3-Nitro-1,2-Xylene: In-Depth Commentary
Historical Development
Chemical compounds often carry a long story, and 3-nitro-1,2-xylene is no exception. Back in the early days of synthetic chemistry, aromatic nitro compounds like this one attracted the curiosity of pioneers looking for new dyes, explosives, and intermediates for pharmaceuticals. The journey of 3-nitro-1,2-xylene traces back to the drive for deeper control over substitution patterns on benzene rings. Early patent literature and academic publications pointed to the growing awareness of its value as a building block for more advanced molecules. Labs in Germany and the United States ramped up work on xylene derivatives in the mid-20th century, exploring their reactivity and potential. It’s remarkable how much today's chemical toolbox owes to the doggedness of those early chemists armed with glassware, basic reagents, and handwritten notebooks.
Product Overview
3-Nitro-1,2-xylene belongs to the nitroaromatic family, a group that underpins much of organic synthesis and applied chemistry. In this compound, two methyl groups have settled onto the aromatic ring alongside a nitro group. This structure helps explain its value, since substituted aromatic rings expand the possibilities in drug development, materials science, and even agricultural chemistry. The molecule sees use both on its own and as a stepping stone to more complex compounds. Industrial players look for this compound either in research settings or in plans for full-scale production, so purity, labeling, and chain of custody matter every step of the way.
Physical & Chemical Properties
In the lab, 3-nitro-1,2-xylene stands out due to its pale yellow crystalline appearance. It doesn’t dissolve well in water, but it blends readily with many common organic solvents, including ethanol and ether. The melting point sits roughly in the range of 60 to 62°C, which helps in handling and storage. Boiling this chemical requires pushing temperatures north of 290°C. The arrangement of its functional groups shapes how it reacts. The nitro group introduces strong electron-withdrawing power, making the ring less reactive toward electrophilic aromatic substitution and more susceptible to nucleophilic attack under special conditions. Both the physical and chemical traits make it easy to spot during chromatography or spectroscopy.
Technical Specifications & Labeling
Any reputable supplier should display a clear product code, CAS number, chemical formula (C8H9NO2), and lot number on the packaging. Purity levels above 98% matter for most high-precision work, and suppliers must back this up with supporting analytical data, usually via HPLC or GC-MS. A well-run lab checks for byproducts, water content, and the absence of other xylene isomers. Packaging often means dark glass bottles to shield the compound from light, with tight seals to minimize exposure to air and moisture. Detailed safety and hazard labeling goes beyond regulatory requirements and protects personnel on the job.
Preparation Method
Getting a clean batch of 3-nitro-1,2-xylene typically involves direct nitration of ortho-xylene. Reacting o-xylene with a mixture of nitric and sulfuric acid under controlled temperatures brings on selective substitution. The process demands close monitoring of temperature, acid ratios, and mixing time to keep unwanted byproducts in check. After nitration, extracting and purifying the product with solvents, followed by recrystallization, gives workers a product that meets research-grade standards. Skilled chemists have studied ways to fine-tune yields and safety by tinkering with reaction conditions, including continuous flow setups and greener reagents.
Chemical Reactions & Modifications
This molecule serves as a springboard in organic synthesis, thanks to that nitro group. Reduction turns the nitro group into an amine, unlocking routes to an entire spectrum of aromatic amines useful in both dyes and pharmaceuticals. Catalytic hydrogenation over palladium or tin(II) chloride reduction stand out as common methods. The methyl groups can be oxidized, setting up even more options for chemical manipulation. Some researchers have tested coupling reactions, including Suzuki and Heck modifications, tapping into the reactivity of the aromatic core. If you’ve spent time in a synthetic chemistry lab, you know that a simple change on a benzene ring can open doors to dozens of reaction pathways.
Synonyms & Product Names
You run into a fair number of names for 3-nitro-1,2-xylene in both academic and commercial settings. It goes by names like o-xylene, 3-nitro-, 1,2-dimethyl-3-nitrobenzene, and also appears simply as 1,2-xylene, 3-nitro derivative. Knowledge of these various synonyms avoids costly mix-ups during procurement, project planning, or regulatory filings.
