3-Nitro-4-Chlorobenzotrifluoride: An Industry Perspective
Historical Development
Modern organic chemistry owes a lot to breakthroughs that took off in the post-war era. During the rise of industrial-scale synthesis in the 1950s, chemists set out to create new molecules for cutting-edge applications in agrochemicals and pharmaceuticals. 3-Nitro-4-chlorobenzotrifluoride didn’t arise from labs by accident—it comes out of a long line of halogenated aromatics that track closely with the changing needs of these industries. Synthetic chemists sought halogenated building blocks with both robustness and versatility, especially where electron-withdrawing groups like trifluoromethyl could open new reaction pathways. From those efforts, 3-nitro-4-chlorobenzotrifluoride found its place as a clutch intermediate by the 1970s and has since taken root in a range of commercial applications.
Product Overview
What you find with 3-nitro-4-chlorobenzotrifluoride is a specialized intermediate featuring a benzene ring substituted with a trifluoromethyl group, nitro group, and chlorine atom. Manufacturers use it as a workhorse for making active ingredients in everything from crop protection formulas to specialty dyes. Its real value shows up during multi-step custom syntheses, where the electron-withdrawing power of the nitro and trifluoromethyl groups creates reactivity options not present in plainer aromatics.
Physical & Chemical Properties
This aromatic compound shows up as a yellowish solid, melting between 50°C and 53°C. It puts off a sharp, pungent odor—a reminder of its nitro and chloro roots. It dissolves well in most organic solvents, but you won’t find it mixing with water. The combination of strong electron-withdrawing groups means it’s less reactive in certain substitutions, but also more predictable in targeted modifications. The compound’s high density and resistance to hydrolysis support its use in processes that demand chemical resilience.
Technical Specifications & Labeling
Industrial-grade shipments run upwards of 98% purity, with clear labeling showing batch identification, net weight, handling instructions, and United Nations identification codes for safe transport. Companies must keep a close eye on residual solvents, moieties like ortho-isomers, and halogen impurities. From experience, supply chain managers and lab techs lean hard on Certificates of Analysis here since final-product safety often depends on raw material traceability. Every barrel must have hazard pictograms alerting handlers to its toxic and irritant nature, and labeling needs to align with global standards such as GHS (Globally Harmonized System).
Preparation Method
Synthesis usually starts with 4-chlorobenzotrifluoride, pulling in strong nitrating agents like nitric and sulfuric acid under careful temperature control. Stoichiometry, agitation rate, and heat removal matter a lot—a slip in any of these can tank a run or create unwanted by-products. Plant managers face a choice here: batch versus continuous processing. Both come with their own trade-offs in terms of safety, throughput, and energy costs. Following the reaction, the product goes through washing, crystallization, and vacuum drying steps. In-house teams watch for nitrophenol contaminants and check melting point ranges to ensure the end product matches tight specifications.
Chemical Reactions & Modifications
Chemists like to turn 3-nitro-4-chlorobenzotrifluoride into other bespoke ingredients. Its electron-poor ring means it takes part in nucleophilic aromatic substitution, which isn’t true for unsubstituted benzenes. The chlorine can leave under the right conditions, letting other nucleophiles take its place—this mechanism underpins the creation of several key herbicides and imaging chemicals. Reduction of the nitro group, often using metals or catalytic hydrogenation, generates amines crucial for dye manufacturing. The combination of substituents has also opened up metal-catalyzed cross-coupling reactions. I’ve seen R&D teams develop high-yield Suzuki or Buchwald couplings using this compound, reducing waste and capturing value for downstream applications.
Synonyms & Product Names
This molecule hides behind a string of aliases, which sometimes causes headaches for procurement pros. Synonyms include 1-chloro-2-nitro-5-(trifluoromethyl)benzene, 3-nitro-4-chlorobenzotrifluoride, 4-chloro-3-nitrobenzotrifluoride, and sometimes simple trade names like NCBTF or CNBT. Distributors and regulatory databases keep tabs on these to prevent mix-ups or missed compliance marks—especially critical for shipments across borders where customs paperwork must match chemical inventory listings exactly.
