3-Nitro-4-Hydroxybenzenearsonic Acid: From Past to Present and Beyond
Historical Development
Chemistry has walked a long and challenging road on the way to discovering compounds that push forward agriculture, pharmaceuticals, and science itself. Among the less talked-about, yet crucial substances is 3-Nitro-4-Hydroxybenzenearsonic Acid, a compound with roots going back to the drive for improved animal health and feed efficiency. Starting in the early 20th century, researchers looked for substances that could enhance animal growth and fight diseases, primarily in poultry and swine. This acid, known for its growth-promoting properties, surfaced out of that push, following on the heels of similar arsonic acids like p-arsanilic acid. Early patents and scientific documents chart a course of steady improvement—each step refined the synthetic pathways, improved product quality, and adjusted use to meet changing regulations and ethical demands.
Product Overview
3-Nitro-4-Hydroxybenzenearsonic Acid stepped into the spotlight through its prominent application in veterinary medicine, acting as a feed additive for decades and giving producers a tool to improve feed conversion and control disease. The bright orange crystals serve as a testament to the age of synthetic feed additives, bridging the gap between old-school animal husbandry and modern targeted interventions. Several commercial formulations spun out of this compound, sold under product names that are still familiar to anyone who’s worked in industrial animal production or agricultural chemistry labs.
Physical & Chemical Properties
The acid forms as bright orange-red crystalline powder, showing stability and durability under storage but dissolving in alkaline water and appearing only sparingly soluble in cold water. Its empirical formula, C6H6AsNO5, reflects a structure where a nitro group and hydroxy function alter the benzene ring, and an arsonic acid group anchors the compound’s unique capabilities. Its melting point sits fairly high, commonly above 200°C, which reflects its robustness during processing and transport. This thermal stability means it can withstand lengthy storage and various environmental conditions without decomposing quickly, which matters for any bulk product sent across long distances.
Technical Specifications & Labeling
If anyone browses a chemical catalogue or import database, labeling follows strict conventions. Certificates of analysis rigorously check purity, commonly requiring levels above 98%. Trace metals need inspection, not just for legal compliance but for practical safety, given the risks heavy metals pose as contaminants. Commercial batches are often color-coded with batch numbers and safety data sheets to flag hazard classes and handling protocols. Workers who handle this acid know that a well-labeled, tightly sealed drum saves time and reduces mistakes on the shop floor.
Preparation Method
Lab notebooks tell of multiple methods to prepare 3-Nitro-4-Hydroxybenzenearsonic Acid, but a classic approach uses hydroxy-nitrobenzenes as a starting material. Chemists introduce arsenic acid under acidic or neutral conditions and then induce the arsonic group’s attachment through controlled oxidation and nitration steps. Success often depends on precise temperature management, slow reagent addition, and careful pH adjustment—shortcuts invite impurities or poor yield. Industrial operations scale up these steps using reactors equipped with strict ventilation and monitoring, reflecting years of trial and error to streamline crystallization and minimize hazardous by-products.
Chemical Reactions & Modifications
Chemists appreciate the versatility this molecule offers. Its arsonic group lends itself to reduction, giving derivatives valuable in synthetic pathways. Nitro reduction or diazotization spins off other functionalized benzene rings, opening up routes to specialty chemicals and bioactive compounds. Oxidation, acetylation, or even coupling with diazonium salts produces a series of related compounds, showing why arsonic acids maintain a niche following in research even after regulations shifted usage patterns away from direct feed additives. Anyone who has spent late nights in a university lab can recall the pungent smell and bright colors as telltale signs of these reactions in progress.
Synonyms & Product Names
People may know 3-Nitro-4-Hydroxybenzenearsonic Acid by many names depending on country, supplier, or application. Scientists often encounter it as Roxarsone, the most widespread trade name. Other synonyms include 4-Hydroxy-3-nitrobenzenearsonic acid, (p-Hydroxy-m-nitrophenylarsonic acid), and shorter forms like 3-Nitro-4-Arsonicacidphenol. Each synonym tells a story about its discovery, markets served, and the interplay of commercial branding and chemical nomenclature. Having worked with old literature, I noticed that early patents and product bulletins often ran several synonyms together, reflecting the sometimes-chaotic state of chemical information management before modern databases took the lead.