Safety & Operational Standards
Handling nitroaromatics demands caution, since these compounds often bring with them toxicity risks and reactivity issues. 3-Nitro-1,2-xylene requires storage in cool, dry environments away from oxidizing agents and open flames. Regular personal protective equipment includes gloves, goggles, and a lab coat, and many seasoned chemists add face shields and fume hoods as extra measures. Spills must be wiped up right away, since tracing an unknown residue to a nitroaromatic involves tiresome analysis and cleanup. The compound can cause skin and eye irritation, and inhaling dust or vapors brings respiratory risks. Proper training makes a world of difference here, from emergency preparedness to understanding the latest changes in hazard communication protocols.
Application Area
Most of the interest in 3-nitro-1,2-xylene comes from its job as a synthetic intermediate. Fine chemical producers convert it to other derivatives more amenable to pharmaceuticals, pesticides, dyes, and specialty materials. Some research has focused on developing new ligands for catalysis, while others target oxime and amine modifications. Practical applications include new pigment formulations, where substituents on the benzene ring change the light absorption profile. Companies in the electronics industry test nitroaromatics for use in plastics and specialized resins. Years working with synthetic intermediates makes clear that adapting the process for each new field means going back to the drawing board with both safety protocols and documentation requirements.
Research & Development
Teams digging into new chemistry know the importance of robust starting materials, and 3-nitro-1,2-xylene consistently comes up in this regard. Recent efforts cover everything from alternative reaction pathways for its production to the development of greener solvents and catalysts. Scientists try out biocatalytic methods to cut down on the need for aggressive mineral acids. Advanced characterization tools like NMR, LC-MS, and crystallography help pin down microstructural details and confirm the absence of byproducts. Funding agencies want to see progress on both basic science and scalability, especially with the rise of stricter environmental regulations worldwide.
Toxicity Research
Work with nitroaromatics brings toxicology into sharp focus. 3-Nitro-1,2-xylene, like many related compounds, carries risks of acute and chronic effects. Exposure studies published over the past decade point to the need for tight limits on airborne concentrations and regular health monitoring for workers. Animal studies have shown doses causing liver and kidney issues at high exposures, and evidence links repeated skin contact to dermatitis and inflammatory responses. Environmental research traces how trace amounts from production sites break down or persist in air, soil, and groundwater, raising broader public health questions. Tight engineering controls, strict waste disposal standards, and real-time monitoring technologies all help close the gap between risk and safety.
Future Prospects
The coming years look to stretch the capabilities of 3-nitro-1,2-xylene on multiple fronts. New synthetic routes harnessing both renewable feedstocks and cleaner reagents signal a shift towards sustainability. As materials science keeps evolving, nitroaromatic intermediates can play a role in crafting advanced polymers and responsive materials for sensors. Advances in computation promise safer handling protocols by predicting reactivity patterns before hands-on work even begins. Tighter regulations on hazardous substances will both challenge and guide the industry’s evolution. Experience shows that collaboration between research teams, manufacturers, and regulators sets the pace for smarter, safer chemicals. Those who learn from past lessons and listen to new science will stay ahead.
What are the main uses of 3-Nitro-1,2-Xylene?
Chemicals Hiding in Plain Sight
Most people walk through life never thinking about compounds like 3-Nitro-1,2-Xylene. To a chemist or someone who’s worked in industrial sectors, though, this material is a familiar tool. This xylene derivative—yellow, pretty pungent—has a set of uses that connect straight to things plenty of us use or touch every day.
Making Dyes and Pigments Pop
Factories often use 3-Nitro-1,2-Xylene as an ingredient to build more complex organic molecules. Its structure makes it a favorite for making certain azo dyes. These dyes wind up in textiles, plastics, paper, and, sometimes, inks. If you’ve watched a red T-shirt fade slower than an off-brand equivalent, it’s no coincidence. The chemical structure, fine-tuned through compounds like this, lets manufacturers lock in bold color and help it last through repeated washes and sunshine.
Key Step in Specialty Chemicals
Years ago, I worked at a plant handling aromatic nitro compounds. There, 3-Nitro-1,2-Xylene stood out as a key intermediate. This meant that even if a consumer never saw it or even read its name, they still handled its legacy—whether through herbicides or pharmaceuticals.