Safety & Operational Standards
Workers handle this substance with care given its toxic and irritant profile. Acute exposure brings eye and skin irritation, and inhalation of dust or vapors can inflame airways. Environmental safety standards—set by agencies like the EPA and the European Chemicals Agency—set tight limits on permissible exposure and emissions. Facilities use closed transfer systems, negative-pressure workspaces, local exhaust ventilation, and glove-box manipulations. Regular PPE training, spill drills, and real-time air monitoring cut the risk of on-site release. Companies must maintain robust documentation, not only for their workforce’s well-being but to stay ahead of regulatory enforcement actions tied to hazardous chemicals.
Application Area
The molecule’s story cuts across several sectors. Agrochemical manufacturers rely on it as a backbone for synthesizing active herbicidal and pesticidal compounds. In dye and pigment production, chemists tweak its substituents to create colors that stick to textiles or plastics without losing intensity. Pharmaceutical research taps the molecule’s rigid skeleton—its halogen and nitro substitutions tune bioactivity in ways that plain benzene rings can’t match. Solid familiarity with its chemistry helps both industrial-scale and specialty chemical makers deliver products that meet today’s performance, safety, and environmental targets.
Research & Development
Academic teams and private R&D groups keep probing new ways to push this aromatic platform. Some aim to lower the ecological footprint of its synthesis, trimming the need for hazardous reagents. Others seek greener solvents and milder conditions during downstream transformations, aiming to reduce worker risk and emissions. There’s buzz about functionalizing its core under flow chemistry or using selective enzymatic methods to swap in new groups—a sign of growing synergy between synthetic chemistry and biotechnology. Tech transfer between academia and industry remains a high priority, especially as companies look for proprietary processes or unique molecular derivatives that sidestep expiring patents.
Toxicity Research
Data collected over decades paints a clear picture: 3-nitro-4-chlorobenzotrifluoride must be respected for its toxicological potential. Rodent studies point out that ingestion and inhalation at high doses cause liver and kidney stress, and chronic exposure raises questions about mutagenic risks, though public data remains inconclusive. Skin testing shows moderate irritation and occasional sensitization. Environmental fate studies show low biodegradability and persistence, signaling a risk for aquatic toxicity if released. Waste treatment regimes must fully oxidize residues or capture them in secure landfill operations. Regulatory reviews, especially those in the EU under REACH, keep pressing for more transparent hazard assessments, making real-time toxicity monitoring and accident tracking essential for any large-volume user.
Future Prospects
Despite regulatory challenges and growing environmental scrutiny, 3-nitro-4-chlorobenzotrifluoride holds a spot in the chemical supply chain. The future will likely see it in cleaner syntheses—possibly under continuous-flow systems or using catalytic shortcuts that steer clear of corrosive acids. Companies will keep investing in real-time emissions control and recycling, both to clear regulatory hurdles and to reduce operating costs. Bio-based paths might emerge, thanks to advances in enzyme engineering and green solvents, although these approaches remain on the horizon. As chemists continue to chase better selectivity and higher atom economy, derivatives of this molecule will turn up in specialty segments—custom pharmaceuticals, advanced imaging agents, and next-generation agrochemicals that cut use rates and environmental impact. The compound’s flexibility means it will stick around, provided that producers match technical prowess with top-tier compliance and responsible stewardship.
What is 3-Nitro-4-Chlorobenzotrifluoride used for?
Unpacking Its Role in Today's Chemical Industry
A name like 3-Nitro-4-Chlorobenzotrifluoride might sound complicated, but the reality behind it is pretty straightforward. This compound plays a significant part in the production of dyes, pigments, pharmaceuticals, and agricultural chemicals. Most people never hear about it, yet it shapes everything from the color of plastic toys to the pills some folks take for chronic conditions.