Safety & Operational Standards
Handling this compound calls for respect and training. The presence of arsenic in any molecule triggers intense scrutiny, given the well-established health risks associated with chronic exposure. Storage uses tightly sealed containers kept in cool, dry rooms set apart from food production areas. Chemical spill kits, absorbent pads, and respirators never gather dust near any active bench or warehouse stocking this acid. Safety data sheets, personal protective equipment, and regular safety drills keep workers alert and aware. My own time running student lab sessions hammered home the need to treat every grain of chemical with care, since one lapse in judgment could spark not just a mess but a medical emergency.
Application Area
Farming communities, research labs, and pharmaceutical manufacturers each saw unique value in this acid. Poultry and swine feed shifted into new territory in the 1940s, as feed mixing plants added Roxarsone to boost growth rates and improve animal health on a commercial scale. Its effectiveness in coccidiosis control, an infection that could devastate livestock, earned it a place in many standard feeding protocols for generations. Even after some regulatory changes curbed direct animal use, certain niche laboratory and diagnostic fields continue to rely on it. Environmental chemists and toxicologists also use it to model the metabolism and breakdown of arsenic-containing organics—data that help set exposure limits and predict contamination risks.
Research & Development
Much of the knowledge around 3-Nitro-4-Hydroxybenzenearsonic Acid developed through painstaking trial, backed by basic research in chemical properties, microbiology, and veterinary medicine. Universities and corporate labs alike published studies tracking its benefits and the molecular pathways through which it worked. Analytical chemists spent years developing improved methods—such as high-performance liquid chromatography and mass spectrometry—to test residues in tissues and feed. On the product side, manufacturers worked with farmers and veterinarians to refine doses, improve palatability, and prevent overdosing. Looking back, I see clear parallels to the development of antibiotics and other “miracle feed” tools—constant feedback loops between bench science, regulation, and field use.
Toxicity Research
Toxicologists have spent decades unraveling the effects of organoarsenicals, especially given the risk that incomplete breakdown can leave residues in food or the environment. Animal studies raised concerns about arsenic accumulation in organs, impacts on kidney and liver function, and potential carcinogenic risks from long-term exposure. Environmental scientists went further, spotlighting how transformation products, such as inorganic arsenic species, could leach into soil and groundwater. The cumulative weight of this research prompted regulatory shifts, particularly in North America and Europe, to phase out or tightly control uses linked to human food production. Every microbiologist with experience in the field keeps a wary eye on arsenic levels in feed and water sources, knowing that prevention is everything when it comes to public trust.
Future Prospects
The legacy of 3-Nitro-4-Hydroxybenzenearsonic Acid stands as both a triumph and a cautionary tale for chemical innovation. Current restrictions mean few feed formulations use this molecule directly, but its chemistry inspires ongoing work into alternative growth promoters, diagnostics, and environmental remediation tools. Analytical methods born from the need to monitor it in animal tissues now underpin programs testing for hundreds of contaminants. Green chemistry efforts look to design molecules with the benefits of growth promotion and disease control but without the downside risks of arsenical contamination. Young chemists and agricultural scientists can learn plenty from the continued debates—balancing the hunger for efficiency with hard-earned lessons about chemical stewardship.
What is 3-Nitro-4-Hydroxybenzenearsonic Acid used for?
Once Common on the Farm
Once, it wasn’t rare to find 3-Nitro-4-Hydroxybenzenearsonic Acid in bags lining the shelves of livestock feed operations. Most people knew it by its trade name, roxarsone. For decades, it showed up in chicken, turkey, and even swine feed. Roxarsone developers claimed it helped animals gain weight a bit faster, kept their droppings drier, and made the meat look pinker—which, let’s be honest, made it look fresher in supermarkets.
Growth-Promoter in the Animal Industry
Facts back up why the industry leaned so hard on this compound. Adding roxarsone cut down on harmful gut parasites and bacterial infections in crowded barns. It could mean fewer sick birds, less downtime, and more profit per pound of meat. Nobody likes thinking about what commercial farms look like on the inside, and diseases run wild in those packed sheds. Roxarsone became a shortcut for healthier animals and bigger yields. The numbers stuck: In the early 2000s, nearly 70% of U.S. broiler chickens ate some form of arsenical feed additive before heading to market.