Many herbicide manufacturers use nitro aromatics because they can tweak their molecules further, giving them just the right shape or charge. These products end up on fields, helping farmers protect crops and keep weeds under control, helping keep food available and affordable at scale. In pharmaceuticals, chemists seek these nitro derivatives as building blocks to craft molecules that can ease pain or treat disease. Regulatory guides require careful handling for these reasons, plenty of scrutiny follows them from factory to waste stream.
Laboratory Synthesis and Innovation
Academic and commercial labs lean on 3-Nitro-1,2-Xylene for small-batch syntheses, either while mapping new reactions or confirming old ones. Tough, persistent molecules like this one help students learn because reactions with nitroxylenes don’t always behave as textbooks promise. This gap, between theory and practice, fuels curiosity and steady progress.
In some labs, work starts here before branching into cross-coupling chemistry or complex pharmaceutical research. By exploring side reactions, new chemical pathways sometimes appear. These accidental discoveries push innovation forward, though it requires careful safety planning due to the compound’s toxicity and environmental impact.
Hazards and the Push for Responsible Handling
Handling chemicals like 3-Nitro-1,2-Xylene in the plant meant never dropping your guard. The nitro group doesn’t just smell harsh, it signals potential toxicity and fire hazard. Regulatory agencies, like OSHA and the EPA, ask companies to monitor air and water discharge closely to avoid endangering workers or communities.
Safer substitutes sometimes get discussed, especially as green chemistry movements pick up steam. Engineers and chemists now search for less hazardous alternatives that give similar performance. This work isn’t just technical; it’s ethical, as safer workplaces and a healthier environment matter to everyone down the line.
The Next Chapter
3-Nitro-1,2-Xylene may not get much attention outside chemical circles, but it plays a real role in paints, pharmaceuticals, agriculture, and research. Long hours in labs and on factory lines have shown me why every link in this chemical chain deserves respect. Strong safeguards, investment in new ideas, and clear-eyed awareness of risks all keep this compound’s benefits in the spotlight, without denying its dangers. Progress will hinge on what industry, science, and regulators learn together next.
What are the hazards and safety precautions for handling 3-Nitro-1,2-Xylene?
Recognizing the Dangers
3-Nitro-1,2-xylene shows up in the lab as a yellowish crystalline solid, though its appearance doesn’t give away the dangers it carries. Breathing in dust or fumes from this compound irritates airways and lungs. Some years back, I worked with nitroaromatic chemicals like this one—you can end up with a headache or even shortness of breath on careless exposure. Skin contact leads to rashes or something worse if you forget to use gloves. The risk grows when eyes and mucous membranes get involved, because even a splash can sting or damage eyes.
Fire hazards don’t lurk far behind. This compound doesn’t catch fire as fast as gasoline, but heating or sharp impact creates a risk of explosion. Anything with a nitro group deserves respect: heat, friction, or static sparks can trigger something unexpected, so energy, chemistry, and fire all get mixed in its profile.
Routes of Exposure
Folks run into this chemical mainly through inhalation, skin absorption, or swallowing small amounts. Spills that aren’t cleaned up right, or leaky containers, mean vapors build up fast. Once, a colleague developed a nasty cough after an unnoticed leak near a poorly ventilated hutch. Labs with strong airflow and clever spill management give everyone a better shot at avoiding health problems.
Personal Protection Comes First
Handling these crystals means donning gloves—nitrile offers decent protection. Giving up on goggles weighs heavy, because the smallest splash brings serious trouble. Long sleeves and a fitted lab coat reduce skin risks. Don’t forget a particulate mask if there’s any dust, and always aim for local extraction hoods instead of trusting open windows.
Storage deserves more thought than many give it. This isn’t a bench-top chemical for casual use. Air-tight, clearly labeled bottles and secure fireproof cabinets keep accidents rare. Strong acids, open flames, and incompatible reagents belong far away. Your mind rests easier knowing a spill kit sits within reach, and that team members practice drills for cleanup or, if things go badly, evacuation.
Emergency Measures Are Non-Negotiable
If exposure does happen, rinsing eyes or skin with clean water becomes urgent. Every second counts—no room for hesitation. Strip off clothes soaked with powder, move outside if the air feels thick, and never try to gut it out—medical attention always comes next. My memory stretches back to times when someone’s stubbornness made things worse; pride has no place here, only a willingness to ask for immediate help.