The Dye and Pigment Link
Anyone who’s walked through a craft store, bought new car parts, or used industrial-grade paints has probably seen the impact of this chemical up close. 3-Nitro-4-Chlorobenzotrifluoride helps create vibrant, stable dyes that stick around, even in sunlight or tough environments. These colors need to last because nobody wants that red car to turn orange or that new shirt's color to run after a single wash.
In my years working side jobs in manufacturing, I saw how tight deadlines and quality controls could be. Consistency always wins. This compound gives dye makers reliable results instead of patchy finishes or inconsistent hues.
Manufacturing Active Ingredients
3-Nitro-4-Chlorobenzotrifluoride often pops up in pharmaceutical labs. It serves as a building block for synthesizing various drugs—antifungals, arthritis medications, and even some antihistamines. Chemists prize it because its unique structure lets them tweak molecules for new effects or greater safety. New medicine costs a fortune to develop. Chemical shortcuts like these can save years and millions of dollars.
A detail worth mentioning: safety rules around these chemicals keep getting stricter for good reason. Companies need sound procedures, solid PPE, and good ventilation. Workers have seen firsthand what neglect leads to—headaches, breathing issues, and far worse. Precaution isn't just policy, it's common sense in industrial chemistry.
Supporting Crop Health
Farmers depend on products made with 3-Nitro-4-Chlorobenzotrifluoride, even if they’ve never heard of it. The chemical helps create pesticides and herbicides that protect against unwanted insects and weeds. Someone who’s grown their own tomatoes knows how quickly a bug problem spreads or invades the leaves. Making these protective agents accessible helps keep yields up and food supplies stable.
On the flip side, strict regulation matters here, too. People want food free from harsh residues. I remember my neighbor’s orchard—local testers came around every harvest to check for unwanted chemicals. Trust comes from following the rules and keeping public health in mind.
Environmental Concerns and Smarter Solutions
Environmental worries crop up anytime synthetic chemicals hit the market. Some of these compounds can stick around in soil or water. That’s led to stronger rules to curb their use and prevent accidents. Water contamination spills are devastating; they affect communities, wildlife, and even property values. From my own home near an industrial park, I saw neighbors raise their voices when waste management wasn’t tight enough. It worked—stricter oversight lowered risk for everyone.
Solutions don’t have to be complicated. Investing in better containment, upgrading scrubbing technology, and developing greener alternatives all play a part. When firms team up with environmental chemists, safer substitutes can come out of the lab and into real-world use faster.
The Bottom Line for Industry and Community
3-Nitro-4-Chlorobenzotrifluoride brings value to households and health care. Companies count on its reliability. Communities insist on transparent practices that protect their water and air. Finding a balance—safe processes, honest oversight, and ongoing innovation—serves everyone. Experience on the ground has shown me that positive change often starts with real conversations and accountability, not empty promises or shortcuts.
What is the chemical formula of 3-Nitro-4-Chlorobenzotrifluoride?
Chemical Formula: C7H3ClF3NO2
In the world of industrial chemistry, the name 3-Nitro-4-Chlorobenzotrifluoride might not turn heads like caffeine or aspirin, but this compound shapes how many processes work behind the scenes. At its core, the formula reads C7H3ClF3NO2. Breaking this down gives you a structure built from a benzene ring, jazzed up with chlorine, nitro, and trifluoromethyl groups. This combination creates a molecule packed with both stability and reactivity—something chemical manufacturers often chase.
Before I ever saw a barrel of this stuff, I learned to respect how adding a trifluoromethyl group—CF3—boosts resistance to degradation. In pharmaceuticals, that group can mean the difference between a pill that works and a pill that fizzles out. The same logic applies in agrochemicals. Farmers want solutions that stick around long enough to get the job done but not linger forever in the environment. By tweaking a benzene ring with both nitro and trifluoromethyl, chemists lock in a delicate balance.