The Arsenic Problem: Health and Environment
But after years of use, science caught up. Regular people started asking tough questions about arsenic’s place at the dinner table, since roxarsone contains an arsenic atom—just like the notoriously toxic stuff that led entire mining towns to ruin. Every pound of manure from these animals kept trace levels of roxarsone or its breakdown products, and much of it wound up spread on fields as fertilizer. Researchers discovered that roxarsone’s breakdown in soil and water led to forms of inorganic arsenic, which holds tight links to cancer and nervous system damage. Even the FDA pointed to studies in 2011 showing increased arsenic in the livers of treated chickens. That’s hard to ignore.
Pulled Off the Market and Global Differences
After these findings went public, roxarsone sales slowed, and in the U.S., manufacturers pulled it off the market. Still, practices ride on international differences. In some countries, its use hasn’t faded completely. Regulations change factory by factory and farm by farm, so global feed imports can still bring in meat from animals raised with arsenical feed. For consumers, this fuels debate over food labeling, trade standards, and the rights of people to know exactly what lands on their plate.
Finding Safer Alternatives
The push for healthier poultry—without arsenic—birthed plenty of new ideas. Companies started using probiotics, improved barn hygiene, and better nutrition tailored for flocks. Researchers found that simple changes, like optimizing ventilation and tweaking feed with more fiber, could replace the growth-promoting punch of arsenical additives. It doesn’t always match the easy chemistry roxarsone offered, but consumers expect better and are willing to pay a bit more for safer food.
Where We Go From Here
People want to trust what they’re eating. That trust takes regular inspection and honest labeling, with strict follow-through from government agencies. My own trips to farmers’ markets bring out questions about which farms skip additives, and the difference you taste in a roast chicken from a place that never used roxarsone is real. Building back safe, healthy food supply chains takes a focus on long-term soil and water health, not just fast growth. If anything, the roxarsone story taught us to ask—what’s really in our food and how does it affect the world beyond our dinner table?
What are the storage and handling requirements for 3-Nitro-4-Hydroxybenzenearsonic Acid?
Understanding the Risks
3-Nitro-4-hydroxybenzenearsonic acid isn’t something most people run into on a daily basis. This compound, often called roxarsone, serves a role in animal feed and industrial research. But it also brings a certain set of risks because it contains arsenic and nitro groups. This isn’t just another chemical you can treat lightly in a storeroom. Over the years, I’ve learned to respect anything labeled “arsonic.” A single mistake, like leaving a cap loose or failing to separate incompatible materials, can lead to dangerous outcomes for workers and the environment.
Storage Conditions that Protect People and Products
Humidity creeps up on storage spaces, especially during warm months. Moisture can cause sensitive chemicals to clump, break down, or—worse—react to form gases. I keep roxarsone in a tightly sealed container, outside of high-traffic areas, on clearly labeled shelving made from resistant materials. Nothing metal that could corrode ever goes nearby. A dedicated chemical storage room keeps sunlight and fluctuating temperatures out. Cool, dry, and well-ventilated describes a safe space for this compound. Some facilities set up double containment: a main bottle, inside a corrosion-resistant plastic tub, to catch spills before they cause problems.
It only takes one splash or spill to put staff at risk. Proper chemical-resistant gloves, goggles, and a fitted laboratory coat go on before the bottle comes off the shelf. Clean-up supplies stay within reach—neutralizing agents, spill pads, and labeled disposal bags, ready for any mishap. Training everyone who walks into that storeroom prevents costly and dangerous accidents later.
Fire and Incompatibility Risks
Mix nitro or arsonic compounds with the wrong substances and you’re playing with fire—sometimes literally. Substances like strong oxidizers, acids, and bases set off dangerous reactions. I block out a separate shelf, well away from incompatible materials, in a closed-off area. Chemical safety data sheets live nearby, not filed away in a locked cabinet, because you can never predict when you’ll need to double-check compatibility.
Roxarsone doesn’t ignite easily on its own, but fires in storage areas turn nasty fast if it’s involved. Dry powder extinguishers rated for chemical fires stay close. I check the room every week to catch containers whose seals might have cracked or labels that faded. Rotating stock so older containers get used first limits leaks and surprises. When disposal day comes, hazardous waste experts take over—nobody dumps chemical waste down the drain.
Protecting Health and the Environment
Working with arsenic-based compounds for years taught me that even small mistakes can have long-term consequences. Splashes contaminate surfaces and skin; dust drifts through the building and ends up far from where it started. Continuous air monitoring tells me if airborne levels stay low. Only trained staff, with access to showers and designated wash stations, handle anything related to roxarsone.