Solutions Start with Training and Mindset
Labs that succeed with chemicals like 3-nitro-1,2-xylene focus on regular training, not just paperwork or posters. Plenty of accidents sprout from shortcuts or trusting past success. Walking through updates on handling, storage, and what ifs, builds habits better than rules alone can ever achieve. Investing in reliable ventilation systems, emergency showers, and eye stations isn’t a budget extra; it’s a necessity. If management stays engaged—checking safety gear and reviewing incidents—everyone stays sharper and safer.
On a day with even small amounts of this chemical in use, the weight of responsibility presses down. Anyone handling 3-nitro-1,2-xylene needs to balance caution and respect for the risks, backed up by habits drilled through training and experience. This approach shields everyone—not just from regulatory trouble, but from harm that lingers long beyond a spill or exposure.
What is the chemical structure and formula of 3-Nitro-1,2-Xylene?
Structure at a Glance
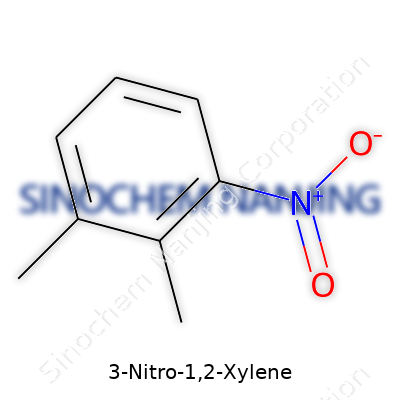
A look at 3-Nitro-1,2-xylene gives us a good snapshot of how small tweaks in a molecule change everything. At its core, this compound grows from xylene, a benzene ring with two methyl groups hanging on neighboring carbon atoms—these are found at the first and second positions. Chemists call this the ortho-configuration. Next, a nitro group (-NO2) latches onto the third carbon in the ring, pushing the boundaries of its reactivity. Put that together, and 3-Nitro-1,2-xylene shows up with the formula C8H9NO2.
Chemical Structure: What It Tells Us
I learned in organic labs that sticky details about structure make or break a target synthesize. On its benzene foundation, 3-Nitro-1,2-xylene carries two methyls (–CH3) placed next to each other, plus a nitro group just one seat away. It's easy to picture: start at the top of the benzene ring and count the carbons—put methyl groups at positions 1 and 2, the nitro hooks on at 3, and hydrogen atoms fill the remaining three carbons. This creates a molecule that's a little more reactive than basic xylene, since the nitro group pushes electron density around, and those methyls offer some shielding from further reactions.
Formula in Practice
This molecule’s formula, C8H9NO2, barely hints at its full story. It signals to folks in labs and industry what to expect in terms of reactivity or safety. Nitro groups pack both energetic and hazardous punch, while those methyls lend some stability. Having seen some nitro-aromatic compounds up close, I found that handling them with care isn't just protocol, it means real-world safety for everyone nearby.
Why This Structure Grabs Attention
Take xylene, a workhorse in paints, solvents, and chemical manufacturing—add a nitro group, and the molecule’s purpose shifts. Now, 3-Nitro-1,2-xylene often becomes an intermediate for dyes, pigments, or more complex drugs. Chemists value it precisely because the exact arrangement of that nitro group creates predictable reactions. Swapping the nitro’s spot changes how the molecule breaks apart or links with others. That saves time, cuts waste, and helps make purer products.
Troubleshooting Safety and Environment
In my time working with industrial chemicals, all nitro compounds set off alarms for safety crews. They sometimes carry explosive properties, especially when mixed with other reactive agents. People need solid training to store, move, and mix these chemicals safely. Regulation helps, but so does pushing for greener reaction methods—using less-toxic solvents or milder temperatures, for example. Universities and manufacturers look for catalysts or enzymes to tame harsh chemical steps, keeping danger and waste down.
Solutions and Forward Thinking
New sensor technology now helps detect nitro-aromatics in soil or water before they can build up into real hazards. Continued investments in stricter monitoring, emergency training, and greener chemistry play just as big a role as knowing the structure on paper. It’s always better to anticipate risks. Every detailed study into molecules like 3-Nitro-1,2-xylene lets chemists fine-tune the balance between usefulness, safety, and sustainability—turning chemical know-how into something that matters outside the lab too.
How should 3-Nitro-1,2-Xylene be stored and transported?