Industrial Significance
This compound doesn't show up in your kitchen cabinet, but it lands right in the middle of chemical synthesis for dyes, pesticides, and pharmaceutical building blocks. Factories produce this chemical in batches and send it through multiple transformations. It’s a stepping stone for making other molecules that wind up in medicines and crop protection agents. Without compounds like 3-Nitro-4-Chlorobenzotrifluoride, these processes would slow down or cost more, which eventually touches every consumer.
The value sits in those groups on the ring. A nitro group adds some heft and directs reactions in predictable paths; a chlorine on the ring fine-tunes how future chemical reactions unfold. The trifluoromethyl group, bulky and electronegative, adds stability and shifts solubility. In a practical sense, that means manufacturers can create advanced molecules with fewer steps, saving energy and reducing waste.
Safety, Health, and Environmental Impact
No chemical commentary gets far without poking at safety and sustainability. Handling chlorinated nitro-aromatics calls for thick gloves and serious training—workers deal with toxic dust, and spills take special cleanup routines. The real challenge comes later, long after factory workers clock out. Trifluoromethyl groups, while useful, don’t break down in the wild as easily as simpler chemicals. Environmentalists have pointed out that these molecules, if released unchecked, can persist. That pushes the industry to find better containment, detoxification approaches, and greener substitutes where possible.
I’ve seen projects where companies tweak their process to capture more of the chemical before it reaches wastewater, and labs spend money chasing bacteria or catalysts that can crack these stubborn compounds. Regulatory agencies now expect tighter reporting and lower emissions, which sometimes hits the bottom line but builds trust with communities.
Seeking Safer Solutions
Science keeps nudging the industry to rethink chemistry. Some researchers design catalysts that work at lower temperatures and pressures, slashing waste and energy use. Others hunt for biodegradable alternatives, though finding a substitute that checks all the boxes isn’t simple. Building a better chemical often means taking risks—trying new processes or reimagining decades-old manufacturing steps.
The story of 3-Nitro-4-Chlorobenzotrifluoride reflects a bigger theme. Chemistry shapes everyday life in quiet ways, from the leading edge of drug design to the roots in sustainable manufacture. Its continued presence in the lab reminds everyone that progress rarely stands still, and the push for safety, efficiency, and responsibility never ends.
What are the safety precautions when handling 3-Nitro-4-Chlorobenzotrifluoride?
Why This Chemical Needs Respect
Working with 3-Nitro-4-Chlorobenzotrifluoride isn't a job anyone should take lightly. It's a yellow crystalline powder, often found in labs making dyes or specialty chemicals. Once you've spent time with strong-smelling organics, your nose recognizes something isn't right. It’s not just about the immediate impact—long-term exposure raises health risks, especially through inhalation or skin contact.
Personal Protection: No Shortcuts
Think back to times you've handled chemicals that burned your skin or made you cough—nobody forgets lessons like that. For this compound, high-quality gloves, splash-proof safety goggles, and solid lab coats are the bare minimum. I once encountered a case where thin disposable gloves failed, and trust me, that quick sting means the compound’s already at work. Respirators rated for organic vapors make a big difference where dust or fumes might form. Careless handling turns even a small spill into a hazard faster than you’d expect.
Ventilation Works Wonders
Stuffy, crowded labs always heighten risks. Good airflow means toxic particles are less likely to stick around for you to breathe in. Fume hoods protect everyone, not just the person pouring. In some older labs, I saw folks work without proper extraction, which always left that chemical taste in the air. Now, OSHA and NIOSH guidelines push for strong engineering controls, with local exhaust systems topping the recommendations. It’s not overkill—it’s insurance.