Documentation matters, too. Every container tracked, every transfer recorded. Regulatory bodies, from OSHA in the U.S. to REACH in Europe, demand detailed paperwork—and for good reason. Traceability helps if anything ever does go wrong.
Practical Solutions for Safer Storage
Never rely on habits or shortcuts. Re-evaluate storage locations each year, rethink spacing, and refresh staff training. Invest in the right shelving, containment tubs, and clear hazard labeling. Prioritize ventilation even if the initial outlay stings: protecting workers and neighbors pays back exponentially over time.
Is 3-Nitro-4-Hydroxybenzenearsonic Acid hazardous and what safety precautions should be taken?
Understanding the Risks
3-Nitro-4-hydroxybenzenearsonic acid, also known as Roxarsone, brings some important health risks to the table. This compound features both nitro and arsonic acid groups, each loaded with danger. Anyone handling this substance in a lab or agricultural setting finds more than just a messy chemical—it poses risks for both people and the environment.
Personal experience tells me these aren’t the sort of risks to brush aside. Even a quick whiff of the powder or a simple spill can escalate into serious contamination if people don’t stay sharp. Skin contact may lead to irritation, and inhaling fine dust has potential to cause respiratory trouble. Over the last decade, researchers have pointed out that compounds containing arsenic ramp up the risk of certain cancers after long-term exposure. That’s not a scare tactic; those are results measured by years of occupational reports and studies tracking arsenic’s route through the body.
The environmental side shouldn’t shrink in importance either. Roxarsone, once widely used to promote growth in poultry, ended up creating water pollution issues. Arsenic from this compound seeps from animal waste into streams, soils, and crops. The consequences ripple out, affecting both wildlife and drinking water supplies. The US Food and Drug Administration moved to limit its use in animal feed after these problems came to light. Knowing arsenic doesn’t just vanish from the earth—once it settles, it enters food chains and sticks around years after its source stops.
Managing Exposure
Simple common sense goes a long way, but strong rules matter even more when dealing with hazardous chemicals. Anyone working with this acid needs to gear up before even approaching it. Chemical-resistant gloves, a proper lab coat, and safety goggles go on before any handling. Dust masks or, even better, respirators help keep harmful particles out of the lungs.
Ventilation stands at the front line. I always check that fume hoods and exhaust systems spin up before work starts. Trying to handle powder in a closed, stuffy room quickly turns from careless to downright dangerous. Spills shouldn’t get wiped with a paper towel or ignored. Spilled material needs an immediate cleanup with proper absorbents and clear hazard signage, keeping the area secure until every trace clears away.
Disposal habits need special care. Pouring leftover material down a drain spells disaster for local water. Roxarsone counts as hazardous waste, so all remnants, PPE, and cleanup materials should go straight into labeled, sealed containers for authorized disposal. Rules get strict here for a good reason: even tiny amounts in the wrong place, over time, contaminate ground and water.
Reducing Future Risk
Education makes a real difference, not just for folks in white lab coats. Support staff, maintenance workers, and new trainees all need clear information on these risks and cleanup routines. Accidents shrink when everyone knows what’s in the bottle and why improper disposal damages more than just the job site.
Substituting safer substances, where possible, also plays a role. Agricultural science moves steadily toward alternatives that help growth but don’t poison water and soil for generations. It takes patience and cooperation between labs, farmers, and regulators to move away from compounds like Roxarsone, but public health gets a lasting boost each time safer practices take root.
At the end of the day, chemicals like 3-nitro-4-hydroxybenzenearsonic acid don’t need to run the show. Awareness, smart handling, strong disposal habits, and looking for alternatives bring the strongest shield against health and environmental fallout.
What is the chemical purity and available packaging sizes of this product?
Understanding Chemical Purity
Chemical purity isn’t just a number on a data sheet—it shapes exactly how a material performs in a real-world setting. You might see the term “purity” measured in percentages, like 98%, 99.5%, or even higher. The higher the purity, the fewer other substances mingle inside the product. This difference often separates a safe lab experiment from a failed reaction or determines whether a manufacturing process runs smoothly or gets bogged down by contaminants.
Let’s use a clear example: laboratories need high-purity chemicals to avoid unwanted reactions. If a researcher aims for repeatable results, even a half percent of impurity may throw off data, skew results, or force a project back to square one. In the electronics world, trace amounts of stray elements can ruin microchips or optical coatings that power our phones and computers.