Getting Serious About Chemical Handling
Anyone who's worked in a lab or at a chemical plant learns that respect for hazardous materials isn't just about rules—it’s about everyone’s well-being. 3-Nitro-1,2-xylene looks like a routine aromatic compound on paper, but its nitro group turns it into a riskier friend. Health concerns, fire hazards, and environmental impact all loom if basic steps get skipped. From years spent on the industrial side, I’ve seen accidents spark up from nothing more than a mislabeled drum or a forgotten warning. Nobody wants to wind up one of those stories.
Direct Handling
The trick to managing 3-nitro-1,2-xylene starts with the right containers. Choose tight-sealing glass bottles or sturdy steel drums with chemical-resistant linings. These aren’t just suggestions—cheap out on packaging and the entire workplace pays the price. Clean labels and large hazard symbols cut down risk even further. Relying on memory, especially during shift changes, invites trouble you don’t need.
Conditions Matter
This chemical doesn’t like heat, sunlight, or sparks. Find a cool, dry space—ideally under 25°C—and shield the storage area from direct sun. Lock it up away from acids, strong oxidizers, or organic peroxides. Storing incompatible chemicals together starts a countdown clock for bad news. Each time regulations shift, dig out those Material Safety Data Sheets and make sure nothing’s changed. Obsessive? Maybe. But ask anyone who’s seen a chemical fire, and they’ll say it’s time well spent.
Transport Smart, Not Fast
Transferring 3-nitro-1,2-xylene brings fresh headaches. Transport vehicles need ventilation and must keep the cargo cool and upright. Barricade those drums—tie-downs or upright racks make shifting during a sudden swerve a non-issue. DOT and ADR rules force every handler to wear protective equipment, from chemical splash goggles to nitrile gloves. That PPE is more than a box-tick; accidental exposure can mean skin burns, eye damage, or lung irritation.
Stripe vehicles with unmistakable hazard markings and documentation. Emergency response guides should ride shotgun with the cargo. It’s easy to skip steps and count on luck, but every trucker and logistics worker owes a safe shift to their family. Fixing a spill on the road means special cleanup crews, possible evacuations, and fines. All avoidable if someone triple-checks before the engine even starts.
Culture Over Compliance
Anyone involved with chemicals has heard about “compliance culture.” The best teams don’t just chase checklists—they ask questions, hold each other accountable, and expect managers to back them up. Training sessions with live scenarios spark more learning than policy memos ever could. Employees speaking up about sketchy storage or shortcuts give a company its backbone. In honest operations, E-E-A-T flows naturally because people know their stuff, act on it, and share openly if something’s off.
Cleaner Futures
Chemicals like 3-nitro-1,2-xylene bring big benefits—if people refuse to cut corners. Investing in better storage racks, electronic tracking, and regular audits pays off, especially for warehouses handling multiple risky substances. From experience, nothing replaces boots-on-the-ground vigilance and caring what happens beyond your own shift. Those lessons stick because lives, health, and the environment stay safer when people give a damn about the details.
Is 3-Nitro-1,2-Xylene available in different grades or purities?
Looking Closer at Purity Choices
The chemical industry runs on details. Take 3-nitro-1,2-xylene, for example. People working with this compound quickly discover the importance of purity. Not every project calls for the same quality, and the available grades reflect that reality. Purity isn’t about prestige—it comes down to the needs of a process, the reliability of results, and, sometimes, safety on the job.
Real-World Experience With Variability
I’ve worked alongside chemists who wouldn’t dream of ordering a drum of something unless the Certificate of Analysis spelled out the impurities, down to the fraction of a percent. Different tasks have different tolerance levels. In academic labs, analytical or “reagent” grade often shows up; researchers want to avoid chasing ghosts in their experiments, so they go for higher purity—even if it costs more. People in large-scale manufacturing might opt for a technical grade if the downstream process doesn’t care about a minor contaminant.
Talking to colleagues in industries like dyes or coatings, I learned that some grades lean toward affordability over purity. Purity jumps up the priority list in pharmaceuticals and electronics, where a small impurity could throw off an entire batch. I remember a case at an electronics firm where a trace contaminant in 3-nitro-1,2-xylene ruined a production run and cost many hours in troubleshooting.