Storage Isn’t Just About Convenience
Some workers stash chemicals wherever there’s shelf space. This stuff deserves more respect. Store it in a cool, dry place, away from acids and bases—reactive chemicals never play nice together. Proper labeling in corrosion-proof containers keeps things safe for the next shift. After a misplaced drum led to a serious chemical reaction overnight, my team learned to double-check compatibility charts and fire safety rules. Simple habits like those stop bigger headaches.
Clean Up Right to Prevent Problems
Spills do happen, no matter how careful people act. The difference comes down to how quickly you respond and what you use. Commercial absorbents, nitrile gloves, and sealed waste bags beat old rags every time. Training pays off—if your crew freezes up or improvises with towels, they risk exposure. At companies following EPA guidelines, every new hire practices spill drills. We saw far fewer accidents in places where proper cleanup was second nature.
Invest in Training for Long-Term Safety
People who understand the risks act smarter. It’s not just about one-off training, either—staying sharp means regular refreshers and honest talk about incidents. Safety data sheets need to be more than shelf décor. At every chemical plant I’ve toured, the best records always came from teams who talked openly about what could go wrong. Sharing near-misses and fixes saves time and pain down the line.
Don’t Ignore Health Monitoring
Years of fieldwork taught me: symptoms sometimes creep up. Skin checks, lung function tests, and bloodwork spot early warning signs. Places investing in employee health catch more problems before they escalate. Keeping track protects not just workers, but families who share homes with them.
Bottom line: Respect the danger, build good habits, and keep people first. That’s what keeps everyone safe in the long run.What is the CAS number of 3-Nitro-4-Chlorobenzotrifluoride?
Getting Specific: CAS Number for 3-Nitro-4-Chlorobenzotrifluoride
3-Nitro-4-Chlorobenzotrifluoride goes by the CAS number 121-17-5. For those working with chemicals, a CAS number serves as the most straightforward way to identify exactly which compound sits in front of you. Many chemicals have long and sometimes confusing names. Laboratory staff, regulatory agencies, and manufacturing companies rely on the CAS system to avoid confusion and errors. Having a specific number—like 121-17-5—makes the paperwork clearer, the safety data sheets more reliable, and storage simpler.
The Role of 3-Nitro-4-Chlorobenzotrifluoride
This compound appears as a yellowish solid with a sharp, noticeable odor. People working in pharmaceutical, agricultural, or pigment industries might not realize how often this chemical turns up behind the scenes. Companies use it as an intermediate, a stepping stone that helps build larger, more complex chemical structures. Sometimes, it acts as a building block for medicines or crop chemicals. Other times, it finds a place in colorants or specialty materials that end up in electronics or plastics. That diversity means it supports several supply chains even though it spends most of its life invisible to the end user.
Why Getting the Right Compound Matters
A close friend worked in a small industrial lab, and once a shipment came labeled incorrectly. They needed 3-Nitro-4-Chlorobenzotrifluoride specifically, but a similar-sounding chemical arrived. That mistake could have ended up in ruined product, wasted money, or even health risks. Safety sheets, emergency first aid guidelines, workplace storage, and shipping processes all tie directly into the CAS number. If you walk into a stockroom and spot the number 121-17-5, there’s no guesswork—everyone knows what lives inside the drum.
Health, Regulation, and Responsibility
This compound doesn’t turn up in your home, but those who handle it look out for toxicological risks. Short-term exposure can cause irritation to eyes or skin. Prolonged inhalation demands real caution. Manufacturers must follow guidelines from agencies like the Occupational Safety and Health Administration (OSHA) in the United States, as well as global standards set by REACH in Europe. Rules about restricted uses, proper disposal, personal protective equipment, and traceability all tie back to having solid chemical identification. One small slip or mislabeling can bring regulatory fines, worker harm, or environmental hazards.
Improving Tracking and Safety
Many companies invest in digital inventory tracking. Assigning barcodes to containers, embedding CAS numbers, or linking chemical data sheets with inventory software helps reduce errors. More labs are training staff not just to read chemical names but to memorize the numbers most relevant to their daily work. In my own experience, creating checklists that rely on chemical codes trims down the margin for mistakes. Reviewing shipping records or purchasing orders for the right CAS code often catches issues before chemicals land on the shelf.