Types of Purity Levels
Most suppliers list purity grades like technical, reagent, and analytical. Technical grades might work for industrial cleaning or bulk manufacturing, where a tiny bit of byproduct does no harm. Analytical grade suits sensitive lab work or instrument calibration, where stray chemicals can spoil months of research. Pharmaceutical grade reaches the highest bar, crossing every “t” to make sure no accidental ingredient could hurt a patient down the line.
In my years working alongside researchers and quality control teams, I’ve watched the difference a mere percentage point can make. One supplier’s so-called “reagent grade” meant success for a water analysis; another’s “technical grade” meant extra hours in the lab tracking down an unexpected substance that changed the color of a sample.
Packaging Sizes and Practical Needs
Suppliers offer chemicals in a range of packages: small bottles for labs, larger drums for industry, and bulk totes for big manufacturing. A research chemist handles 100-gram bottles with ease and reduces waste, while a food processing facility might take delivery by the pallet. Size matters for storage, cost, and transport—and reduces the risk of spillage or contamination.
I once worked with a health department that insisted on single-use packaging for their field kits to guarantee sample integrity. Break the seal, use the chemical, and toss the container—no risk of cross-contamination. But large-scale industrial processors might do the opposite, using bulk containers to cut cost and avoid running out between deliveries.
Matching Purity and Size to The Job
Picking the right purity level and package size should match the real demands of the job. High-purity materials keep critical experiments reliable and medical applications safe. Industrial users often find a balance between cost and acceptable impurity levels, relying on supplier transparency and trustworthy data sheets.
It pays to ask suppliers about not only purity percentages but also the actual impurities present. Sometimes one type of trace element spells disaster for a process. Clear communication with vendors and robust documentation help avoid costly surprises down the road.
Meeting Safety and Environmental Expectations
With stricter rules around chemical handling, packaging matters even more. Leak-proof seals, chemical-resistant containers, and proper labeling protect both workers and the environment. Many companies now offer eco-friendly packaging, too, cutting down on plastic and hazardous waste.
Staying informed about chemical purity and packaging options benefits everyone—from a high school teacher prepping a science demo, to a manufacturer producing food, to a researcher pushing boundaries in the lab. Getting the details right up front keeps projects safe, reliable, and efficient.
How should 3-Nitro-4-Hydroxybenzenearsonic Acid be disposed of after use?
Disposing of 3-Nitro-4-Hydroxybenzenearsonic Acid can make anyone feel uneasy. This compound, once used in poultry farming and research, isn't something you pour down the drain or toss in the trash. The main thing that pops into my head is health and safety—both for people and the environment. Exposure to anything with arsenic means risk, and soil or water doesn’t have much forgiveness when heavy metals are involved. The stories that hit the news about chemicals in groundwater drive that point home.
Why This Matters
Many rural communities rely on well water. Once arsenic compounds find their way into these supplies, health starts to slide. Arsenic exposure links to cancer, nerve problems, and all sorts of long-term damage. Children and pregnant women face the biggest threats, and those effects stick around for generations. I watched neighbors spend years frustrated by chemical spills that never seem to clear out of their water sources. Costly filters turn into the only way forward, but not everyone can afford those solutions.
Legal Rules and Realities
The EPA and local authorities make the rules pretty clear: skip local landfills and avoid home disposal. That acid needs the hazardous waste label, sealed in containers rated for toxic and acidic chemicals. There's rarely any leeway, and for good reason. The wrong move means fines, but more importantly, lasting environmental harm.
In real life, people sometimes cut corners. They hope a splash won’t matter, but that thought fades once bad smell or sick pets bring regret. Regulatory mistakes often stem from a lack of information. Not enough reminders about which chemicals need special handling means bad habits hang on.
Responsible Paths Forward
Safe handling always starts at the storage step. Never leave this compound uncapped or in a cracked jar. I saw one case where a soggy cardboard box of old chemicals leaked after a storm, and the cleanup cost thousands. Secure shelving and climate control seem like a hassle, but they keep everyone safe for the long haul.
Look for local hazardous waste programs. Many cities offer take-back days for chemicals. Institutions and larger farms usually arrange pickups with certified disposal firms. These companies safely burn or chemically treat waste, making the by-products less dangerous. I have called in specialty teams before during my own lab days, and it cost us some cash, but we slept easier afterwards.
