of causing cancer. Very toxic to aquatic life. |
| GHS labelling | GHS02, GHS07, GHS09 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331: Toxic if swallowed, in contact with skin or if inhaled. |
| Precautionary statements | P261, P264, P272, P280, P302+P352, P305+P351+P338, P310, P362+P364 |
| NFPA 704 (fire diamond) | 2-3-2-爆 |
| Flash point | Flash point: 113°C |
| Autoignition temperature | 470°C |
| Explosive limits | Explosive limits not found. |
| Lethal dose or concentration | LD50 oral rat 1410 mg/kg |
| LD50 (median dose) | LD50 (median dose) Oral - rat - 1,410 mg/kg |
| NIOSH | JN8225000 |
| REL (Recommended) | REL: 0.5 mg/m³ |
| IDLH (Immediate danger) | IDLH: 100 mg/m³ |
| Related compounds | |
| Related compounds |
3-Nitroaniline 4-Nitro-o-toluidine 2-Nitro-4-toluidine 3-Nitrotoluene |