Real-World Solutions and Smarter Management
Manufacturers and suppliers should keep open lines of communication, especially if there’s ever any doubt about what’s about to be shipped. Asking for data sheets that reference the CAS number can catch mistakes ahead of time. Secondary labeling on containers, spot checks in the lab, and secure digital files all encourage better handling of specialty chemicals. Trust gets built not through fancy branding but accurate labeling and a dash of caution with every shipment. As an added step, connecting the CAS code to training and compliance programs helps crews stay aware, safe, and prepared for any change in supply.
How should 3-Nitro-4-Chlorobenzotrifluoride be stored?
The Everyday Reality of Handling Specialty Chemicals
I’ve spent years among dark bottles and safety labels. Anyone who’s worked chemical storage rooms knows two types of people: those who respect the rules, and those who think the rules are overkill—until something leaks or smokes. Specialty substances, like 3-Nitro-4-Chlorobenzotrifluoride, separate the careful from the careless pretty quickly. 3-NCBTF, found in labs and factories, carries enough risks to earn careful treatment, because mistakes sneak up quietly but hit fast.
Why Location and Environment Matter
Shelving choices aren’t arbitrary. Picking a cool, dry, ventilated place saves headaches later. Even the best chemical doesn’t like sitting in a hot, muggy spot or soaking up sunlight. Elevated temperatures and sunlight push reactions you don’t want—sometimes that’s discoloration, sometimes something nastier. Humid rooms? They encourage corrosion on containers, meaning someday you reach for a bottle and discover the screw cap has fused shut or, worse, started to leak.
Separation Saves Lives
Early in my career, I learned to check not just the label, but also what sits beside each bottle. 3-Nitro-4-Chlorobenzotrifluoride should never stand next to strong oxidizers, bases, or reducers. Cross-contamination doesn’t announce itself until a headache, a cloud, or a ruined experiment gives it away. Many chemical incidents trace back to storing incompatible chemicals together. At best, that means lost materials. At worst, everyone clears the building.
Containment: Respect for the Routine
Good containers earn trust. Glass and specialized plastics handle 3-NCBTF’s quirks. The closures seal tight but also open without violent effort—no one wants to wrestle a lid off. Labels should stay legible, because playing “mystery bottle” never ends well. Spill trays under shelving help too; a small leak escapes notice quickly unless you check regularly, but sealed secondary containment stops a problem from spreading.
Temperature Control Over Carelessness
Most labs pick somewhere between 15-25°C for storing chemicals like this. I heard jokes about “cold storage for everything” but too much chill brings condensation and cracked bottles, so moderation works best. Even the light fixtures in a chemical room matter: fluorescent tubes don’t warm up shelves or add UV the way sunlight can. Each piece of the environment works to slow down decomposition and keep fumes down.
Personal Protective Equipment and Training
Nobody wants a whiff of 3-Nitro-4-Chlorobenzotrifluoride or the irritation it brings. Handling always calls for gloves, goggles, and sometimes a lab coat—no shortcuts, even for “quick” storage jobs. Training saves fingers and eyes. At my old shop, anyone caught handling bottles barehanded owed the rest of us donuts. It became a point of pride to follow the right steps, because skin or eye troubles from exposure don’t wash off in a hurry.
Planning for Accidents Before They Happen
Not every spill is a crisis, but planning for one makes a difference. Absorbent pads, neutralizing agents, and a clear walk to the eyewash station all keep small slipups from turning into scenes you remember for years. In some places, workers keep extra containers close at hand for transferring small amounts—this reduces the size of any accident.