Facts on Grades and Testing
Producers offer various purities because no one likes to pay more than they need to. Chemists and purchasing managers can choose between “technical,” “laboratory,” and high-purity grades, depending on what they’re making. Technical grade usually fits for bulk work, but testing and strict documentation become a must once regulations or consumer safety get involved.
Analyses can pinpoint even trace levels of byproducts or related isomers. Quality control teams pore over gas chromatography charts, making sure something labeled 98% actually means what it says. If the paperwork fails, buyers find out fast—resulting in lost money and possibly product recalls. In some regions, laws cover certain applications, demanding minimum specifications for things like pharmaceuticals or food packaging.
Safety and Environmental Factors
Ignoring the grade can give more than just bad results. Lower purities sometimes bring along hazardous additives or unreacted starting materials. Experienced technicians know to ask pointed questions about trace ingredients—not just the main chemical itself. These hidden extras can trigger workplace incidents or headaches for environmental compliance; my own experience reviewing safety data sheets has taught me that lower-grade solvents sometimes come with unexpected risk warnings.
Moving Toward Fewer Surprises
With so much riding on the right chemical, more companies look for full transparency from suppliers. Reliable sources work hard to keep their batches consistent and are not shy to share data sheets and analysis details. Some buyers request third-party certificates or even perform their internal tests, just to stay one step ahead. Tight budgets can tempt a team to accept a less-refined product, but the cost of fixing problems later usually outweighs the savings.
Smart Strategies for Better Outcomes
Choosing the right purity grade brings headaches down the road—or keeps them away. Users who talk directly to their suppliers about intended uses, tolerable levels of contaminants, and delivery expectations usually get fewer surprises. Regulatory guidelines also play a growing role in decision-making. As stricter standards roll out globally, the chemical industry as a whole leans into traceability and reliable record-keeping, driven by both law and hard-learned lessons.
| Names | |
| Preferred IUPAC name | 2-Methyl-3-nitrotoluene |
| Other names |
1,2-Dimethyl-3-nitrobenzene 3-Nitroxylene 3-Nitro-o-xylene m-Nitro-o-xylene 3-Nitro-1,2-dimethylbenzene |
| Pronunciation | /ˈθriː ˈnaɪtroʊ waɪˈliːn/ |
| Identifiers | |
| CAS Number | 603-82-3 |
| Beilstein Reference | 1598737 |
| ChEBI | CHEBI:77256 |
| ChEMBL | CHEMBL22998 |
| ChemSpider | 170358 |
| DrugBank | DB02141 |
| ECHA InfoCard | 100.015.493 |
| EC Number | 202-716-6 |
| Gmelin Reference | 80892 |
| KEGG | C14365 |
| MeSH | D016555 |
| PubChem CID | 13655 |
| RTECS number | XZ9625000 |
| UNII | R2J151FU85 |
| UN number | UN1664 |
| Properties | |
| Chemical formula | C8H9NO2 |
| Molar mass | 150.15 g/mol |
| Appearance | Light yellow solid |
| Odor | aromatic |
| Density | 1.17 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 1.98 |
| Vapor pressure | 0.019 mmHg (25 °C) |
| Acidity (pKa) | pKa = 11.15 |
| Basicity (pKb) | 12.43 |
| Magnetic susceptibility (χ) | -64.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.595 |
| Viscosity | 2.13 cP (20 °C) |
| Dipole moment | 3.37 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 229.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 143.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3882.1 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07,GHS08 |
| Signal word | Danger |
| Hazard statements | H302 + H315 + H319 + H332 |
| Precautionary statements | P261, P264, P273, P280, P301+P312, P302+P352, P305+P351+P338, P337+P313, P330, P501 |
| NFPA 704 (fire diamond) | 3*2*0 |
| Flash point | 81 °C |
| Autoignition temperature | 579°C |
| Explosive limits | 1.6–?% (at 130°C) |
| Lethal dose or concentration | LD50 oral rat 1970 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1850 mg/kg (rat, oral) |
| NIOSH | SN4525000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of 3-Nitro-1,2-Xylene is "Not established". |
| REL (Recommended) | 0.1 ppm |
| IDLH (Immediate danger) | IDLH: 100 mg/m³ |
| Related compounds | |
| Related compounds |
1-Nitro-2,3-xylene 2-Nitro-1,3-xylene 3-Nitrotoluene 1,2-Dimethylbenzene (o-Xylene) Nitrobenzene |