Cutting Down the Problem
Prevention counts for a lot. Switch away from old-school arsenic-based solutions where modern alternatives work just as well or better. The industry is shifting toward safer compounds as regulations tighten. I keep thinking about education, too. If more people clearly understood the risks, safer disposal wouldn’t seem like a chore—it would just be common sense.
Making Safety Routine
At the end of the day, there’s no trick or shortcut for this kind of waste. Take time to double-check storage, contact the right disposal service, and teach those handling these materials. We owe it to the next family using that land or water, and to our own health. The risks stay invisible for too long before damage appears, but the solutions only take a few concrete steps.
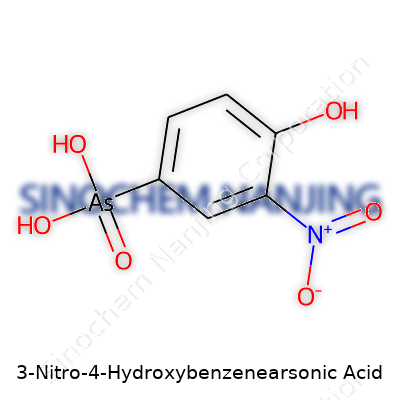
| Names | |
| Preferred IUPAC name | 4-hydroxy-3-nitrophenyl-arsonic acid |
| Other names |
Roxarsone 4-Hydroxy-3-nitrophenylarsonic acid p-Hydroxy-m-nitrobenzenearsonic acid 3-Nitro-4-hydroxyphenylarsonic acid Arsonic acid, (4-hydroxy-3-nitrophenyl)- p-Hydroxy-m-nitrobenzenearsonic acid |
| Pronunciation | /ˈθriː ˈnaɪtroʊ ˈfɔːr haɪˈdrɒksi ˈbɛnzin ɑːrˈsɒnɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | 121-24-9 |
| 3D model (JSmol) | `3-nitro-4-hydroxybenzenearsonic acid JSmol string`: ``` mol3d = loadInline("data:text/plain;mol3d,C1=CC(=C(C=C1[N+](=O)[O-])O)[As](=O)(O)O"); ``` |
| Beilstein Reference | 1722051 |
| ChEBI | CHEBI:76260 |
| ChEMBL | CHEMBL514187 |
| ChemSpider | 24774 |
| DrugBank | DB03675 |
| ECHA InfoCard | 100.000.089 |
| EC Number | 215-036-8 |
| Gmelin Reference | 144458 |
| KEGG | C18606 |
| MeSH | D014839 |
| PubChem CID | 7039 |
| RTECS number | CY8575000 |
| UNII | Q3W7DRB56J |
| UN number | Not regulated |
| Properties | |
| Chemical formula | C6H6AsNO5 |
| Molar mass | 265.05 g/mol |
| Appearance | white to light yellow crystalline powder |
| Odor | Odorless |
| Density | 1.74 g/cm3 |
| Solubility in water | soluble |
| log P | 0.39 |
| Vapor pressure | 7.98E-10 mmHg at 25°C |
| Acidity (pKa) | 6.81 |
| Basicity (pKb) | 7.03 |
| Magnetic susceptibility (χ) | -67.4e-6 cm³/mol |
| Refractive index (nD) | 1.668 |
| Viscosity | Viscous liquid |
| Dipole moment | 4.81 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 146 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -657.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1242.5 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | QG12AC01 |
| Hazards | |
| Main hazards | H301 + H331 + H373 + H410 |
| GHS labelling | GHS labelling: `"Warning; H302, H315, H319, H335"` |
| Pictograms | GHS07, GHS08 |
| Signal word | Danger |
| Hazard statements | H302 + H312 + H332: Harmful if swallowed, in contact with skin or if inhaled. |
| Precautionary statements | P264, P270, P273, P301+P312, P330, P501 |
| NFPA 704 (fire diamond) | 3-2-0-OX |
| Lethal dose or concentration | LD50 oral rat 4100 mg/kg |
| LD50 (median dose) | LD50 (median dose): 500 mg/kg (Rat, oral) |
| NIOSH | NA0450000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 3-Nitro-4-Hydroxybenzenearsonic Acid: Not established |
| REL (Recommended) | 0.1 mg As/kg |
| Related compounds | |
| Related compounds |
3-Amino-4-hydroxybenzenearsonic acid 4-Hydroxybenzenearsonic acid Arsanilic acid |