Responsible Disposal
Old inventory becomes a risk over time. I’ve seen bottles older than some of the students in the lab—never a good sign. Scheduled audits, disposal lists, and proper hazardous waste pickup keep these specialty chemicals from outstaying their welcome. Keeping records of incoming and outgoing substances not only matches good regulatory practice, it helps every colleague sleep at night.
Pushing for a Culture of Safety
Everything circles back to respect: for the chemical, for your coworkers, and for good order. Storing 3-Nitro-4-Chlorobenzotrifluoride safely is more than rules. It’s an attitude visible in every uncluttered shelf and every checked label. Safety isn’t a product you open from a box; it’s a habit you build and pass along to the next person who takes over your shift.
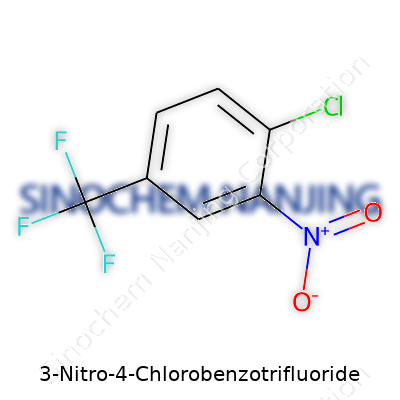
| Names | |
| Preferred IUPAC name | 1-chloro-2-nitro-4-(trifluoromethyl)benzene |
| Other names |
3-Chloro-4-nitrobenzotrifluoride Benzotrifluoride, 3-chloro-4-nitro- 1-Chloro-2-nitro-4-(trifluoromethyl)benzene 2-Nitro-1-chloro-4-(trifluoromethyl)benzene 4-Chloro-1-nitro-2-(trifluoromethyl)benzene |
| Pronunciation | /ˈθriːˌnaɪtroʊˌfɔːrˌklɔːroʊˌbɛnzoʊˌtraɪˈfluːəraɪd/ |
| Identifiers | |
| CAS Number | 121-17-5 |
| Beilstein Reference | Beilstein 1887922 |
| ChEBI | CHEBI:82259 |
| ChEMBL | CHEMBL3622381 |
| ChemSpider | 140262 |
| DrugBank | DB08717 |
| ECHA InfoCard | 03b147e2-58af-46c5-99bc-f2b4f1f0ba4d |
| EC Number | 121-11-3 |
| Gmelin Reference | 82151 |
| KEGG | C19233 |
| MeSH | D015424 |
| PubChem CID | 85978 |
| RTECS number | GN8575000 |
| UNII | V81Q8A64YX |
| UN number | UN2674 |
| CompTox Dashboard (EPA) | DTXSID7054226 |
| Properties | |
| Chemical formula | C7H3ClF3NO2 |
| Molar mass | 259.56 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Pungent odor |
| Density | 1.54 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 2.94 |
| Vapor pressure | 0.11 mmHg (25°C) |
| Acidity (pKa) | pKa = -2.09 |
| Basicity (pKb) | 12.49 |
| Magnetic susceptibility (χ) | -68.3×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.5430 |
| Viscosity | 1.53 mPa·s (25°C) |
| Dipole moment | 3.41 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 353.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −154.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1531.7 kJ/mol |
| Pharmacology | |
| ATC code | |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07, GHS09 |
| Signal word | Danger |
| Hazard statements | H315, H319, H335 |
| Precautionary statements | P261, P264, P271, P273, P301+P312, P304+P340, P305+P351+P338, P312, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2-2-1 |
| Flash point | Flash point: 113°C |
| Autoignition temperature | 540°C |
| Lethal dose or concentration | LD50 Oral Rat 1870 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 3190 mg/kg |
| NIOSH | NA222 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 10 mg/m³ |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
3-Nitrobenzotrifluoride 4-Chlorobenzotrifluoride 3-Chloro-4-nitrobenzotrifluoride 3-Nitro-2-chlorobenzotrifluoride 3-Nitro-4-chlorotoluene 3-Nitro-4-fluorobenzotrifluoride |

